ORIGINAL RESEARCH ARTICLE
Staged dissection reduces blood loss in surgery for metopic synostosis
Anna Sundelina,b, Madiha Bhatti-Søftelanda,b, Ingrid Stubeliusa,c, Tobias Hallén MDd,e, Robert Olssond,e, Giovanni Maltesea,b, Peter Tarnowa,b, Karin Säljöa,b and Lars Kölbya,b
aDepartment of Plastic Surgery, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden; bDepartment of Plastic Surgery, Sahlgrenska University Hospital, Region Västra Götaland, Gothenburg, Sweden; cDepartment of Anaesthesiology and Intensive Care, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Sahlgrenska University Hospital, Gothenburg, Sweden; dDepartment of Clinical Neuroscience, Institute of Neuroscience and Physiology, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden; eDepartment of Neurosurgery, Sahlgrenska University Hospital, Region Västra Götaland, Gothenburg, Sweden
ABSTRACT
INTRODUCTION: Fronto-orbital remodelling for metopic synostosis is an extensive operation with substantial blood loss, particularly from emissary veins in the glabellar region. One possibility to reduce blood loss may be to stage dissection and cauterise anomalous emissary veins before dissecting in the subperiostal plane.
OBJECTIVE: The aim of the present study was to compare perioperative bleeding using a staged dissection in the glabellar region with the traditional subperiostal dissection technique during surgery for metopic synostosis.
METHODS: All consecutive patients operated for metopic synostosis with the new staged dissection technique (T2) were included. For comparison, the most recent equal number of cases operated with the traditional dissection technique (T1) were included. Age, sex, weight, surgical technique (spring or bone graft), perioperative blood loss, perioperative blood pressure, per- and postoperative blood transfusion, operation time and length of hospital stay were registered.
RESULTS: A total of 80 patients were included; 40 T1 and 40 T2, respectively. Perioperative blood loss was significantly reduced with the new staged dissection technique. Blood loss in absolute numbers was reduced from 160.0 (120-240) (median and (interquartile range)) ml to 150 (102.5-170.0) ml, p=0.028. Blood loss in relation to patient weight was reduced from 24.2 ml/kg (15.3-33.3) to 18.7 (16.6-23.1) ml/kg, p=0.024. As percentage of total blood volume, blood loss was reduced from 32.3 (20.3-32.3) % to 29.9 (18.4-30.8) %, p=0.024. No other variables differed significantly between the techniques.
CONCLUSION: In summary, staged dissection technique in the glabellar region, allowing control of the emissary veins, reduces perioperative blood loss during surgery for metopic synostosis.
KEYWORDS: Craniosynostoses; metopic synostosis; haemostasis; blood loss; surgical; haemostatic techniques
Citation: Journal of Plastic Surgery and Hand Surgery 2024; 59: 146–152. DOI: https://doi.org/10.2340/jphs.v59.42160.
Copyright: © 2024 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 26 August 2024; Accepted: 1 October 2024; Published: 12 November 2024
CONTACT Anna Sundelin anna.sundelin@gu.se Sahlgrenska University Hospital, Department for Transplant Surgery, 413 45 Gothenburg, Sweden
Competing interests and funding: The authors have no financial conflicts of interest to declare.
Introduction
Metopic synostosis (MS) or premature fusion of the metopic suture results in lateral growth restriction of the frontal bones and is characterised by trigonocephaly, ridge over the metopic suture, hypotelorism, temporal hollowing and increased biparietal distance [1, 2]. The incidence of MS in Sweden is 1.7 out of 10,000 births [3]. Over the last decades, there has been a significant increase in the number of patients with MS, both in absolute numbers and in proportion to the total number of craniosynostosis [4–6]. This increase now confirms that MS is the second most frequent type of isolated craniosynostosis after sagittal synostosis [7, 8].
Surgical treatment of MS commonly includes frontal remodelling and correction of hypotelorism, aiming to correct the shape of the skull and reduce the risk of increased intracranial pressure [9]. Different surgical methods are used for surgical treatment in different centres. Blood loss is a concern in the surgical management of MS and has been analysed to be the main cause of morbidity in craniosynostosis surgery [10]. Substantial blood loss during craniosynostosis surgery is managed by allogenic blood transfusion. Even though allogenic blood transfusions are generally safe, there are associated risks in morbidity and mortality in paediatric patients, for example transfusion-related lung injury (TRALI), allergic reactions, haemolytic and non-haemolytic reactions, transfusion-related circulatory overload (TRCO) [11]. There is also a risk for development of antibodies against red blood-cell antigen and alloimmunisation associated to repeated transfusions; therefore, it is of importance to reduce transfusions [12].
The MS skull does not only present a different shape and volume proportion than a normal skull, it also differs in vascular morphology. In a previous study of preoperative computer tomography (CT) images, it was confirmed that almost half of the infants (45%) with MS have transosseous emissary veins with origin from extracranial vessels that run through the cranium a few centimetres above the nasofrontal suture and terminate in superior sagittal sinus (SSS) [13]. These emissary veins were not observed in a control group of patients without synostosis; the controls were however, younger than the MS cohort (mean age 5.5 months, range 2 weeks-11 months in the controls and median age 11.1 months, range 6 days-19 months in the patient cohort) [13]. The morphology of diploic veins has also been studied. It was found that craniosynostosis skulls have less large diploic veins than anatomical normal skulls [14]. During open surgery of MS, substantial blood loss typically occurs when dissecting in the subperiosteal plane over the glabellar region. The dissection results in tearing of emissary veins and consequently substantial bleeding. With the intention to reduce the perioperative bleeding in MS surgery, Di Rocco et al. described a surgical technique leaving a triangular shaped bone segment intact in order to facilitate access to the area and control haemostasis before detaching the bone triangle [15].
The present study aims to evaluate a modified surgical technique for MS, with the intention to reduce perioperative blood loss.
Materials and methods
Patients
This is a retrospective study based on data extracted from the Gothenburg Craniofacial Registry, in which data regarding all patients treated for craniosynostosis at Sahlgrenska University Hospital, Gothenburg, Sweden are collected.
All consecutive patients operated for MS with a new dissection technique in the glabellar region between January 2018 and January 2022 were included. For comparison, an equal number of the most recent consecutively operated patients with the traditional technique were included, operated between February 2017 and March 2022.
Demographic and surgical data such as age at surgery, sex, weight at surgery, surgical technique (spring or bone transplant), perioperative blood loss, per- and post-operative blood transfusion, operation time and length of stay was extracted from the registry. Direct continuous intraarterial blood pressure was measured during MS surgery using an arterial catheter placed in any upper limb. Values for intraoperative heart rate, systolic and diastolic blood pressures were monitored continuously and registered in 5-min intervals. Anaesthetic records were used for data extraction. Exclusion criteria were presence of combined synostosis, a craniofacial syndrome or diagnosed haematological disorder.
Surgical techniques
Traditional technique (T1)
With the patient in supine position, a zigzag bi-coronal incision is made and a subperiosteal dissection of the scalp is undertaken (illustrated in Figure 1). The frontal bones are exposed and the dissection continues into the orbits to expose the orbital roofs. The forehead is removed with a craniotome burr, and the frontal bones are separated and moulded into a more rounded shape. The supraorbital complex is detached and split in the midline in patients older than 6 months of age, and a bone graft is placed between the two halves of the supraorbital complex for correction of forehead contour and hypotelorism. In patients younger than 6 months of age, the lateral forehead is advanced by a temporal tongue-in-groove incision preserving the bony attachment only in the glabellar region. In younger patients, an 8N spring is placed in the glabellar region in order to correct hypotelorism and lateralise the frontotemporal contour.
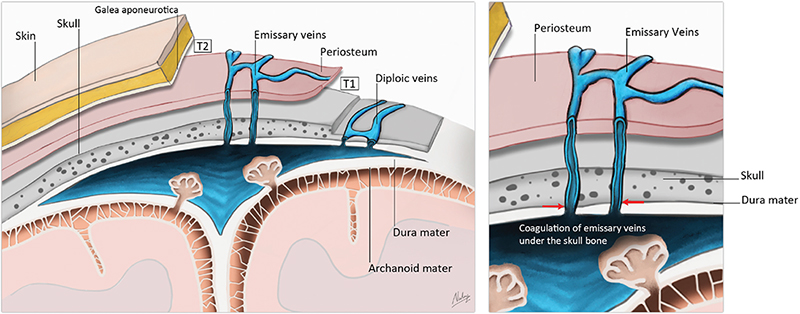
Figure 1. Illustration of anatomy in the glabellar region. Emissary veins pass through foramina in the skull and provide a venous connection between extracranial veins of the skull and intracranial dural venous sinuses (SSS). Diploic veins run through the spongy part of the skull bone, between the outer and inner layer of the cortical skull bone and are drained in emissary veins or dural venous sinuses. With subgaleal frontal dissection, emissary veins remain intact, and coagulation of those veins can be performed under the skull bone.
New dissection technique (T2)
With the new technique, the scalp dissection is undertaken in the subgaleal plane down to about 15 mm above the orbital rims (illustrated in Figure 1). The frontal bones are then removed, followed by meticulous haemostasis with special attention to the emissary veins under the supraorbital complex (illustrated in Figure 2). Thereafter, the dissection continues in the subperiosteal plane into the orbits to expose the orbital roofs, and the operation continues as described above.

Figure 2. (A) Intraoperative images. Access to emissary veins under the bone for haemostasis before further dissection. (B) After emissary vein haemostasis the dissection continues in a subperiosteal plane into the orbits.
Statistical analysis
All statistical analyses were carried out using Statistical Package for the Social Sciences (SPSS) Statistics Software version 28 (IBM Corp., Armonk, NY, USA). Patient characteristics and operative outcomes for continuous normally distributed variables are presented as mean values with standard deviation (SD), significance analyses were based on Student’s t-test. Categorical variables are presented with frequencies and percentages with significance analyses based on Chi-Square test. The variables for estimated blood loss (mL, mL/kg and %) are presented as median with interquartile range (IQR) (25th–75th percentiles) and significance analysis was based on Mann-Whitney test since the data were not normally distributed. Histograms and boxplots were used to visualise the difference in distribution of blood loss (mL/kg) and distribution of SDs in differentiated blood pressure values.
Blood pressure was analysed statistically in two ways. The time frame for blood pressure analysis was set to 1 h from start of the operation because the frontal dissection and blood loss from emissary veins was supposed to appear within that period. Firstly, the fundamental principle that blood pressure drops as a reaction to a significant and sudden blood loss was analysed in the perioperative blood pressure curves for systolic blood pressure (SBP) to identify occurrence of intraoperative hypotension (IOH). The SBP baseline was calculated as a mean value of up to five intraarterial values for SBP measured during anaesthesia before the start of the operation. This resulted in an individualised baseline value for each patient. A relative drop of 20% in mmHg from baseline was used as threshold for IOH. The frequency of IOH was dichotomised and analysed as other categorical variables.
Secondly, as the blood pressure drops as a reaction to blood loss; the anaesthetist manages the situation and corrects the blood pressure by administrating fluids and medications. In theory, that would result in more variability in blood pressure for patients with large and sudden blood loss. An attempt was made to model blood pressure data as time series using statistical autoregressive moving-average (ARMA) models. Linear interpolation was performed for missing values. Time series were tested with different statistical parameters and AIC (Akaike information criterion) was compared across models. The estimated SDs from the model with the best AIC were identical to the SDs obtained by differencing and taking the SD of the differences for each series. For analysis of variability the SDs from differenced time series were analysed as other continuous normally distributed variables.
Ethics
The study was approved by the Gothenburg Ethics Committee (Dnr 784:11) and conducted according to the principles stated in the Declaration of Helsinki.
Results
Patient characteristics
In total there were 43 patients operated with the new dissection technique (T2) between January 2018 and January 2022. Of those, 3 patients were excluded because of combined craniosynostosis which left us with a cohort of 40 patients in T2. The remaining cohort of 40 patients in T2 were compared with 40 patients operated with the traditional technique (T1) between February 2017 and March 2022. Descriptive characteristics are presented in Table 1. The age at surgery was 188 ± 56 (mean ± SD) days for T1 and 179 ± 49 days for T2, respectively. Overall, female: male ratio was 1:3.7. Patients in T1 showed a slightly higher average weight (7.9 ± 1.3 kg vs. 7.8 ± 1.2 kg) but the difference was not significant. Surgical technique related to age below or above 6 months, i.e. spring or bone transplant, showed an even distribution between the groups (62.5% spring and 37.5% bone transplant in both groups). Operation time was 150.4 ± 31.1 min in T1, and 140.9 ± 22.2 min in T2 (p = 0.122).
Blood loss
Outcome variables are presented in Table 2. Estimated blood loss showed significantly lower values after the new dissection technique (T2) compared to the traditional dissection technique (T1) for blood loss in absolute numbers (mL), in relation to patient weight (mL/kg) and as percentage of total blood volume (TBV). Blood loss in absolute numbers was 150.0 (102.5–170.0) (median and IQR) mL in T2 and 160.0 (120.0–240.0) mL in T1, respectively; p = 0.028. Blood loss in relation to patient weight was 18.7 (16.6–23.1) mL/kg in T2 and 24.2 (15.3–33.3) mL/kg in T1, p = 0.024. For visualisation of the distributions see Figures 3 and 4. Blood loss as percentage of total blood volume was calculated assuming a total blood volume of 80 mL/kg. The percentual blood loss was 23.3 (17.1–28.9) % in T2 and 30.3 (19.1–41.6) % in T1, p = 0.024.
| T1 (n = 40) | T2 (n = 40) | p | |
| Blood loss (mL)1 | 160.0 (120.0–240.0) | 150.0 (102.5–170.0) | 0.028 |
| Blood loss, (mL/kg)1 | 24.2 (15.3–33.3) | 18.7 (13.7–23.1) | 0.024 |
| Blood loss (%)1 | 30.3 (19.1–41.6) | 23.3 (17.1–28.9) | 0.024 |
| Transfusion perioperative, n (%) | 0.077 | ||
| Yes | 40 (100) | 37 (92.5) | |
| No | 0 (0) | 3 (7.5) | |
| Transfusion perioperative volume, (mL) | 201.3 ± 92.5 (171.7–230.9) n = 40 |
177.8 ± 74.3 (152.6–202.9) n = 37 |
0.228 |
| Transfusion perioperative (mL/kg) | 25.8 ± 11.3 (22.2–29.5) n = 40 |
23.2±10.1 (19.8–26.6) n = 37 |
0.279 |
| Transfusion postoperative, n (%) | 0.576 | ||
| Yes | 7 (17.5) | 9 (22.5) | |
| No | 33 (82.5) | 31 (77.5) | |
| Hospital stay (days) | 6.0 ± 1.7 (5.4–6.5) | 6.0 ± 1.7 (5.4–6.5) | 1.000 |
| Frequency of IOH, n (%) Yes No |
11 (28.2) 28 (71.8) n = 39 |
7 (17.5) 33 (82.5) n = 40 |
0.257 |
| Variability (mmHg) | 6.4 ± 3.4 (5.3–7.5) n = 39 |
5.8 ± 1.9 (5.2–6.4) n = 40 |
0.316 |
| Data are presented as mean ± SD (95% confidence interval) unless otherwise stated. IOH: intraoperative hypotension. 1Not normally distributed variable presented as; median (25th–75th percentile). |
|||
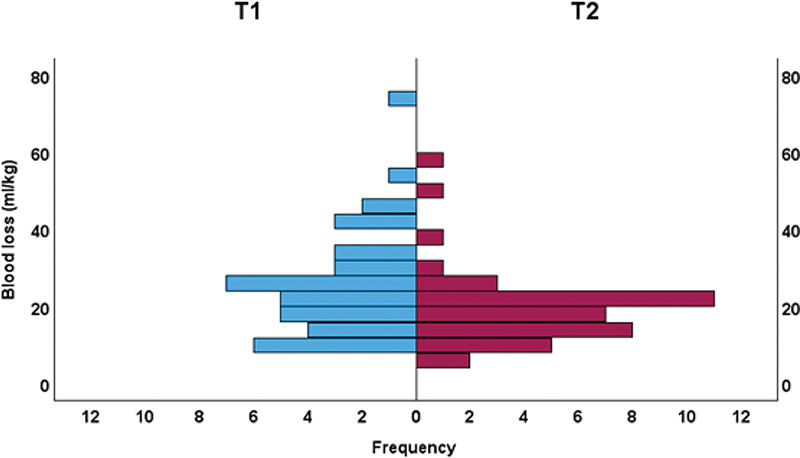
Figure 3. Histogram of blood loss (mL/kg) distribution. Blue T1; Red T2.
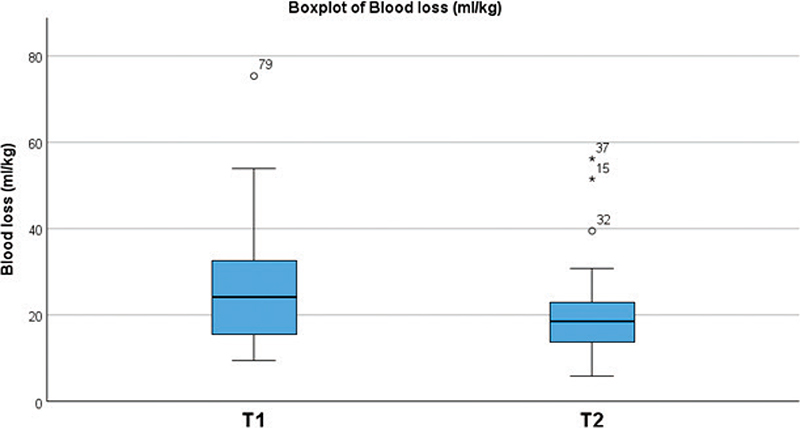
Figure 4. Boxplot of blood loss (mL/kg) distribution. Blue T1; Red T2.
Blood transfusion
Following T2, a lower number of patients required perioperative transfusion than after T1 (92.5% vs. 100%, p = 0.077). Also, after T2 the transfused volumes in mL and mL/kg were smaller but not to a significant level, compared to T1 (T2 177.8 ± 74.3 mL vs. T1 201.3 ± 92.5 mL, p = 0.228 and T2 23.2 ± 10.1 mL/kg vs. T1 25.8 ± 11.3 mL/kg, p = 0.279, respectively). There was no significant difference in frequency of postoperative transfusion (22.5 in T2 and 17.5% in T1, p = 0.576) or length of hospital stay (6.0 ± 1.7 days in both groups, p = 1.00).
Blood pressure
One patient was excluded from blood pressure analysis because of missing data. There was no significant difference in frequency of IOH between the groups (28.2% in T1 and 17.5% in T2, p = 0.293). The result from differenced time series and analysis of SDs showed that patients in T1 had a volatility of 6.4 ± 3.4 mmHg and patients in T2 5.8 ± 1.9 mmHg (p = 0.316). The distribution is plotted in a histogram (Figure 5).
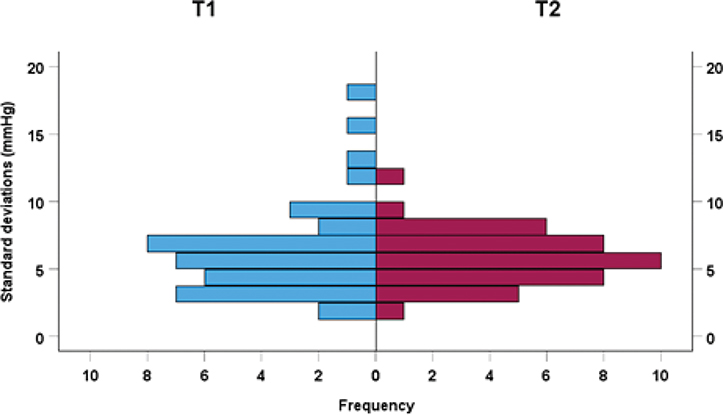
Figure 5. Histogram of distribution of standard deviations in differentiated blood pressure values. Blue T1; Red T2.
Discussion
Surgical correction of MS is associated with significant perioperative blood loss that requires blood transfusion. To address this problem, a novel modified dissection technique (T2) was developed at the craniofacial unit at Sahlgrenska University Hospital in Gothenburg, enabling electrocautery/thermal cautery of the emissary veins under the supraorbital complex before going subperiosteally in the glabellar region. This study aimed to evaluate a cohort of consecutively operated patients with the new technique (T2, n = 40) and traditional dissection technique (T1, n = 40), respectively. The main outcome was significantly lower perioperative blood loss following T2 compared to T1.
Large perioperative blood loss is a known complication to fronto-orbital remodelling in MS surgery [16]. Previous studies have shown that there is an association between MS and presence of frontal emissary veins [13], which is of importance when planning surgical intervention. It is suggested that the development of emissary veins in MS is a result of increased pressure on the venous SSS by the closed suture, predisposing development of emissary veins [15].
The underlying principle with this modified technique is to preserve the emissary veins by performing dissection the subgaleal plane in the glabellar region, allowing haemostasis in a controlled manner before raising the frontal bone flap. By clinical experience, it is during glabellar dissection that substantial blood loss from emissary veins may occur, but other mechanisms related to dissection in the subgaleal plane might favour the modified technique as well. Hallén et al. analysed blood loss in PI-Plasty surgery for sagittal synostosis and showed significantly less blood loss with strict subgaleal dissection compared to subperiosteal dissection over the craniotomy lines [17]. Findings in the present study show that the modified technique reduces perioperative blood loss and does not affect the operation time. The results in this study are supported by a previously published technical note by Di Rocco et al. [15]. In the technical note, a slightly different technique is presented with leaving a triangular bone flap in the area where emissary veins occur and performing haemostasis under the skull bone. Both techniques are based on the same fundamentals of venous morphology in the patient group.
A strength of this study is that the surgical technique for correction of MS has been identical over the study time, except for the introduction of the studied detail in frontal dissection. All surgical procedures were performed by a small number of surgeons (n = 4), working in a team. Furthermore, the cases as well as the controls were collected from an overlapping time period.
Values for estimated blood loss used in the present study are based on suction volumes and surgical gauze saturation. Perioperative blood loss is difficult to calculate accurately and should be considered as estimates and not as absolute numbers [18, 19]. However, the values for blood loss are gross estimations that reliably separates large from small blood volumes. Within T2, there were certainly fewer cases with severe perioperative blood loss (see Figure 3). Visually, there is an uneven distribution of blood loss, that is the upper tail is heavier in T1 compared to T2. A blood loss of 40 mL/kg equals about half the total blood volume for a child around the age of 6 months. There were seven patients in T1 that had a blood loss >40 mL/kg, whereas there were only two patients in T2 with blood loss that large. It seems like the modification in surgical technique reduces the risk for severe blood loss. From the clinical perspective, it is of large importance to reduce the number of cases with severe blood loss. Another possible confounding factor could be minor alterations in perioperative anaesthesiologic fluid management over the study time.
Significant blood loss results in physiological response in heart rate and blood pressure [20]. However, there is no general definition of IOH in anaesthesiology [21]. Intraoperative hypotension is often based on relative (%) or absolute (mmHg) thresholds in SBP or mean arterial pressure. According to a systematic review by Beijker et al., 140 definitions of IOH were identified in 130 articles [22]. The most widely used definition was a decrease in SBP of 20% from the baseline value as threshold of IOH [22, 23]. Intraoperative hypotension is in clinical practice used as a marker for tissue perfusion [23]. The goal of perioperative fluid replacement therapy and blood transfusion is to maintain normovolemia and replace intraoperative losses [24].
Statistical analyses of estimated blood loss in the present study were based on Mann Whitney test and described with IQR since the cohort was not normally distributed. To reliably assess the tails of the distribution, a larger sample would be needed. The analysis of intraoperative IOH and volatility in blood pressure was an attempt to triangulate the variable since it is imprecise. There are however few studies of IOH in children. As for adults, there is no general definition for thresholds. For this study, the most widely accepted definition was used.
With the intention to reduce perioperative blood loss in surgery for MS, a modified surgical technique was evaluated based on the theory of coagulating emissary veins before glabellar dissection. Traditionally, frontal dissection in MS surgery is performed in the subperiosteal plane (T1). With the modified technique (T2), frontal dissection was performed in the subgaleal plane until about 15 mm above the orbital rim and haemostasis of emissary veins was performed under the supraorbital complex. In total 80 patients were collected, n = 40 in T1 and T2, respectively. Analysis showed significantly lower perioperative blood loss volumes in absolute numbers (mL), weight related volume (mL/kg) and in percentage of total blood volume (%) in T2 compared to T1.
Conclusion
Staged frontal dissection in surgery for MS is associated with lower perioperative blood loss volumes. This is because of improved haemostatic control of the emissary veins in the glabellar region.
Financial Disclosure Statement
This study was supported by grants from the Swedish State under the agreement between the government and the county councils, the ALF-agreement (ALFGBG-716621, ALFGBG-971618).
References
[1] Karabagli P. Pathology in metopic synostosis. Childs Nerv Syst 2013; 29(12): 2165–2170. https://doi.org/10.1007/s00381-013-2284-4
[2] Speltz ML, Kapp-Simon KA, Cunningham M, et al. Single-suture craniosynostosis: a review of neurobehavioral research and theory. J Pediatr Psychol 2004; 29(8): 651–668. https://doi.org/10.1093/jpepsy/jsh068
[3] Tarnow P, Kölby L, Maltese G, et al. Incidence of non-syndromic and syndromic craniosynostosis in Sweden. J Craniofac Surg 2022; 33(5): 1517–1520. https://doi.org/10.1097/SCS.0000000000008457
[4] van der Meulen J, van der Hulst R, van Adrichem L, et al. The increase of metopic synostosis: a Pan-European observation. J Craniofac Surg 2009; 20(2): 283–286. https://doi.org/10.1097/SCS.0b013e31818436be
[5] Selber J, Reid RR, Chike-Obi CJ, et al. The changing epidemiologic spectrum of single-suture synostoses. Plastic Reconstruct Surg 2008; 122(2): 527–533. https://doi.org/10.1097/PRS.0b013e31817d548c
[6] Cornelissen M, den Ottelander B, Rizopoulos D, et al. Increase of prevalence of craniosynostosis. J Cranio-Maxillofac Surg 2016; 44(9): 1273–1279. https://doi.org/10.1016/j.jcms.2016.07.007
[7] Mathijssen IM. Guideline for care of patients with the diagnoses of craniosynostosis: Working Group on Craniosynostosis. J Craniofac Surg 2015; 26(6): 1735–1807. https://doi.org/10.1097/SCS.0000000000002016
[8] Wójcicki P, Prudel B. Trigonocephaly: long-term results after surgical correction of metopic suture synostosis. Adv Clin Exp Med 2019; 28(5): 625–35. https://doi.org/10.17219/acem/90763
[9] Gault DT, Renier D, Marchac D, et al. Intracranial pressure and intracranial volume in children with craniosynostosis. Plastic Reconstruct Surg. 1992;90(3):377–381. https://doi.org/10.1097/00006534-199209000-00003
[10] Czerwinski M, Hopper RA, Gruss J, et al. Major morbidity and mortality rates in craniofacial surgery: an analysis of 8101 major procedures. Plast Reconstr Surg 2010; 126(1): 181–6. https://doi.org/10.1097/PRS.0b013e3181da87df
[11] Lavoie J. Blood transfusion risks and alternative strategies in pediatric patients. Paediatr Anaesth 2011; 21(1): 14–24. https://doi.org/10.1111/j.1460-9592.2010.03470.x
[12] Bhuva DK, Vachhani JH. Red cell alloimmunization in repeatedly transfused patients. Asian J Transfus Sci 2017; 11(2): 115–120. https://doi.org/10.4103/0973-6247.214347
[13] Di Rocco F, Garcia-Gonzalez O, Szathmari A, et al. Emissary veins and pericerebral cerebrospinal fluid in trigonocephaly: do they define a specific subtype? Childs Nerv Syst 2021; 37(4): 1159–1165. https://doi.org/10.1007/s00381-020-04982-z
[14] Eisová S, Velemínský P, Velemínská J, et al. Diploic vein morphology in normal and craniosynostotic adult human skulls. J Morphol 2022; 283(10): 1318–1336. https://doi.org/10.1002/jmor.21505
[15] Di Rocco F, Gleizal A, Lohkamp L, et al. Control of metopic emissary veins in trigonocephaly surgery. Technical note. Child’s Nervous System 2018; 34(12): 2481–2484. https://doi.org/10.1007/s00381-018-3928-1
[16] Seruya M, Oh AK, Rogers GF, et al. Factors related to blood loss during fronto-orbital advancement. J Craniofac Surg 2012; 23(2): 358–362. https://doi.org/10.1097/SCS.0b013e31824b9c45
[17] Hallén T, Maltese G, Olsson R, et al. Cranioplasty without periosteal dissection reduces blood loss in pi-plasty surgery for sagittal synostosis. Pediatr Neurosurg 2017; 52(4): 284–287. https://doi.org/10.1159/000477444
[18] Gerdessen L, Meybohm P, Choorapoikayil S, et al. Comparison of common perioperative blood loss estimation techniques: a systematic review and meta-analysis. J Clin Monit Comput 2021; 35(2): 245–58. https://doi.org/10.1007/s10877-020-00579-8
[19] Seruya M, Oh AK, Rogers GF, et al. Blood loss estimation during fronto-orbital advancement: implications for blood transfusion practice and hospital length of stay. J Craniofac Surg 2012; 23(5): 1314–1317. https://doi.org/10.1097/SCS.0b013e31825bd02a
[20] Pacagnella RC, Souza JP, Durocher J, et al. A systematic review of the relationship between blood loss and clinical signs. PLoS One 2013; 8(3): e57594. https://doi.org/10.1371/journal.pone.0057594
[21] Saugel B, Sessler DI. Perioperative blood pressure management. Anesthesiology 2021; 134(2): 250–261. https://doi.org/10.1097/ALN.0000000000003610
[22] Bijker JB, van Klei WA, Kappen TH, et al. Incidence of intraoperative hypotension as a function of the chosen definition: literature definitions applied to a retrospective cohort using automated data collection. J Am Soc Anesthesiol 2007; 107(2): 213–220. https://doi.org/10.1097/01.anes.0000270724.40897.8e
[23] Nafiu OO, Voepel‐Lewis T, Morris M, et al. How do pediatric anesthesiologists define intraoperative hypotension? Pediatr Anesth 2009; 19(11): 1048–53. https://doi.org/10.1111/j.1460-9592.2009.03140.x
[24] Bhardwaj N. Perioperative fluid therapy and intraoperative blood loss in children. Indian J Anaesth 2019; 63(9): 729–36. https://doi.org/10.4103/ija.IJA_493_19