SYSTEMATIC REVIEW
Schwannomas of the deep motor branch of the ulnar nerve: a systematic review of the literature with an additional case report and anatomical analysis
Filippo Pantaleonia  , Mariia Kotovab
, Mariia Kotovab  , Marta Montanaria
, Marta Montanaria  , Marco Passiatorec
, Marco Passiatorec  , Davide Gravinaa
, Davide Gravinaa  , Paolo Boccolaria
, Paolo Boccolaria  , Danilo Donatid
, Danilo Donatid  , Marta Starnonie
, Marta Starnonie  and Andrea Leti Acciaroa
and Andrea Leti Acciaroa 
aDepartment of Hand Surgery and Microsurgery, University Hospital of Modena, Modena, Italy; bInstitute of Traumatology and Orthopedics of NAMS of Ukraine, Kyiv, Ukraine; cDepartment of Medical and Surgical Specialties, Radiological Sciences and Public Health, University of Brescia ASST Spedali Civili, Brescia, Italy; dPhysical Therapy and Rehabilitation Unit, University Hospital of Modena, Modena, Italy; ePlastic Surgery and Burn Center, Hospital of Parma, Parma, Italy
ABSTRACT
Purpose: Schwannomas originating from the deep motor branch of the ulnar nerve (DMBUN) are exceptionally rare, presenting with variable and often heterogeneous clinical manifestations depending on their precise anatomical location. While these tumors have been described in the literature, reports specifically involving the DMBUN are exceedingly limited, rendering postoperative prognostication challenging. This study aims to systematically review the existing literature and present a novel case, with the objective of delineating anatomical characteristics and clarifying potential postoperative outcomes.
Methods: A systematic literature search was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, using the MEDLINE and Scopus databases. Data regarding patient demographics, clinical presentation, anatomical localization, and surgical outcomes were extracted and analyzed.
Results: Seven articles met the inclusion criteria. A total of nine tumors were initially identified as schwannomas of the DMBUN over a 73-year period; however, only five cases – including the newly reported one – were confirmed. The median patient age was 59.6 years; complete recovery was achieved in three cases, with time to functional improvement ranging from 4 to 6 months.
Conclusion: In this limited series, available data suggest that accurate preoperative imaging and careful intraoperative microsurgical dissection may lead to favorable outcomes, with complete recovery achievable in schwannomas of the DMBUN. No clear association was observed between postoperative nerve function and tumor size, patient age, or time since symptom onset.
KEYWORDS: Schwannoma; neurinoma; deep motor branch; ulnar nerve; peripheral nerve tumor; microsurgery
Citation: Journal of Plastic Surgery and Hand Surgery 2026; 61: 65–71. DOI: https://doi.org/10.2340/jphs.v61.45668.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 21 September 2025; Accepted: 13 February 2026; Published: 27 March 2026
CONTACT: Mariia Kotova, MD uaot@ukr.net Institute of Traumatology and Orthopedics of NAMS of Ukraine, Bulvarno-Kudriavska St, 27, Kyiv, Ukraine
Competing interests and funding: The authors report that there are no competing interests to declare.
The authors declare no conflicts of interest.
The authors received no financial support for the research, authorship, and/or publication of this article.
Introduction
Benign peripheral nerve sheath tumors (BPNSTs) account for less than 5% of all upper extremity neoplasms and are frequently misdiagnosed as other soft-tissue masses, such as ganglion cysts, myxomas, or fibromas. Among BPNSTs, schwannomas (neurilemmomas) are the most common, followed by neurofibromas, lipofibromatous hamartomas, and perineuriomas. Schwannomas arise from the pathological proliferation of myelinating Schwann cells, typically due to biallelic inactivation of a tumor suppressor gene on chromosome 22 encoding the merlin protein. Although genetic conditions such as neurofibromatosis type 2 and schwannomatosis (previously referred to as neurofibromatosis type 3) may predispose to multiple tumors, most schwannomas are solitary.
Myelinating Schwann cells envelop axons and facilitate saltatory conduction. As a result, schwannomas typically grow adjacent to nerves without infiltrating axons – unlike neurofibromas, which originate from non-myelinating Schwann cells and are intraneural. This distinction has significant implications for both clinical presentation and surgical management. Schwannomas are usually round or oval and grow eccentrically along the nerve, allowing en bloc excision while preserving fascicular integrity. In contrast, neurofibromas are fusiform, centrally located within the nerve, and often require nerve resection and reconstruction. Although schwannomas are benign, malignant transformation is extremely rare [1].
Clinically, patients usually present with a slow-growing, painless mass. Compression may lead to paresthesia or numbness in the sensory distribution of the affected nerve or discomfort upon palpation, often eliciting a positive Tinel’s sign. Motor symptoms, such as weakness or muscle atrophy, may also occur. Schwannomas are more frequently found on the flexor aspect of the forearm but can arise in any peripheral nerve.
Ultrasound is the preferred initial diagnostic tool, typically revealing a well-encapsulated, hypoechoic mass with minimal vascularity, mobile with respect to surrounding tissues. Magnetic resonance imaging (MRI) remains the gold standard, showing a hyperintense signal on T2-weighted images and iso- or slightly hyperintense signal on T1-weighted sequences. The presence of a ‘target sign’, suggestive of an ancient schwannoma, represents a distinctive pattern characterized by a peripheral hyperintense rim and a central hypo- or iso-intense focus, reflecting differences in tissue composition between the myxomatous outer layers and the fibrocollagenous inner core [2].
Surgical excision is recommended due to the lesion’s progressive growth and the inability to confirm diagnosis without histopathology. Needle biopsy is contraindicated due to the high risk of axonal injury and subsequent functional impairment.
It is essential to inform patients of the potential for transient nerve dysfunction lasting 6 to 12 months after schwannoma removal. In the case of neurofibromas, nerve resection and microsurgical grafting may be necessary. For critical nerves or main trunks, if a neurofibroma is identified intraoperatively, excision may be reconsidered in favor of preserving nerve function.
Microsurgical techniques are employed to optimize outcomes. After longitudinal epineurial incision, schwannomas can be dissected and removed en bloc by isolating and protecting unaffected axons. Local recurrence is rare. Histologically, schwannomas exhibit two characteristic patterns: Antoni type A (high cellularity) and Antoni type B (low cellularity). Immunohistochemically, S100 protein is a sensitive marker for identifying Schwann cell tumors [3].
Although schwannomas may involve any peripheral nerve, their occurrence within the deep motor branch of the ulnar nerve (DMBUN) is exceedingly rare. The DMBUN plays a crucial role in hand function, innervating most of the intrinsic muscles, including the hypothenar group, interossei, the two ulnar-sided lumbricals, the deep head of the flexor pollicis brevis, and the adductor pollicis [4]. Tumors affecting this branch can impair fine motor coordination and grip strength.
Despite several reviews having already examined neurilemmomas of the upper limb [5, 6], the exceptional rarity of schwannomas of the DMBUN means that the existing literature consists primarily of isolated case reports, with no prior systematic review and limited evidence available to guide optimal surgical management.
The primary aim of this study is to conduct a systematic review of the literature, supplemented by a new case report, in order to map all documented anatomical sites of tumor origin. A secondary objective is to elucidate preoperative clinical features, prognostic factors, and optimal surgical strategies for these rare tumors.
Materials and methods
This study was conducted as a systematic review with an accompanying case report, in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [7]. It was designed to identify all published cases of schwannomas involving the DMBUN. A comprehensive literature search was performed using two major electronic databases: MEDLINE via Ovid (from 1946 to April 30, 2025) and Scopus (all years available). The search strategy employed a combination of Medical Subject Headings (MeSH) and free-text terms.
To capture relevant studies, search terms included variations of ‘schwannoma’, such as ‘neurilemmoma’, ‘neurilemoma’, ‘neurinoma’, ‘nerve sheath neoplasm’, ‘nerve sheath tumour’, ‘peripheral nerve tumour’, and ‘benign peripheral nerve sheath tumour’, combined with anatomical terms such as ‘ulnar nerve’, ‘deep motor branch’, ‘deep branch’, ‘motor branch’, ‘deep motor branch of the ulnar nerve’, ‘DMBUN’, ‘Guyon canal’, ‘Guyon’s canal’, ‘hypothenar’, ‘interosseous’, and ‘adductor pollicis’. These were further refined with location-specific terms including ‘hand’, ‘palm’, ‘palmar’, and ‘wrist’. The three concept groups were linked using the Boolean operator ‘AND’.
Eligible studies included original case reports, case series, or cohort studies that reported schwannomas involving the DMBUN, published in English and available in full text, and that provided detailed information on patient demographics, clinical presentation, diagnostic workup, treatment modality, and outcomes.
Studies were excluded if they were review articles without original cases, lacked adequate clinical or surgical detail, focused solely on schwannomas of the main trunk of the ulnar nerve without deep motor branch involvement, were published in languages other than English without accessible translations, or were conference abstracts, editorials, or duplicate publications without primary case data.
Two independent reviewers screened all titles and abstracts to assess potential eligibility. Full-text articles of selected studies were retrieved and evaluated for final inclusion based on the aforementioned criteria. Disagreements were resolved through discussion, and if consensus could not be reached, a third reviewer was consulted.
Data were extracted using a standardized form. For each case, the following variables were collected: patient age and sex, presenting symptoms and clinical findings, diagnostic methods including imaging and electromyography, tumor characteristics such as size and anatomical location within the DMBUN, surgical approach and technique, histopathological features, postoperative outcomes and complications, and follow-up duration.
The methodological quality of the included case reports was assessed using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Case Reports [8]. Each study was evaluated across eight domains: reporting of patient demographics, clinical history and presentation, current clinical status, diagnostic process, details of the intervention, clinical outcomes following intervention, documentation of adverse events, and the presence of key clinical insights or lessons.
Due to the rarity of DMBUN schwannomas and the expected heterogeneity across included studies, a narrative synthesis approach was adopted. Findings were summarized qualitatively according to patient characteristics, clinical features, diagnostic and surgical approaches, and postoperative outcomes.
Artificial intelligence tools were employed solely to assist with literature organization, reference formatting, and optimization of manuscript structure. No AI was used in the processes of data collection, clinical interpretation, or content generation. All clinical judgments, analyses, and conclusions represent the original work of the authors.
This study involved no animal subjects. All procedures involving human participants were conducted in accordance with the ethical standards of the institutional research committee and the 1964 Declaration of Helsinki and its later amendments or comparable ethical guidelines.
New case report
In addition to the systematic review, we present a novel case of a schwannoma arising from the DMBUN, treated at our institution. Written informed consent was obtained from the patient for the publication of clinical details and images related to the case.
Results
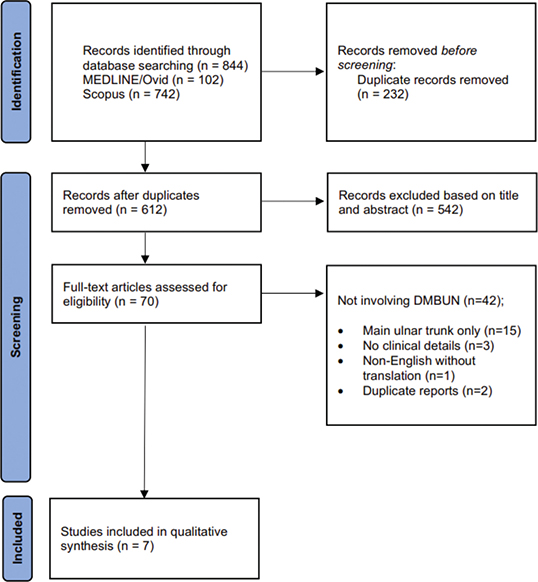
The systematic literature search retrieved 844 records through database queries (MEDLINE: 102; Scopus: 742). After removing 232 duplicates, 612 records remained for title and abstract screening. Of these, 542 were excluded as irrelevant, leaving 70 full-text articles assessed for eligibility.
The primary reasons for exclusion included the absence of DMBUN involvement (n = 42), exclusive involvement of the main ulnar trunk (n = 15), insufficient clinical details (n = 3), non-English language without available translation (n = 1), and duplicate reports (n = 2).
Ultimately, seven studies reporting eight cases of schwannomas involving the DMBUN met the inclusion criteria and were incorporated into the qualitative synthesis, spanning a period of 73 years (1952–2022) (Figure 1) [9–15]. Including our newly described case, a total of nine cases were analyzed (Table 1) and anatomically mapped (Figure 2).
| Author | Year | Age, gender and side | Presenting symptoms | Outcome of surgery | Considerations | |
| 1 | Jenkins A [9] | 1952 | 51, F, left hand | Intrinsic minus hand | Partial recovery after 6 months | Probably neuroma of the ulnar nerve proximal to VT1 trunk |
| 2 | Cavanagh NP [10] | 1977 | 7, F, right hand | Loss of finger adduction | Partial recovery after 4 months | Neurofibroma of DMBUN at VT1 trunk division |
| 3 | < 1, F, right hand | Swelling of hypothenar eminence | Minimal recovery after 13 years | Probably plexiform neurofibroma of ulnar nerve in Guyon’s canal | ||
| 4 | Bowers WH [11] | 1979 | 50, M, right hand | Intrinsic minus hand and pain in hypothenar eminence | Minimal recovery after 5 months | Probably intraneural ganglion cyst of DMBUN between VT1 and VT2 |
| 5 | Sarangapani K [12] | 1982 | 63, M, left hand | Ache over hypothenar eminence and tingling of ring and little fingers | Full recovery | Schwannoma of DMBUN proximal to VT1 trunk |
| 6 | Oguma H [13] | 1999 | 61, M, left hand | Acute tenderness in palm | No recovery | Schwannoma of DMBUN distal to HT2 trunk |
| 7 | Moran J [14] | 2021 | 78, M, right hand | Giant mass in first web space | No recovery | Schwannoma of AD or DI1 branches |
| 8 | Giunta G [15] | 2022 | 35, F, right hand | Moderate pain | Full recovery after 6 months | Schwannoma of VT3 trunk |
| 9 | Pantaleoni F | 2025 | 61, F, right hand | Hypothenar mass and reduced hand dexterity | Full recovery after 5 months | Schwannoma of DMBUN at VT2 and VT3 trunks divisions |
| VT: Vertical Trunk; HT: horizontal trunk; AD: adductor, DI1: first dorsal interosseous; DMBUN: deep motor branch of the ulnar nerve. | ||||||
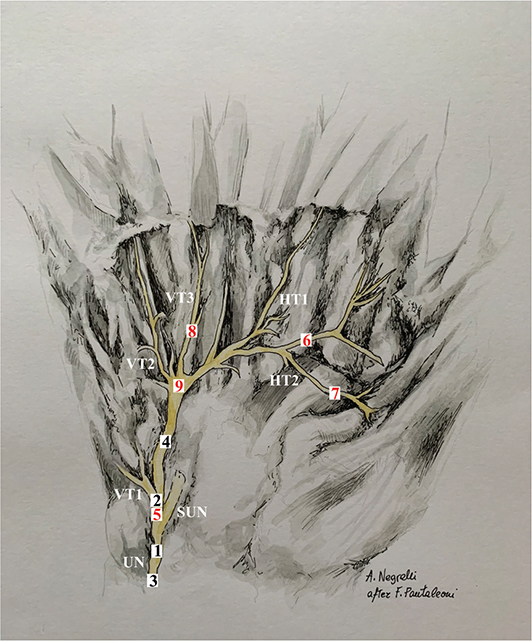
Figure 2. Anatomical illustration of the DMBUN summarizing the distribution of schwannomas reported in the literature to date: instances confirmed by histology or immunohistochemistry are marked in red, while unconfirmed cases are indicated in black. DMBUN: deep motor branch of the ulnar nerve; UN: ulnar nerve; SUN: superficial branch of ulnar nerve; VT1: vertical trunk for abductor digiti minimi muscle and flexor digiti minimi brevis muscle; VT2: vertical trunk for opponens digiti minimi muscle, 4th palmar interosseous muscle and 4th lumbrical muscle; VT3: vertical trunk for 3rd palmar interosseous muscle and 3rd lumbrical muscle; HT1: horizontal trunk for 2nd palmar interosseous muscle and 2nd dorsal interosseous muscle; HT2: horizontal trunk for adductor pollicis muscle and flexor pollicis brevis muscle.
Histologically confirmed schwannomas were diagnosed in five cases (55.6%), including the present one, with positive immunohistochemical staining for S100 protein. Four cases were not included in the statistics: two (22.2%) were diagnosed as neurofibromas, one (11.1%) as an intraneural cyst, and one (11.1%) remained a probable neuroma based on clinical characteristics.
The median age at presentation was 60 years (range: 35–78 years), including three males and two females. The right hand was affected more frequently (three of five cases). The most common presenting features were pain or aching (three of five), a hypothenar mass (two of five), loss of hand dexterity (two of five), and ulnar paresthesia (one of five). The duration of symptoms prior to presentation ranged from acute onset to several years.
Complete functional restoration was achieved in three cases (60%), while no recovery was observed in the remaining two cases (40%). Among those with complete recovery, the time to functional improvement ranged from 4 to 6 months. No clear association was observed between postoperative nerve function and tumor size, patient age, or time since symptom onset, however, the most recent schwannoma excisions, performed with microsurgical techniques and the aid of pre-operative MRI, yielded the most favorable results. Thus far, no cases of local recurrence have been reported for neurinomas involving the DMBUN.
Quality assessment
Quality appraisal revealed considerable temporal variability, reflecting changing standards of clinical reporting over time. More recent cases (2021–2025) demonstrated high methodological quality, with JBI scores ranging from 7 to 8 out of 8. In contrast, earlier reports (1952–1982) received moderate scores (4–6 out of 8), primarily limited by outdated diagnostic tools and less detailed documentation. Preoperative MRI was employed only in the four most recent schwannoma cases, while the preceding five did not include imaging.
New case presentation
A 61-year-old woman with no significant medical history was referred to the Hand Surgery and Microsurgery Department at the University Hospital of Modena for evaluation of a mass on the ulnar aspect of the right palm. She reported discomfort during gripping and decreased hand dexterity.
On examination, a firm, mobile nodule was palpable. Palpation elicited mild numbness in the ulnar digits and discomfort radiating across the hand (Figure 3). Ultrasound imaging revealed a well-encapsulated lesion adjacent to the ulnar nerve in the distal segment of Guyon’s canal. MRI demonstrated a hyperintense signal on T2-weighted sequences and an isointense signal on T1-weighted sequences (Figure 4). Following informed consent, surgical excision was performed.
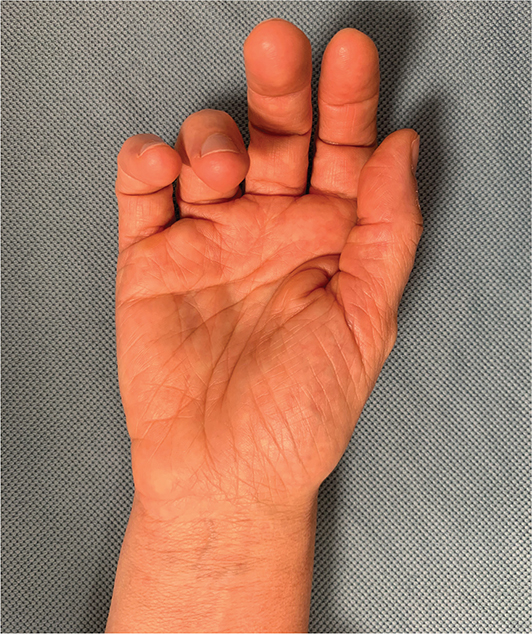
Figure 3. Preoperative clinical image showing a slight ulnar bump.
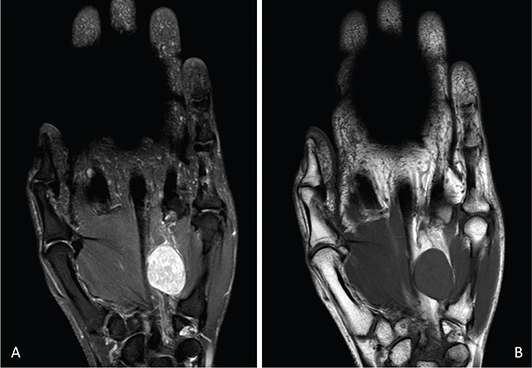
Figure 4. On MRI, the schwannoma appears: (A) hyperintense relative to muscle on T2-weighted sequences and (B) isointense on T1-weighted sequences. MRI: magnetic resonance imaging.
Through an extended approach to Guyon’s canal, the ulnar artery and the sensory branches to the fourth and fifth digits were identified and gently retracted. The lesion originated from the DMBUN at the VT2 and VT3 trunk divisions (Figure 5A). Intraoperative findings were highly suggestive of a schwannoma (Figure 5B). Under magnification and using microsurgical instruments, a longitudinal incision of the epineurium was made, and the tumor was excised en bloc (Figure 6A). The axons, stretched by the lesion’s significant size (2.5 × 1.9 × 2.0 cm), were preserved intact (Figure 6B). Histopathological analysis confirmed a benign neurilemmoma, with strong positivity for S100 protein.
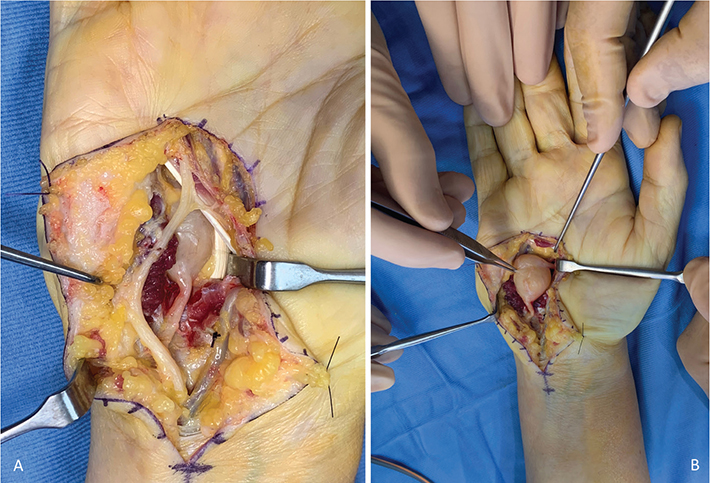
Figure 5. (A) From proximal to distal: on the left, the ulnar nerve divides into the motor branch (deep) and the sensory branch (superficial), the latter gives rise to the common digital nerve to 4th and 5th fingers distally isolated. On the right, the ulnar artery is retracted along with flexor digitorum superficialis V leaving the schwannoma of the DMBUN between the hypothenar muscles and the flexor digitorum profundus V. (B) The tumor is isolated from all surrounding anatomical structures. DMBUN: deep motor branch of the ulnar nerve.
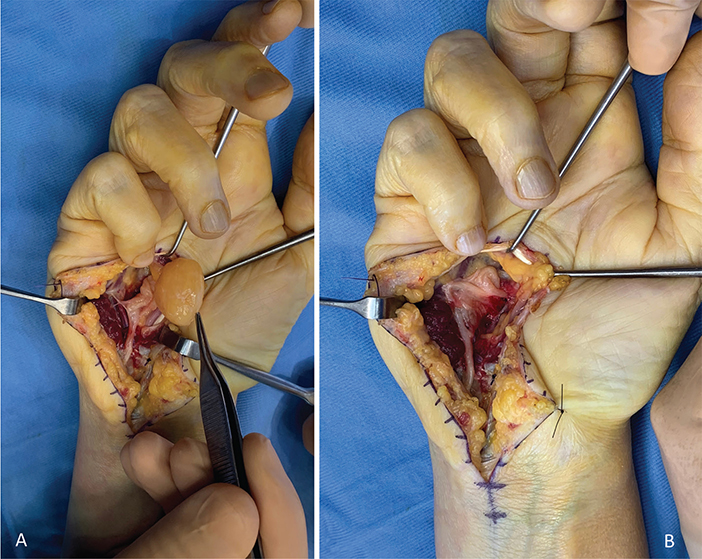
Figure 6. (A) The neurilemmoma is shelled out from the DMBUN without causing axonal injury. (B) The nerve fascicles of the DMBUN appear elongated but remain continuous and unharmed. DMBUN: deep motor branch of the ulnar nerve.
Postoperatively, the patient exhibited an intrinsic-minus hand posture for 2 months, without sensory deficits. An intrinsic-plus night splint was fabricated to assist in passive correction. After this period, clawing of the ring and little fingers resolved spontaneously, and the patient gradually regained dexterity and hand function. At 5 months post-surgery, she had achieved full functional recovery. At 2-year follow-up, she remained asymptomatic, with no signs of recurrence or residual discomfort (Figure 7).
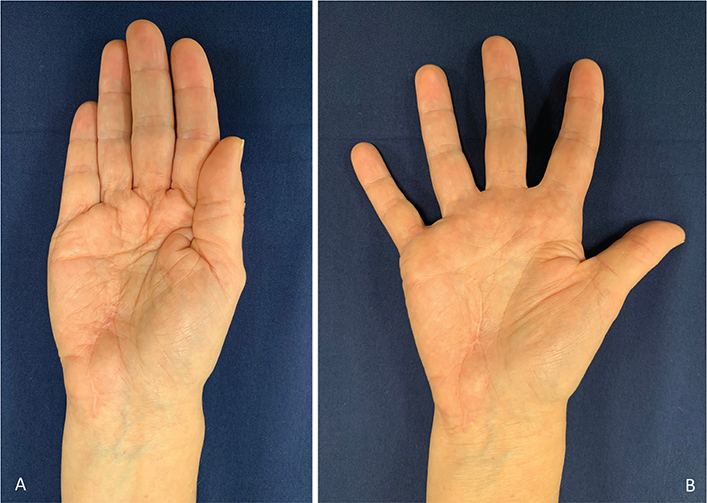
Figure 7. Clinical image demonstrating complete intrinsic muscle functionality at the 2-year follow-up: (A) adduction and (B) abduction of the fingers.
Discussion
This systematic review presents the most comprehensive analysis to date of schwannomas involving the DMBUN, encompassing five histologically confirmed cases reported over a 73-year period [9–15]. The exceptional rarity of these tumors is underscored by the limited number of publications, despite the widespread adoption of high-resolution imaging and heightened awareness of peripheral nerve pathologies in recent decades.
The slight male predominance and right-hand involvement observed in this series are likely attributable to sampling bias rather than indicative of true epidemiological trends. Most symptoms reported by patients were subjective and primarily related to neuropathic compression of either the superficial branch of the ulnar nerve or the DMBUN.
Our case is the first to describe a schwannoma simultaneously involving both the VT2 and VT3 trunks. Despite the tumor’s relatively large size (2.5 × 1.9 × 2.0 cm) and the involvement of multiple nerve branches, complete functional recovery was achieved. This outcome suggests that tumor size alone may not be a dominant prognostic factor in the limited cases reported to date; rather, preservation of nerve fascicle integrity during surgical excision appears to be critical. Furthermore, no correlation was observed between patient age or time since tumor onset and final outcome.
The literature review highlights several potential prognostic factors. Firstly, the time period in which surgery was performed: more recent cases demonstrated better outcomes, likely due to advances in preoperative imaging, microsurgical techniques, and the improved surgical understanding of peripheral nerve pathologies. Secondly, anatomical location: tumors arising from more proximal segments of the DMBUN appeared to be easier to resect, possibly owing to the larger caliber of the nerve trunk, which may facilitate fascicular dissection. Thirdly, histological subtype: confirmed schwannomas were associated with better clinical outcomes compared to neuromas, neurofibromas, or intraneural cysts. This may be attributed to the peripheral and encapsulated nature of schwannomas, which allows en bloc excision without sacrificing functional fascicles. Lastly, the surgical approach: resections involving transection of the nerve trunk led to poorer outcomes, whereas meticulous microsurgical dissection with preservation of fascicles correlated with superior recovery – emphasizing the importance of precise surgical technique [9–15].
Based on these findings, we propose an experience based management algorithm aimed at optimizing outcomes in suspected DMBUN schwannomas. Preoperative evaluation should consistently include high-resolution MRI to assess the lesion’s relationship to surrounding nerve fascicles. This may facilitate differential diagnosis and reveal proximity to the DMBUN branching pattern.
The recommended surgical strategy involves an extended Guyon’s canal approach, utilizing microsurgical instruments under magnification. A longitudinal epineurial incision should be followed by gentle blunt dissection of the lesion, with maximal preservation of functional fascicles [16].
Currently, there is no evidence supporting the superiority of any specific postoperative regimen. Therefore, we recommend early active mobilization during the day, combined with nocturnal splinting in an intrinsic-plus position to prevent the development of a fixed intrinsic-minus posture during the potential transient neuropraxic phase, which may last up to 6 months postoperatively. Follow-up should include serial assessments at 1, 3, 6, and 12 months to monitor neurological recovery and functional restoration [17].
Limitations and clinical significance
This review is subject to inherent limitations due to the rarity of the condition and the nature of the available data. Publication bias is a concern, as more remarkable or favorable outcomes are more likely to be reported, potentially inflating success rates. Observer bias may also be present, as methodological quality was assessed using the JBI checklist, which involves some degree of subjectivity despite dual independent review. Information bias is introduced by the heterogeneity among reported cases in terms of tumor location, surgical technique, follow-up duration, and outcome metrics, which complicates direct comparisons. Additionally, the small and heterogeneous sample size limits statistical power and increases the influence of individual cases on the overall findings.
Nevertheless, this study addresses critical gaps in the literature by offering the first anatomical mapping of DMBUN schwannomas and demonstrating that even large tumors exceeding two centimeters in diameter can be safely excised with complete functional preservation, provided that a meticulous microsurgical technique is applied.
Conclusion
Schwannomas of the DMBUN are exceedingly rare lesions with highly variable clinical outcomes. Nonetheless, although evidence is limited, available data suggest that accurate preoperative imaging assessment and careful intraoperative microsurgical dissection may allow favorable outcomes, even in large lesions. This study lays the groundwork for evidence based management by presenting the largest systematic analysis to date and by proposing a structured experience based treatment algorithm.
Acknowledgments
The authors extend their gratitude to Arianna Negrelli, talented artist, for her enthusiastic and dedicated collaboration in this project.
Ethics approval
All procedures performed were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to participate and to publish
Written informed consent was obtained from the patient for her anonymized information to be published in this article (including figures).
Authors’ contributions
Filippo Pantaleoni performed the surgical procedure and drafted the majority of the manuscript.
Mariia Kotova proposed the concept of a systematic review on this rare topic following discussion of the index case, revised the manuscript, and provided substantive editorial input.
Marco Passiatore and Marta Starnoni conducted the literature review and drafted the Methods section.
Marta Montanari contributed to completion of the literature review, critically revised the manuscript, and handled submission.
Davide Gravina completed postoperative follow-up and acquired the clinical images.
Paolo Boccolari supervised rehabilitation, performed physiotherapy, and designed custom splints.
Danilo Donati supervised the physiatric management, coordinated rehabilitation, and evaluated the patient following nerve palsy.
Andrea Leti Acciaro served as second surgeon, provided senior oversight and revised the manuscript in accordance with Journal guidelines.
References
[1] Paterson C, Bozic I, Smith MJ, et al. A mechanistic mathematical model of initiation and malignant transformation in sporadic vestibular schwannoma. Br J Cancer. 2022;127:1843–1857. https://doi.org/10.1038/s41416-022-01955-8
[2] Kim DH, Murovic JA, Tiel RL, et al. A series of 397 peripheral neural sheath tumors: 30-year experience at Louisiana State University Health Sciences Center. J Neurosurg. 2005;102:246–255. https://doi.org/10.3171/jns.2005.102.2.0246
[3] Stefansson K, Wollmann R, Jerkovic M. S-100 protein in soft-tissue tumors derived from Schwann cells and melanocytes. Am J Pathol. 1982;106:261–268.
[4] Gil YC, Shin KJ, Lee SH, et al. Anatomy of the deep branch of the ulnar nerve. J Hand Surg Eur Vol. 2016;41:843–847. https://doi.org/10.1177/1753193415622188
[5] Pantaleoni F, Petrella G, Colopi S, et al. Update on peripheral nerve sheath tumors in upper extremity: a descriptive review. J Hand Surg Asian Pac Vol. 2021;26:309–318. https://doi.org/10.1142/S2424835521400051
[6] Pantaleoni F, Marcoccio I, Adani R. Microsurgical approach to peripheral nerve tumors of the upper limb. Minerva Orthop. 2023;74:257–267. https://doi.org/10.23736/S2784-8469.23.04282-7
[7] Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2020;372:n71. https://doi.org/10.1136/bmj.n71
[8] Munn Z, Barker TH, Moola S, et al. Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid Synth. 2020;18:2127–2133. https://doi.org/10.11124/JBISRIR-D-19-00099
[9] Jenkins SA. Solitary tumours of peripheral nerve trunks. J Bone Joint Surg Br. 1952;34-B:401–411. https://doi.org/10.1302/0301-620X.34B3.401
[10] Cavanagh NPC, Pincott JR. Ulnar nerve tumours of the hand in childhood. J Neurol Neurosurg Psychiatry. 1977;40:795–800. https://doi.org/10.1136/jnnp.40.8.795
[11] Bowers WH, Doppelt SH. Compression of the deep branch of the ulnar nerve by an intraneural cyst. J Bone Joint Surg Am. 1979;61:612–613. https://doi.org/10.2106/00004623-197961040-00024
[12] Sarangapani K, More AR. Neurilemmoma of deep branch of ulnar nerve presenting as ulnar tunnel syndrome. Hand. 1983;15:216–217. https://doi.org/10.1016/S0072-968X(83)80017-5
[13] Oguma H, Aoki M, Nagoya S. Neurilemmoma originating from a deep branch of the ulnar nerve. Orthopedics. 1999;22:359–360. https://doi.org/10.3928/0147-7447-19990301-15
[14] Moran J, Kahan J, Schneble CA, et al. Surgical excision of a giant schwannoma of the hand: a case report. JBJS Case Connect. 2021;11:e21.00318. https://doi.org/10.2106/JBJS.CC.21.00318
[15] Giunta G, De Baerdemaeker R, Zeltzer AA, et al. Schwannoma arising from the deep motor branch of the ulnar nerve – a case report and review of literature. J Hand Surg Asian Pac Vol. 2022;27:366–369. https://doi.org/10.1142/S2424835522720109
[16] Mackinnon SE, Dellon AL. Surgery of the peripheral nerve. New York, NY: Thieme Medical; 1988.
[17] Sunderland S. Nerves and nerve injuries. Edinburg, London and New York, NY: Churchill Livingstone; 1978.