ORIGINAL RESEARCH ARTICLE
Clinical outcomes in cutaneous squamous cell carcinoma of the head and neck: a retrospective analysis of risk factors, recurrence and metastasis
Veera Korhonen, MDa,b, Esko Veräjänkorva, MD, PhDc, Mari Koivisto, MScd and Susanna Pajula, MD, PhDb
aDepartment of Surgery, Satasairaala Hospital, Pori, Finland; bDepartment of Plastic and General Surgery, Turku University Hospital, Turku, Finland; cHospital Mehiläinen-NEO, Turku, Finland; dDepartment of Biostatistics, University of Turku and Turku University Hospital, Turku, Finland
ABSTRACT
Introduction: Cutaneous squamous cell carcinoma (cSCC) is a common non-melanoma skin cancer primarily affecting the head and neck area, with possibility of local recurrence and metastasis. Surgical excision is the standard treatment. The objective of this study was to investigate the treatment outcomes of cSCC including local recurrence, late-onset metastasis and mortality as well as to identify the associated risk factors.
Material and methods: A retrospective single-center analysis was conducted involving patients with cSCC of the head and neck treated by surgical excision at Turku University Hospital between 2000 and 2021 with a minimum 2-year follow-up from primary surgery.
Results: A total of 195 patients, with a mean age of 79.1 years, were included. During follow-up, 17 (8.7%) recurrences were diagnosed, nine (4.6%) patients developed late-onset metastasis, and 100 (51.3%) patients died. The significant risk factors for recurrences included age ≥ 80 years (p = 0.006), poor tumor differentiation (p = 0.02) and tumor invasion depth (p = 0.02). Tumor depth also increased metastasis risk (p < 0.0001). Poor tumor differentiation (p = 0.004) and metastasis (p = 0.04) were associated with higher mortality. Greater histological deep margins decreased the risk of recurrence (p = 0.01) and metastasis (p = 0.02) and improved survival (p = 0.006). All-cause 3-year and 5-year mortality rates were 27.7 and 36.4%, respectively.
Conclusion: Deeper growing, poorly differentiated cSCC tumors of the head and neck carry a higher risk of recurrence and metastasis. Pursuing greater deep excision margins could reduce these risks. Given the high age and all-cause mortality rates of cSCC patients, close surveillance should be individualized.
KEYWORDS: cutaneous squamous cell carcinoma; basosquamous carcinoma; head and neck; skin cancer; recurrence rate; local metastasis; mortality
Citation: Journal of Plastic Surgery and Hand Surgery 2026; 61: 17–23. DOI: https://doi.org/10.2340/jphs.v61.45372.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 7 September 2025; Accepted: 19 December 2025; Published: 24 February 2026
CONTACT: Veera Korhonen veeman@utu.fi Department of Plastic and General Surgery, Turku University Hospital, Kiinamyllynkatu 4-8, 20500, Turku Finland
Competing interests and funding: The authors report there are no competing interests to declare.
Introduction
Cutaneous squamous cell carcinoma (cSCC) is the second most common non-melanoma skin cancer (NMSC) after basal cell carcinoma (BCC), accounting for approximately 20% of cutaneous malignancies [1, 2]. Approximately 70–80% of cSCCs involve the head and neck area, where the most common sites are scalp, forehead, temple, cheek and ear [3– 5]. The average onset age for cSCC is around 65–70 years [6, 7]. Risk factors for cSCC include age, male gender, sun exposure, immunosuppression, and fair skin [4, 6, 8, 9]. The incidence of cSCC has been rising [4, 6, 10], with an expected annual increase of 2% [11]. The high-risk tumor features of cSCC are associated with greater risk of metastasis, recurrence and death. These features include poor histological differentiation, greater invasion thickness and tumor size, perineural invasion, and high-risk location [1, 6, 8, 11].
The standard treatment for cSCC is surgical excision with histologically confirmed tumor removal [1, 12]. For inoperable tumors radiation monotherapy is an alternative, although usually not efficient for locally advanced and metastatic tumors [1, 3]. Adjuvant radiotherapy is considered in cases of very high-risk, locally advanced or metastatic disease [3, 12]. Regional lymph node dissection (LND) is traditionally recommended for lymph node metastases [1].
Although head and neck cSCC has been studied previously, there is limited real-world data on elderly patients in the Nordic population, particularly regarding long-term outcomes. In addition, procedures such as sentinel lymph node biopsy, imaging, and adjuvant therapies may be burdensome for elderly patients, and their prognostic benefit remains unclear. Furthermore, no clear depth threshold has been established in previous studies to define when staging investigations are necessary.
This retrospective single-center study aimed to investigate and describe the clinical and histopathological features of head and neck cSCC tumors, including basosquamous cell carcinomas (BSC), which display clinical behavior similar to cSCC and are therefore managed with comparable treatment and follow-up strategies [13]. Additionally, the study aimed to identify risk factors associated with recurrence and development of metastasis, exploring their impact on prognosis.
Material and methods
The hospital’s institutional review board approved this retrospective chart review study and its protocol.
We retrospectively collected data on patients treated surgically for head and neck cSCC or BSC at Turku University Hospital, Finland between 2000 and 2021. The patient data were searched from Miranda®, an electronic database, identifying 1360 cases with skin cancerous or pre-cancerous lesions. Patients with unclear histology, or missing pathologic or surgical reports, were excluded.
Of the 305 randomly selected patients reviewed, 195 were confirmed as cSCC or BSC cases, with random sampling applied to minimize selection bias and preserve the representativeness of the study population. Some patients had more than one cSCC or BSC; we only included the first tumors for the statistical analysis. This approach was chosen to keep the analyses straightforward, to ensure independence of observations, and to avoid clustering effects caused by multiple tumors in the same patient. Data collection occurred between 2021 and 2023, ensuring a minimum surveillance time of 2 years for all patients.
Collected data included patient’s age, gender, tumor details including size and histological features, operation date, reconstruction technique, surgical and histological margins, sentinel lymph node biopsies (SLNB), regional LND, parotidectomies, adjuvant treatments, surveillance, recurrences, occurrence of late-onset metastases and survival.
Statistical analyses
The data were described using percentages and frequencies or with medians and an interquartile range (IQR) together with minimum and maximum values. Age was described with mean and standard deviation (SD). Categorical variables were compared between recurrence groups and between late-onset metastasis groups using Pearson’s χ2 test or Fisher’s exact test. Continuous variables were compared with the Mann-Whitney U test. ROC (receiver operating characteristic) curve analysis was used to obtain the optimal cutoff value for tumor thickness in recurrence risk and late-onset metastasis risk.
Patient survival between recurrence groups and between late-onset metastasis groups was explored with the log-rank test (illustrated with a Kaplan–Meier plots). Cox proportional hazard models were used in assessing the risk for recurrence and for late-onset metastasis. Independent variables in the models were categorized as age, tumor thickness, tumor differentiation, histological deep margins, histological side margins, surgical side margins and immunodeficiency. Since the recurrence and late-onset metastasis groups were small, only univariate models were conducted. A Cox proportional hazard model was used with all-cause mortality with the same independent variables, however, only univariate analyses were made.
All tests were performed as two-sided with the significance level set at 0.05. The analyses were carried out using an SAS System, version 9.4 for Windows (SAS Institute Inc., Cary, NC, US).
Results
The specific inclusion criteria resulted in a sample of 195 patients, consisting of 80 (41.0%) women and 115 (59.0%) men. The mean age was 79.1 ± 10.0 years, Table 1. The women were older than the men (81.0 vs. 77.7 years; p = 0.02).
| Total n = 195 (100%) | No recurrence n = 178 (91.3%) | Recurrence n = 17 (8.7%) | P-value | |
| Age | ||||
| Mean (SD) | 79.1 ± 10.0 | 78.8 ± 9.8 | 81.4 ± 11.5 | |
| Range | 49.0–100.0 | 49.0–100.0 | 55.0–96.0 | |
| ≥ 80 | 104 (53.3) | 93 (52.25) | 11 (64.7) | 0.33 |
| < 80 | 91 (46.7) | 85 (47.75) | 6 (35.3) | |
| Gender n (%) | ||||
| Male | 115 (59.0) | 106 (59.55) | 9 (52.9) | 0.60 |
| Female | 80 (41.0) | 72 (40.45) | 8 (47.1) | |
| Follow-up time (months) in special health care | ||||
| Median (IQR) | 4.0 (0.0–18.0) | 3.0 (0.0–18.0) | 10.0 (4.0–15.0) | 0.05 |
| Prior Bowen’s disease in site n (%) | 5 (2.6) | 4 (2.2) | 1 (5.9) | 0.37 |
| Tumor differentiation n (%) | ||||
| Good | 81 (41.5) | 78 (43.82) | 3 (17.65) | 0.05 |
| Moderate | 77 (39.5) | 67 (37.64) | 10 (58.82) | |
| Poor | 16 (8.2) | 13 (7.30) | 3 (17.65) | |
| NA | 21 (10.8) | 20 (11.24) | 1 (5.88) | |
| Perineural invasion n (%) | ||||
| Yes | 6 (3.1) | 4 (2.25) | 2 (11.76) | 0.06 |
| No | 7 (3.6) | 6 (3.37) | 1 (5.88) | |
| NA | 182 (93.3) | 168 (94.38) | 14 (82.35) | |
| Tumor diameter (mm) | n = 149 | n = 135 | n = 14 | |
| Median (IQR) | 12.0 (8.0–20.0) | 10.0 (7.0–20.0) | 25.0 (10.0–40.0) | 0.002* |
| Tumor histological characteristics | ||||
| cSCC | 181 (92.8) | 164 (92.1) | 17 (100.0) | |
| BSC | 14 (7.2) | 14 (7.9) | 0 | 0.62 |
| Invasion thickness (mm) | n = 119 | n = 111 | n = 8 | 0.02* |
| Median (IQR) | 2.7 (1.5–5.5) | 2.5 (1.5–5.0) | 5.0 (3.5–9.0) | |
| Histological tumor diameter (mm) | n = 105 | n = 94 | n = 11 | 0.05 |
| Median (IQR) | 10.5 (5.0–19.0) | 10.0 (5.0–18.0) | 16.0 (8.0–32.0) | |
| Side margins of invasive carcinoma (mm) | n = 135 | n = 128 | n = 13 | 0.43 |
| Median (IQR) | 3.0 (1.5–5.0) | 3.0 (1.9–5.0) | 3.5 (0.2–5.0) | |
| Deep margins of invasive carcinoma (mm) | n = 117 | n = 111 | n = 12 | 0.009* |
| Median (IQR) | 1.2 (0.5–2.5) | 1.5 (0.6–3.0) | 0.4 (0–1.4) | |
| Immunodeficiency n (%) | 25 (12.8) | 22 (12.4) | 3 (17.7) | 0.46 |
| Surgical side margins (mm) | n = 136 | n = 127 | n = 9 | |
| Median (IQR) | 5.0 (5.0–10.0) | 5.0 (5.0–10.0) | 10.0 (5.0–10.0) | 0.06 |
| Reconstruction method n (%) | ||||
| Direct closure | 112 (57.4) | 105 (59.0) | 7 (41.2) | 0.07 |
| Flap | 53 (27.2) | 49 (27.5) | 4 (23.5) | |
| Skin graft | 30 (15.4) | 24 (13.5) | 6 (35.3) | |
| Re-excision after primarya surgery n (%) | 38 (19.5) | 33 (18.5) | 5 (29.4) | 0.33 |
| Late-onset metastasis n (%) | 9 (4.6) | 5 (2.8) | 4 (23.5) | 0.004* |
| Deathb n (%) | 100 (51.3) | 86 (48.3) | 14 (82.4) | 0.007* |
| Death after 3 years surveillance n (%) | 54 (27.7) | 46 (25.8) | 8 (47.1) | 0.09 |
| Death after 5 years surveillance n (%) | 71 (36.4) | 62 (34.8) | 9 (52.9) | 0.14 |
| P-values indicate the correlation between recurrence and no recurrence groups. *Indicates a statistically significant finding. cSCC: Cutaneous squamous cell carcinoma; BSC: basosquamous cell carcinomas; IQR: interquartile range. aPrimary stands for the first surgical excision performed in Turku University Hospital; bDeath before data collection. |
||||
Most patients, 158 (81.0%), had one tumor, 26 (13.3%) had two tumors, and 11 (5.6%) had more than two tumors in the head and neck area. A total of 181 (92.8%) were cSCCs and 14 (7.2%) were BSCs. Figure 1 illustrates the anatomical distribution of all, recurrent and metastatic tumors.
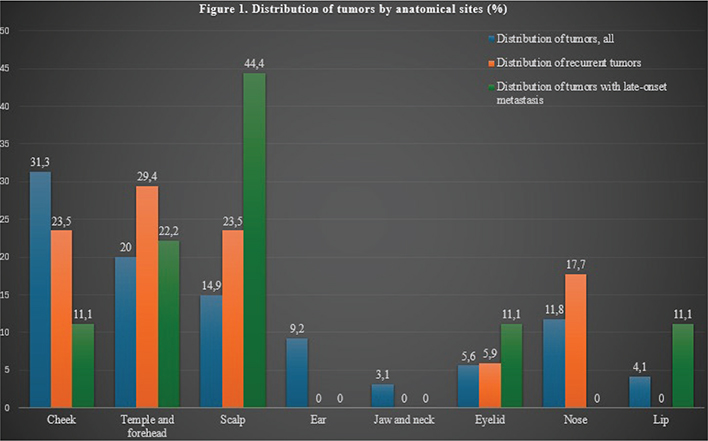
Figure 1. Distribution of tumors by anatomical sites (%): all tumors, recurrent tumors and metastatic tumors.
Treatment of cSCC
Imaging
Imaging studies were performed for 49 (25.1%) patients during the initial treatment, showing regional lymph node metastasis for three (6.1%) patients. One of these transpired to be a false positive. No distant metastases were diagnosed with imaging. The ultrasonography of the head and neck was performed for 34 (69.4%) patients. Other imaging methods included nine (18.4%) computed tomography (CT), five (10.2%) magnetic resonance imaging (MRI) and one (2.0%) positron emission tomography (PET).
Surgical details
For 30 (15.4%) patients, the surgical operation was re-excision, meaning that the first non-radical excision had been made at a local health care center or private clinic, Table 1. SLNB was performed for eight (4.1%) patients. One (0.5%) patient had a positive SLNB for metastasis and four (2.1%) patients had negative sentinels. The cervical LND was done for four (2.1%) patients. One of these patients had cervical lymph node metastasis, and this patient was previously diagnosed with metastasis by imaging. Two patients had parotid metastasis and one patient did not have a reported reason for the LND.
Three (1.5%) patients underwent partial or total removal of the parotid gland (parotidectomy) during initial treatment. Two of them had parotid metastasis and one had a sentinel lymph node in the parotid gland, which turned out to be healthy.
Histological details
There was still invasive carcinoma found in 146 (74.9%) patients in the pathology report after the primary operation in Turku University Hospital. The remaining 49 (25.1%) carcinomas were diagnosed by biopsy or excision prior to the operation at the university hospital. The histological details are reported in Table 1.
Oncological treatment
Radiation therapy was used as a postoperative treatment for two (1.0%) patients. One patient was primarily diagnosed with parotid metastasis, and one patient had very small (0.1 mm) histological deep margins and re-excision was not considered possible for anatomical reasons.
Recurrence risk of cSCC
The cSCC recurred in 17 (8.7%) patients, and altogether 14 (82.4%) of them underwent re-surgery, Table 1. Two patients were treated palliatively, and one received radiation monotherapy for the recurring tumor. Overall, four (23.5%) patients received radiation therapy for the recurrent disease. Four (2.1%) patients developed both recurrence and late-onset metastasis. The all-cause mortality rate was greater in recurrent tumors (p = 0.007).
The statistically significant risk factors for the recurrence of cSCC included age ≥ 80 years (p = 0.006), tumor thickness (p = 0.02), a poorly versus well-differentiated tumor (p = 0.006), greater surgical side margins (p = 0.002) and the occurrence of late-onset metastasis (p = 0.0002). Greater histological deep margins decreased the recurrence risk (p = 0.01), Table 2.
| Predictive factors | Recurrence | Late-onset metastasis | ||
| HR (95% CI) | P-value | HR (95% CI) | P-value | |
| Age ≥ 80 | 4.15 (1.51–11.45) | 0.006* | 3.50 (0.87–13.98) | 0.08 |
| Invasion thickness of tumor | 1.20 (1.03–1.39) | 0.02* | 1.33 (1.16–1.53) | < 0.0001* |
| Tumor differentiation | ||||
| Good vs. poor | 0.11 (0.02–0.53) | 0.006* | 0.06 (0.006–0.68) | 0.02* |
| Moderate vs. poor | 0.49 (0.14–1.79) | 0.28 | 0.50 (0.10–2.47) | 0.39 |
| Histological deep margins | 0.44 (0.23–0.84) | 0.01* | 0.23 (0.07–0.82) | 0.02* |
| Histological side margins | 0.88 (0.70–1.10) | 0.25 | 0.83 (0.62–1.12) | 0.23 |
| Surgical side margins | 1.33 (1.11–1.60) | 0.002* | 0.94 (0.68–1.30) | 0.70 |
| Immunodeficiency | 2.66 (0.76–9.29) | 0.13 | 3.19 (0.66–15.34) | 0.15 |
| Late-onset metastasis | 9.09 (2.88–28.72) | 0.0002* | - | - |
| Recurrence | - | - | 14.41 (3.84–54.09) | < 0.0001* |
| HR: Hazard ratio; CI: Confidence interval. *Indicates a statistically significant finding. |
||||
We used ROC analysis to determine a 3.0 mm cutoff value to predict recurrence risk. All-in-all, eight (47.1%) of the recurred tumors had tumor thickness values reported, and seven (87.5%) were at least 3.0 mm thick. In the survival analysis, the difference in recurrence risk between tumors ≥ 3.0 mm thick and < 3.0 mm thick was not statistically significant (HR (hazard ratio)) 7.24; 95% CI (confidence interval) [0.89–58.94], p = 0.06).
Risk for late-onset metastasis of cSCC
Late-onset metastasis was diagnosed for nine (4.6%) patients: seven (77.8%) then underwent re-surgery and two (22.2%) received radiation monotherapy instead. In total, seven (77.8%) out of the nine patients received radiation therapy for the metastases, including one who also received chemotherapy. Late-onset metastasis was associated with a greater 3-year mortality (p = 0.02), Table 3.
| No late-onset metastasis n = 186 (95.4%) | Late-onset metastasis n = 9 (4.6%) | P-value | |
| Age | |||
| Mean (SD) | 79.0 ± 10.1 | 81.4 ± 8.6 | 0.51 |
| Range | 49.0–100.0 | 64.0–93.0 | |
| ≥ 80 | 98 (52.7) | 6 (66.7) | |
| < 80 | 88 (47.3) | 3 (33.3) | |
| Gender n (%) | |||
| Male | 108 (58.1) | 7 (77.8) | 0.31 |
| Female | 78 (41.9) | 2 (22.2) | |
| Follow-up time (months) in special healthcare | |||
| Median (IQR) | 3.0 (0.0–18.0) | 10.0 (6.0–13.0) | 0.11 |
| Prior Bowen’s disease in site n (%) | 5 (2.7) | 0 | 1.00 |
| Tumor differentiation n (%) | |||
| Good | 80 (43.0) | 1 (11.1) | 0.07 |
| Moderate | 71 (38.2) | 6 (66.7) | |
| Poor | 14 (7.5) | 2 (22.2) | |
| NA | 21 (11.3) | 0 | |
| Perineural invasion n (%) | |||
| Yes | 4 (2.15) | 2 (22.2) | 0.03* |
| No | 7 (3.76) | 0 | |
| NA | 175 (94.09) | 7 (77.8) | |
| Tumor diameter (mm) | n = 141 | n = 8 | |
| Median (IQR) | 10.0 (7.0–20.0) | 30.0 (20.0–30.0) | 0.0005* |
| Tumor histological characteristics | |||
| cSCC | 172 (92.5) | 9 (100.0) | |
| BSC | 14 (7.5) | 0 | 1.00 |
| Invasion thickness (mm) | n = 111 | n = 8 | 0.0003* |
| Median (IQR) | 2.5 (1.5–5.0) | 10.0 (5.3–11.0) | |
| Histological tumor diameter (mm) | n = 98 | n = 7 | 0.007* |
| Median (IQR) | 9.5 (5.0–18.0) | 18.0 (16.5–32.0) | |
| Side margins of invasive carcinoma (mm) | n = 132 | n = 9 | 0.50 |
| Median (IQR) | 3.3 (1.5–5.0) | 3.0 (1.5–3.5) | |
| Deep margins of invasive carcinoma (mm) | n = 116 | n = 7 | 0.009* |
| Median (IQR) | 1.6 (0.5–3.0) | 0 (0–1.1) | |
| Immunodeficiency n (%) | 23 (12.4) | 2 (22.2) | 0.32 |
| Surgical side margins (mm) | n = 130 | n = 6 | |
| Median (IQR) | 5.0 (5.0–10.0) | 5.0 (5.0–7.0) | 0.48 |
| Reconstruction method n (%) | |||
| Direct closure | 110 (59.1) | 2 (22.2) | 0.05 |
| Flap | 48 (25.8) | 5 (55.6) | |
| Skin graft | 28 (15.1) | 2 (22.2) | |
| Re-excision after primarya surgery n (%) | 35 (18.8) | 3 (33.3) | 0.38 |
| Recurrence n (%) | 13 (7.0) | 4 (44.4) | 0.004* |
| Deathb n (%) | 94 (50.5) | 6 (66.7) | 0.50 |
| Death after 3 years surveillance n (%) | 48 (25.8) | 6 (66.7) | 0.02* |
| Death after 5 years surveillance n (%) | 65 (35.0) | 6 (66.7) | 0.08 |
| P-values indicate the correlation between late-onset metastasis and no late-onset metastasis groups. *Indicates a statistically significant finding. cSCC: Cutaneous squamous cell carcinoma; BSC: basosquamous cell carcinomas; IQR: interquartile range. aPrimary stands for the first surgical excision performed in Turku University Hospital; bDeath before data collection. |
|||
Statistically significant risk factors for metastasis were tumor thickness (p < 0.0001) and recurrence (p < 0.0001). The greater histological deep margins decreased metastasis risk (p = 0.02), Table 2.
The cutoff value for tumor thickness predicting metastasis risk was defined as 5.5 mm by ROC analysis. Tumors 5.5 mm thick or thicker were in significantly greater risk for developing late-onset metastasis in the survival analysis (HR 10.63; 95% CI [2.14–52.81], p = 0.004). In our study, the invasion thickness was ≥ 5.5 mm in six (66.7%) of the nine tumors, which resulted in late-onset metastases (two were < 5.5 mm thick and one was missing invasion thickness data).
Mortality and survival
Overall, 100 (51.3%) patients had died before reviewing the patient data between 2021 and 2023. All mortality details are reported in Tables 1 and 3.
The statistically significant risk factors for mortality were the development of late-onset metastasis (p = 0.04) and poor tumor differentiation (p = 0.004). Greater histological deep margins improved survival (p = 0.006), Table 4. The survival probabilities in relation to recurrence and late-onset metastasis are demonstrated in Figure 2.
| Predictive factors | HR (95% CI) | P-value |
| No recurrence | 0.60 (0.34–1.06) | 0.08 |
| No late-onset metastasis | 0.42 (0.18–0.96) | 0.04* |
| Invasion thickness of tumor | 1.04 (0.98–1.10) | 0.20 |
| Tumor differentiation | ||
| Good vs. poor | 0.38 (0.20–0.74) | 0.004* |
| Moderate vs. poor | 0.61 (0.32–1.15) | 0.12 |
| Histological deep margins | 0.99 (0.99–0.998) | 0.006* |
| Histological side margins | 1.00 (0.99–1.00) | 0.18 |
| Surgical side margins | 1.04 (1.00–1.09) | 0.08 |
| Immunodeficiency | 0.67 (0.40–1.13) | 0.13 |
| HR: Hazard ratio; CI: Confidence interval. *Indicates a statistically significant finding. |
||
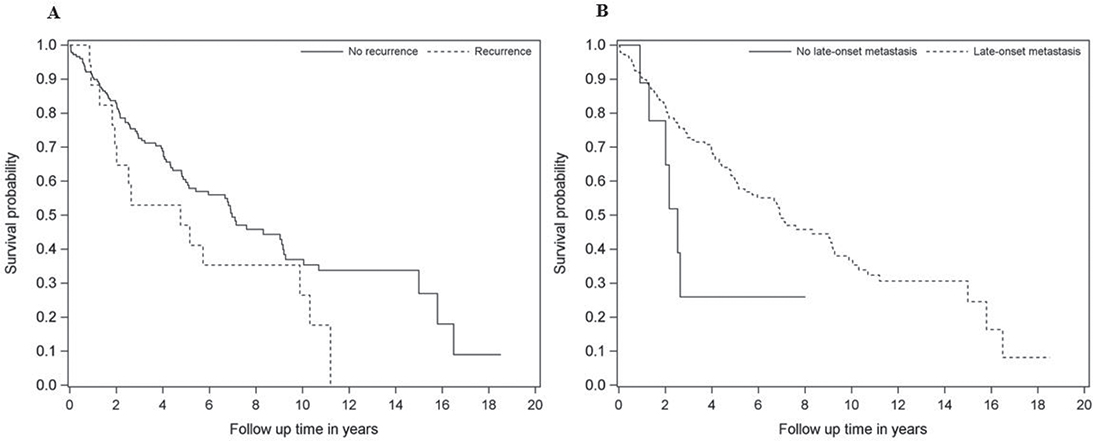
Figure 2. Survival of patients in relation to (A) recurrence and (B) late-onset metastasis.
Discussion
This single-center retrospective study analyzed 195 patients treated for cSCC and BSC of the head and neck region, focusing on recurrence and late-onset metastases. Our results indicate that these tumors are more prevalent in elderly patients, with a high average age contributing to significant all-cause mortality rates, and they occurred more frequently in males. Recurrence of cSCC was uncommon but associated with risk factors such as age 80 years and older, deeper invasion thickness and poor tumor differentiation. Metastasizing was even rarer, yet strongly linked to tumor’s greater depth. These results underscore the need for tailored follow-up and treatment strategies.
The head and neck cSCC tumors are common with an increasing incidence, and potential for recurrence, metastasis and disease-specific death [1, 3, 6, 11]. Radical surgical excision is the treatment of choice [1, 12]. In this study, all tumors were primarily treated with surgical excision, and direct closure of the wound was possible in 57.4% of the cases, indicating that smaller head and neck cSCC tumors can often be removed without the need for specialized plastic surgery expertise. However, the face consists of several anatomical units with specific characteristics and shapes. Therefore, achieving optimal functional and aesthetic outcomes with appropriate closure techniques requires experience and thorough anatomical understanding, when operating on larger tumors.
Our study mainly comprised elderly patients, with a mean age of 79.1 years, and over half were aged 80 or older. This increases the likelihood of comorbidities and reduced life expectancy, as reflected in our high mortality rates. With this high average age, the mortality rates were relatively high, as the 3-year mortality was 27.7% and the 5-year mortality was 36.4%. However, there was no data on disease-specific death rates, which have been as low as 1.5–4% in previous studies [3, 14, 15]. In the literature, the all-cause 3-year and 5-year mortality rates have been 23–38% and 34–59%, respectively [16, 17]. Patients with cSCC are reported to have a 25% increased overall risk for death compared to the general population, which may be partially explained by common risk factors such as immunosuppression and smoking [18].
This study showed a higher local recurrence rate than previous studies (8.7% vs. 3.0–5.6%) [1, 3, 8, 15]. Factors such as age 80 years or older (p = 0.006), deeper invasion thickness (p = 0.02) and poor tumor differentiation (p = 0.006) increased the risk of recurrence, which is consistent with literature [6, 8]. Greater surgical side margins paradoxically increased recurrence risk (p = 0.002), potentially due to the limited number of recurrent tumors with reported surgical margins. Moreover, larger and thicker tumors, with already higher risk for recurrence, probably required more extensive margins. Recurrent tumors were mostly (82.4%) treated surgically, and 23.5% of patients with recurrent tumors received radiation therapy.
Approximately 85% of metastatic cSCC cases spread to lymph nodes, and the remaining 15% develop distant or in-transit metastases without nodal involvement [1]. In the head and neck area, the risk of nodal metastasis is 4–5%, potentially affecting the neck or the parotid gland [2, 4]. For high-risk tumors, metastasis rates are reported as high as 30–40% [8, 11]. In our cohort, the late-onset metastasis rate was 4.6%, consistent with previous studies, 0.5–6% [1, 2, 8, 15]. Tumor depth was a clear risk factor for metastasis (p = 0.004), with the thickness of 5.5 mm and deeper. In addition, our results show that each 1 mm increase in tumor thickness increases the risk of metastasis by 1.4 times. Que et al. described that tumors of 6.0 mm thickness or thicker have a 16% risk of metastasis, while on the other hand, a tumor thickness of 2.0 mm or less is associated with minimal metastasis risk [6].
Late-onset metastasis significantly increased the risk of death (p = 0.04), with an all-cause mortality rate of 66.7%. All patients with late-onset metastasis who died during the follow-up period did so within 3 years of primary surgery. The literature describes over 70% disease-specific mortality rates and a median survival of fewer than 2 years with distant metastases [1, 6, 11, 19]. The 5-year survival rate for regionally metastatic cases is around 70–80% [11, 20]. Although our study did not separate regional and distant metastasis categories, the prognosis of metastatic cSCC was poor, with a life expectancy of less than 3 years in the majority of cases. Poor tumor differentiation further worsened the prognosis (p = 0.004), as has also been shown in previous studies [21].
Greater histological deep margins decreased both metastasis (p = 0.02) and recurrence risk (p = 0.01), and increased survival (p = 0.006). One retrospective review by Phillips et al. showed that more than 5 mm histological margins improved disease-specific survival [22], although not differentiating between side and deep margins. Another study by Brinkman et al. proved that incomplete tumor excision increased the risk of disease-specific death compared to minimum 1 mm histological excision margins [21]. In our results, side margins did not significantly impact the risk of recurrence, metastasis or death. While the literature recommends 5 mm surgical excision margins for low-risk tumors and 10 mm for high-risk cases [1], we suggest reconsidering the necessity for extensive surgical side margins in challenging head and neck areas.
In this study, imaging revealed metastatic disease in three (6.1%) out of 49 patients. According to the literature, ultrasonography is currently the best imaging method for detecting occult metastases in head and neck cSCC [23], while neck and parotid imaging is recommended for high-risk cSCC tumors at the time of initial diagnosis [3]. Some recommend nodal ultrasound in addition to clinical examination during the first 2 years of follow-ups for high-risk cases, as most metastases occur within this period [3, 8].
We recommend initial ultrasonography of regional lymph nodes for patients with high-risk cSCC tumors. If metastasis is suspected, a body CT or PET scan should be considered. Given the advanced age of cSCC patients, the decision of additional imaging should be made on a case-by-case basis, carefully weighing the burdens and risks against the potential prognostic benefits.
SLNB has become common practice in the treatment of malignant melanoma; however, its prognostic value in cSCC remains uncertain due to limited controlled studies and lack of standardized indications [1, 2, 9, 20]. In this study, SLNB was performed on eight patients with a 12.5% positive result rate. Previous studies have shown that SLNB finds lymph node metastasis in 6–21% of high-risk cSCC cases, with low morbidity and few false-negative results [3, 9, 20]. However, high-risk cSCC has the potential to metastasize despite a negative SLNB [8, 9], highlighting the importance of surveillance in high-risk cases.
Regular follow-ups are recommended for cSCC patients. Guzman et al. suggest follow-ups every 3–6 months for the first 2 years, every 6–12 months for the next 3 years, and annually for life after 5 years, depending on the tumor staging [24]. At Turku University Hospital, patients receive an individualized surveillance program after surgery, recommending follow-up at primary healthcare units. We collected surveillance data only from specialized healthcare and do not know the extent of follow-up in primary healthcare. Due to advanced age and comorbidities, intensive treatment and follow-up may be too burdensome for some patients. Especially in metastatic or recurrent cases, it remains uncertain whether surgery and oncological treatments improve life expectancy or quality of life in elderly patients. Nevertheless, low-risk tumors might not require extensive excision margins or close follow-ups. We recommend that in units where cSCC of the head and neck area is operated on, a multidisciplinary team, potentially including an oncologist, surgeon, radiologist, dermatologist and pathologist, should individually consider the patient’s follow-up plan.
The limitations of this study include its retrospective design, as our data were solely collected from medical records from a single center. The relatively small sample size and number of outcomes increase the possibility of confounding factors. In addition, only a random sample of the eligible cases was reviewed in detail, of which a subset was confirmed as cSCC or BSC. While random sampling was applied to minimize selection bias and preserve representativeness, this approach may have led to underrepresentation of some uncommon outcomes such as recurrence and metastasis. Furthermore, the follow-up period was only 2 years for the most recently operated patients.
To conclude, this study and previous research underscore the importance of intensive surveillance for high-risk cSCC tumors. The patient population is mostly elderly with limited life expectancy, making it challenging to establish a standardized adjuvant treatment and follow-up protocol for all. We recommend that surveillance for head and neck cSCC patients be individually tailored by a multidisciplinary team, considering the patient’s age, comorbidities, and life expectancy, as these follow-ups should not impose an excessive burden on elderly patients.
Acknowledgments
The authors acknowledge the Satasairaala Central Hospital District, Pori, Finland, and Tyks Säätiö, for the personal grants awarded to the response author, which enabled the writing of this article.
References
[1] Stratigos A, Garbe C, Lebbe C, et al. Diagnosis and treatment of invasive squamous cell carcinoma of the skin: European consensus-based interdisciplinary guideline. Eur J Cancer. 2015;51(14):1989–2007. https://doi.org/10.1016/j.ejca.2015.06.110
[2] Silberstein E, Sofrin E, Bogdanov-Berezovsky A, et al. Lymph node metastasis in cutaneous head and neck squamous cell carcinoma. Dermatol Surg. 2015;41(10):1126–1129. https://doi.org/10.1097/DSS.0000000000000488
[3] Yan F, Tillman BN, Nijhawan RI, et al. High-risk cutaneous squamous cell carcinoma of the head and neck: a clinical review. Ann Surg Oncol. 2021;28(13):9009–9030. https://doi.org/10.1245/s10434-021-10108-9
[4] Lubov J, Labbé M, Sioufi K, et al. Prognostic factors of head and neck cutaneous squamous cell carcinoma: a systematic review. J Otolaryngol Head Neck Surg. 2021;50(1):54. https://doi.org/10.1186/s40463-021-00529-7
[5] Harris BN, Bayoumi A, Rao S, et al. Factors associated with recurrence and regional adenopathy for head and neck cutaneous squamous cell carcinoma. Otolaryngol Head Neck Surg. 2017;156(5):863–869. https://doi.org/10.1177/0194599817697053
[6] Que SKT, Zwald FO, Schmults CD. Cutaneous squamous cell carcinoma: incidence, risk factors, diagnosis, and staging. J Am Acad Dermatol. 2018;78(2):237–247. https://doi.org/10.1016/j.jaad.2017.08.059
[7] Simonacci F, Bertozzi N, Grieco MP, et al. Surgical therapy of cutaneous squamous cell carcinoma: our experience. Acta Biomed. 2018;89(2):242–248.
[8] Krediet JT, Beyer M, Lenz K, et al. Sentinel lymph node biopsy and risk factors for predicting metastasis in cutaneous squamous cell carcinoma. Br J Dermatol. 2015;172(4):1029–1036. https://doi.org/10.1111/bjd.13508
[9] Ilmonen S, Sollamo E, Juteau S, et al. Sentinel lymph node biopsy in high-risk cutaneous squamous cell carcinoma of the head and neck. J Plast Reconstr Aesthet Surg. 2022;75(1):210–216. https://doi.org/10.1016/j.bjps.2021.08.041
[10] Lomas A, Leonardi-Bee J, Bath-Hextall F. A systematic review of worldwide incidence of nonmelanoma skin cancer. Br J Dermatol. 2012;166(5):1069–1080. https://doi.org/10.1111/j.1365-2133.2012.10830.x
[11] Burton KA, Ashack KA, Khachemoune A. Cutaneous squamous cell carcinoma: a review of high-risk and metastatic disease. Am J Clin Dermatol. 2016;17(5):491–508. https://doi.org/10.1007/s40257-016-0207-3
[12] Kim JYS, Kozlow JH, Mittal B, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78(3):560–578. https://doi.org/10.1016/j.jaad.2017.10.007
[13] Fotiadou C, Apalla Z, Lazaridou E. Basosquamous carcinoma: a commentary. Cancers (Basel). 2021;13(23):6146. https://doi.org/10.3390/cancers13236146
[14] Waldman A, Schmults C. Cutaneous squamous cell carcinoma. Hematol Oncol Clin North Am. 2019; 33(1): 1–12. https://doi.org/10.1016/j.hoc.2018.08.001
[15] Thiem DGE, Scharr K, Pabst AM, et al. Facial cutaneous squamous cell carcinoma – microscopic safety margins and their impact on developing local recurrences. J Craniomaxillofac Surg. 2020;48(1):49–55. https://doi.org/10.1016/j.jcms.2019.11.022
[16] Tuominen S, Ukkola-Vuoti L, Riihilä P, et al. Retrospective, registry-based, cohort investigation of clinical outcomes in patients with cutaneous squamous cell carcinoma and basal cell carcinoma in Finland. Acta Derm Venereol. 2022;102:adv00693. https://doi.org/10.2340/actadv.v102.2073
[17] Eigentler TK, Leiter U, Häfner H-M, et al. Survival of patients with cutaneous squamous cell carcinoma: results of a prospective cohort study. J Invest Dermatol. 2017;137(11):2309–2315. https://doi.org/10.1016/j.jid.2017.06.025
[18] Wehner MR, Serrano WC, Nosrati A, et al. All-cause mortality in patients with basal and squamous cell carcinoma: a systematic review and meta-analysis. JAAD. 2018;78(4):663–672. https://doi.org/10.1016/j.jaad.2017.11.026
[19] Thompson AK, Kelley BF, Prokop LJ, et al. Risk factors for cutaneous squamous cell carcinoma recurrence, metastasis, and disease-specific death: a systematic review and meta-analysis. JAMA Dermatol. 2016;152(4):419–428. https://doi.org/10.1001/jamadermatol.2015.4994
[20] Ross AS, Schmults CD. Sentinel lymph node biopsy in cutaneous squamous cell carcinoma: a systematic review of the English literature. Dermatol Surg. 2006;32(11):1309–1321. https://doi.org/10.1111/j.1524-4725.2006.32300.x
[21] Brinkman JN, Hajder E, van der Holt B, et al. The effect of differentiation grade of cutaneous squamous cell carcinoma on excision margins, local recurrence, metastasis, and patient survival: a retrospective follow-up study. Ann Plast Surg. 2015;75(3):323–326. https://doi.org/10.1097/SAP.0000000000000110
[22] Phillips TJ, Harris BN, Moore MG, et al. Pathological margins and advanced cutaneous squamous cell carcinoma of the head and neck. J Otolaryngol Head Neck Surg. 2019;48:55. https://doi.org/10.1186/s40463-019-0374-3
[23] Richards PS, Peacock TE. The role of ultrasound in the detection of cervical lymph node metastases in clinically N0 squamous cell carcinoma of the head and neck. Cancer Imaging. 2007;7(1):167–178. https://doi.org/10.1102/1470-7330.2007.0024
[24] Guzman AK, Schmults CD, Ruiz ES. Squamous cell carcinoma: an update in staging, management, and postoperative surveillance strategies. Dermatol Clin. 2023;41(1):1–11. https://doi.org/10.1016/j.det.2022.07.004