ORIGINAL RESEARCH ARTICLE
Clinical and histopathological effects of topical pentoxifylline on autonomisation processes in rat dorsal randomised skin flaps
Melike Zeynep Kuşçu, MDa, Selman Hakkı Altuntaş, MD, Assist. Prof.a, Özlem Özmen, DVM, Professorb and Simge Garlı, DVMc
aDepartment of Plastic, Reconstructive and Aesthetic Surgery, Süleyman Demirel University Faculty of Medicine, Isparta, Türkiye; bDepartment of Pathology, Burdur Mehmet Akif Ersoy University Faculty of Veterinary Medicine, Burdur, Türkiye; cAnimal Experiments Production and Experimental Research Laboratory, Burdur Mehmet Akif Ersoy University Faculty of Veterinary Medicine, Burdur, Türkiye
ABSTRACT
Background: Staged pedicled skin flaps that require pedicle division are frequently employed in reconstructive surgery, rely on their pedicle for vascular supply following transfer until neovascularisation occurs at the recipient site, a phenomenon referred to as autonomisation (pedicle-independent flap viability following neovascularisation). This study sought to investigate the application of topical pentoxifylline to expedite the autonomisation process.
Methods: Caudal-based skin flaps were surgically created in the dorsal region of 60 Wistar rats. On the fifth day, the bases of the flaps were surgically cut, and the flaps were subsequently monitored for an additional 7 days. The rats were divided into three groups: (1) Control group (Vaseline for 5 + 7 = 12 days), (2) Experimental-1 group (topical pentoxifylline for 5 days + vaseline for 7 days), and (3) Experimental-2 group (topical pentoxifylline for 5 + 7 = 12 days). Flap viability was assessed both macroscopically and microangiographically, and histopathological examinations were conducted.
Results: On day 12, the viable flap areas were 40.7% in the control group, 95.5% in experimental-1 group, and 100.0% in experimental-2 group (p < 0.05). Microangiography was conducted on day 5 and 12, and both experimental groups exhibited markedly greater vascularisation compared to the control group. Histopathological and immunohistochemical analyses revealed that pentoxifylline significantly enhanced angiogenesis, new vessel formation, and epithelisation, while mitigating early-stage inflammation, edema, and late-stage fibrosis (p < 0.05).
Conclusion: The application of topical pentoxifylline significantly promoted angiogenesis, thereby expediting the autonomisation of rat dorsal randomised skin flaps. Consequently, its use in reconstructive surgery may be considered, as it has the potential to reduce reliance on the pedicle, minimise complications, and enhance patient outcomes.
KEYWORDS: Flap autonomisation; flap viability; neoangiogenesis; topical pentoxifylline
Citation: Journal of Plastic Surgery and Hand Surgery 2025; 61: 56–64. DOI: https://doi.org/10.2340/jphs.v61.45582.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 05 September 2025; Accepted: 27 January 2026; Published: 25 March 2026.
CONTACT: Selman Hakkı Altuntaş, MD., Assist. Prof. shaltuntas@gmail.com; selmanaltuntas@sdu.edu.tr Department of Plastic, Reconstructive and Aesthetic Surgery, Süleyman Demirel University Faculty of Medicine, 32260 Çünür, Isparta / Türkiye
Competing interests and funding: The authors declare that there is no conflict of interest.
Introduction
Distant or regional pedicled skin flaps play a crucial role in reconstructive surgery for addressing skin and soft tissue defects when local flaps are insufficient. These tissue units maintain their vascular supply during transfer from the donor to the recipient site, thereby ensuring their initial survival. The pedicle is a temporary structure and necessitates a second stage for its division [1]. The pedicle, which encompasses blood-supplying vessels, is essential for flap viability in the early post-transfer phase. Over time, neoangiogenesis between the flap and recipient site diminishes this dependency, a process known as autonomisation, wherein the flap survives independently of its pedicle due to new vessel formation. Research has focused on accelerating autonomisation, with pharmacological agents being investigated to enhance flap viability and reduce reliance on pedicle [2, 3].
Pentoxifylline, a methylxanthine derivative and non-selective phosphodiesterase inhibitor, facilitates vasodilation by relaxing the vascular smooth muscle and enhancing blood flow [4]. Its hemorheological effects include reducing platelet adhesion and aggregation, increasing erythrocyte flexibility, and promoting fibrinolysis, all of which contribute to improved blood fluidity [5]. Furthermore, pentoxifylline demonstrates anti-inflammatory properties by decreasing proinflammatory cytokine production, reducing leukocyte sensitivity to cytokines, and inhibiting endothelial adhesion, chemotaxis, and neutrophil degranulation [6]. It also mitigates ischemia-reperfusion injury, thereby supporting tissue survival [7]. Pentoxifylline, approved by the Food and Drug Administration (FDA) for intermittent claudication, is systemically used to enhance flap survival in surgical contexts [8]. However, its systemic administration is constrained by side effects, primarily gastrointestinal (nausea, vomiting, and diarrhoea) and cardiovascular (flushing, hypotension, tachycardia, and arrhythmia), which diminish patient compliance [9].
In light of these constraints, topical pentoxifylline is a viable alternative. Prior animal studies have indicated that parenteral pentoxifylline enhances flap survival, likely attributable to its vasodilatory, hemorheological, and anti-inflammatory properties [10–12]. This study aimed to examine the efficacy of topical pentoxifylline in flap surgery, positing that it could replicate these advantages without inducing systemic side effects. By facilitating neovascularisation, mitigating inflammation, and promoting wound healing, topical pentoxifylline can expedite flap autonomisation and enhance flap viability following pedicle division. This strategy may reduce complications associated with extended pedicle dependence, such as increased morbidity, thereby improving patient outcomes in reconstructive procedures.
Materials and methods
Ethical approval and animals
This study was approved by the Burdur Mehmet Akif Ersoy University Ethics Committee on Animal Research (Date: 11.09.2024, No: 127-1370). Sixty female Sprague-Dawley rats weighing between 250 and 300 g were sourced from the Animal Research Laboratory vivarium at Burdur Mehmet Akif Ersoy University for conducting this research. This study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals and the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines. The rats were individually housed in a temperature-controlled environment maintained at 21°C with a 12-hour light/dark cycle and were provided with unrestricted access to food and water. They were divided randomly into three groups: (1) Control Group (n = 24): 5 days + 7 days topical vaseline application group, (2) Experimental-1 Group (n = 24): 5 days topical pentoxifylline + 7 days topical vaseline application group, (3) Experimental-2 Group (n = 12): 5 days + 7 days topical pentoxifylline application group. On the fifth day, which was the pedicle cutting day, half of the rats (n = 12) from the control and experimental-1 groups were sacrificed for microangiography (n = 4) and histopathological analysis (n = 8).
Surgical procedure and postoperative care
The rats were anesthetised via intraperitoneal administration of ketamine (90 mg/kg) and xylazine (10 mg/kg). After shaving the dorsal region, the rats were positioned in a prone orientation on a board for the surgical procedure.
On day 0, the dorsal area was prepared antiseptically with povidone-iodine. A caudal-based skin flap, measuring 4 cm in width and 3 cm in length, was delineated at the level of the iliac crest, incised, and elevated to encompass the skin, subcutaneous tissue, and panniculus carnosus. The flap was then repositioned and sutured using 3-0 polypropylene. Until day 5, the control group received vaseline application twice daily, whereas the experimental-1 and experimental-2 groups were treated with 5% pentoxifylline cream. On day 5, the flap base was incised above the muscle fascia and subsequently sutured back with 3-0 polypropylene. Following pedicle severance, all groups were monitored for 7 days. During this observation period, topical vaseline was applied to the control and experimental-1 groups, whereas topical pentoxifylline was administered to the experimental-2 group until the 12th day.
Pentoxifylline 5% cream was prepared according to the recipe by Najafi et al. in 2018. Osrin (79 gms) and paraffin (8 gms) were mixed until a uniform mixture was obtained. The powder obtained from 400 mg pentoxifylline tablets (Pentox SR®, Abdi İbrahim Pharmaceutical Industry and Trade Inc.) (5 gms) was completely dissolved in distilled water (8 gms) using a magnetic stirrer. Than the drug was finally added to the cream base [13].
Macroscopic evaluation
On the 12th day, 12 rats from each group were euthanised, and flap photographs were analysed using ImageJ software (National Institute of Health, USA). The total flap, necrotic, and viable areas were quantified in sq mm and expressed as percentages.
Microangiographic evaluation
Four rats from each group were used in this experiment. The rats were anesthetised using ketamine (90 mg/kg) and xylazine (10 mg/kg) and positioned in the supine position. A 6 cm midline chest incision and sternotomy were performed to expose the heart and inferior vena cava. The vena cava was incised, and a 20-gauge catheter was inserted into the apex of the heart for cannulation. The vascular bed was flushed with 50 cc of 25,000 IU heparin in 500 cc saline until the organs appeared lighter. A mixture comprising 50 g barium sulfate, 5 g gelatine, and 70 cc saline was heated to 50°C, homogenised, and injected into the left ventricle. During injection, the coronary, liver, and mesenteric vessels turned white. Following the injection, the rats were refrigerated at +2°C for 4 h to solidify the contrast. Flaps, excised with 2 cm of surrounding tissue, were refrigerated and subsequently imaged using a mammography device (Viola DBT(Digital Breast Tomosynthesis), GMM(General Medical Merate) Group, Italy).
Histopathological evaluation
The flap tissue, including a 1 cm margin of the surrounding skin, was excised and fixed in formaldehyde for 24 h. The samples were subjected to hematoxylin-eosin, Masson’s trichrome, and immunohistochemical staining (VEGF, CD31). Histopathological scoring, as adapted from Mehrabani et al., was used to evaluate epithelialisation, fibrosis, edema, haemorrhage, angiogenesis, leukocyte infiltration, fibroblast count, and collagen degradation [14]. Angiogenesis was quantified by counting the capillary vessels in four randomly selected fields at x 40 magnification, and the mean value was calculated per section [15]. Immunohistochemical staining was scored on a scale of 0–3, based on the intensity under a x 20 objective.
Statistical analysis
The data analysis was conducted utilising Statistical Package for the Social Sciences (SPSS) version 19.00. The assumption of normality was verified through the Shapiro-Wilk test, with a significance level of p < 0.05. For the purpose of comparing groups, both one-way analysis of variance (ANOVA) and the Student’s t-test were employed, with statistical significance determined at p < 0.05. For the analysis of paired groups, the Student’s t-test was utilised, while the one-way ANOVA post hoc-Duncan test was applied to examine triple groups.
Results
Macroscopic results
After making a full-thickness incision at the flap base on day 5, 12 rats from each of the control, experimental-1, and experimental-2 groups were monitored for an additional 7 days, during which they received different topical treatments. On day 12, photographs were taken and analysed using the ImageJ software (National Institute of Health, USA). The areas of the total flap, viable tissue, and necrotic tissue were measured in sq mm and calculated. The viable tissue areas in the experimental-1 and experimental-2 groups were significantly larger than those in the control group (p = 0.009 and p = 0.003, respectively), with the experimental-2 group showing complete viability (Figure 1).
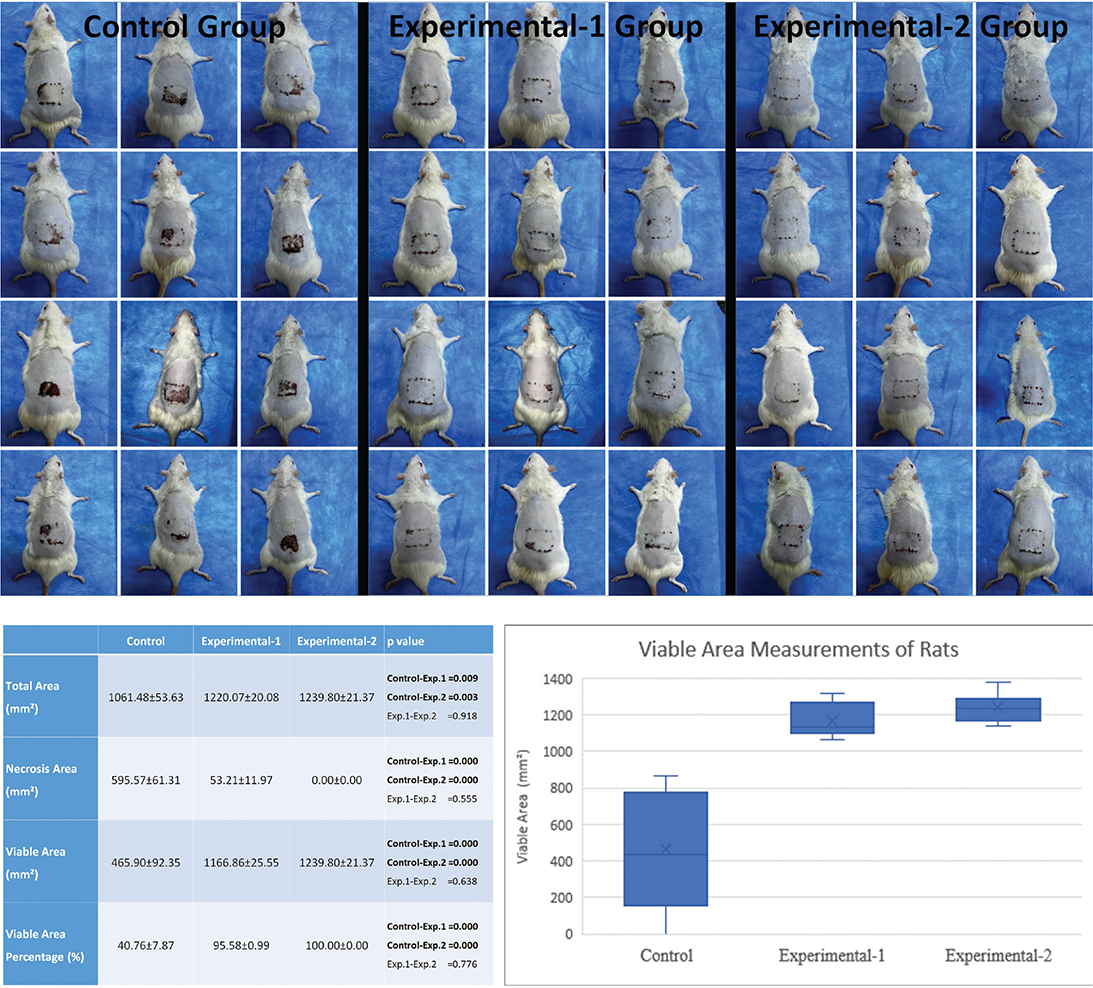
Figure 1. Images of flaps in the control, experimental-1, and experimental-2 groups on the 12th day after flap elevation and 7th day after pedicle transection. A graph and table showing the results of the viable area measurements of the experimental groups at the bottom (Data are given as mean ± standard error (SE). One-Way ANOVA post hoc-Duncan test was used. P < 0.05: Statistically significant.). ANOVA: analysis of variance.
Microangiographic results
On day 5, microangiographic imaging revealed no new vessel growth from the surrounding tissues to the flap body in the control group. In contrast, the experimental-1 group exhibited new vessel formation advancing from the upper and lateral edges of the flap, indicating early angiogenic activity stimulated by pentoxifylline. By day 12, two of the four rats in the control group still showed no new vessel progression, whereas the others displayed minimal growth. Both the experimental-1 and experimental-2 groups demonstrated significant vessel formation from the upper and lateral edges, with the experimental-2 group showing greater vascularisation than that in experimental-1 (Figure 2).
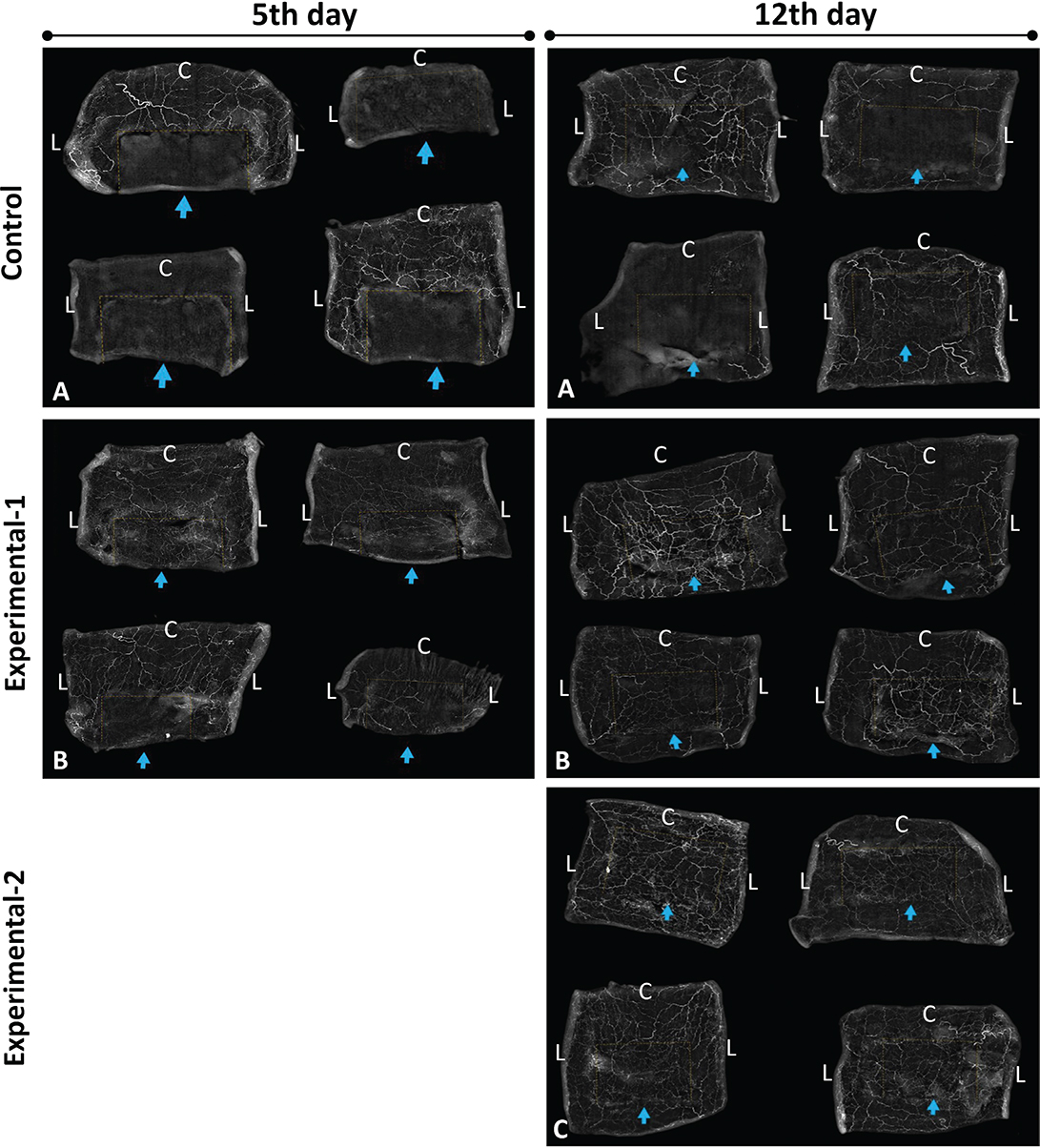
Figure 2. Contrast-enhanced microangiography images were taken on the 5th day of the experiment. (A) Control group; (B) Experiment-1 group. Contrast-enhanced microangiography images were taken on the 12th day of the experiment. (A) Control group (B) Experiment-1 group (C) Experiment-2 group. (Dashed lines: flap boundaries; arrow: base edge of the flap, C: Cranial side, L: Lateral side).
Histopathological results
On day 5, sections from the lateral and cranial flap regions in both the control and experimental-1 groups were assessed for epithelialisation, fibrosis, edema, haemorrhage, angiogenesis, leukocyte infiltration, fibroblast count, and collagen degradation (Table 1). Across all groups, the lateral regions consistently demonstrated more significant improvements than those in the cranial regions. The experimental-1 group showed enhanced epithelialisation, increased fibrosis, greater angiogenesis, and reduced inflammation compared to the control group. In the control group, inflammatory cells were denser and more widespread, whereas in experimental-1 group, they were milder and more localised (Figure 3). Although epithelialisation and fibrosis were incomplete in both groups, they were more advanced in experimental-1 group.
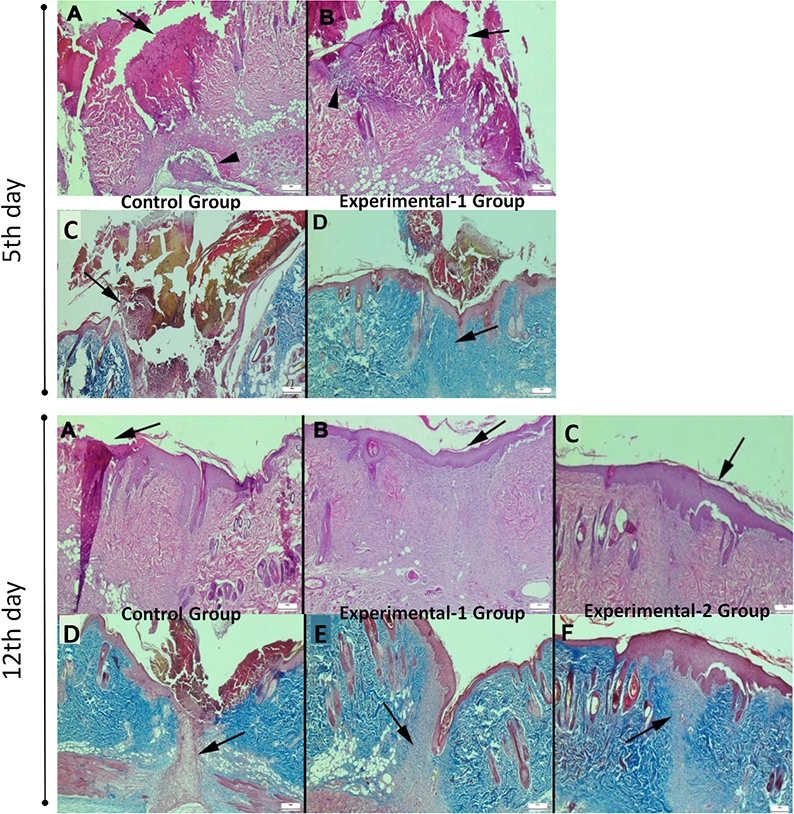
Figure 3. On the 5th day of the experiment, histopathological examination of the control and experimental-1 groups (bars = 200 µm) (A) Significant necrosis and crust formation in the flap in the control group (arrow), (H&E) (B) In the Experiment-1 group, necrosis and crust formations (arrow) are observed less at the cranial edge of the flap compared to the control, (H&E) (C) Appearance of very mild and immature fibrosis in the flap in the control group (arrow), (Masson’s trichrome) (D) Mature blue coloured fibrosis formation in Experiment-1 group (arrow), (Masson’s trichrome). On the 12th day of the experiment, histopathological examination of the control, experimental-1, and experimental-2 groups (bars = 200 µm). (A) Necrosis and crust formation in the flap in the control group (arrow), (H&E) (B) Epithelialisation (arrow) and fully formed fibrosis at the cranial edge of the flap in the experiment-1 group, (H&E) (C) In the experiment-2 group, significant epithelialization (arrow) and fibrosis, (H&E) D) In the control group, incompletely mature, pink-stained fibrosis (arrow), (Masson’s trichrome) (E) Mature, blue-stained fibrosis in experiment-1 group (arrow), (Masson’s trichrome) (F) Mature, blue-stained fibrosis in experiment-2 group (arrow), (Masson’s trichrome).
On day 12, all groups were assessed using the same histopathological criteria as described above. Healing was more evident in the lateral regions than in the cranial regions. The experimental-1 and experimental-2 groups exhibited significantly enhanced epithelialisation, fibrosis, and angiogenesis, with a marked reduction in inflammation compared to the control group. In these experimental groups, epithelialisation and fibrosis were fully developed, whereas the control group showed incomplete formation in several areas (Table 2). Inflammatory cells were notably present in the control group but were significantly reduced in both the experimental groups (Figure 3).
Immunohistochemical results
Biopsy samples from all groups on days 5 and 12 were stained with VEGF and CD31 antibodies to assess the expression of angiogenic markers. In all groups, the cranial regions exhibited less pronounced expression than the lateral regions. On day 5, the experimental-1 group showed significantly higher VEGF and CD31 expression than the control group (Table 3) (Figure 4), highlighting the early angiogenic benefits of pentoxifylline. By day 12, the experimental-2 group demonstrated the highest VEGF and CD31 expression, followed by experimental-1 group, both exceeding the control group (Figure 4).
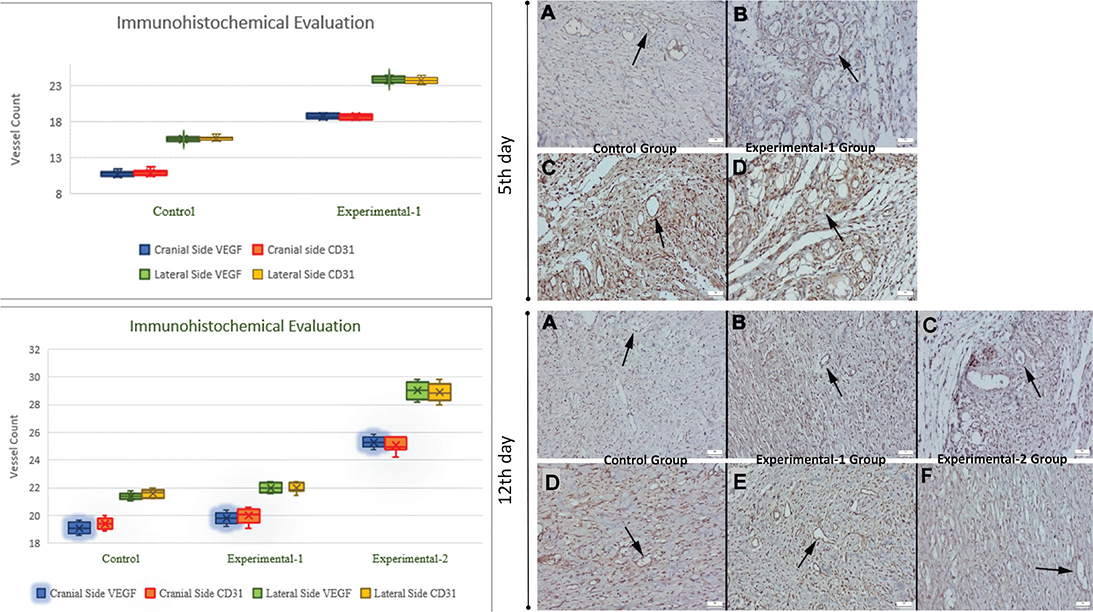
Figure 4. On the 5th day of the experiment, CD31 and VEGF immunohistochemical findings of the control and experimental-1 groups. (Streptavidin biotin peroxidase method) (Bars = 50 µm) (A) Few capillary vessel formations in the control group (arrow), (CD31) (B) Increased capillary vessel formation in experiment-1 group (arrow), (CD31). (C) Few capillary vessel formations in the control group (arrow) (VEGF). (D) Increased capillary vessel formation in the experiment-1 group (arrow), (VEGF). On the 12th day of the experiment, CD31 and VEGF immunohistochemical findings of the control, experimental-1, and experimental-2 groups. Streptavidin biotin peroxidase method. (Bars = 50 µm) (A) Few capillary vessel formations in the control group (arrow), (CD31), (B) Increased capillary vessel formation in the experiment-1 group (arrow), (CD31), (C) Increased capillary vessel formation in the experiment-2 group (arrow), (CD31), (D) Few capillary vessel formations in the control group (arrow), (VEGF), (E) Increased capillary vessel formation in the experiment-1 group (arrow), (VEGF), (F) Increased capillary vessel formation in the experiment-2 group (arrow), (VEGF). In upper graph; VEGF and CD31 immunohistochemical staining vessels counts on the 5th day of the experiment. (The Experimental-1 Group was significantly higher than the Control group (p < 0.001), and the lateral sides of the flaps were higher than the cranial sides in both groups (p < 0.05)). In lower graph; VEGF and CD31 immunohistochemical staining vessels counts on the 12th day of the experiment. (The Experimental-2 Group was significantly higher than the Control group and Experimental-1 group (p < 0.001), and the lateral sides of the flaps were higher than the cranial sides in all groups (p < 0.05)).
Statistical results
On day 12, photographs of the flaps from all groups were analysed to assess the total, viable, and necrotic areas (for detailed results and measurements of the viable areas see Figure 1. Histopathological assessments of biopsy specimens obtained from the lateral and cranial regions on days 5 and 12 were performed, and the corresponding p-values are detailed in Tables 1 and 2. Statistical analyses of the VEGF and CD31 staining scores and vessel counts on days 5 and 12 are shown in Table 3 and Figure 4. All comparisons demonstrated significant improvements in the experimental groups compared to the control group (p < 0.05), particularly in terms of viable area, angiogenesis, epithelialisation, and reduced inflammation.
Discussion
Pedicled skin flaps (distant or regional) requiring the second stage for pedicle division are essential for reconstructing defects when local or free flaps are not feasible [1]. Following their transfer to the recipient site, neovascularisation occurs, allowing the flap to sustain itself independently of its pedicle, a process known as autonomisation. Enhancing neovascularisation can reduce the duration of pedicle dependence, thereby minimising functional impairments and morbidity associated with prolonged attachment to the donor site until the second surgery, when the pedicle is severed. This study examined whether the application of topical pentoxifylline promotes neovascularisation and expedites autonomisation in a caudal-based dorsal skin flap model in rats, potentially offering an innovative strategy to improve the outcomes of flap surgery. Although the experimental model used in the present study does not fully replicate some pedicled flaps (e.g. interpolation flaps) as defined clinically, it was designed to simulate the autonomisation process with neoangiogenesis observed in pedicled skin flaps.
The dorsal skin flap model in rats was developed to simulate the pedicled flaps required for pedicle division, employing a flap measuring 4 cm in width and 3 cm in length, based on the iliac crest landmark [16]. These dimensions were chosen to assess vascularisation and histological changes following the initiation of neovascularisation [17, 18] and subsequent to pedicle division on day 5, ensuring the presence of sufficient viable tissue for analysis without complete necrosis.
The effect of parenteral pentoxifylline on flap survival has been demonstrated in numerous animal studies [10–12]. These research involving parenteral pentoxifylline has primarily focused on evaluating changes in flap vascularity by measuring blood flow through the pedicle. In contrast, the present study examined the effect of new vascularisation that develops around the flap following pedicle separation. There are studies in the literature on the effects of topical pentoxifylline on wound healing in burn and pressure ulcer [13, 19]. However, there are no studies in the literature on the effect of topical pentoxifylline on flap survival. Consequently, this study diverges from the existing literature in these two specific aspects.
The experimental model revealed that the application of topical pentoxifylline significantly improved flap survival in rats. Following the division of the pedicle on day 5 and a subsequent 7-day observation period, the viable flap area in the experimental-1 group (pentoxifylline for 5 days, vaseline for 7 days) and the experimental-2 group (pentoxifylline for 12 days) was significantly greater than that in the control group (vaseline for 12 days) (p < 0.05). Contrast microangiography conducted on day 5 indicated the initiation of vascularisation in the experimental groups, particularly from the lateral edges of the flap, whereas no such progression was observed in the control group. By day 12, vascularisation was markedly enhanced in both the experimental-1 and experimental-2 groups compared to the control group, with the experimental-2 group demonstrating superior vascularisation, suggesting that extended application of pentoxifylline optimises angiogenic effects. Histopathological and immunohistochemical analyses further corroborated the role of pentoxifylline in augmenting angiogenesis, reducing inflammation, and promoting tissue repair, thereby supporting its potential to expedite autonomisation. Biopsies conducted on days 5 and 12 indicated that the lateral edges of the flap exhibited superior healing compared to the cranial edges across all groups; this may be attributed to the proximity of the lateral edges to the flap base [20]. The lateral regions consistently demonstrated enhanced vascularisation and epithelialisation, along with reduced inflammation, underscoring the significance of regional vascular dynamiInterleukin-10
Vessel counts were significantly elevated in the experimental-1 group compared to the control group on day 5 (p < 0.001). By day 12, the experimental-2 group demonstrated the highest vessel counts, exceeding those of both experimental-1 group and the control group (p < 0.001). These findings align with those of Pedretti et al., who documented reduced necrosis and increased vessel numbers and VEGF expression in flaps treated with pentoxifylline, with 5-day treatments proving more effective than shorter durations [23]. Similarly, Seo et al. observed enhanced angiogenesis in osteoradionecrosis models following the oral administration of pentoxifylline [24]. The elevated expression of Vascular Endothelial Growth Factor (VEGF) and Cluster of Differentiation 31 (CD31) in the experimental groups, particularly in experimental-2 group on day 12, further substantiates the angiogenic advantages of pentoxifylline, which is likely attributable to its capacity to improve microcirculation and stimulate endothelial cell proliferation.
On day 12, the fibrosis scores for the cranial flap edges were recorded as 1.30 ± 0.15 for the control group, 2.60 ± 0.16 for the experimental-1 group, and 1.90 ± 0.23 for the experimental-2 group. The corresponding fibroblast counts were 22.80 ± 0.61 for the control group, 27.90 ± 0.34 for the experimental-1 group, and 21.30 ± 0.39 for the experimental-2 group. Although pentoxifylline is recognised for its ability to reduce myofibroblasts and fibrosis [25–27], fibrosis was greater in the experimental-1 group than in the control group and experimental-2 group. This discrepancy may be attributed to variations in treatment duration, which could influence the fibroblast activity or collagen deposition. Fang et al. have demonstrated that pentoxifylline inhibits fibroblast mitosis and collagen production in a dose-dependent manner in vitro and reduces silica-induced peritoneal fibrosis in vivo (p < 0.05) [25]. The observed differences in fibrosis scores suggest that the extended application of pentoxifylline may more effectively modulate fibroblast activity, indicating the need for further research into optimal dosing regimens.
The findings of this study indicate that topical pentoxifylline may serve as a valuable adjunct in flap surgery by reducing necrosis, enhancing angiogenesis, and improving the wound healing process. By accelerating autonomisation, the duration of pedicle dependence may be shortened, thereby reducing patient morbidity and improving the functional outcomes. Compared to systemic pentoxifylline, which is constrained by gastrointestinal and cardiovascular side effects [9], topical application provides a targeted approach with potentially fewer adverse effects, thus enhancing patient compliance.
Limitations
The use of rat skin in this study, which differs from human skin in terms of structure, metabolism, and healing capacity, presents limitations for direct clinical applications [28, 29]. Human skin is characterised by a thicker epidermis and slower healing processes, necessitating clinical trials to validate the efficacy of topical pentoxifylline in humans. Clinical studies are also needed to understand differences in people’s age, additional diseases, skin thickness and quality, and the application area of the cream on the body. The 12-day experimental period, including a 7-day post-pedicle division observation, may not adequately capture the remodelling phase of wound healing, including long-term fibrosis or wound maturation. Extended studies could provide insights into the effects of pentoxifylline on these processes. The magistral pentoxifylline cream used in this study may be less effective than advanced formulations, such as nanoparticle-based systems, which could enhance drug delivery and efficacy [30]. The effectiveness of the medicine may vary depending on the preparation method of the magistral medicine and other substances that may be used in the cream base, and this needs to be standardised in studies. Future research should investigate optimised topical formulations, longer follow-up periods, and human clinical trials to confirm these findings and establish their practical applications in reconstructive surgery.
Conclusion
This study demonstrates that the application of topical pentoxifylline significantly enhances the outcomes of two-stage random-pedicled skin flap surgery by expediting autonomisation and promoting tissue repair in a rat model. When applied to caudal-based dorsal skin flaps, pentoxifylline markedly increased angiogenesis, as evidenced by higher vessel counts and enhanced expression of angiogenic markers in the treated groups compared to the control group. Beyond angiogenesis, pentoxifylline improves wound healing by enhancing epithelialisation and mature collagen formation. These findings underscore the multifaceted role of pentoxifylline in enhancing vascular supply, minimising necrosis, and accelerating tissue integration, which are critical for successful flap surgery outcomes.
The potential application of topical pentoxifylline in clinical practice appears promising, as it circumvents systemic side effects, thereby enhancing patient adherence. Future clinical trials and investigations into advanced formulations, such as nanoparticle-based creams, could substantiate these benefits in human subjects. This may establish topical pentoxifylline as a valuable asset in reconstructive surgery, optimising flap survival, reducing complications, and expediting patient recovery.
References
[1] Pederson WC, Lister GD. Local and regional flap coverage of the hand. In: Wolfe SW, editor. Green’s operative hand surgery. Philadelphia, PA: Elsevier Churchill; 2011. p. 1648–1713.
[2] Pang CY, Neligan PC. Flap pathophysiology and pharmacology. In: Gurtner GC, Neligan PC, editors. Plastic surgery e-book. Volume I. Oxford: Elsevier Health Sciences; 2017. p. 573–586.
[3] Pang CY, Forrest CR, Morris SF. Pharmacological augmentation of skin flap viability: a hypothesis to mimic the surgical delay phenomenon or a wishful thought. Ann Plast Surg. 1989;22(4):293–306. https://doi.org/10.1097/00000637-198904000-00003
[4] Ward A, Clissold SP. Pentoxifylline. A review of its pharmacodynamic and pharmacokinetic properties, and its therapeutic efficacy. Drugs. 1987;34(1):50–97. https://doi.org/10.2165/00003495-198734010-00003
[5] Angelkort B, Maurin N, Boateng K. Influence of pentoxifylline on erythrocyte deformability in peripheral occlusive arterial disease. Curr Med Res Opin. 1979;6(4):255–258. https://doi.org/10.1185/03007997909109432
[6] Zhang M, Xu YJ, Mengi SA, et al. Therapeutic potentials of pentoxifylline for treatment of cardiovascular diseases. Exp Clin Cardiol. 2004;9(2):103–111.
[7] Ciuffetti G, Mercuri M, Ott C, et al. Use of pentoxifylline as an inhibitor of free radical generation in peripheral vascular disease. Results of a double-blind placebo-controlled study. Eur J Clin Pharmacol. 1991;41(6):511–515. https://doi.org/10.1007/BF00314976
[8] Aviado DM, Dettelbach HR. Pharmacology of pentoxifylline, a hemorheologic agent for the treatment of intermittent claudication. Angiology. 1984;35(7):407–417. https://doi.org/10.1177/000331978403500703
[9] Liang X, Wang Y, Zhao C, et al. Systematic review the efficacy and safety of cilostazol, pentoxifylline, beraprost in the treatment of intermittent claudication: a network meta-analysis. PLoS One. 2022;17(11):e0275392. https://doi.org/10.1371/journal.pone.0275392
[10] Roth AG, Briggs PC, Jones EW, et al. Augmentation of skin flap survival by parenteral pentoxifylline. Br J Plast Surg. 1988;41(5):515–520. https://doi.org/10.1016/0007-1226(88)90010-0
[11] Takayanagi S, Ogawa Y. Effects of Pentoxifylline on flap survival. Plast Reconstr Surg. 1980;65(6):763–767. https://doi.org/10.1097/00006534-198006000-00008
[12] Topalan M, Arıncı A, Erer M, et al. The effect of parenteral pentoxifylline therapy on skin flap survival. Eur J Plast Surg. 1993;16:33–35. https://doi.org/10.1007/BF00192704
[13] Najafi E, Ahmadi M, Mohammadi M, et al. Topical pentoxifylline for pressure ulcer treatment: a randomised, double-blind, placebo-controlled clinical trial. J Wound Care. 2018;27(8):495–502. https://doi.org/10.12968/jowc.2018.27.8.495
[14] Mehrabani M, Najafi M, Kamarul T, et al. Deferoxamine preconditioning to restore impaired HIF-1α-mediated angiogenic mechanisms in adipose-derived stem cells from STZ-induced type 1 diabetic rats. Cell Prolif. 2015;48(5):532–549. https://doi.org/10.1111/cpr.12209
[15] Payette JR, Kohlenberg E, Leonardi L, et al. Assessment of skin flaps using optically based methods for measuring blood flow and oxygenation. Plast Reconstr Surg. 2005;115(2):539–546. https://doi.org/10.1097/01.PRS.0000148415.54546.CA
[16] McFarlane RM, Deyoung G, Henry RA. The design of a pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965;35:177–182. https://doi.org/10.1097/00006534-196502000-00007
[17] Ordman LJ, Gillman T. Studies in the healing of cutaneous wounds. 3. A critical comparison in the pig of the healing of surgical incisions closed with sutures or adhesive tape based on tensile strength and clinical and histological criteria. Arch Surg. 1966;93(6):911–928. https://doi.org/10.1001/archsurg.1966.01330060055003
[18] Velander E. Vascular changes in tubed pedicles. An animal experimental study. Acta Chir Scand Suppl. 1964;322(Suppl.):1–142.
[19] Yucel B, Coruh A, Deniz K. Salvaging the zone of stasis in burns by Pentoxifylline: an experimental study in rats. J Burn Care Res. 2019;40(2):211–219. https://doi.org/10.1093/jbcr/irz005
[20] Lancien U, Charbonnier B, Weiss P, et al. Rat perforator and skin vessels vascular mapping: an original anatomical study about 140 vessels and literature review. J Surg Res. 2023;288:298–308. https://doi.org/10.1016/j.jss.2023.03.015
[21] Abdel-Salam OM, Baiuomy AR, El-Shenawy SM, et al. The anti-inflammatory effects of the phosphodiesterase inhibitor pentoxifylline in the rat. Pharmacol Res. 2003;47(4):331–340. https://doi.org/10.1016/S1043-6618(03)00002-1
[22] Marques LJ, Zheng L, Poulakis N, et al. Pentoxifylline inhibits TNF-alpha production from human alveolar macrophages. Am J Respir Crit Care Med. 1999;159(2):508–511. https://doi.org/10.1164/ajrccm.159.2.9804085
[23] Pedretti S, Rena CL, Orellano LAA, et al. Benefits of pentoxifylline for skin flap tissue repair in rats. Acta Cir Bras. 2020;35(11):e301105. https://doi.org/10.1590/acb351105
[24] Seo MH, Myoung H, Lee JH, et al. Effects of pentoxifylline and tocopherol on an osteoradionecrosis animal model. J Craniomaxillofac Surg. 2020;48(7):621–631. https://doi.org/10.1016/j.jcms.2020.02.008
[25] Fang CC, Lai MN, Chien CT, et al. Effects of pentoxifylline on peritoneal fibroblasts and silica-induced peritoneal fibrosis. Perit Dial Int. 2003;23(3):228–236. https://doi.org/10.1177/089686080302300303
[26] Lin SL, Chen RH, Chen YM, et al. Pentoxifylline attenuates tubulointerstitial fibrosis by blocking Smad3/4-activated transcription and profibrogenic effects of connective tissue growth factor. J Am Soc Nephrol. 2005;16(9):2702–2713. https://doi.org/10.1681/ASN.2005040435
[27] Paulis G, Barletta D, Turchi P, et al. Efficacy and safety evaluation of pentoxifylline associated with other antioxidants in medical treatment of Peyronie’s disease: a case-control study. Res Rep Urol. 2016;8:1–10. https://doi.org/10.2147/RRU.S97194
[28] Bayramiçli M. Laboratuar sıçanının anatomisi. Ameliyat. Bayramiçli M. Deneysel mikrocerrahi. Temel Araştırma, Doku ve Organ Nakil Modelleri Argos iletişim Hizmetleri. 2005;119–184:75–98.
[29] Scherer-Pietramaggiori SS, Pietramaggiori G, Orgill DP. Skin graft. In: Gurtner GC, Neligan PC, editors. Plastic surgery e-book. Volume I. Oxford: Elsevier Health Sciences; 2017. p. 573–586.
[30] Wang W, Lu KJ, Yu CH, et al. Nano-drug delivery systems in wound treatment and skin regeneration. J Nanobiotechnol. 2019;17(1):82. https://doi.org/10.1186/s12951-019-0514-y