ORIGINAL RESEARCH ARTICLE
Advances in epidermolysis bullosa hand deformity management: a 20-year journey of techniques and outcomes
Francesco Ciancioa, Antonio Cuzzocreaa, Caterina Albertia, Dario Melitaa, Paolo Marchicaa, Rosario Rannoa and Giuseppe A.G. Lombardoa,b
aBurn & Plastic, Reconstructive and Aesthetic Surgery, Azienda Ospedaliera Cannizzaro, Catania, Italy; bUniversità Unikore di Enna, Piazza dell’Università, Enna, Italy
ABSTRACT
Introduction: Epidermolysis bullosa (EB) is a rare disease involving the skin with the formation of synechiae and cicatricial retractions, especially on the hands.
The objective of this study is the evaluation of the surgical technique of debridement of the amorphous hand in patients suffering from EB.
Material and methods: All patients were treated according to the ethical principles of the Helsinki declaration. Data collection was performed in terms of patient’s hospitalization time, healing time (healing for more than 90% of the epidermis), possible recurrence and reoperation times, Visual Analogue Scale (VAS) for pain evaluation, and Disability of Arm-Shoulder-Hand (DASH) score in preoperative time and after 60 days from surgery. The clinical follow-up ranges from a minimum of 36 months up to 10 years.
Results: Complete reepithelialization of the hands (<90% of the bloody surface) occurred in 19.2 days, with a range of 14–23 days. In our study, the improvement in the DASH score and pain evaluation with VAS scale were statistically significant (P < 0.05), and the goal of this treatment was the functional recovery of the hands with resumption of daily autonomy.
Conclusion: The surgical technique shown is safe, simple, and repeatable with a rapid learning curve, and no complex instruments or large operating spaces are needed, which is why it should be easy to make it repeatable in other plastic or hand surgery centers. Our data regarding discomfort and the DASH scale give us encouraging premises to continue in this direction.
KEYWORDS: Epidermolysis; bullosa; blisters; pseudosyndactyly
Citation: Journal of Plastic Surgery and Hand Surgery 2026; 61: 1–5. DOI: https://doi.org/10.2340/jphs.v61.45064.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 20 June 2025; Accepted: 4 November 2025; Published: 26 January 2026
CONTACT: Francesco Ciancio francescociancio01@gmail.com via umbria n 9 Paternò (Ct), 95047 Italy
Competing interests and funding: None of the authors has a financial interest in any of the products, devices, or drugs mentioned in this article.
Introduction
Epidermolysis bullosa (EB) is a group of rare and currently incurable congenital genetic conditions that result in painful blistering of the skin and mucous membranes, which occur with minor trauma or friction.
EB is due to mutation in a number of genes, some types are autosomal dominant, while others are autosomal recessive. The underlying mechanism of this disease is a defect in the attachment between or within the epidermis and dermis of the skin. A broad phenotypic spectrum has been described, with potentially severe extracutaneous manifestations, morbidity, and mortality. Over 30 subtypes are recognized, grouped into four main categories, based on the plane of ultrastructural fragility and blister formation, reflecting which protein abnormality is present. The management and prognosis of each can vary significantly depending on the type, and they are EB simplex, dystrophic epidermolysis bullosa (DEB), junctional epidermolysis bullosa (JEB), and Kindler syndrome (KS). The severity of disease varies within and between major subtypes and reflects the extent of cutaneous disease and extracutaneous complications as well as the risk of early mortality. For instance, severe junctional EB has been associated with early mortality following birth, whereas localized EB simplex does not affect longevity [1–3].
The incidence of EB is of 1.4–25.0 per million live births with a prevalence of 2.82–54.0 per million population; there are approximately 500,000 people in the world living with EB.
Most forms of EB result from recessive or dominant mutations in genes encoding structural proteins at or close to the dermal-epidermal junction (DEJ).
Clinically, in some forms of EB, such as localized epidermolysis bullosa simplex (EBS), there may only be a small number of seasonal blisters, perhaps confined to the toes. In contrast, other forms of EB may have more extensive blistering, often involving various mucosae as well as extracutaneous involvement, such as muscle, gastrointestinal tract, lung, and kidney [4].
Among the several secondary complications that are encountered in EB, hand deformities are one of the most disabling conditions that require treatment. Despite utmost protection, hands are exposed to repetitive minor trauma in daily life, and this leads to blister formation and ulceration. Repeated cycles of blisters, ulcers, and healing are thought to be the cause of dystrophic scarring, yet the exact mechanism remains unknown. The resultant deformity includes progressive digital fusion (pseudosyndactyly), flexion contractures, and adduction contracture of the thumb. Hand deformities in patients with EB have a serious impact on psychomotor and social development, leading to low self-esteem, social isolation, and an overall poor quality of life [5]. The evidence as to whether surgery improves hand function is not clear, due to the lack of large, controlled studies; however, most data indicate surgery can improve hand function in DEB with severe and moderate stages of deformity [6–13]. For cases where the adduction is more proximal, an additional incision over the carpal tunnel may be necessary to release the carpometacarpal contracture. The surgery employs both sharp and blunt dissection to carefully separate interdigitations while preserving the neurovascular bundles, which may be displaced by scar tissue. In the first web space, dissection extends down to the level of the muscle fascia while ensuring neurovascular protection. If the fascia is excessively tight, it may also be released. Longitudinal incisions in the second to fourth web spaces are made as needed. Since the wounds are left open to heal and therapy is initiated immediately, zig-zag incisions are unnecessary. Remaining digital flexion contractures are addressed with transverse palmar incisions at the affected joints. Additionally, hyperextension of the metacarpophalangeal joints may require dorsal-releasing incisions [8]. Epidermal degloving, which involves removing the entire outer layer of epidermis from the hand, has been performed prior to making releasing incisions. However, its effectiveness and long-term outcomes remain uncertain. While some opt for this approach, others prefer to retain the natural epidermis. In general, older reports are more inclined to perform epidermal degloving, whereas more recent cases describe preserving innate epidermal tissue because sparing of the epidermis limits surface area that requires healing after surgery [9].
In 1997, Campiglio et al. described a new protocol for the treatment of hand deformities in recessive DEB based on the association of various internationally developed techniques: brachial plexus anesthesia with ketamine sedation, dynamic splinting, and coverage of the wounds with allogenic keratinocytes sheets [10]. Several studies have demonstrated the safety of general, neuraxial, and regional anesthetics in patients with EB [11].
It is important to note that perioperative aspiration has been reported in EB patients with esophageal disease despite preoperative fasting. This may be due to abnormal esophageal peristalsis and stiffening of the lower esophageal sphincter due to recurrent scarring [12].
In this paper, we present our 20-year experience in the treatment of mitten hand in patients suffering from EB using a surgical technique, and we also propose a surgical treatment protocol in patients with pseudosyndactyly and EB.
Materials and methods
Population study
Forty-two EB patients were operated for varying degrees of hand deformities in the Burn Center Unit of Cannizzaro Hospital, Catania, Italy, between 2001 and 2023. All patients suffered by severe pseudosyndactylies or mitten hands, with the functional impairment. Exclusion criteria were as follows: hemoglobin < 8 g/dl and suspected infection or neoplasms in the hand. Inclusion criteria were as follows: hemoglobin > 8 g/dl and Birmingham score > 3. The group included 18 males and 24 females, with a median age of 12.4 years (range 8–37 years old). The population under study was divided according to the four canonical forms of EB: JEB, EBS, DEB, and KS. The population is homogeneous for continuous variables (Table 1).
Written informed consent was provided by all patients or to their legal tutors, and medical information and charts along with photographic data were cumulated throughout each patient’s treatment period. All patients were treated according to the ethical principles of the Helsinki declaration. Data collection was performed in terms of patient’s hospitalization time, healing time (healing for more than 90% of the epidermis), possible recurrence and reoperation times, Visual Analogue Scales (VAS) scale for pain evaluation, and Disability of Arm-Shoulder-Hand (DASH) score in preoperative time and after 60 days from surgery. The clinical follow-up ranges from a minimum of 36 months up to 10 years.
Our protocol
The management of these patients is as follows: (1) preoperative evaluation, (2) surgical treatment, and (3) follow-up.
Preoperative evaluation and preparation
Routine preoperative blood tests were performed, and a multidisciplinary team including plastic-hand surgeons, anesthesiologists, and pediatricians evaluated the patients before the surgery. Hand deformities were evaluated by hand surgeons through physical examination to determine the level of pseudosyndactyly and joint contracture. The deformities of each digit in EB patients were categorized according to Birmingham Epidermolysis Bulls Severity Score [5] for guidance during the surgical treatment. Depending on the severity of each digital deformity, the pseudosyndactyly was categorized into five groups: I milia and/or atrophic scar; II just detectable contracture or webbing; III obvious contracture or proximal webbing; IV between III and V; V mitten hand with finger all fused. Pseudosyndactyly involving less than 50% of the interdigital space and/or proximal interphalangeal (PIP) joint contracture up to 15° was considered mild (III group), and pseudosyndactyly involving more than 50% of the interdigital space and PIP joint contracture between 15° and 60° was considered moderate (IV group). Finally, pseudosyndactyly of the digit involving more than 50% of the interdigital space and PIP joint contracture more than 60° was considered severe (V group) (Figure 1).
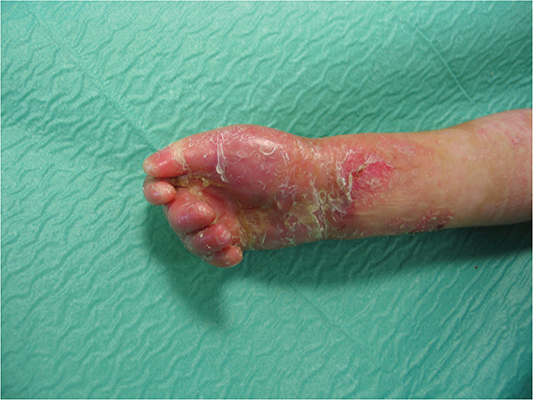
Figure 1. Pseudosyndactyly of the digit involving more than 50% of the interdigital space and PIP joint contracture more than 60° was considered severe (V group).
Surgical technique
For this type of technique, we do not use limb ischemia, and we proceed with chemical hemostasis using saline solution with adrenaline, hemostasis using bipolar forceps. All treatments were conducted under local regional anesthesia using 2% mepivacaine, with analgosedation to avoid orotracheal intubation. The technique begins with an incision with the 15 blade of the epidermis at the proximal side of the hand and then continues with degloving of the hand. The initial phase of degloving begins with the use of blunt curved scissors in order to detach the proximal portion of the palm and then proceed with manual traction to deglove all the rays (Figure 2). We then proceed with the traction of the digital rays through the use of hooks in order to allow debridement of the rays through transverse incisions at the level of the interphalangeal joints first and then the metacarpophalangeal joints, if necessary also at the palmar level. The fingers are then immobilized in extension using Kirshner wires or cannula needles (Figure 3). We have rarely employed the use of skin grafts or dermal substitutes in areas of tissue deficiency, especially at the palm level. Then, we proceed with the dressing with non-stick gauze and antibiotic ointments, which will be left in place for approximately 7 days. Postoperative medications will be performed under sedation with the removal of the K-wires on the 15th day.
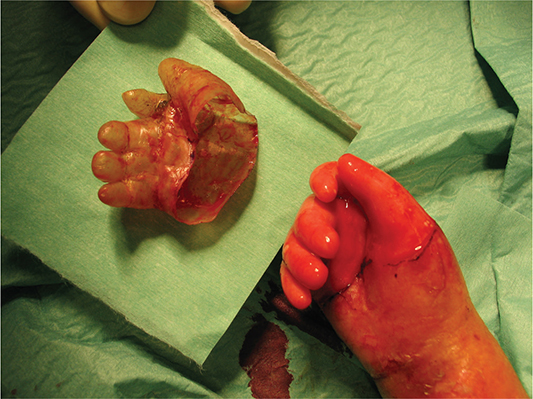
Figure 2. Degloving of the hand.
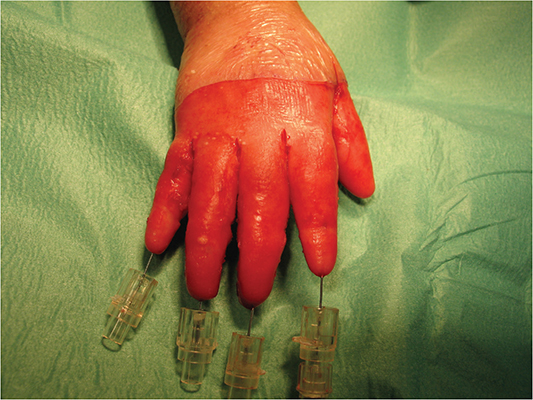
Figure 3. Immobilization of long fingers with cannula needles after surgical debridement.
Follow-up
It is essential to evaluate the DASH questionnaire, which is an outcomes’ data collection instrument that has been developed by the ‘American Academy of Orthopaedic Surgeons’, the Council of the ‘Musculoskeletal Specialty Societies’, and the ‘Institute for Work and Health’, in order to assess outcomes among patient groups with musculoskeletal disorders. Using a self-report system, patients attribute scores on 1 to 5 on 30 items relating to functional activities and symptoms; a further optional module contains four items relating to disability levels among musicians and athletes. Patients answer to the DASH score before surgery and after 60 days [14] (Figure 4).
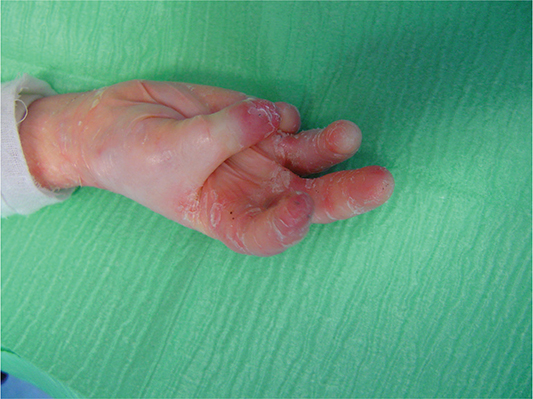
Figure 4. 60 dd post operative evaluation.
In our study, the VAS was used to assess patients’ discomfort after surgery (1 week) and after outpatients procedures (2 week) (VAS = 1 minor discomfort; VAS = 10 max discomfort). VAS scales of sensory intensity and affective magnitude are validated as ratio scale measures for both chronic and experimental pain [15].
The rate of recurrence was calculated during follow-up as a condition in which the patient showed cicatricial contractures again and returned to the previous group according to the Birmingham classification. Then, we evaluated the reoperation rate.
Data record and statistical analyses
Patients’ demographics, DASH questionnaire results, and VAS records were collected with Microsoft Excel for Mac OS, Ver.16.45 (© 2021 Microsoft). Statistical analyses were provided by SPSS® Statistics for Mac OS, Ver.25 (© IBM Corporation). Descriptive statistics were obtained. Kolmogorov-Smirnov and Shapiro-Wilks tests evaluated the distribution of the values obtained via DASH and VAS. Since DASH values presented a normal distribution and VAS values had a non-normal distribution, a two tailed paired samples t-test and the Wilcoxon signed-rank test were used to obtain inferential statistics, investigating differences between preoperative and postoperative DASH and VAS result at 1 week and 2 weeks and considering a statistically significant difference if p < 0.05 (α = 0.05 as a threshold value).
Results
The average hospitalization time was 2.2 days, with a range of 2–4 days as patients were admitted on the morning of the procedure and discharged after approximately 48 h.
Complete reepithelialization of the hands (<90% of the bloody surface) occurred in 19.2 days, with a range of 14–23 days. The average value of the DASH scale before surgery was 90.2%, while 60 days after scar debridement, we recorded an average value of 46.7%. The procedures were all conducted under regional anesthesia and analgosedation with VAS scale values for discomfort at 1 week of 3.8 and at 2 weeks of 2.8. The recurrence rate was 22.6 months in all patients with reoperation in the majority of them (Table 2).
Algorithm: based on our experience and the data extrapolated from our study, we can formulate a therapeutic algorithm. The first step is the evaluation of the patient and the classification according to the Birmimgham scale. If the patient belongs to grades I–II, we do not use surgical treatment, while it is indicated for patients in grade III–IV–V and with hemoglobin > 8 g/dl.
The patient then begins a follow-up, and if he presents a recurrence, understood as a return to a Birmimgham class of III or greater after 12 months from the previous surgery, he can undergo surgical treatment again (Figure 5).
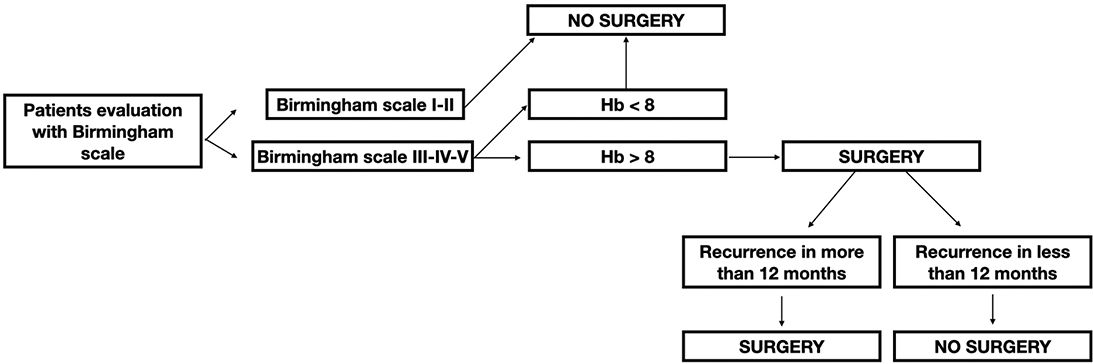
Figure 5. Decision-making algorithm in patients with epidermolysis bullosa with mitten hand.
Discussion
Optimizing nutritional status may be one excellent method of improving the morbidity and mortality associated with EB and ultimately improving the overall quality of life [16, 17].
Chronic Anemia represents one of the most important complications of EB patients; therefore, regular serum iron dosing could be particularly useful in these patients as well as blood transfusions in case hemoglobin is <8 g/dl. To date, there are no guidelines outlining best clinical practices to manage anemia in the EB population [18, 19].
In our series, patients with hemoglobin values less than 8 g/dl were excluded for surgery and referred after adequate therapy with intravenous iron.
EB is characterized not only by a difficult systemic management but also by an anesthesiologic one. It is essential to be as invasive as possible, to maintain a spontaneous breath and to use appropriate venous accesses. Our anesthetic management of these patients involved the use of local, loco-regional anesthesia and never orotracheal intubation.
The multidisciplinary approach to these patients is crucial as it allows us to quickly provide pediatric, hematological, nutritional, and anesthetic assessments with greater comfort for the patient.
From a reconstructive point of view, grafts are generally non-implied in EB patients’ surgeries. In a study of 2021, Dr. Lembo et al. proposed the following options for EB patients: they divided patients into two groups: those who underwent surgical release of the digital rays and dressing with Vaseline gauze (group A) and those who underwent surgical release of the web spaces, finger, and wrist contractions, followed by reconstruction with gloves of INTEGRA dermal regeneration template. In their protocol, Integra-gloves were used as an advanced wound dressing, avoiding the second phase of skin graft and obtaining an almost complete and spontaneous re-epithelization.
Surgical treatment of these patients represents a great challenge for the hand surgeon. The process involves degloving and separating the digits, followed by the incision of interphalangeal joints to release the contractures. Obtaining successful results and avoiding recurrences are still major problems. In the literature, surgical approaches have been described to correct hand malformations in EB [16–22].
In our study, the improvement in the DASH score was statistically significant (P < 0.05), and the goal of this treatment was the functional recovery of the hands with resumption of daily autonomy of basic functions such as washing and eating. The quality of life of these patients is very compromised due to the disease, and the decision to carry out postoperative medications under analgosedation and pain assessment with a VAS-scale allows us to state that our therapeutic protocol is well accepted in our series.
Conclusions
Our experience with EB is among the most important in terms of number of cases in the scientific literature. Few studies have such a large and long-lasting database; however, these are small numbers to be able to have absolute evidence so one of the limits is certainly represented by the small sample. Other multicenter studies should be conducted for this clinical condition as the rarity of the disease makes it difficult for a single center to have a representative sample. The surgical technique shown is safe, simple, and repeatable with a rapid learning curve, and no complex instruments or large operating spaces are needed, which is why it should be easy to make it repeatable in other plastic or hand surgery centers. Our data regarding discomfort and the DASH scale give us encouraging premises to continue in this direction.
Clinical question/Level of evidence
Case series with pre/post-test or only post-test IV Level Therapeutics.
All aspects of this study were conducted in accordance with the declaration of Helsinki and the subsequent revisions.
References
[1] Hon KL, Chu S, Leung AKC. Epidermolysis bullosa: pediatric perspectives. Curr Pediatr Rev. 2022;18(3):182–190. https://doi.org/10.2174/1573396317666210525161252
[2] Bardhan A, Bruckner-Tuderman L, Chapple ILC, et al. Epidermolysis bullosa. Nat Rev Dis Primers. 2020;6(1):78. https://doi.org/10.1038/s41572-020-0210-0
[3] Feinstein JA, Bruckner AL, Chastek B, et al. Clinical characteristics, healthcare use, and annual costs among patients with dystrophic epidermolysis bullosa. Orphanet J Rare Dis. 2022;17(1):367. https://doi.org/10.1186/s13023-022-02509-0
[4] Hou PC, Wang HT, Abhee S, et al. Investigational treatments for epidermolysis bullosa. Am J Clin Dermatol. 2021;22(6):801–817. https://doi.org/10.1007/s40257-021-00626-3
[5] Moss C, Wong A, Davies P. The Birmingham Epidermolysis Bullosa Severity Score: development and validation. Br J Dermatol. 2009;160(5):1057–1065.
[6] Box R, Bernardis C, Pleshkov A, et al. Hand surgery and hand therapy clinical practice guideline for epidermolysis bullosa. Orphanet J Rare Dis. 2022;17(1):406. https://doi.org/10.1186/s13023-022-02282-0. Erratum in: Orphanet J Rare Dis. 2022;17(1):438.
[7] Greider JL, Flatt AE. Surgical restoration of the hand in epidermolysis bullosa. Arch Dermatol. 1988;124(5):765–7.
[8] Marín-Bertolín S, Amaya Valero JV, Neira Giménez C, et al. Surgical management of hand contractures and pseudosyndactyly in dystrophic epidermolysis bullosa. Ann Plast Surg. 1999;43(5): 555–9. https://doi.org/10.1097/00000637-199911000-00017
[9] Villavisanis DF, Perrault DP, Kiani SN, et al. Current treatment landscape for dystrophic epidermolysis bullosa: from surgical management to emerging gene therapies and novel skin grafts. J Hand Surg Am. 2024;49(5):472–480. https://doi.org/10.1016/j.jhsa.2023.10.022
[10] Campiglio GL, Pajardi G, Rafanelli G. A new protocol for the treatment of hand deformities in recessive dystrophic epidermolysis bullosa (13 cases). Ann Chir Main Memb Super. 1997;16(2):91–100; discussion 101. https://doi.org/10.1016/s0753-9053(97)80025-7
[11] Mittal BM, Goodnough CL, Bushell E, et al. Anesthetic management of adults with epidermolysis bullosa. Anesth Analg. 2022;134(1):90–101. https://doi.org/10.1213/ANE.0000000000005706
[12] Griffin RP, Mayou BJ. The anaesthetic management of patients with dystrophic epidermolysis bullosa. A review of 44 patients over a 10 year period. Anaesthesia. 1993;48:810–815.
[13] Tuncer S, Sezgin B, Kaya B, et al. An algorithmic approach for the management of hand deformities in dystrophic epidermolysis bullosa. J Plast Surg Hand Surg. 2018;52(2):80–86. https://doi.org/10.1080/2000656X.2017.1338183
[14] Germann G, Wind G, Harth A. Der DASH-Fragebogen – Ein neues Instrument zur Beurteilung von Behandlungsergebnissen an der oberen Extremität [The DASH(Disability of Arm-Shoulder-Hand) Questionnaire – a new instrument for evaluating upper extremity treatment outcome]. Handchir Mikrochir Plast Chir. 1999;31(3):149–52. https://doi.org/10.1055/s-1999-13902
[15] Price DD, McGrath PA, Rafii A, et al. The validation of visual analogue scales as ratio scale measures for chronic and experimental pain. Pain. 1983;17(1):45–56. https://doi.org/10.1016/0304-3959(83)90126-4
[16] Trent JT, Kirsner RS. Epidermolysis bullosa: identification and treatment. Adv Skin Wound Care. 2003;16(6):284–90. https://doi.org/10.1097/00129334-200311000-00009
[17] Lechner-Gruskay D, Honig PJ, Pereira G, et al. Nutritional and metabolic profile of children with epidermolysis bullosa. Pediatr Dermatol. 1988;5(1):22–7. https://doi.org/10.1111/j.1525-1470.1988.tb00879.x
[18] Liy-Wong C, Tarango C, Pope E, et al. Consensus guidelines for diagnosis and management of anemia in epidermolysis bullosa. Orphanet J Rare Dis. 2023;18(1):38. https://doi.org/10.1186/s13023-022-02448-w
[19] Zhou X, Zhang Y, Zhao M, et al. Surgical management of hand deformities in patients with recessive dystrophic epidermolysis bullosa. J Plastic Surg Hand Surg. 2020;54(1):33–39. https://doi.org/10.1080/2000656X.2019.1661846
[20] Lembo F, Parisi D, Cecchino LR, et al. Release of pseudosyndactyly in recessive dystrophic epidermolysis bullosa using a dermal regeneration template glove: the Foggia experience. Orphanet J Rare Dis. 2021;16(1):52. https://doi.org/10.1186/s13023-021-01697-5
[21] Lombardo GAG, Tamburino S, Tarico MS, et al. Reverse flow shunt restricted arterialized venous free flap. J Hand Surg Am. 2018;43(5):492.e1–492.e5. https://doi.org/10.1016/j.jhsa.2018.02.023
[22] Stupka I, Dvorak Z, Vesely J, et al. Reconstruction of a nose bitten and eaten by a dog: a ten-year follow-up of a bitten-off nose replantation performed on an eleven-year-old boy. Ann Plast Surg. 2015;75(6):585–7. https://doi.org/10.1097/SAP.0000000000000643