ORIGINAL RESEARCH ARTICLE
Minor modification of soft palate surgical technique resulted in a considerable increase of residual cleft dimensions for unilateral cleft lip and palate
Midia Najar Chaliena, d  , Hans Markb
, Hans Markb  , Josef Rizellc
, Josef Rizellc  and Sara Rizella, d
and Sara Rizella, d 
aDepartment of Orthodontics, Institute of Odontology, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden; bDepartment of Plastic surgery, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden; cDepartment of Physics, Chalmers University of Technology, Gothenburg, Sweden; dClinic of Ortodontics, Gothenburg, Public Dental Service, Region Västra Götaland, Sweden
ABSTRACT
Introduction: A two-stage surgical approach, starting with the soft palate, has been practiced in the Gothenburg cleft team for unilateral cleft lip and palate (UCLP) since decades. To improve the velopharyngeal function, a modified soft palate closure (SPC) technique was introduced. Clinical observations did however indicate that the novel technique resulted in a deviant residual cleft shape, potentially contributing to future compromised dental arch form.
Aim: This study aimed to compare residual cleft dimensions (area, width and length) between different SPC techniques and at various age, in individuals born with UCLP.
Material and methods: The sample consisted of three groups of children with non-syndromic UCLP:
27 individuals who had original SPC and hard palate closure (HPC) at 3 years (GBG3)
37 individuals who had a modified SPC and HPC at 2 years (GBGmod)
29 individuals who had original SPC and HPC at 2 years (GBG2).
Residual cleft area, length and width were measured on digitised cast models using Blender software. Multiple linear regression analysis was used to compare the groups.
Results: Larger residual cleft dimensions (area and length) were found for GBGmod compared to both GBG3 (p < 0.001, p = 0.009) and GBG2 (p = 0.009, p = 0.009). In addition, GBGmod had increased cleft width in comparison with GBG3 and GBG2 in quartile 1.
Conclusion: The modified SPC technique resulted in larger residual clefts in comparison with the groups operated with the original SPC method. Timing differences between GBG3 and GBG2 are likely one explanation of cleft width variations anteriorly.
KEYWORDS: cleft lip and palate; soft palate surgery; hard palate surgery; residual cleft; residual cleft dimension
Citation: Journal of Plastic Surgery and Hand Surgery 2026; 61: 6–11. DOI: https://doi.org/10.2340/jphs.v61.45063.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 12 June 2025; Accepted: 4 November 2025; Published: 4 February 2026
CONTACT: Midia Najar Chalien midia.najar.chalien@vgregion.se Department of Orthodontics, Institute of Odontology, Sahlgrenska Academy, University of Gothenburg, Medicinaregatan 12E, 413 90, Gothenburg, Sweden
Introduction
A two-stage surgical procedure was introduced by the Gothenburg cleft team in 1975 and has been practised ever since [1, 2]. Initially, the hard palate was closed at 8 years, which has demonstrated favourable long-term maxillary growth, nevertheless with retracted oral articulation. Consequently, the age for hard palate closure (HPC) was reduced to 3 years, to decrease the speech problems [1, 3]. Long-term evaluation has shown a corresponding favourable craniofacial growth between the 3 and 8 years HPC protocols and in addition decreased retracted oral articulation for the 3 years HPC [4]. To further improve the speech, an additional decrease of timing for the HPC until 2 years of age, together with a small modification of the Gothenburg original method for soft palate closure (SPC), was introduced in 2005. This modification was implemented to optimise the direction of the soft palate and to favour the velopharyngeal function (Figure 1). However, clinical observations indicated an altered residual cleft shape, requiring more invasive hard palate surgery and later also a compromised dental arch relation. This method was therefore abandoned in 2014, and the original SPC technique was reassumed together with HPC at 2 years of age. The consequence of the modified method requires however to be analysed and evaluated. To our knowledge, no study has related the technique for SPC to the size and shape of the residual cleft and its possible effect on the craniofacial growth. Present literature focuses mainly on infant cleft severity related to the effect on maxillofacial growth and shows contradictory results [5–7].
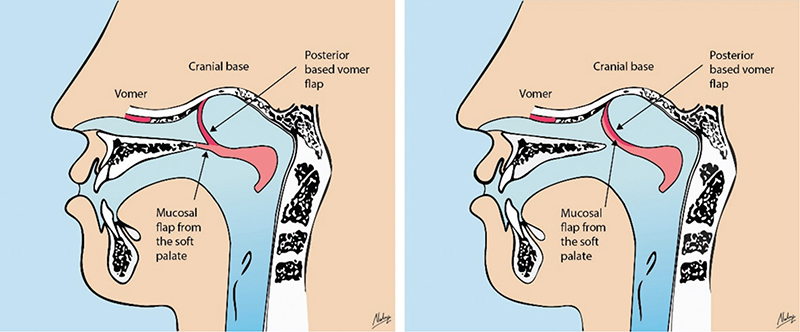
Figure 1. (a) Sagittal view of the original soft palate closure (SPC) method in a two-stage palatal technique, with anterior flaps sutured to the posterior margin of the hard palate. (b) Sagittal view of the modified SPC, with the anterior flaps sutured to the base of the posterior-based vomerine flap. Illustration from thesis of Johnna Sahlsten Schölin.
The aim of this study was therefore to evaluate and compare residual cleft dimensions for different SPC techniques and at various age points, in individuals born with unilateral cleft lip and palate (UCLP).
The null hypothesis was that (1) there is no difference in residual cleft dimension between the different techniques for SPC (2) nor has the age for assessment, any influence on the residual cleft dimension.
Materials and methods
The inclusion criteria comprised non-syndromic individuals born with UCLP, in which a soft-tissue bridge (Simonart’s band) of less than 5 mm was permitted, and who had exclusively undergone surgical interventions according to the Gothenburg CLP treatment protocol. Patients with missing or poor quality of study cast were excluded.
This study included three groups that followed different protocols for palatal surgery.
The first group, born 1993–1996, had lip-adhesion at 3–4 months of age, SPC with original Gothenburg method (Figure 1a) at 6 months of age together with lip plasty and HPC at 3 years of age [8]. This group, referred to as GBG3, comprised 27 individuals (18 males and 9 females), and four of these displayed a soft-tissue bridge of less than 5 mm.
The second group, born 2004–2012, had modified Gothenburg SPC (Figure 1b) at 6 months together with lip plasty and HPC at 2 years of age. This group, referred to as GBGmod, comprised 37 individuals (25 males and 13 females), of whom seven displayed a soft-tissue bridge < 5 mm.
The third group, born 2013–2020, had the original Gothenburg SPC method (Figure 1a) at 6 months together with lip plasty and HPC at 2 years of age. This group, referred to as GBG2, comprised 29 individuals (13 males and 18 females), of whom eight displayed a soft-tissue bridge < 5 mm.
The same soft palate surgical approach was accordingly utilised for both GBG3 and GBG2, whilst GBGmod had a modified surgical technique for the soft palate, where the oral layer of mucoperiosteal flaps was sutured down to the skull base instead of the bony part of the posterior border of the hard palate (Figure 1) [9, 10]. Study casts were obtained at two occasions, immediately before the soft palate surgery (T0) and at the time for the hard palate surgery (T1), which differed between the three groups (Table 1).
Due to missing or bad quality of study casts, five patients were excluded, one from GBG3, two from GBGmod and two individuals from GBG2.
The width of the infant cleft (baseline width) was measured with a digital calliper along the inter-tuberosity line on study casts obtained before SPC (T0). The cast models obtained before HPC (T1) were digitised using a plaster model scanner (OrthoX Scan) with the base of the cast, resting at the model holder and fitted on the magnet retention. The STL files were imported and analysed in the software program Blender 3.6 LTS, to assess area, width and length of the residual cleft. The three-dimensional model of the maxilla was aligned by rotating the model to position the cusp tips of one primary tooth in each lateral segment and one primary central to fit the same horizontal plane (Figure 2a). Viewing the cleft along the axis orthogonal to the horizontal plane, the outline of the cleft was sketched. The contour of the cleft was delimited anteriorly by the outline of the most anterior part of the clinically visible cleft and the inter-tuberosity line posteriorly, and the area was calculated by the software program (Figure 2b). The nonvisible cleft was considered to be functionally closed and was therefore not included in the measurable cleft. The length and the width of the cleft were quantified using a Matlab script (Figure 2c), where the length of the cleft was defined by its extent in the sagittal direction. The width was assessed dividing the cleft into four quartiles (Q1–Q4) perpendicular to the sagittal line, and the maximum width of the cleft in each of these quartiles was calculated. Clinical photographs taken at the same time as the study cast (T1) were examined to identify residual clefts occurring bilaterally of vomer (Figure 3). The assessment was repeated after 3 weeks, for 30 cases to calculate the intra-rater reliability.
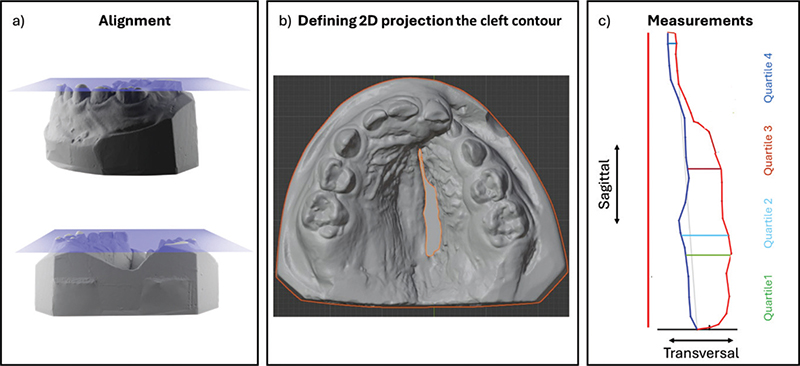
Figure 2. Cleft measurements. (a) Alignment by rotating the digitised model to position the cusp tips of one primary tooth in each lateral segment and one primary central to fit the same horizontal plane (blue). (b) The contour of the cleft, drawn with a viewing direction orthogonal to the plane. (c) The contour of the cleft with the measurements of total length (red) and maximum width in the four quartiles (Q1–Q4).
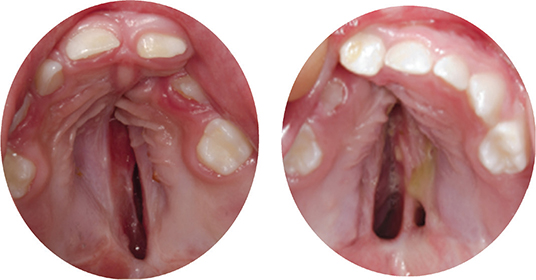
Figure 3. Examples of residual clefts prior to hard palate closure, occurring (a) unilateral of vomer and (b) bilateral of vomer posteriorly.
Ethics
This study was conducted in adherence to the Helisinki Declarations, and ethical approval was approved from the Ethical Committee in Gothenburg (Dnr 1020-12 and 2024-02525-02).
Statistics
The statistical analyses were conducted using SPSS Version 28 (IBM, Inc., NY, USA). All statistical tests were two-sided, and differences were considered statistically significant at p < 0.05. The intraclass correlation coefficient (ICC) was used to calculate the intra-rater reliability for the measurements.
The ICC and 95% confidence interval were based on a single measurement, two-way mixed-effects model with absolute agreement. A one-way analysis of variance (ANOVA) was conducted to examine the baseline width differences amongst the three groups. Multiple linear regression analyses were used to estimate whether the choice of the soft palatal surgical technique or age for assessment had an impact on the residual cleft regarding area, width and length. The analysis was adjusted for differences in baseline width (T0), to compensate for presumed effect from possible initial variations, on residual cleft dimensions.
Results
The mean ages for SPC and HPC are presented in Table 1. The intrarater reliability was 0.991 with a 95% confidence interval ranging between 0.981 and 0.996 (Table 2).
| Intraclass Correlationb | 95% Confidence Interval | F Test with True Value 0 | |||||
| Lower Bound | Upper Bound | Value | df1 | df2 | Sig | ||
| Single Measures | 0.991a | 0.981 | 0.996 | 220.901 | 29 | 29 | 0.000 |
| Two-way mixed-effects model where people effects are random, and measures effects are fixed. aThe estimator is the same, whether the interaction effect is present or not. bType A intraclass correlation coefficients using an absolute agreement definition. |
|||||||
Baseline
The mean initial cleft width at its posterior border varied between the three groups, revealing the smallest baseline width in the GBG3 group compared with the two other groups but with only statistically significant difference compared with GBG2 (p < 0.001), which was the broadest amongst the groups (Table 3, Figure 4).
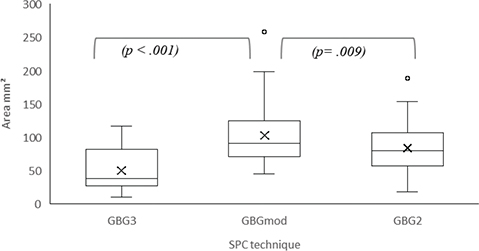
Figure 4. Box plots comparing the area in mm2 in the three groups (GBG3, GBGmod and GBG2). The boxes represent the IQR, with the horizontal line indicating the median and ‘x’ marking the mean value. Whiskers extend to the minimum and maximum values. Individual points represent outliers. Significant differences are indicated by the brackets and p-values, which were considered statistically significant at p < 0.05.
Area
The mean residual cleft area in GBGmod was statistically significantly larger than in GBG3 and GBG2 (Table 3, Figure 4).
Width
The mean residual cleft width at Q1 in GBGmod was statistically significantly wider than in GBG3 and GBG2. At Q2-Q4, the residual cleft width in GBGmod and GBG2 was statistically significantly wider than in GBG3 (Table 3, Figure 5).
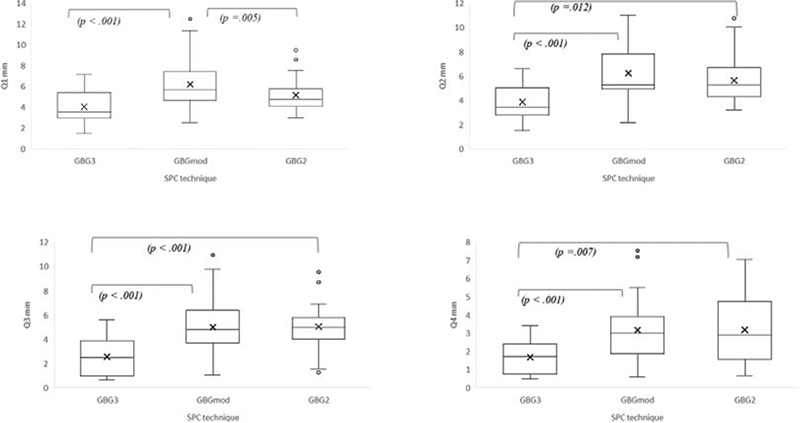
Figure 5. Box plots comparing the residual cleft width in the three groups (GBG3, GBGmod and GBG2) for four different quartiles (Q1, Q2, Q3 and Q4 in mm). The boxes represent the IQR, with the horizontal line indicating the median and ‘x’ marking the mean value. Whiskers extend to the minimum and maximum values. Individual points represent outliers. Significant differences are indicated by the brackets and p-values, which were considered statistically significant at p < 0.05.
Length
The mean residual cleft length in GBGmod was statistically significantly longer than in GBG3 and GBG2 (Table 3, Figure 6).
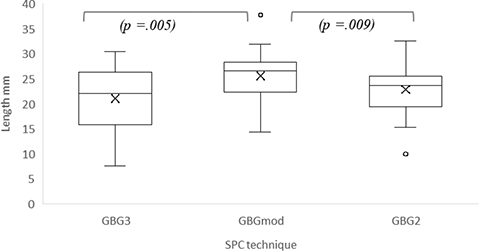
Figure 6. Box plots comparing the residual cleft length in the three groups (GBG3, GBGmod and GBG2). The boxes represent the IQR, with the horizontal line indicating the median and ‘x’ marking the mean value. Whiskers extend to the minimum and maximum values. Individual points represent outliers. Significant differences are indicated by the brackets and p-values, which were considered statistically significant at p < 0.05.
Assessment of clinical photographs
The assessments of the clinical photographs identified eight cases out of 37 solely in GBGmod, with residual clefts occurring posteriorly, bilaterally of vomer.
Discussion
Main finding
The main findings from this study were that GBGmod displayed larger residual cleft dimensions for area, length and width, in comparison with GBG3 (Table 3, Figures 4–6). In addition, GBGmod was found to have an increased residual cleft area, length as well as width in quartile 1 in comparison with GBG2, whilst no differences were detected for residual cleft width in the more anterior quartiles (Table 3, Figures 4–6). The residual cleft area was more than doubled for GBGmod and 4.5 mm longer in comparison with GBG3, whilst the differences versus GBG2 were smaller, and the length discrepancy is only 2.9 mm (Table 3). The residual cleft area, length and width in quartile 1 equalled for the two groups with the same SPC, that is, GBG3 and GBG2 (Table 3, Figures 4–6). However, in the quartiles 2–4, the residual cleft was wider for GBG2 relatively to GBG3 (Table 3, Figure 5).
Other studies on residual cleft dimensions are limited [11, 12]. However, comparisons with our results are unfeasible due to different aims as well as methodology, and to our knowledge, no study has related the residual cleft dimensions with different SPC methods.
The increased cleft dimensions revealed in the GBGmod group in comparison with GBG3 are interpreted by the authors as a consequence from the changes in the technique for SPC. In the modified SPC, a small part of the anterior portion of the soft palate was sutured to the skull base rather than to the posterior edge of the hard palate. This change brought mucosa towards the nasal cavity in the posterior part of the vomer, causing, thus, an increase of the residual cleft length. In addition, due to the orientation of the anterior mucoperiostal flaps towards the skull base, not only a wider residual cleft occurred, but almost also a quarter of the individuals in GBGmod exhibited a residual cleft posteriorly and bilateral of vomer, whilst no patients with the original Gothenburg SPC technique, that is, GBG3 and GBG2, demonstrated this finding.
The GBG3 and GBG2 exhibited similar results except for residual cleft width at Q2-Q4, where the GBG2 was statistically significantly wider (Table 3, Figure 5). The time difference, that is, one year, between the groups when the residual cleft was assessed is likely one probable explanation, since both groups had the original Gothenburg SPC technique. The GBG3 group followed the prevailing Gothenburg surgical protocol, wherein the hard palate was closed at the age of three, giving GBG2 1 year less for the residual cleft to narrow spontaneously, thus resulting in a wider cleft. It has been shown that the main reduction of the residual cleft width occurs within the first year after veloplasty for all cleft types [12]. This study also revealed a continuous reduction of the cleft area and length from 1.5 to 3 years of age, in the UCLP group, which supports our theory of time as one contributing factor for the differences between GBG3 and GBG2.
Study design
The cleft width was measured on infant casts obtained before SPC at around 6 months of age (baseline width) to assure that all three groups had comparable presumptions. The initial cleft width measured at the posterior border of the tuber on the infant casts revealed a 1.3 and 2.4 mm narrower mean width in the GBG3 group compared to GBGmod (ns) and GBG2 (p < 0.001), respectively. A reasonable explanation could be the more common use of the plates theoretically preventing the tongue from interfering with cleft size reduction and also due to the fact that a number of individuals in this group had a lip-adhesion as a first surgery already at 3 months of age [13]. The initial cleft width difference was however compensated for in the statistical analyses to avoid bias. Initial cleft area was discussed as an option for baseline comparison. However, a linear measurement was considered sufficient to identify differences between the groups.
A handful of papers are found presenting the volume and ratio measurements of the initial cleft [5, 11, 14]. The present investigation focuses on the residual cleft and quantifies its dimensions by calculating the area rather than the volume, based on the hypothesis that cleft area is more directly related to the technical difficulty and choice of method for hard palate repair. With regard to the impact of surgical repair on maxillary growth, the severity of the infant cleft has been proposed to be best expressed as a ratio between the palatal cleft area and the tissue available for closure [11, 15]. In this study, such ratio and volumetric measures were not applied; instead, the residual defect was characterized by its area, width, and length, reflecting that the hard palate repair technique used derive tissue from a vomer flap rather than from the surrounding palatal tissues.
Although infant cleft ratio is deemed suitable for defining cleft size, it fails to represent cleft shape. The shape of the cleft is therefore better represented when the defect is measured linearly at different anteroposterior levels [16]. The linear measurements in this study represent two-dimensional distances on the 3-dimensional object, as we aimed to characterise shape independently of the cleft’s depth.
Clinical consequences
This study was focusing on how the dimensions of the residual cleft were affected from different surgical SPC-techniques in the first of a two-stage-surgery approach and henceforth having a probable clinical impact on the choice of surgical method for HPC. The residual cleft dimensions will plausibly affect the method of choice for hard palate repair since it is considered unfeasibly to perform a less invasive one-layer ‘tuck-in vomer-flap’ in an extensive residual cleft [9]. Preliminary results from the CLP centre in Gothenburg has indicated an increased need for one or two mucoperiosteal flaps to cover the larger residual cleft after the modified SPC, in contrast to the original SPC technique, which required the one-layer “tuck-in vomer flap”. Additionally, the occurrence of the residual clefts bilaterally of vomer prevented the choice of the less traumatic HPC method. The modified SPC technique was thus discarded in 2014, as a consequence of early signs concerning adverse maxillary growth such as more severe lateral crossbites and speech disorders. To investigate whether the surgical modification might generate complications as fistulas or impaired speech is essential, and therefore, a study with this objective is initiated in our CLP centre.
Strength and limitations
With a retrospective design, the risk for selection bias increases. A prospective approach with randomised allocation to the different surgical protocols would have been beneficial to minimise the possible bias with effect from different surgeons and different cleft severity at T0. However, such a design in the field of CLP is demanding and scarcely feasible due to the rarity of the malformation [17]. Nevertheless, the surgical protocol as well as the collection of cast models has been controlled by a strict care program, since decades, why consecutive series of patients could be included. The sample consisted of 98 consecutive patients born between 1993 and 1996 and between 2004 and 2019. No patients born between 1997 and 2004 were included, as the Gothenburg cleft team recruited patients to the multicentre study Scandcleft during these years. Since this a retrospective study, a power analysis was not performed. Due to the retrospective approach and scarce prevalence of UCLP [17], inclusion of additional participants was not realistic. However, a substantial sample was nonetheless possible to collect. The dropouts, due to missing or poor cast models, were few and evenly distributed amongst the three groups. The time span for the included surgical interventions covered more than three decades, and several surgeons were involved in surgical interventions. However, two main surgeons were consistent in both original and modified SPC techniques.
The main author (MNC) identified and plotted the outlines of the residual cleft after a thorough examination of the cast models and clinical photographs. In cases of insecurity, the outline of the residual cleft was determined together with an experienced cleft surgeon (HM) to assure that the contour of the cleft was defined correctly prior to measurement. The resolution of scanners used to digitise dental casts presents a fundamental limitation, with higher-resolution scanners producing more accurate digital representations. The observer in this study was however not misled by digital artefacts when the outline of the cleft was drawn. If such artefacts were indeed problematic, the random error in the investigation would have been notably higher than observed. This suggests that the digitisation process maintained sufficient accuracy for reliable cleft delineation, despite potential resolution constraints.
The intra-rater reliability was calculated by re-assessment after 3 weeks, and the ICC value was 0.991 with a 95% confidence interval ranging between 0.981 and 0.996, which indicates excellent reliability. The correlation coefficients followed the cut-off limits of Koo and Li [17], and the high F-value (220.9) also indicates strong consistency in measurements between the two occasions.
Conclusion
With the findings in this study, we can reject the null hypothesis. The modified SPC method resulted in wider and longer residual clefts with increased area in comparison with the groups operated with the original SPC method. The different age for assessment, that is, the time elapsed after SPC, is considered one probable reason for an anteriorly wider residual cleft in GBG2 compared with GBG3. The hypothesis is that the increased dimension of the residual cleft in the GBGmod group is assumed to require more invasive HPC surgery. This may in turn lead to clinical consequences such as impaired speech, maxillofacial growth and, hence, potentially increase the need for orthognathic surgery.
Additional studies are planned to evaluate the long-term consequences on maxillofacial growth and speech from the more invasive HPC surgery.
Acknowledgement
The main author wishes to express sincere gratitude to Adnan Noor Baloch, statistician, for valuable guidance and assistance in performing the statistical analyses for this study.
References
[1] Friede H, Lilja J, Lohmander A. Long-term, longitudinal follow-up of individuals with UCLP after the Gothenburg primary early veloplasty and delayed hard palate closure protocol: maxillofacial growth outcome. Cleft Palate Craniofac J. 2012;49(6):649–656. https://doi.org/10.1597/10-252
[2] Lilja J, et al. Analysis of dental arch relationships in Swedish unilateral cleft lip and palate subjects: 20-year longitudinal consecutive series treated with delayed hard palate closure. Cleft Palate Craniofac J. 2006;43(5):606–611. https://doi.org/10.1597/05-069
[3] Friede H, Lilja J, Lohmander A. Two-stage palatal surgery with early veloplasty and delayed hard palate repair: a balanced view on speech and midfacial growth outcome, in cleft lip and palate, diagnosis and management. Berkowitz S, editor. Springer; 2013. p. 413–436.
[4] Najar Chalien M, et al. Long-term outcome for two-stage palatal closure with different timings for hard palate surgery: craniofacial growth and dental arch relation. Cleft Palate Craniofac J. 2023;60(9):1140–1148. https://doi.org/10.1177/10556656221140676
[5] Botticelli S, et al. Do infant cleft dimensions have an influence on occlusal relations? A subgroup analysis within an RCT of primary surgery in patients with unilateral cleft lip and palate. Cleft Palate Craniofac J. 2020;57(3):378–388. https://doi.org/10.1177/1055665619875320
[6] Peltomäki T, et al. Associations between severity of clefting and maxillary growth in patients with unilateral cleft lip and palate treated with infant orthopedics. Cleft Palate Craniofac J. 2001;38(6):582–586. https://doi.org/10.1597/1545-1569_2001_038_0582_absoca_2.0.co_2
[7] Wiggman K, et al. The influence of the initial width of the cleft in patients with unilateral cleft lip and palate related to final treatment outcome in the maxilla at 17 years of age. Eur J Orthod. 2013;35(3):335–340. https://doi.org/10.1093/ejo/cjr144
[8] Mark, H., J. Lilja, and C. Havstam, Long-term longitudinal follow-up of individuals with UCLP after Gothenburg twostage palate closure: surgical and speech outcomes. J Plast Surg Hand Surg, 2023. 58: p. 19-25.
[9] Mark, H. and J. Lilja, Staged Palate Repair – Soft Palate First, in Interdisciplinary Cleft Care, H. U, et al., Editors. 2022/2023, Plural Publishing: San Diego. p. 327-334.
[10] Sahlsten Schölin, J., H. Mark, and S. Rizell, Craniofacial Growth in Children With Unilateral Cleft Lip and Palate: Adopted From China Versus Born in Sweden. J Craniofac Surg, 2025. 36(3): p. 882-886.
[11] Berkowitz S, et al. Timing of cleft palate closure should be based on the ratio of the area of the cleft to that of the palatal segments and not on age alone. Plast Reconstr Surg. 2005; 115(6):1483–1499. https://doi.org/10.1097/01.PRS.0000161673.31770.23
[12] Owman-Moll P, Katsaros C, Friede H. Development of the residual cleft in the hard palate after velar repair in a 2-stage palatal repair regimen. J Orofac Orthop. 1998;59(5):286–300. https://doi.org/10.1007/BF01321795
[13] de Jong JP, Breugem CC. Early hard palate closure using a vomer flap in unilateral cleft lip and palate: effects on cleft width. Clin Oral Investig. 2014;18(4):1285–1290. https://doi.org/10.1007/s00784-013-1091-3
[14] Botticelli S, et al. Palatal morphology in unilateral cleft lip and palate patients: sssociation with infant cleft dimensions and timing of hard palate repair. Orthod Craniofac Res. 2019;22(4):270–280. https://doi.org/10.1111/ocr.12318
[15] Johnson N, et al. Initial cleft size does not correlate with outcome in unilateral cleft lip and palate. Eur J Orthod. 2000;22(1): 93–100. https://doi.org/10.1093/ejo/22.1.93
[16] Botticelli S, et al. Novel 3-D analysis for the assessment of cleft dimensions on digital models of infants with unilateral cleft lip and palate. Cleft Palate Craniofac J. 2019;56(1):127–133. https://doi.org/10.1177/1055665618770795
[17] Cornefjord, M., K. Källén, K. Klintö, et al., Birth prevalence of cleft lip and/or palate - a register study of all children born in Sweden years 2000-2020. J Plast Surg Hand Surg, 2025. 60: p. 120-126. https://doi.org/10.2340/jphs.v60.43739
[18] Koo TK, Li MY. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med. 2016;15(2):155–163. https://doi.org/10.1016/j.jcm.2016.02.012