ORIGINAL RESEARCH ARTICLE
Evaluation of modified abdominoplasty for excess skin in post-bariatric surgery patients with residual obesity
Jonas Ockell, PhD, MDa  , Christina Biörserud, PhD, RNb
, Christina Biörserud, PhD, RNb  , Monika Fagevik Olsén, PhD, RPTb,c
, Monika Fagevik Olsén, PhD, RPTb,c  and Anna Elander, PhD, MDa,d
and Anna Elander, PhD, MDa,d 
aDepartment of Plastic Surgery, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden; bDepartment of Clinical Sciences/Gastrosurgical Research and Education, The Sahlgrenska Academy, Gothenburg University, Gothenburg, Sweden; cDepartment of Health and Rehabilitation/Physiotherapy, Institute of Neuroscience and Physiology, The Sahlgrenska Academy, Gothenburg University, Göteborg, Sweden; dDepartment of Plastic Surgery, Region Västra Götaland, Sahlgrenska University Hospital, Gothenburg, Sweden
ABSTRACT
Excess skin after massive weight loss is especially discomforting on the abdomen due to physical and psychosocial symptoms, and many patients with excess skin have a lowered Quality of Life (QoL). Abdominoplasty may improve symptoms as well as QoL and is offered to post-bariatric patients in Sweden with a post-operative body mass index (BMI) < 30 kg/m2. However, since > 50% never reach a BMI < 30, they are often left with an abdominal pannus that may hinder further weight loss. The reluctance to operate on patients with BMI > 30 is the increased risk of complications reported in several studies. Contradictorily, many studies have not found BMI > 30 to be an independent risk factor. Thus, the aim of this study was to prospectively evaluate a modified abdominoplasty technique on patients with a residual BMI 30–40, regarding complications and patient satisfaction. To contextualize, this was compared to a group of post-bariatric patients with a BMI < 30 that underwent standard abdominoplasties.
A total of 110 patients underwent either a standard abdominoplasty (BMI < 30 group) or a modified abdominoplasty (BMI 30–40 group). Pre-, peri-, and post-operative data were analyzed.
The complication rates were similar, apart from that the patients with BMI 30–40 had significantly more bleedings requiring re-interventions. There were no other significant differences in the complication panorama. The results from the questionnaires implied significant improvements in QoL and perception of excess skin, particularly in the BMI 30–40 group. In conclusion, this modified technique for abdominoplasty may be an acceptable compromise to a standard abdominoplasty for post-bariatric patients with residual obesity.
Clinical trials registry: https://www.researchweb.org/is/sverige/project/203961.
KEYWORDS: bariatric surgery; body contouring surgery; reconstructive surgery; excess skin; surplus skin; redundant skin; plastic surgery; abdominoplasty; panniculectomy
Citation: Journal of Plastic Surgery and Hand Surgery 2025; 61: 24–34. DOI: https://doi.org/10.2340/jphs.v61.45541.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 26 November 2024; Accepted: 20 January 2026; Published: 16 March 2026.
CONTACT: Jonas Ockell jonas.ockell@gu.se Department of Plastic Surgery, Sahlgrenska University Hospital, 413 45 Gothenburg Sweden
Competing interests and funding: The authors report that there are no competing interests to declare.
The work was supported by The Health and Medical Care Committee of the Region Västra Götaland under Grant Nr: 755461 and 471641.
Introduction
Excess skin is a well-documented adverse effect of massive weight loss, particularly after bariatric surgery [1–3]. Several studies indicate that most post-bariatric patients have excess skin on several body parts, with the abdomen being one of the most common locations [1, 2, 4]. Excess skin may be the cause of severe discomfort due to symptoms such as itching, rashes, intertriginous eczemas, and fungal infections; physical impairments, such as back pain and difficulties with running and sports; and psychosocial impairments, such as intimacy issues and difficulties finding clothes that fit properly [3]. Bariatric surgery may improve quality of life (QoL), particularly the physical domains [5]. Nevertheless, post-bariatric patients often have a lower QoL than the general population [6]. Unfortunately, it has even been reported that some patients regret their weight loss surgery because of the excess skin [7].
Body contouring surgery (BCS) may further improve QoL, body image and self-esteem in post-bariatric patients [8, 9]. An increasing number of studies in recent decades have indicated that abdominoplasty, for instance, effectively decreases the symptoms and impairments of excess abdominal skin, and patients’ QoL, as well as body image and mobility, is often significantly improved [3, 10]. Therefore, this operation is currently offered to post-bariatric patients in Sweden, provided that the patient meets the eligibility criteria (i.e. previous bariatric surgery, body mass index (BMI) < 30 kg/m2, reduced QoL, ≥ 3 cm ptosis of the abdominal skin fold, American Society of Anesthesiologists (ASA) Physical Status Classification ≤ 2).
However, most of the patients who undergo bariatric surgery never reach a BMI < 30 kg/m2 [11]. A common reason is that their already large weight loss causes a large abdominal skin pannus that can both physically and psychologically pose an obstacle for further weight loss. Additionally, the skin itself constitutes a large part of the total body weight. The skin and subcutis excised during body contouring procedures may weigh up to 20 kg, and the effect of the resected tissue on the patients, on average, is 1.5 BMI units [12].
The BCS not only improves discomfort and subjective symptoms of excess skin but may also help patients to obtain long-term weight control [12, 13]. In 2020, Sandvik et al. compared secondary weight regain in post-bariatric patients who underwent publicly funded abdominoplasty with that in post-bariatric patients who did not. Patients who underwent bariatric surgery and subsequent abdominoplasty had reduced weight regain, particularly in the subgroup that did not achieve a nadir BMI of < 30 kg/m2 [13]. There is also increasing evidence that lipectomy on the abdomen causes positive effects on insulin sensitivity and lowers the risk of cardiovascular disease [14, 15]. A review from 2019 summarizes the increasing amount of research on the topic: a significant reduction in inflammatory markers, a significant increase in adiponectin, a decrease in low-density lipoprotein (LDL) and triglycerides, an increase in high-density lipoprotein (HDL), a normalization in insulin and glucose, and an improvement in cardiac function [14]. The authors concluded that ‘mounting evidence indicates that reduction in adipose tissue volume alone may confer significant cardiovascular benefit’.
One reason for the restrictiveness of surgery on this group of patients with a BMI > 30 kg/m2 is the increased rates of wound complications, bleeding, and venous thromboembolism (VTE), which are associated with higher BMI according to several studies [8, 16–18]. Conflictingly, other studies have not identified BMI as a risk factor for complications [19–22]. In addition to the diversity of the scientific data, the definitions of complications also differ. Furthermore, ‘major complications’ are defined differently in different studies. Some studies have performed other procedures (liposuction, mammaplasty, brachioplasty, etc.) simultaneously or used different techniques for abdominoplasty in the same study. Finally, most studies have been retrospective.
Other researchers have proposed and evaluated modified techniques for abdominal contouring surgery, with the aim of reducing the risk of complications while still reducing symptoms associated with a large abdominal pannus. In 2009, Friedman et al. performed fleur-de-lis abdominoplasties with both horizontal and vertical resections but with limited undermining laterally from the vertical supraumbilical incision to spare perforators [23]. The authors concluded that fleur-de-lis abdominoplasty can be safely performed in massive weight loss patients.
Due to the large diversity in the results, the difference in definitions of complications and the predominance of retrospective study designs, the scientific basis for categorically excluding patients with BMI > 30 kg/m2 is inconclusive [19, 21, 24]. Furthermore, with increasing evidence of the benefits of abdominoplasty in this patient group, it is important to continue evaluating surgical techniques and perioperative protocols to improve safety for patients with residual obesity after bariatric surgery. Thus, the aim of this prospective study was to evaluate a modified abdominoplasty technique in an adequately sized patient group of obese post-bariatric patients, with standardized implementation of the operation and careful and structured follow-up. The secondary aim was to examine the patients’ QoL, physical and mental health and symptoms related to excess skin and weight before and after abdominoplasty.
Materials and methods
Patients
The study has been preregistered at https://www.researchweb.org/is/sverige/project/203961 under the project name ‘Evaluation of post operative complications and patients satisfaction after abdominoplasty on post bariatric patients with BMI ≥ 30–40 kg/m2’. A total of 130 post-bariatric patients who were referred to the Department of Plastic Surgery at Sahlgrenska University Hospital, Gothenburg, Sweden, were recruited. All participants were provided written and oral information about the study and gave their written informed consent. The study was approved by the local ethics committee in Gothenburg, Sweden (Dnr: 590-14). Figure 1 presents a flow chart of the included patients. The patients were divided into two groups depending on their BMI: the BMI < 30 group and the BMI 30–40 group. After exclusions, 110 participants were operated on between 2015 and 2020 at Sahlgrenska University Hospital and Art Clinic (BMI < 30-group) or Sahlgrenska University Hospital and Carlanderska Hospital (BMI 30–40-group), all in Gothenburg, Sweden.
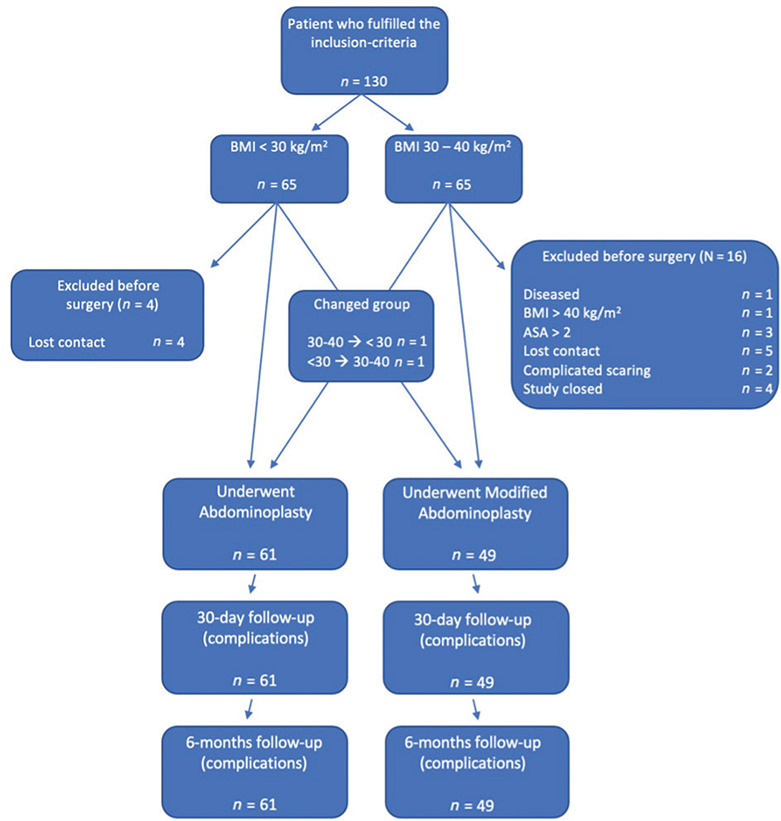
Figure 1. Flow chart of included patients.
Inclusion and exclusion
The inclusion criteria were as follows:
- Previous bariatric surgery ≥ 2 years ago
- Weight stability for at least 6 months (fluctuation of BMI less than ±1 kg/m2)
- ≥ 3 cm of ptosis of the skin on the abdomen
The exclusion criteria were as follows:
- Inability to understand and/or write Swedish
- ASA-score > 2 (or assessed as ineligible for surgery by anesthesiologist)
- Preoperatively discovered hernia in need of surgical repair with a mesh (physical examination and, for the BMI 30–40-group, pre-operative ultrasound or computer tomography of the abdominal wall).
- Serious mental illness
- Ongoing treatment with corticosteroids or other immunosuppressant medication
- Patients with neurological, orthopedic or rheumatological disease or impairment as well as patients with scaring on the abdomen were discussed within the research group before inclusion. If deemed necessary, the patients were discussed with their respective specialist physician.
- Smoking. All participants had to stop smoking at least 6 weeks before surgery.
Patient history and physical measurements
The measurements and patient history data collected consisted of the following:
- Demographic data, including age, sex, maximum BMI in life, preoperative BMI, pregnancies, months since bariatric surgery and educational level.
- Preoperative measurements consisted of laboratory tests, months of weight stability (1 BMI step), patient health history (health status, medication, allergies, smoking) and ptosis of the abdominal skin.
Questionnaires
All patients were asked to fill out questionnaires on two occasions, i.e. before and 6 months after abdominoplasty:
- Sahlgrenska Excess Skin Questionnaire (SESQ) – Patients’ rating of the amount of excess skin on different body parts (0–4) as well as the discomfort (0–10) and specific symptoms/impairments it causes (0–4). In addition, a combined scoring, ‘SESQ-score’, is calculated based on the summarized score of certain symptom/impairment scores (0–28). The SESQ has been used in a multitude of studies and has been shown to be reliable and valid [1, 3, 25].
- EuroQoL 5-dimensions (EQ-5D) – measurement of Health-Related Quality of Life (HRQoL) with Swedish value set. As recommended by the authors, the time trade-off (TTO) score was used in this study [26].
- Hospital Anxiety and Depression Scale (HAD) – Self-rating instrument to assess psychological distress in nonpsychiatric patients [27].
- Patient Specific Functional Scale (PSFS) – Self-reported, valid, reliable outcome measure instrument that evaluates functional disability caused by pain or injuries based on patient-reported activities and the difficulty in performing them [28].
Surgical technique and perioperative data collection
BMI < 30 group
The surgical technique for the BMI < 30 group was unaltered from the routine abdominoplasty at our department.
Suprapubic incision under the overhang at the bottom of the fold, dissection down to the fascia and undermining lateral as well as cranially to the umbilicus. The umbilicus was cut out from the skin. The abdominal skin and subcutis were then dissected superficially to the fascia up to the processus xiphoideus. A diastasis of the rectus muscle was then plicated if necessary. Diastasis measures (cm) and plication (yes/no) were observed in the operation report. The excess skin was measured in place and cut away. If necessary and if deemed possible, a skin resection was performed in the middle line to reduce the excess horizontally (inverted T-incision). The wounds were sutured, and the umbilicus was implanted unless otherwise agreed upon by the patient. Two subcutaneous drains were placed caudally and laterally, and abdominal dressings were applied.
BMI 30–40 group
The surgical technique in the BMI 30–40 group was altered to facilitate optimal blood supply to the abdominal skin flap.
Suprapubic incision under the overhang dissection down to the fascia and undermining lateral as well as cranially to the umbilicus. The umbilicus was cut out from the flap. The resection above the umbilicus included a triangle up to the processus xiphoideus with a base of 8–10 cm just above the umbilicus along the midline. The skin flaps lateral to the midline were bluntly dissected with the fingers so that the perforators were not divided. In all patients, a skin resection was performed in the middle line to reduce the excess horizontally (inverted T-incision) as well as a resection of the caudal part of the flaps removing the ptosis. The wounds were sutured, and the umbilicus was implanted unless otherwise agreed upon by the patient. Two subcutaneous drains were placed caudally and laterally, and abdominal dressings were applied.
Both groups
During and after the surgery, the length of the operation (min), the amount of perioperative bleeding (mL), weight of the resected tissue (g), and whether a T-incision was performed were recorded (yes/no). All patients received prophylactic treatment with antibiotics according to the normal routine at our department, i.e. flucloxacillin or clindamycin, until the drainage was removed (when < 50 mL of secretion or for a maximum of 10 days). Furthermore, all patients were fitted for compression stockings that were worn from just before the operation and day and night for 7 days and then daytime until fully mobilized (class 2, JOBST Opaque, Essity Hygiene and Health AB, Mölndal, Sweden). All patients received a girdle to wear day and night for 6 weeks and then day or night for 6 additional weeks.
Complications
Complications were recorded and categorized into three time periods: during surgery, within 30 days and from 31 days to 6 months. Complications were then categorized as either minor or major and defined as follows.
Minor complications: Suspected seroma/hematoma, minor wound ruptures as well as wound infections, fat necrosis or skin necrosis where conservative treatment (wound revision, drainage) in the outpatient clinic and/or oral antibiotic treatment was sufficient.
Wound infections were categorized as suggested by Lewin et al. [29], into:
Level 1: Secretion from the wound
Level 2: Redness, swelling, heat and exudate
Level 3: Redness, swelling, and heat and purulent drainage or induration
Level 4: Fewer and/or septicemia as well as Grade 3.
Major complications: Deep vein thrombosis (DVT), pulmonary embolism, pneumonia, sepsis, bleeding with the need for blood transfusion and/or reoperation, hematoma/seroma that needs to be drained with other than puncture, abscess/cellulitis/infection requiring surgery and/or inpatient treatment with or without intravenous antibiotics, or death within 30 days of surgery.
Follow-up
The patients were assessed by a doctor before discharge and then again by a nurse after 1 week. The non-resorbable sutures were removed after 2 weeks. After 5–7 weeks, the patients were contacted and were either assessed at the outpatient clinic or contacted by phone, and the patient journal was reviewed to record any complications during the first 30 days post-operatively. After 6 months, the patients were called to the outpatient clinic to assess the cosmetic and functional results of the surgery as well as to record any late complications.
Statistics
For demographics, physical measurements, and complications, means, standard deviations (SDs), medians, minimal and maximal are presented for continuous variables. For categorical variables, n (%) of reported data are presented. For comparisons within groups, the Wilcoxon signed rank test was used. For comparisons between groups, the Mann–Whitney U test was used for continuous variables, and the chi-square test or Fisher’s exact test was used for categorical variables. All significance tests were two-sided, and a p-value < 0.05 was considered significant. The statistical analyses were performed using SPSS 28.0.1.1 (IBM Corp. 2023).
Results
Demographic data
The demographics are presented in Table 1. As expected, the participants in the BMI 30–40 group had higher BMI at all points in time, they had also been weight-stable for a significantly shorter time (20.4 months compared to 21.1 months, p = 0.014), had waited longer for BCS since their bariatric surgery (87.8 months compared to 63.1 months, p < 0.001) and had a larger ptosis of the abdominal skin (7.6 cm compared to 4.8 cm, p < 0.001).
Perioperative data
The data from the operation and the duration of the hospital stay are presented in Table 2. The BMI 30–40 group had a longer mean operating time (mean 136.7 min compared to 108.0 min) and more resected tissue (3024.0 g compared to 1945.9 g) than the BMI < 30 group (p < 0.001 for both). Additionally, the hospital stay was significantly longer for the BMI 30–40 group (1.8 days compared to 1.1 days, p < 0.001). The participants in the BMI < 30 group more often had their subcutaneous drainage removed before dismissal from the ward, 92 compared to 41% (< 0.001). Only one patient in the BMI 30–40 group had an unexpected finding during surgery: a preoperatively unknown hernia requiring repair with a mesh.
Early and late complications
The data from the follow-ups (early and late complications) are presented in Table 3. Up to 30 days postoperatively, approximately 63% of the participants in the BMI 30–40 group had any minor complications, and 20% had any major complications. In the BMI < 30-group, the corresponding numbers were approximately 72 and 7%, respectively. The minor complications in both groups were mainly seromas and wound infections requiring orally administered antibiotics. The major complications in the BMI 30–40-group were mainly bleeding and infections, whereas in the BMI < 30-group they constituted mainly bleeding, VTE’s and sepsis/infected seroma. The BMI 30–40-group had significantly more major complications than the BMI < 30-group (p = 0.043), but there were no significant differences regarding minor complications (p > 0.05).
From 31 days to 6 months, about 18% of the participants in the BMI 30–40 group had any new minor complication, and none had any new major complication. For the BMI < 30 group, nearly 25% of the participants had any new minor complication, and approximately 7% had any new major complication. In both groups, seromas were the most common minor complication. Only the BMI < 30 group had any new major complications, mainly related to severe infections, VTE and wound dehiscence requiring reintervention. There were no significant differences between the groups regarding minor or major complications (p > 0.05).
Finally, the total amount of minor and major complications, as well as any complications throughout the entire study duration (6 months), were analyzed. Seventy-four per cent of the participants in the BMI 30–40 group had any minor complication, compared to 72% in the BMI < 30 group. The corresponding numbers for major complications were 20% and 12%, respectively. Finally, approximately 80% in the BMI 30–40 group and 75% in the BMI < 30 group had any complication within 6 months of the surgery. We found no significant difference between the groups regarding any minor, any major or any complication at all (p > 0.05).
Post-operative data
As described in Table 2, the BMI < 30 group did not decrease in BMI 6 months after abdominoplasty (p > 0.05), while the BMI 30–40 group lost a mean of 1.0 kg/m2 (p = 0.003).
Results from the questionnaires
SESQ
The scores from the SESQ-questions specific for the abdomen (rating of the amount of excess skin from 0 to 4 and the discomfort it causes from 0 to 10) are presented in Table 4, and the results indicate that both groups improved significantly. The rating of the perceived amount of excess skin decreased from a median of 3.5 to 0 in the BMI < 30 group and from 4.0 to 1.0 in the BMI 30–40 group (both p < 0.001). The discomfort decreased from a median of 9.0 to 0.0 in both groups (both p < 0.001). The BMI 30–40 group scored significantly more excess skin before abdominoplasty than the BMI < 30 group (median 4 compared to 3.5, p = 0.044), but there were no other significant differences between the groups.
The results from the SESQ-scores are presented in Figure 2. Both groups improved their SESQ scores significantly (p < 0.001 for both). The BMI 30–40 group scored significantly lower both before and after abdominoplasty (p < 0.001 and < 0.01, respectively), but the difference between before and after abdominoplasty did not differ significantly between the groups.
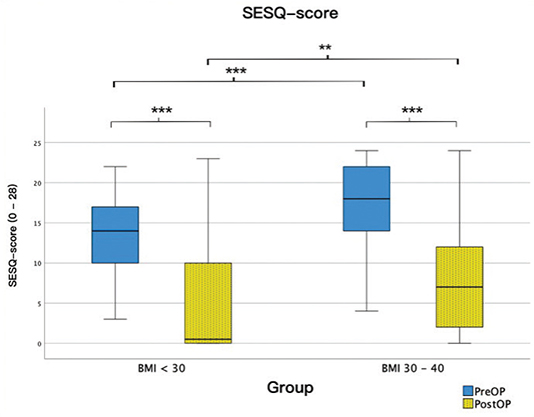
Figure 2. Boxplot of SESQ scores for the two groups pre- and post-operatively. The thick horizontal line is the median, the box edges are Q1 and Q3, and the whiskers are the minimum and maximum. SESQ: Sahlgrenska Excess Skin Questionnaire.
The results from the symptoms and impairment questions are presented in Figure 3. Both groups improved significantly regarding all symptoms and impairments (p < 0.05 – < 0.001), and the median for most patients post-operatively was 0. The BMI 30–40 group scored significantly higher (worse) pre-operatively for all questions except ‘hindrance in intimate situations’ and ‘unattractive body’. There were no significant differences between the groups post-operatively.
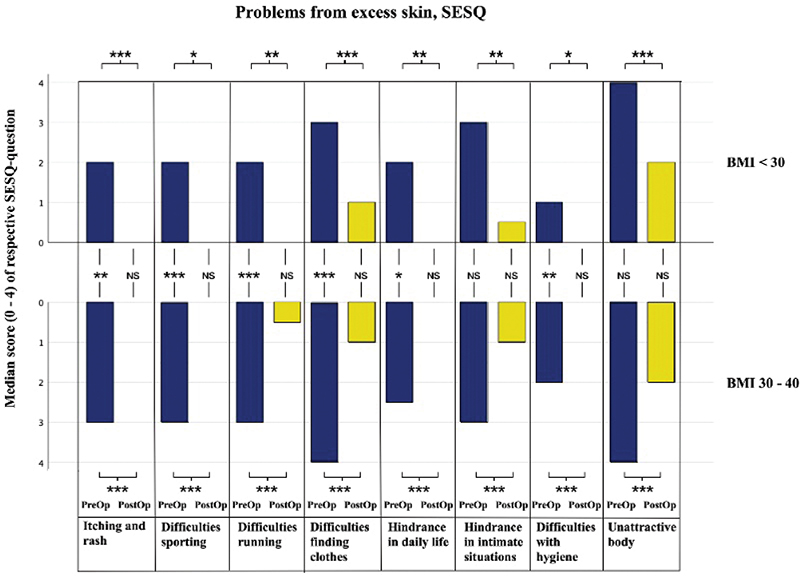
Figure 3. Bar chart with median scores from the symptoms and hindrance questions of the SESQ preoperatively (PreOp) and postoperatively (PostOp) for the two study groups. NS: non-significant; SESQ: Sahlgrenska Excess Skin Questionnaire. *p < 0.05, **p < 0.01, ***p < 0.001.
EQ-5D
The results from the EQ-5D TTO scores are presented in Figure 4. The BMI 30–40 group had significantly improved scores post-operatively (p < 0.05), which the BMI < 30 group did not. Furthermore, the BMI 30–40 group scored significantly lower scores (worse) than the BMI < 30 group both pre- and post-operatively (both p < 0.01). There was no significant difference between the groups, however, regarding the difference from pre- to post-operatively.
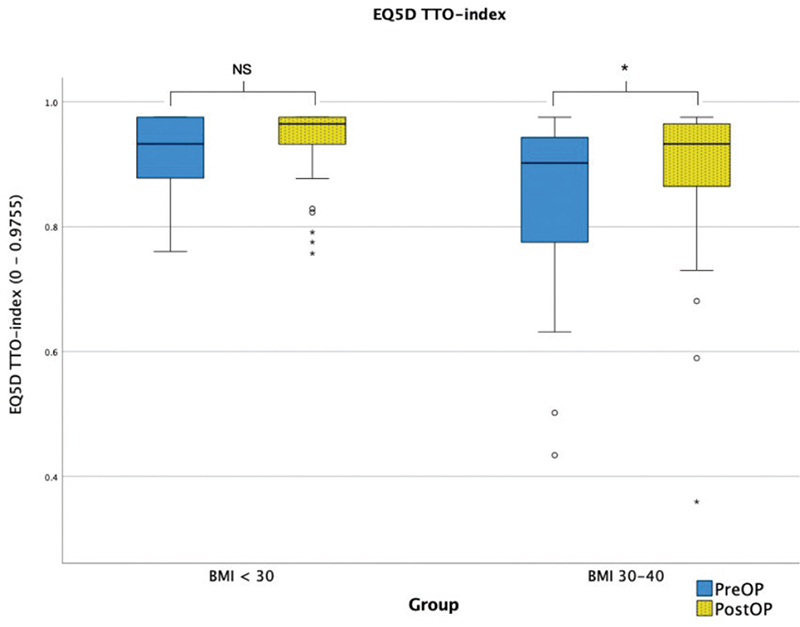
Figure 4. Boxplot of EQ5D-TTO scores for the two groups pre- and post-operatively. The thick horizontal line is the median, box edges are Q1 and Q3, whiskers are the minimum and maximum, o are outliers, and + are extremes. NS: non-significant. *p < 0.05, **p < 0.01, ***p < 0.001.
PSFS
Commonly participant-reported examples of activities where the excess skin constituted a hindrance were activities associated with showing oneself without covering clothes, such as bathing in bathhouses, public beaches, and intimate situations, as well as finding clothes, performing one’s personal hygiene and exercising. The results from the PSFS scores are presented in Figure 5. Both groups improved significantly from pre- to postoperatively? (both p < 0.001), but there were no significant differences between the groups regarding pre- or post-operative scores or the difference between the two.
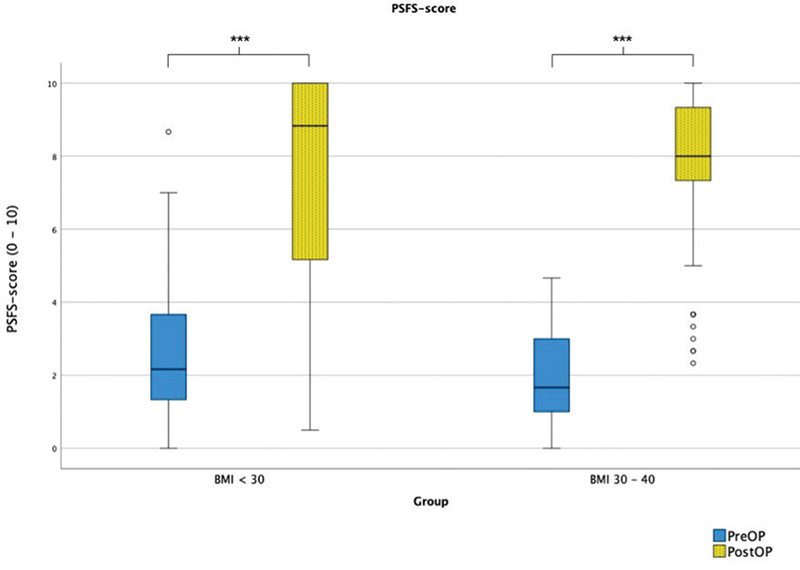
Figure 5. Boxplot of PSFS scores for the two groups pre- and post-operatively. The thick horizontal line is the median, box edges are Q1 and Q3, whiskers are the minimum and maximum, o are outliers, and + are extremes. NS: non-significant; PSFS: Patient Specific Functional Scale. *p < 0.05, **p < 0.01, ***p < 0.001.
HAD
We found no significant difference regarding the HAD scores pre- or post-operatively within the groups or between the groups (data not shown).
Discussion
Although evidence is mounting on the benefits of post-bariatric abdominoplasty, the procedure is not suitable for everyone. Apart from financial barriers, medical conditions, such as obesity or obesity-related diseases, may render the procedure dangerous. Because of this, a BMI > 30 kg/m2 is often used as a cut off for surgery, leaving more than 50% of post-bariatric patients ineligible for this additional step toward their ‘second chance’ in life. As described above, this cut off has been debated, and several studies have concluded that abdominoplasty, traditional or modified, can be performed with the same level of safety in patients with obesity [19, 20].
The main aim of this study was to prospectively map the complication panorama of a modified abdominoplasty technique in a post-bariatric group of patients with residual obesity (BMI 30–40 kg/m2). To put the results into context, the results were compared to a group of patients who underwent an operation according to the current routine (BMI < 30 kg/m2, full abdominoplasty). Our results suggest that modifications in the operation technique and perioperative protocol may render it safe to operate on post-bariatric patients with a BMI of 30–40 kg/m2. The purpose of not performing a full abdominoplasty was to spare important perforators located in the upper quadrants of the abdomen, thus lowering the risk of wound complications related to reduced tissue perfusion, such as wound dehiscence, skin necrosis and infections. Merely offering a standard panniculectomy may leave a discomforting pannus on the flanks, especially in obese patients, possibly resulting in residual discomfort and impairments. The inverted T-incision enables the mobilization and contouring of the lateral skin pannus, resulting in a better aesthetic and functional outcome. Our results show that the two groups had similar numbers of wound complications, and no patients in the BMI 30–40 group had any wound complications needing reintervention. Local wound infections were similar in the two groups, although more patients (8.2% vs. 3.3%) in the BMI 30–40 group needed readmission due to severe infection. Finally, the frequencies of seromas were similar in both groups, possibly with a slight advantage for the BMI 30–40 group. The more careful undermining in the modified technique, compared to a full abdominoplasty, may be a reason for this. All interpretations regarding specific complications should be done with caution, however, as the power calculation of the study was based on the total number of complications in each group, and the study sample is not large enough to draw detailed conclusions about each specific complication. When categorizing the complications of the entire study into minor, major and any complication, however, we found no significant difference between the groups by the end of the study, suggesting that the modified technique combined with the perioperative protocol may offer a safe compromise for patients with a BMI of 30–40 kg/m2.
To minimize the risk of VTE, the perioperative protocol for the BMI 30–40 group included early post-operative mobilization, compression stockings and chemoprophylaxis (dalteparin) at an increased dose (7500E). The reason for choosing an increased dose was to compensate for the high BMI in this group, a decision made in agreement with coagulation specialists as well as obesity surgeons. Indeed, the only VTE cases in the study were found in the BMI < 30 group, in which most patients (79%) did not receive chemoprophylaxis. However, the BMI 30–40-group had a higher frequency of post-operative bleeding requiring transfusion and/or reintervention. The high number resulted in a pause in the study to investigate possible causes for this and to compare the outcome regarding bleeding in the BMI < 30 group. Although the BMI < 30 group also had bleeding complications, the higher number in the BMI 30–40 group resulted in an early closure of the study to further investigate the complications in detail and re-evaluate the peri-operative protocol. However, we found that the patients with bleeding complications in the BMI 30–40 group had a relatively low preoperative BMI (30–36 kg/m2), possibly implying that the increased chemoprophylaxis dose, as calculated per kg, may have been too high for these patients. Further investigation is needed to decide whether to reduce the dose for all patients or to raise the cutoff for the increased dose.
The rate of minor complications in this study was high compared to earlier studies [18, 22, 30], just over 70% for both groups. There may be several reasons for this. First, a prospective design enables detailed documentation of complications compared to retrospective studies, where the risk of missing minor complications is much higher. Second, almost any deviation from a flawless recovery was documented as a complication, with the goal of achieving a detailed documentation of the complication panorama. For instance, any fluctuation under the skin was defined as a suspected seroma or hematoma, and any secretion from the wound was defined as a possible level 1 infection. This level of detail should ultimately impact the number of complications. Finally, all complications in all patients were recorded, with the result that many patients had several different complications recorded, in some cases both before and after 30 days.
Regarding major complications, the rates of approximately 12% for the BMI < 30 group and 20% for the BMI 30–40 group are in line with earlier publications [18, 22, 30]. Schlosshauer, for instance, reported rates of 12.8 and 10.8% for major complications with revision and bleeding, respectively [18]. Finally, the rates of VTE were comparable to those in several previous studies [18, 30–32]. As described above, none of the patients in the BMI 30–40 group had any VTE, possibly due to the increased dose of chemoprophylaxis, which, in turn, may have caused an increased number of post-operative bleeding events. Several studies report a high risk of VTE in body contouring procedures, especially for obese patients [33], and the importance and efficiency of prophylactic measures such as early mobilization, compressive stockings and chemoprophylaxis has been indicated and discussed in several studies [34, 35]. Due to the possible fatal outcome of VTE, we believe that it is important to keep evaluating perioperative protocols with a focus on prophylactic measures.
There is strong support for the benefits of bariatric surgery. Likewise, the procedures are generally safe and beneficial from a social economic perspective. The benefits and the acceptable risk/reward ratio explain the increasing popularity of the procedures worldwide and, unfortunately, the corresponding increase in the demand for BCS, such as abdominoplasty. The complication rate of abdominoplasty, as discussed earlier, is relatively high; thus, the reward-side of the scale must be heavy enough to weigh up for the risk of major complications. For that reason, the secondary aims of this study were to examine and compare the patients’ scores regarding symptoms and impairments of excess skin, physical and mental health, and QoL and, finally, potential weight loss after abdominoplasty.
We found that both groups improved significantly regarding all aspects of the SESQ, strongly suggesting that both surgical techniques adequately treated what they were supposed to treat. This is further strengthened by the results from the PSFS, where the scores almost normalized after surgery. As the PSFS allows the participant themselves to choose the situations where the excess skin causes problems, these results also strongly imply that both procedures significantly reduce the discomfort, related symptoms, and limitations of excess skin.
When focusing on the difference in the participants’ assessments of the two groups, the BMI 30–40 group scored significantly worse both before and after surgery on the SESQ-score, the EQ-5D TTO-score and before surgery on most SESQ symptom-specific questions. It may be that a higher BMI causes more discomfort and physical impairments of excess skin. The larger mean ptosis in this group may be the cause of this relationship, as described in previous studies [3]. While the BMI < 30 group had a seemingly higher (better) mean TTO score compared to Swedish norm values (0.898 for men and 0.886 for women) [36] both pre- and post-operation, the BMI 30–40 group scored lower than the norm values pre-operatively and similar to the norm values post-operatively, suggesting a normalization of HRQoL as measured by this instrument. Furthermore, in accordance with previous findings, we also found indications that abdominoplasty in this group of patients may facilitate further weight loss [12, 13]. In summary, our results suggest that treating these physical impairments with BCS may be particularly effective in reducing symptoms and discomfort and increasing QoL in post-bariatric patients with residual obesity.
There are some limitations to this study, which should be considered when interpreting the results. Firstly, the study was terminated earlier than planned due to an unexpectedly high number of bleeding complications in the BMI 30–40 group. When examining these complications in detail, a possible cause of the high dose of chemoprophylaxis was found. Nevertheless, we do not know for sure if these events were actually caused by chemoprophylaxis, the higher BMI in this group or merely by chance. This will be investigated further. Regardless, 92% of the planned 120 participants (as decided after power calculations prestudy) were operated on, and the follow-up regarding complications were 100%, which we believe allows us to draw conclusions from the results. Secondly, since the power of this study was calculated on the difference in total complications at the group level, detailed conclusions may not be drawn from each specific complication. As some of the complications are rare, the study sample would need to be very large to find any significant differences. Thus, we chose only to compare grouped complications (minor, major, any) and not to compare each complication, as we cannot know if a lack of significance is due to an actual difference between the groups or due to insufficient sample size. Nevertheless, the table of complication frequencies (Table 3) should allow us to draw conclusions from the results and, hopefully, serve as material for future studies on the topic. Finally, the groups of patients were operated on in different clinics, which may influence the perioperative results as well as the complications. For instance, the culture of the clinics may differ regarding how peri-operative bleeding is calculated, patient mobilization routines, discharge time from the ward, removal of drainage, use of antibiotics and handling of complications (e.g. puncture of seromas). Thus, differences in perioperative data should be interpreted with caution, as they may be influenced by both different routines and the differences between the groups. Nonetheless, the complication data were recorded in a prospective and standardized manner, thus ensuring high reliability in these results.
In conclusion, we operated on post-bariatric patients with residual obesity with a modified abdominoplasty (BMI 30–40 group) and compared the results with a group with BMI < 30 who underwent full abdominoplasty (BMI < 30 group). We found significantly more early major complications in the BMI 30–40 group but no significant difference in complication rates between the groups otherwise, suggesting that our modifications in operating technique as well as perioperative protocol may contribute to the development of standardized safety routines in future post-bariatric BCS of obese patients. Furthermore, the results from the questionnaires as well as BMI change from pre- to post-operation imply that post-bariatric patients with a BMI over 30 benefits from abdominoplasty with a reduction in physical impairments and discomfort, an increase in QoL and further weight loss. Hopefully, our results may contribute to rendering these patients eligible for abdominal BCS in the future, thereby providing them with the same possibilities as non-obese post-bariatric patients.
Ethics
Approved by the local ethics committee of Gothenburg, Sweden, D-nr: 590-14
Trial registration
The study has been preregistered at https://www.researchweb.org/is/sverige/project/203961 under the project name ‘Evaluation of post-operative complications and patients satisfaction after abdominoplasty on post-bariatric patients with BMI ≥ 30–40 kg/m2’.
References
[1] Biorserud C, Nielsen C, Staalesen T, et al. Sahlgrenska Excess Skin Questionnaire (SESQ): a reliable questionnaire to assess the experience of excessive skin after weight loss. J Plast Surg Hand Surg. 2013;47(1):50–59. https://doi.org/10.3109/2000656X.2012.731001
[2] Klassen AF, Kaur M, Breitkopf T, et al. Using the BODY-Q to understand impact of weight loss, excess skin, and the need for body contouring following bariatric surgery. Plast Reconstr Surg. 2018;142(1):77–86. https://doi.org/10.1097/PRS.0000000000004461
[3] Elander A, Biorserud C, Staalesen T, et al. Aspects of excess skin in obesity, after weight loss, after body contouring surgery and in a reference population. Surg Obes Relat Dis. 2019;15(2):305–311. https://doi.org/10.1016/j.soard.2018.10.032
[4] Biorserud C, Olbers T, Staalesen T, et al. Understanding excess skin in postbariatric patients: objective measurements and subjective experiences. Surg Obes Relat Dis. 2016;12(7):1410–1417. https://doi.org/10.1016/j.soard.2016.01.033
[5] Magallares A, Schomerus G. Mental and physical health-related quality of life in obese patients before and after bariatric surgery: a meta-analysis. Psychol Health Med. 2015;20(2):165–176. https://doi.org/10.1080/13548506.2014.963627
[6] Driscoll S, Gregory DM, Fardy JM, et al. Long-term health-related quality of life in bariatric surgery patients: a systematic review and meta-analysis. Obesity (Silver Spring, Md). 2016;24(1):60–70. https://doi.org/10.1002/oby.21322
[7] Klassen AF, Cano SJ, Scott A, et al. Satisfaction and quality-of-life issues in body contouring surgery patients: a qualitative study. Obes Surg. 2012;22(10):1527–1534. https://doi.org/10.1007/s11695-012-0640-1
[8] Sadeghi P, Duarte-Bateman D, Ma W, et al. Post-bariatric plastic surgery: abdominoplasty, the state of the art in body contouring. J Clin Med. 2022;11(15):4315. https://doi.org/10.3390/jcm11154315
[9] Jiang Z, Zhang G, Huang J, et al. A systematic review of body contouring surgery in post-bariatric patients to determine its prevalence, effects on quality of life, desire, and barriers. Obes Rev. 2021;22(5):e13201. https://doi.org/10.1111/obr.13201
[10] Toma T, Harling L, Athanasiou T, et al. Does body contouring after bariatric weight loss enhance quality of life? A systematic review of QOL studies. Obes Surg. 2018;28(10):3333–3341. https://doi.org/10.1007/s11695-018-3323-8
[11] Varban OA, Cassidy RB, Bonham A, et al. Factors associated with achieving a body mass index of less than 30 after bariatric surgery. JAMA Surg. 2017;152(11):1058–1064. https://doi.org/10.1001/jamasurg.2017.2348
[12] Froylich D, Corcelles R, Daigle CR, et al. Weight loss is higher among patients who undergo body contouring procedures after bariatric surgery. Surg Obes Relat Dis. 2016;12(9):1731–1736. https://doi.org/10.1016/j.soard.2015.09.009
[13] Sandvik J, Hole T, Klöckner C, et al. The impact of post-bariatric abdominoplasty on secondary weight regain after Roux-en-Y Gastric Bypass. Front Endocrinol (Lausanne). 2020;11:459. https://doi.org/10.3389/fendo.2020.00459
[14] Rocic P. Comparison of cardiovascular benefits of bariatric surgery and abdominal lipectomy. Curr Hypertens Rep. 2019;21(5):37. https://doi.org/10.1007/s11906-019-0945-8
[15] Cuomo R, Russo F, Sisti A, et al. Abdominoplasty in mildly obese patients (BMI 30-35 kg/m2): metabolic, biochemical and complication analysis at one year. In vivo (Athens, Greece). 2015;29(6):757–761.
[16] Rogliani M, Silvi E, Labardi L, et al. Obese and nonobese patients: complications of abdominoplasty. Ann Plast Surg. 2006;57(3):336–338. https://doi.org/10.1097/01.sap.0000221460.43861.6b
[17] Kalmar CL, Park BC, Kassis S, et al. Functional panniculectomy vs cosmetic abdominoplasty: multicenter analysis of risk factors and complications. J Plast Reconstr Aesth Surg. 2022;75(9):3541–3550. https://doi.org/10.1016/j.bjps.2022.04.057
[18] Schlosshauer T, Kiehlmann M, Jung D, et al. Post-bariatric abdominoplasty: analysis of 406 cases with focus on risk factors and complications. Aesth Surg J. 2021;41(1):59–71. https://doi.org/10.1093/asj/sjaa067
[19] Christopher AN, Morris MP, Patel V, et al. Abdominal body contouring: does body mass index affect clinical and patient reported outcomes? J Surg Res. 2022;270:348–358. https://doi.org/10.1016/j.jss.2021.09.035
[20] de Macedo JLS, Rosa SC, Canedo LR, et al. What is the impact of residual obesity on the risk for postoperative body-contouring surgery complications in postbariatric patients? Obes Surg. 2020;30(10):4149–4154. https://doi.org/10.1007/s11695-020-04711-0
[21] Hasanbegovic E, Sorensen JA. Complications following body contouring surgery after massive weight loss: a meta-analysis. J Plast Reconstr Aesth Surg. 2014;67(3):295–301. https://doi.org/10.1016/j.bjps.2013.10.031
[22] Staalesen T, Olsen MF, Elander A. Complications of abdominoplasty after weight loss as a result of bariatric surgery or dieting/postpregnancy. J Plast Surg Hand Surg. 2012;46(6):416–20. https://doi.org/10.3109/2000656X.2012.717898
[23] Friedman T, O’Brien Coon D, Michaels J, et al. Fleur-de-Lis abdominoplasty: a safe alternative to traditional abdominoplasty for the massive weight loss patient. Plast Reconstr Surg. 2010;125(5):1525–1535. https://doi.org/10.1097/PRS.0b013e3181d6e7e0
[24] Gunnarson GL, Froyen JK, Sandbu R, et al. Plastic surgery after bariatric surgery. Tidsskr Nor Laegeforen. 2015;135(11):1044–1049. https://doi.org/10.4045/tidsskr.14.0814
[25] Ockell J, Elander A, Staalesen T, et al. Evaluation of excess skin in Swedish adults 18-59 years of age. J Plast Surg Hand Surg. 2017;51(2):99–104. https://doi.org/10.1080/2000656X.2016.1187157
[26] Burström K, Sun S, Gerdtham UG, et al. Swedish experience-based value sets for EQ-5D health states. Qual Life Res. 2014;23(2):431–442. https://doi.org/10.1007/s11136-013-0496-4
[27] Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67(6):361–370. https://doi.org/10.1111/j.1600-0447.1983.tb09716.x
[28] Nicholas P, Hefford C, Tumilty S. The use of the Patient-Specific Functional Scale to measure rehabilitative progress in a physiotherapy setting. J Man Manip Ther. 2012;20(3):147–152. https://doi.org/10.1179/2042618612Y.0000000006
[29] Lewin R, Göransson M, Elander A, et al. Risk factors for complications after breast reduction surgery. J Plast Surg Hand Surg. 2014;48(1):10–14. https://doi.org/10.3109/2000656X.2013.791625
[30] De Paep K, Van Campenhout I, Van Cauwenberge S, Dillemans B. Post-bariatric abdominoplasty: identification of risk factors for complications. Obes Surg. 2021;31(7):3203–3209. https://doi.org/10.1007/s11695-021-05383-0
[31] Griffin M, Akhavani MA, Muirhead N, et al. Risk of thromboembolism following body-contouring surgery after massive weight loss. Eplasty. 2015;15:e17.
[32] Gupta V, Winocour J, Rodriguez-Feo C, et al. Safety of aesthetic surgery in the overweight patient: analysis of 127,961 patients. Aesth Surg J. 2016;36(6):718–729. https://doi.org/10.1093/asj/sjv268
[33] Kalmar CL, Thayer WP, Kassis S, et al. Pulmonary embolism risk after cosmetic abdominoplasty and functional panniculectomy. Ann Plast Surg. 2022;89(6):664–669. https://doi.org/10.1097/SAP.0000000000003281
[34] Reish RG, Damjanovic B, Colwell AS. Deep venous thrombosis prophylaxis in body contouring: 105 consecutive patients. Ann Plast Surg. 2012;69(4):412–414. https://doi.org/10.1097/SAP.0b013e31824a45e0
[35] Brito ÍM, Meireles R, Baltazar J, et al. Abdominoplasty and patient safety: the impact of body mass index and bariatric surgery on complications profile. Aesth Plast Surg. 2020;44(5):1615–1624. https://doi.org/10.1007/s00266-020-01725-y
[36] Teni FS, Gerdtham UG, Leidl R, et al. Inequality and heterogeneity in health-related quality of life: findings based on a large sample of cross-sectional EQ-5D-5L data from the Swedish general population. Qual Life Res. 2022;31(3):697–712. https://doi.org/10.1007/s11136-021-02982-3