ORIGINAL RESEARCH ARTICLE
Anatomical study of vulnerable sensory and expendable motor nerves for targeted muscle reinnervation in the upper extremity
Kenji Kawamuraa  , Hideo Hasegawaa, Shohei Omokawab, Pasuk Mahakkanukrauhc and Yasuhito Tanakaa
, Hideo Hasegawaa, Shohei Omokawab, Pasuk Mahakkanukrauhc and Yasuhito Tanakaa
aDepartment of Orthopaedic Surgery, Nara Medical University, Nara, Japan; bDepartment of Hand Surgery, Nara Medical University, Nara, Japan; cDepartment of Anatomy, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
ABSTRACT
Purpose: Targeted muscle reinnervation (TMR) is a new technique for treating symptomatic neuroma, in which a sensory nerve after resection of a painful neuroma is coapted to an expendable motor nerve. There has been little information about optimal motor nerves for TMR of the vulnerable sensory nerves in the upper extremity.
Methods: Fourteen upper extremities of fresh-frozen cadavers were dissected to describe the anatomical course of the vulnerable sensory nerves, which included the superficial radial nerve, the dorsal branch of the ulnar nerve, and the medial and lateral antebrachial cutaneous nerves. The bifurcation, diameter, and entry points in muscles of expendable motor nerves suitable for TMR of the sensory nerves were investigated.
Results: The distal anterior interosseus nerve was available as a donor for TMR of the superficial radial nerve and the dorsal branch of the ulnar nerve in the distal third of the forearm. The motor branch to the pronator teres muscle was suitable for TMR of the medial antebrachial cutaneous nerve. It was possible to transfer the lateral antebrachial cutaneous nerve to the motor branch of the brachioradialis or extensor carpi radialis longus muscles.
Conclusions: The results of this anatomical study provide useful information when TMR is applied for neuromas of the sensory nerves in the upper extremity.
KEYWORDS: Neuroma; sensory nerve; targeted muscle reinnervation; upper extremity
Citation: Journal of Plastic Surgery and Hand Surgery 2025; 60: 35–39. DOI: https://doi.org/10.2340/jphs.v60.42655.
Copyright: © 2025 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 23 August 2024; Accepted: 3 December 2024; Published: 13 February 2025
CONTACT: Kenji Kawamura kkenji@naramed-u.ac.jp Department of Orthopaedic Surgery, Nara Medical University, 840 Shijo-cho, Kashihara, Nara 634-8522, Japan
Competing interests and funding: All authors have no potential conflicts of interest, including financial interests, activities, relationships, and affiliations, to disclose.
No funding was received for conducting this study.
Introduction
It is estimated that transected sensory nerves develop into symptomatic neuromas, which can cause chronic pain, in up to 25% of patients [1]. Neuroma formation is caused by aberrant regeneration of the proximal nerve end when it lacks a distal target and pathway [2]. Traditional surgical treatment options have variable efficacy, and include neurectomy, neurolysis, relocation of the nerve stump into deeper tissues such as muscle or bone, and capping of the nerve stump [3]. However, up to 30% of painful neuromas do not improve with traditional surgical treatment [4, 5]. Targeted muscle reinnervation (TMR) is a new technique used for treatment of symptomatic neuromas [6]. TMR was originally described as the transfer of mixed motor/sensory nerves to motor nerves for myoelectric control of a prosthesis, although it was also found to prevent neuroma formation and improve pain after major limb loss [7, 8]. Furthermore, TMR was reported to be more effective for treatment of postamputation neuroma pain compared with traditional neurectomy and muscle burying [9].
Traumatic peripheral neuroma formation is a major cause of neuropathic pain, which remains a challenging problem faced by surgeons. Neuromas of the upper extremity can cause considerable and debilitating pain, which significantly decreases patients’ quality of life [10]. The superficial radial nerve (SRN) is the most vulnerable nerve to injury from trauma or surgery in the forearm, and is a common site for painful peripheral neuromas [11]. The dorsal branch of the ulnar nerve (DBUN) is also vulnerable to injury from trauma or surgery on the ulnar side of the wrist [12, 13]. The medial antebrachial cutaneous nerve (MACN) can be damaged by numerous iatrogenic sources, including injection for medial epicondylitis, cubital nerve surgery, elbow arthroscopy, and open elbow surgery [14, 15]. Furthermore, the lateral antebrachial cutaneous nerve (LACN) is easily injured during vein puncture [16, 17]. Importantly, TMR is also applicable to neuromas following such peripheral nerve injury via transfer of a transected sensory nerve to a motor entry point (MEP) of an expendable nearby muscle, which provides a pathway and target for regeneration to reduce neuroma formation [18–20]. However, there is currently little information available on which motor nerves are anatomically suitable targets for TMR of the vulnerable sensory nerves in the upper extremity. TMR requires precise anatomical knowledge of innervation of possible motor targets, which is essential for efficient and safe nerve harvest and transfer. The aim of this anatomical study was to analyze the anatomical course of the SRN, DBUN, MACN, and LACN to identify the optimal motor nerves for TMR.
Materials and methods
After institutional ethical approval was obtained, we examined 14 upper extremities of seven fresh-frozen adult cadavers without surgical or traumatic scars in the upper extremity. All skin of the upper arm and forearm was carefully excised, while the sensory nerves were preserved on the cadaveric side. Next, dissection was performed to describe the anatomical course of the SRN, DBUN, MACN, and LACN. Measurements were made with a ruler or caliper and presented as mean ± standard deviation.
To evaluate the course of the SRN in the forearm, the distance between the radial styloid process and subcutaneous emergence between the brachioradialis (BR) and the extensor carpi radialis longus (ECRL) tendons were measured. The ulnar nerve and its branch (the DBUN) were dissected, and the distance between the branching point and the radial styloid process was measured. The distal anterior interosseus nerve (AIN) was reported as a candidate target for TMR [21, 22]. Because the motor branch to the flexor pollicis longus (FPL) muscle of the AIN must be preserved, the distance between the motor branch to the FPL and the radial styloid process was measured. The distance between the motor branch to the FPL and the MEP of the pronator quadratus (PQ) muscle was also measured. Finally, we measured the diameters of the SRN and DBUN at the radial styloid process, and the diameter of the AIN just distal to the motor branch to the FPL.
In the medial side of the elbow, the MACN was dissected, and its diameter was measured at the medial epicondyle of the humerus. The median nerve was dissected to identify the motor branch to the pronator teres (PT) muscle as a target for TMR of the MACN because the motor branch to the PT is the most proximal branch of the median nerve. Another reason is that the PT has multiple motor branches to enter the two muscle bellies, one motor branch is expendable. The motor branch closest to the medial epicondyle of the humerus was selected as a target. The distance between the medial epicondyle of the humerus and the motor branch to the PT was also measured. Finally, we measured the diameter and MEP of the motor branch to the PT.
In the lateral side of the elbow, the distance between the lateral epicondyle of the humerus and subcutaneous emergence of the LACN between the biceps brachii and the BR muscles was measured. The diameter of the LACN at the lateral epicondyle of the humerus was also measured. The radial nerve was dissected to identify the motor branches to the BR and ECRL as targets for TMR of the LACN because both BR and ECRL are expendable. When there were multiple motor branches to the BR and ECRL, the motor branch closest to the lateral epicondyle of the humerus was selected in each. The distances between the BR and ECRL motor branches and the lateral epicondyle of the humerus were measured, respectively. Finally, the diameters and MEPs of the motor branches to the BR and ECRL were measured.
Results
The findings of this cadaveric study are summarized in Table 1. The SRN emerged superficially at 86 ± 20 mm proximal to the radial styloid process (Figure 1). The DBUN branched from the ulnar nerve at 77 ± 10 mm proximal to the radial styloid process (Figure 2). The AIN branched off its motor branch to the FPL at 117 ± 16 mm proximal to the radial styloid process, and then traveled 61 ± 17 mm distally before entering to the PQ (Figure 3). The MEP of the PT was located at 56 ± 7 mm proximal to the radial styloid process. The diameters of the SRN and DBUN at the radial styloid process were 1.9 ± 0.2 mm and 1.8 ± 0.3 mm, respectively. The diameter of the AIN, just distal to the motor branch to the FPL, was 1.4 ± 0.2 mm. From the anatomical findings, the distal AIN was available as a donor for TMR of the SRN and the DBUN in the distal third of the forearm.
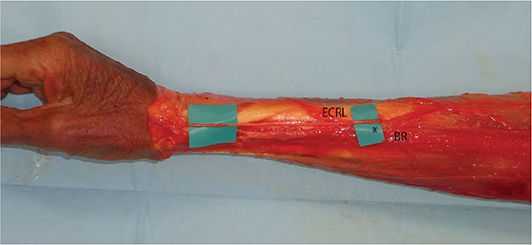
Figure 1. Anatomical course of the superficial radial nerve (SRN). The SRN (*) emerged superficially between the brachioradialis and extensor carpi radialis longus tendons at a mean distance of 86 mm proximal to the radial styloid process.
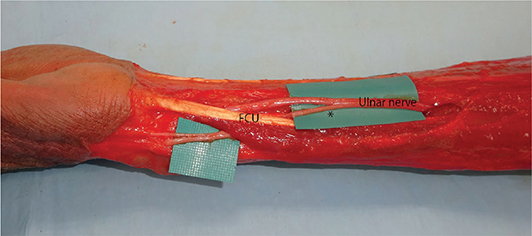
Figure 2. Anatomical course of the dorsal branch of the ulnar nerve (DBUN). The DBUN (*) branched from the ulnar nerve at a mean distance of 77 mm proximal to the radial styloid process, and traveled distally under the flexor carpi ulnaris (FCU) muscle.
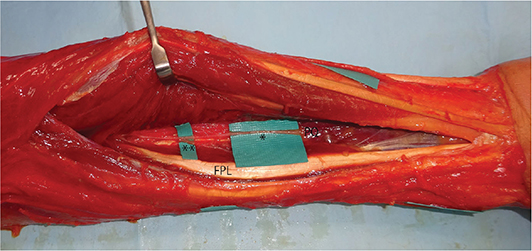
Figure 3. Anatomical course of the anterior interosseus nerve (AIN). The AIN (*) branched off its motor branch to the flexor pollicis longus (FPL) muscle (**), and then traveled distally before entering the pronator quadratus (PQ) muscle.
The MACN ran anterior to the medial epicondyle of the humerus (diameter, 1.6 ± 0.3 mm). The motor branch to the PT closest to the medial epicondyle of the humerus branched from the median nerve at 5 ± 14 mm distal to the medial epicondyle of the humerus, and entered to the PT at 22 ± 11 mm distally (Figure 4). The MEP of the PT was located at 27 ± 11 mm distal to the medial epicondyle of the humerus. The diameter of the motor branch to the PT was 1.6 ± 0.3 mm. The anatomical findings revealed that the motor branch to the PT was suitable for TMR of the MACN at a location distal to the medial epicondyle of the humerus.

Figure 4. Anatomical course of the medial antebrachial cutaneous nerve (MACN) and motor branches to the pronator teres (PT) muscle. The MACN (*) ran anterior to the medial epicondyle of the humerus. The motor branches to the PT (**) branched from the median nerve and entered the PT distally.
The LACN emerged superficially at 25 ± 9 mm proximal to the lateral epicondyle of the humerus (diameter, 2.0 ± 0.3 mm). The motor branches to the BR and ECRL closest to the lateral epicondyle of the humerus branched from the radial nerve at 33 ± 17 mm and 14 ± 9 mm proximal to the lateral epicondyle of the humerus, and entered each muscle at 20 ± 11 mm and 25 ± 8 mm distally, respectively (Figure 5). The MEPs of the BR and ECRL were located at 13 ± 20 mm proximal and 11 ± 11 mm distal to the lateral epicondyle of the humerus, respectively. The diameters of the motor branches to the BR and ECRL muscles were 1.3 ± 0.2 mm and 1.4 ± 0.2 mm, respectively. It was possible to transfer the LACN to the motor branch of the BR or ECRL.
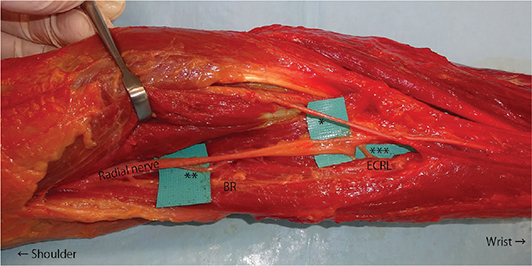
Figure 5. Anatomical course of the lateral antebrachial cutaneous nerve (LACN) and motor branches to the brachioradialis (BR) and extensor carpi radialis longus (ECRL) muscles. The LACN (*) emerged superficially at a mean distance of 25 mm proximal to the lateral epicondyle of the humerus. The motor branches to the BR (**) and ECRL (***) branched from the radial nerve and entered each muscle distally.
Discussion
There are a number of important considerations when performing TMR for the treatment of sensory nerve neuromas, which include that the sensory nerve is coapted to a nearby expendable motor nerve without tension, an expendable motor nerve is easily accessible, and that the diameter of an expendable nerve is matched to the sensory nerve diameter (Figure 6). In this anatomical study, we examined the optimal motor nerves for performing TMR of the vulnerable sensory nerves in the upper extremity. The SRN is the most easily injured cutaneous nerve in the upper extremity and has the highest frequency of painful neuroma formation [11, 23]. Neuromas of the DBUN are less common, but can occur from traumatic and iatrogenic injuries [12, 13]. The distal AIN was reported to be a suitable donor target when considering TMR for neuromas of the SRN, DBUN, and the palmar cutaneous branch of the median nerve [21, 22]. Cavadas et al. reported a case of symptom improvement after distal AIN transfer for a neuroma of the DBUN [13]. Grome et al. also reported a case of complete symptom resolution after using the distal AIN as a motor target for a neuroma of the palmar cutaneous branch of the median nerve [18]. The distal AIN is anatomically consistent and expendable in patients with a functioning PT. The results of this study indicated that it was possible to transfer the AIN a mean distance of 61 mm from the MEP of the PQ, while avoiding damage to the motor branch to the FPL. The freely transferable length of 61 mm was similar to the 56.4 mm of the previous study by Langeveld et al. [21]. As reported by Langeveld et al., this study also showed that the distal AIN had sufficient length to perform TMR for the SRN and the DBUN in the distal third of the forearm. The 1.4 mm diameter of the distal AIN was acceptable for suturing with the 1.9 mm diameter SRN and the 1.8 mm diameter DBUN.
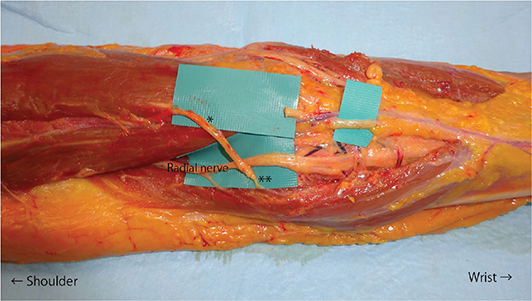
Figure 6. Targeted muscle reinnervation of the lateral antebrachial cutaneous nerve (LACN) with the motor branch to the brachioradialis (BR). The LACN (*) was transected just proximal to intersection with the cephalic vein and coapted to the motor branch to the BR (**) without tension.
The anterior and posterior branches of the MACN are located at the medial side of the elbow. Because of the marked variability in their anatomic positions, especially of the posterior branches, they are highly susceptible to iatrogenic damage during cubital tunnel surgery or other medial approaches to the elbow [24]. Mackinnon et al. showed an incidence of MACN neuromas in 73 of 100 patients who underwent revision surgery after cubital tunnel release [25]. The posterior branch of the MACN distal to the medial epicondyle of the humerus is vulnerable to injury during cubital tunnel surgery. The results of our study showed that the motor branch to the PT was a suitable donor target when considering TMR for neuromas of the MACN. The PT has two different points of origin: the humeral head and the ulnar head. The humeral head is larger and more superficial, while the ulnar head is deeper and joins at an acute angle to form one muscle belly. Each muscle head typically has a motor branch. The motor branch to the PT closest to the medial epicondyle of the humerus branched from the median nerve at a mean distance of 5 mm distal to the medial epicondyle of the humerus and entered the PT at a mean distance of 22 mm distally. The freely transferable length of 22 mm was sufficient to perform TMR for the MACN at a location distal to the medial epicondyle of the humerus. The mean diameter of the motor branch to the PT was 1.6 mm, which was well matched with the mean 1.6 mm diameter of the MACN.
The LACN is located in close proximity to the cephalic vein all along its course [17], and is thus susceptible to injury during venipuncture [16]. When considering TMR for neuromas of the LACN in the distal third of the forearm, the distal AIN is a suitable donor target. However, other donor targets are necessary for neuromas in the proximal two-thirds of the forearm and elbow. The results of this study revealed that motor branches of the BR and ECRL were suitable for TMR of the proximal LACN. Both the BR and ECRL are expendable. The motor branches of the BR and ECRL branched from the radial nerves around the elbow, which are easily accessible. The 2.0 mm diameter LACN was acceptable for suturing with the 1.3 mm diameter BR and the 1.4 mm diameter ECRL. Neuromas of the proximal SRN are less common because of protection of the nerve under the BR, but may occur following sharp trauma [5]. For neuromas of the SRN in the proximal two-thirds of the forearm, the motor branch to the BR or ERCL may be a suitable motor target [21].
A limitation of this study was the small number of cadavers. Nevertheless, our results are reliable because the investigated expendable motor nerves are anatomically consistent. Another limitation is that although TMR of vulnerable sensory nerves in the upper extremity was proved to be anatomically possible, clinical outcomes remain unknown because few clinical cases have been reported to date.
Conclusion
The results of this anatomical study provide useful information when TMR is applied for neuromas of the vulnerable sensory nerves in the upper extremity. The distal AIN was available as a donor for TMR of the SRN and DBUN in the distal third of the forearm. The motor branch to the PT was suitable for TMR of the MACN. It was possible to transfer the LACN to the motor branch of the BR or ECRL.
Acknowledgments
The authors would like to thank the donors and their families, the staff of the Excellence in Osteology Research and Training Center, the Surgical Training Center, and the Department of Anatomy and Orthopedics of Chiang Mai University for their assistance in this study.
Institutional review board statement
The study protocol involving human cadavers was approved by the Chiang Mai University Faculty of Medicine Research Ethics Committee, Chiang Mai, Thailand (code: ANA-2567–0011, 0013). The cadavers used in the study were provided by the Department of Anatomy at Chiang Mai University. Consent to use the cadavers and submit for publication was obtained from the patients before death.
Data availability statement
Data are available upon reasonable request by contacting the corresponding author.
References
[1] Ducic I, Mesbahi AN, Attinger CE, et al. The role of peripheral nerve surgery in the treatment of chronic pain associated with amputation stumps. Plast Reconstr Surg. 2008; 121: 908–914. https://doi.org/10.1097/01.prs.0000299281.57480.77
[2] Lee SK, Wolfe SW. Peripheral nerve injury and repair. J Am Acad Orthop Surg. 2000; 8: 243–252. https://doi.org/10.5435/00124635-200007000-00005
[3] Poppler LH, Parikh RP, Bichanich MJ, et al. Surgical interventions for the treatment of painful neuroma: a comparative metaanalysis. Pain. 2018; 159: 214–223. https://doi.org/10.1097/j.pain.0000000000001101
[4] Guse DM, Morannnn SL. Outcomes of the surgical treatment of peripheral neuromas of the hand and forearm: a 25-year comparative outcome study. Ann Plast Surg. 2013; 71: 654–658. https://doi.org/10.1097/sap.0b013e3182583cf9
[5] Langeveld M, Hundepool CA, Duraku LS, et al. Surgical treatment of peripheral nerve neuromas: a systematic review and meta-analysis. Plast Reconstr Surg. 2022; 150: 823e–834e. https://doi.org/10.1097/prs.0000000000009545
[6] Bishay J, Yeap I, Wang T. The effectiveness of targeted muscle reinnervation in reducing pain and improving quality of life for patients following lower limb amputation. J Plast Reconstr Aesthet Surg. 2024; 92: 288–298. https://doi.org/10.1016/j.bjps.2024.03.013
[7] Kuiken TA, Dumanian GA, Lipschutz RD, et al. The use of targeted muscle reinnervation for improved myoelectric prosthesis control in a bilateral shoulder disarticulation amputee. Prosthet Orthot Int. 2004; 28: 245–253. https://doi.org/10.3109/03093640409167756
[8] Hoyt BW, Gibson JA, Potter BK, et al. Practice patterns and pain outcomes for targeted muscle reinnervation: an informed approach to targeted muscle reinnervation use in the acute amputation setting. J Bone Joint Surg Am. 2021; 103: 681–687. https://doi.org/10.2106/jbjs.20.01005
[9] Dumanian GA, Potter BK, Mioton LM, et al. Targeted muscle reinnervation treats neuroma and phantom pain in major limb amputees: a randomized clinical trial. Ann Surg. 2019; 270: 238–246. https://doi.org/10.1097/sla.0000000000003088
[10] Dellon AL, Mackinnon SE. Radial sensory nerve entrapment in the forearm. J Hand Surg Am. 1986; 11: 199–205. https://doi.org/10.1016/s0363-5023(86)80051-x
[11] Dellon AL, Mackinnon SE. Susceptibility of the superficial sensory branch of the radial nerve to form painful neuromas. J Hand Surg Br. 1984; 9: 42–45. https://doi.org/10.1016/0266-7681(84)90012-3
[12] Tsu-Hsin Chen E, Wei JD, Huang VW. Injury of the dorsal sensory branch of the ulnar nerve as a complication of arthroscopic repair of the triangular fibrocartilage. J Hand Surg Br. 2006; 31: 530–532. https://doi.org/10.1016/j.jhsb.2006.04.026
[13] Cavadas PC, Téllez Palacios D. Treatment of neuroma of the dorsal branch of the ulnar nerve with transfer to the distal anterior interosseous nerve. J Hand Surg Glob Online. 2020; 2: 363–364. https://doi.org/10.1016/j.jhsg.2020.08.003
[14] Dellon AL, Mackinnon SE. Injury to the medial ante-brachial cutaneous nerve during cubital tunnel surgery. J Hand Surg Br. 1985; 10: 33–36. https://doi.org/10.1016/s0266-7681(85)80011-5
[15] Stahl S, Rosenberg N. Surgical treatment of painful neuroma in medial antebrachial cutaneous nerve. Ann Plast Surg. 2002; 48: 154–158. https://doi.org/10.1097/00000637-200202000-00007
[16] Yuan RT, Cohen MJ. Lateral antebrachial cutaneous nerve injury as a complication of phlebotomy. Plast Reconstr Surg. 1985; 76: 299–300. https://doi.org/10.1097/00006534-198508000-00024
[17] Mikuni Y, Chiba S, Tonosaki Y. Topographical anatomy of superficial veins, cutaneous nerves, and arteries at venipuncture sites in the cubital fossa. Anat Sci Int. 2013; 88: 46–57. https://doi.org/10.1007/s12565-012-0160-z
[18] Grome LJ, Agrawal NA, Wang E, et al. Targeted muscle reinnervation for symptomatic neuromas utilizing the terminal anterior interosseous nerve. Plast Reconstr Surg Glob Open. 2020; 8: e2979. https://doi.org/10.1097/gox.0000000000002979
[19] Daugherty THF, Bueno RA Jr, Neumeister MW. Novel use of targeted muscle reinnervation in the hand for treatment of recurrent symptomatic neuromas following digit amputations. Plast Reconstr Surg Glob Open. 2019; 7: e2376. https://doi.org/10.1097/gox.0000000000002376
[20] Wimalawansa SM, Lygrisse D, Anderson SR, et al. Targeted muscle reinnervation in partial hand amputations. Plast Reconstr Surg Glob Open. 2021; 9: e3542. https://doi.org/10.1097/gox.0000000000003542
[21] Langeveld M, Bruin LL, Hundepool CA, et al. Anatomy of the superficial radial nerve and its target nerves for targeted muscle reinnervation: an anatomical cadaver study. Plast Reconstr Surg. 2024; 153: 95e–100e. https://doi.org/10.1097/prs.0000000000010690
[22] Muneer M. TMR for peripheral sensory nerve neuroma around the wrist utilizing the distal anterior interosseous nerve. Plast Reconstr Surg Glob Open. 2024; 12: e5531. https://doi.org/10.1097/gox.0000000000005531
[23] Chang BL, Harbour P, Mondshine J, et al. Targeted muscle reinnervation to expendable motor nerves for the treatment of refractory symptomatic neuromas in nonamputees. Plast Reconstr Surg Glob Open. 2021; 9: e3436. https://doi.org/10.1097/gox.0000000000003436
[24] Manoukov Y, Herisson O, Sali E, et al. Anatomy of the posterior branch of the medial antebrachial cutaneous nerve: a cadaveric study. Orthop Traumatol Surg Res. 2020; 106: 771–774. https://doi.org/10.1016/j.otsr.2020.02.006
[25] Mackinnon SE, Novak CB. Operative findings in reoperation of patients with cubital tunnel syndrome. Hand (N Y). 2007; 2: 137–143. https://doi.org/10.1007/s11552-007-9037-3