ORIGINAL RESEARCH ARTICLE
Medial femoral condyle flap for ulnar reconstruction and structural bone allograft for radial reconstruction in post-traumatic forearm diaphyseal non-union: a two-case report
Matthias Holzbauera,b, Maximilian Zaussingerb,c, Klaus Strassmairb, Stefan M. Froschauera,b, Tobias Gotterbarma,b and Manfred Schmidtb,c
aDepartment of Orthopaedics and Traumatology, Kepler University Hospital GmbH, Linz, Austria; bMedical Faculty, Johannes Kepler University Linz, Linz, Austria; cDepartment of Plastic, Aesthetic, and Reconstructive Surgery, Kepler University Hospital GmbH, Linz, Austria
ABSTRACT
The successful treatment of forearm non-unions, which occur as a complication in 2–10% of all forearm fractures, poses a significant challenge in reconstructive surgery. Current treatment often involves revision fixation with non-vascularized bone grafts for atrophic non-unions. Large bony defects in both forearm bones, however, necessitate advanced reconstructive techniques. In isolated reconstructions of the radius or ulna, the vascularized medial femoral condyle (MFC) flap has gained popularity, achieving 98–100% union rate. Allogenic bone grafts have also been described for reconstruction of large bone defects, potentially reducing donor site morbidity and operation time.
This two-case report investigates the performance of vascularized MFC cortico-cancellous flap for ulna reconstruction and tricortical allogenic bone graft of the iliac crest for radius reconstruction in excessive, post-traumatic, atrophic non-union of the forearm diaphysis. In May and June 2021, a 62-year-old and a 56-year-old male received reconstruction surgery 9 and 13 months after initial plate osteosynthesis after a motorcycle accident.
The MFC flap showed successful bony union of the ulna after 3 months as well as consecutive remodeling. However, the reconstruction of the radius showed a recurrent non-union, which even led to plate breakage in one case.
In both cases, ulnar reconstruction with a vascularized MFC flap achieved successful union. However, radial reconstruction using structural allograft did not achieve union, causing recurrent non-union, likely reflecting limited biological incorporation.
Level of Evidence: V (Therapeutic)
KEYWORDS: Bone allograft; bone reconstruction; forearm non-union; medial femoral condyle flap; plastic surgery; post-traumatic non-union
Citation: Journal of Plastic Surgery and Hand Surgery 2026; 61: 91–94. DOI: https://doi.org/10.2340/jphs.v61.45795.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 11 November 2025; Accepted: 13 March 2026; Published: 24 April 2026
CONTACT: Manfred Schmidt manfred.schmidt@kepleruniklinikum.at Department of Plastic and Reconstructive Surgery, Kepler University Hospital GmbH Krankenhausstrasse 9, 4020 Linz, Upper Austria, Austria
Supplemental data for this article can be accessed online at https://doi.org/10.2340/jphs.v61.45795
Competing interests and funding: The authors have no competing interests to declare that are relevant to the content of this article.
Introduction
In forearm fractures, non-unions occur in 2–10% of all forearm fractures [1]. The Food and Drug Administration (FDA) defines a no-union as ‘a fracture that persists for a minimum of 9 months without signs of healing for 3 months’ [2]. Weber and Čech classify non-unions into atrophic, oligotropic, and hypertropic non-union [3]. Due to the complex mechanism of fracture healing and its dysfunctions resulting in non-union, the ‘Diamond Concept’ was introduced, emphasizing cellular environment, the growth factors, the bone matrix, and the mechanical stability [4]. More advanced non-union classifications have since been developed [5, 6], although Calori et al. summarize the difficulty in the diagnosis and further successful treatment of non-union ‘as no two cases of non-union are apparently alike’ [6]. While atrophic non-union is considered to be caused by poor vascularity, histological analyses do not completely support this radiological finding [7, 8].
In isolated ulnar or radial diaphyseal non-unions, effective treatment includes debridement of avital bone tissue, removal of failed hardware, restoration of length, alignment, and rotation, as well as stable fixation using compression osteosynthesis methods (e.g. 3.5 mm plates] [9]. For defects up to 6 cm of length, autologous bone grafts – most commonly harvested from the iliac crest are recommended [9, 10]. However, treating non-unions of both the radius and the ulna poses a significant surgical challenge in achieving the aforementioned mechanical objectives while ensuring enough biological healing properties. For the treatment of forearm non-unions, current literature supports reconstruction using compression plating with autologous bone grafts in large bone loss (3–5 cm) [1], while vascularized bone grafts should be considered in defects larger than 5–6 cm [11]. A recent scoping review reports that the vascularized fibula graft is most utilized in diaphyseal osseous gap reconstruction in the upper extremity [12]. Recently, the vascularized medial femoral condyle (MFC) flap, first described by Sakai in 1991, has gained increasing popularity in bony defect reconstructions [13]. MFC flap offers several advantages including a constant pedicle anatomy [14], a maximal flap length up to 13 cm, and a stable cortical bone with various contouring options [15]. In upper extremity reconstructions, the MFC flap yielded an overall flap failure rate of 2% in recent meta-analysis [16]. In terms of isolated reconstructions of either the radius or the ulna using a MFC cortico-cancellous or periosteal flaps, a recent systematic review reported a union rate of 100% [11].
Alternatively, allogenic bone grafts have been suggested as an option for addressing large bone defects of the radius in patients with infected non-unions and after tumor excision [17, 18]. Considering the higher rates of recurring non-unions at single allograft-host bone junctions, this option appears attractive for the simultaneous reconstruction of both forearm bones: If a MFC reconstruction for the ulna is performed, radius reconstruction using a bone allograft aims to reduce donor site morbidity and operation time.
No reports exist in the literature describing simultaneous reconstruction of both forearm bones using a vascularized MFC graft for the ulna combined with a structural graft for the radius in extensive atrophic diaphyseal non-union.
Case presentation
The first case includes a 62-year-old male patient, who suffered an open forearm diaphysis fracture including a two-level ulna fracture, a radial head and a coronoid process fracture following a motorcycle accident. Initial treatment was performed at another clinic using a 12-hole Variable Angle Locking Compression Plate (VA-LCP) for the radius and a 14-hole VA-LCP plate for the ulna including each one lag screw. Due to persistent delayed bony union until 6 months postoperatively, high-energy extracorporeal shockwave therapy was applied. However, CT imaging 9 months postoperatively showed a persistent bony defect zone of at least 2 cm for the radius and 3 cm for ulna with no signs of bony consolidation. Due to the presence of an atrophic non-union, reconstruction surgery was performed 9 months after the initial trauma in May 2021: Excision of the non-union resulted in a bony defect of 3 and 4 cm for the ulna and the radius, respectively. The defects were reconstructed using a MFC flap for the ulna and a sizematched structural allograft for the radius, both stabilized with a 10-hole and an 8-hole VA-LCP (see Figure 1). Following reconstructive surgery, pain and swelling at the harvest site were managed through partial weight-bearing and local treatment. Bony union of the MFC flap could be achieved 3 months postoperatively, while complete remodeling was observed 7 months postoperatively (see Video 1). Twenty months after reconstructive surgery, the patient presented with plate breakage due to a recurrent non-union of the radius (see Figure 1). This patient has no known risk factor for impaired bony healing with arterial hypertension as the only pre-existing condition.
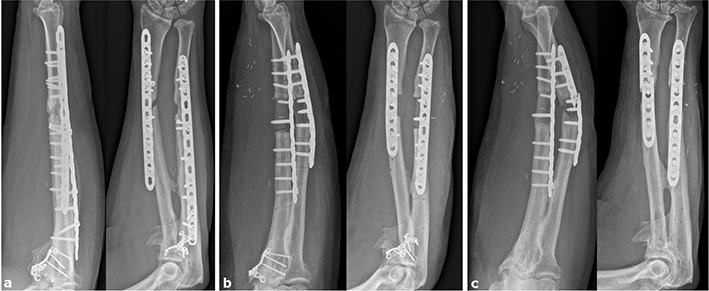
Figure 1. Case 1 Radiographs. Radiographs obtained preoperatively (a) after splint removal (b) as well as 20 months after reconstruction surgery (c).
The second case involves a 56-year-old male polytrauma patient who was transferred to our clinic after a motorcycle accident. He showed an open, comminuted fracture of the radius and ulna diaphysis, while he also suffered a traumatic amputation of the left lower leg, rib fractures, and a fracture of the coracoid process. Initial treatment included a 7-hole VA-LAP osteosynthesis of the ulna, while no compression of the fracture site could be achieved due to a comminution zone of 1 cm. The radius was treated using an 8-hole plate osteosynthesis and one lag screw. A concomitant soft tissue defect necessitated seven wound revisions including Vacuum Assisted Closure cycles before being ultimately covered with a vascularized muscle flap and split skin-grafting 20 days after the accident. Postoperative radiographic imaging showed no signs of bony consolidation resulting in a defect zone of 3 and 4 cm of the radius and ulna, which was verified in a CT scan 10 months postoperatively. Due to atrophic forearm non-union, reconstructive surgery was performed 13 months after the accident in June 2021. This procedure involved the excision of a bony defect of approximately 5 cm in both the radius and the ulna. The defects were again reconstructed using a MFC flap for the ulna and a size-matched structural allograft for the radius, both stabilized with a 10-hole and a 9-hole VA-LCP (see Figure 2). Bony union of the MFC flap could be achieved 3 months postoperatively. While CT scans taken 9 and 19 months after the reconstruction surgery revealed a complete remodeling of the MFC flap, recurrent atrophic non-union of the radius with significant lysis of the allogeneic graft was observed (see Figure 2 and Video 2). This patient showed chronic alcohol abuse and chronic obstructive pulmonary disease as pre-existing conditions in his medical history.
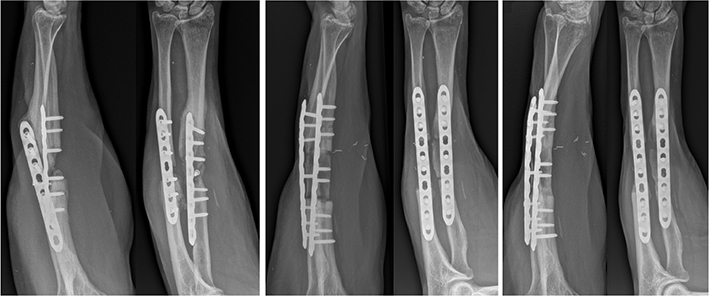
Figure 2. Case 2 Radiographs. Radiographs obtained preoperatively (a) after splint removal (b) as well as 19 months after reconstruction surgery (c).
Surgical technique
Reconstructive surgery involved excision of the non-union area until bleeding cortical margins are achieved. Ulna reconstruction involved a vascularized, cortico-cancellous MFC flap from the contralateral knee. Flap harvesting was aided by a 3D printed model of the defect [19, 20]. The pedicle of the flap was anastomosed to the ulnar artery and vein (see Figure 3). For radius reconstruction, a tricortical allogenic bone graft of the iliac crest was used. Re-osteosynthesis was performed using a 3.5 mm VA-LCP, while excentric drilling was performed on both sides of the graft-host junctions. The tricortical iliac crest allograft was obtained from a certified musculoskeletal tissue bank and processed according to standardized sterilization protocols. Postoperative care involved immobilisation with an upper-arm plaster cast for 4 weeks, followed by a forearm cast for additional 2 weeks. X-ray controls were performed immediately postoperatively, 3 weeks, 6 weeks, 12 weeks, 6 months, 12 months, and 18 months postoperatively.
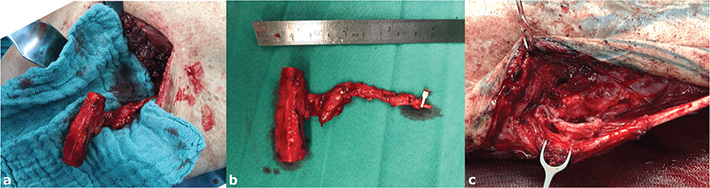
Figure 3. Intraoperative images: An intraoperative photograph of the cortico-cancellous medial femur condyle flap harvesting (a), the dimension of the graft with its pedicle (b), and its anastomosis to the ulnar artery and vein after flap inset and osteosynthesis (c).
Ethics
This retrospective case report was approved by the regional ethical review committee (EK 1093/2023). Patients provided written informed consent for inclusion.
Discussion and conclusions
The procedure examined in this case report includes a simultaneous reconstruction of large bony defects of the ulna and the radius using a vascularized cortico-cancellous MFC flap and an iliac crest allograft for post-traumatic, atrophic non-union of the forearm. Bony union of MFC flap was achieved within 3 months, which is at the lower end of reported healing times [21]. The rapid union observed in our cases is consistent with previous reports indicating thatdiabetes mellitus, a body mass index greater than 30 kg/m², and more than one previous surgery are associated with longer healing times [21].
Recent systematic reviews show an overall bony union of MFC flaps of 98 to 100% [11, 16]. In 2009, de Smet was one of the first authors who reported solid union of four non-unions of the ulna and two non-unions of the radius with prior surgeries that were successfully treated with MFC flaps [22]. In contrast, revision fixation using a bone allograft not only failed to achieve union at the allograft-host junctions, whose occurrence was described between 30 and 50% in large bone defects of the radius [18, 17], but even resulted in an entire failure of allograft incorporation in both cases. The ulna is generally considered to have poorer regenerative properties compared to the radius [23]. Despite comparable sizes of the bone defects in the forearm diaphysis, reconstruction using vascularized MFC flap appeared biologically superior in these cases compared to the iliac crest allograft in all aspects. The absence of intrinsic vascularity, cellularity and growth factors in combination with the high mechanical strain of the forearm during movement pose essential drawbacks of the allograft [4]. Hence, the critical defect size for achieving bony union seemed to be exceeded in these two cases.
The main limitation of this retrospective case report is the sample size including two patients, which may be caused by the rare occurrence of extensive, atrophic non-unions of the forearm. From a technical standpoint, the two cases also differed in fixation strategy, as the radial allograft was secured with one fixed-angle screw in the first patient and with three fixed-angle screws in the second. This variation likely affected construct rigidity and may have influenced graft stability and biological incorporation. The choice of allograft for radius reconstruction failed its intended aim of reducing donor site morbidity and shortening surgical time for simultaneous forearm reconstruction, instead necessitating another complex revision surgery.
For extensive, atrophic forearm non-unions, we conclude that the MFC cortico-cancellous flap represents a favorable option for ulna reconstruction. For simultaneous radius reconstruction, an autologous bone flap seems necessary to provide sufficient biological healing properties, with the defect length determining the necessity for either a vascularized or non-vascularized option. Another surgical option would be a double-barrel vascularized fibular graft for the reconstruction of both forearm bones, while flap trimming and the positioning of the pedicle might imply a surgical challenge [24]. While forearm shortening osteotomy in a case with 1.5 cm resection was reported with satisfactory results, recent studies report considerable loss of range of motion in pronation and supination and in finger flexion in shortening of more than 3 cm [25–27]. Because the vascularized MFC flap significantly outperformed the iliac bone allograft, a vascularized graft seems recommended whenever possible in large bone defects resulting from forearm non-union [28].
Acknowledgments
This study is supported by the Johannes Kepler Open Access Publishing Fund and the federal state of Upper Austria.
During the preparation of this work the author(s) used ChatGPT, in order to enhance the grammar, and linguistic quality of the text. After using this tool/service, the author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
Data availability statement
All relevant data supporting the findings of this report are included within the article.
Ethics declarations & trial registry information
This retrospective case report was approved by the regional ethical review committee (EK 1093/2023).
Authors’ contributions
Conceptualization: Manfred Schmidt, Stefan M Froschauer; Methodology: Manfred Schmidt; Formal analysis and investigation: Matthias Holzbauer, Klaus Strassmair, Maximilian Zaussinger, Manfred Schmidt, Stefan M Froschauer; Writing - original draft preparation: Matthias Holzbauer; Writing - review and editing: Manfred Schmidt; Funding acquisition: n/a; Resources: Manfred Schmidt, Matthias Holzbauer; Supervision: Tobias Gotterbarm; Manfred Schmidt.
References
[1] Srinivasan RC, Hutson RL, Richard MJ. Forearm nonunion: characterization and management. J Hand Surg Am. 2020; 45(11): 1055–1064. https://doi.org/10.1016/j.jhsa.2020.05.030
[2] Cunningham BP, Brazina S, Morshed S, et al. Fracture healing: a review of clinical, imaging and laboratory diagnostic options. Injury. 2017; 48 Suppl 1: S69–S75. https://doi.org/10.1016/j.injury.2017.04.020
[3] Weber BG, Čech O. Pseudarthrosis: pathophysiology, biomechanics, therapy, results. New York: Grune & Stratton; 1976.
[4] Giannoudis PV, Einhorn TA, Marsh D. Fracture healing: the diamond concept. Injury. 2007; 38 Suppl 4: S3–S6. https://doi.org/10.1016/S0020-1383(08)70003-2
[5] Solomin LN, Semenistyy AA, Komarov AV, et al. Universal long bone nonunion classification. Strategies Trauma Limb Reconstr. 2023; 18(3): 169–173. https://doi.org/10.5005/jp-journals-10080-1597
[6] Calori GM, Phillips M, Jeetle S, et al. Classification of non-union: need for a new scoring system? Injury. 2008; 39 Suppl 2: S59–S63. https://doi.org/10.1016/S0020-1383(08)70016-0
[7] Rupp M, Biehl C, Budak M, et al. Diaphyseal long bone nonunions – types, aetiology, economics, and treatment recommendations. Int Orthop. 2018; 42(2): 247–258. https://doi.org/10.1007/s00264-017-3734-5
[8] Bajada S, Marshall MJ, Wright KT, et al. Decreased osteogenesis, increased cell senescence and elevated Dickkopf-1 secretion in human fracture non union stromal cells. Bone. 2009; 45(4): 726–735. https://doi.org/10.1016/j.bone.2009.06.015
[9] Kloen P, Wiggers JK, Buijze GA. Treatment of diaphyseal non-unions of the ulna and radius. Arch Orthop Trauma Surg. 2010; 130(12): 1439–1445. https://doi.org/10.1007/s00402-010-1071-x
[10] Ring D, Allende C, Jafarnia K, et al. Ununited diaphyseal forearm fractures with segmental defects: plate fixation and autogenous cancellous bone-grafting. J Bone Joint Surg Am. 2004; 86(11): 2440–2445. https://doi.org/10.2106/00004623-200411000-00013
[11] Weir JC, Osinga R, Reid A, et al. Free vascularised medial femoral condyle periosteal flaps in recalcitrant long bone non-union: a systematic review. Arch Orthop Trauma Surg. 2020; 140(11): 1619–1631. https://doi.org/10.1007/s00402-020-03354-1
[12] Naga HI, Kim J, Dunworth K, et al. Upper extremity diaphyseal osseous gap reconstruction with free vascularized bone flaps: a scoping review. J Reconstr Microsurg. 2025;41(1):68–76.
[13] Sakai K, Doi K, Kawai S. Free vascularized thin corticoperiosteal graft. Plast Reconstr Surg. 1991; 87(2): 290–298. https://doi.org/10.1097/00006534-199102000-00011
[14] Higgins JP. Vascularized medial femoral condyle flap reconstruction for osseous defects of the hand and wrist. Hand Clin. 2024; 40(1): 151–159. https://doi.org/10.1016/j.hcl.2023.06.003
[15] Iorio ML, Masden DL, Higgins JP. The limits of medial femoral condyle corticoperiosteal flaps. J Hand Surg Am. 2011; 36(10): 1592–1596. https://doi.org/10.1016/j.jhsa.2011.07.015
[16] Scampa M, Mégevand V, Martineau J, et al. Medial femoral condyle free flap: a systematic review and proportional meta-analysis of applications and surgical outcomes. Plast Reconstr Surg Glob Open. 2024; 12(4): e5708. https://doi.org/10.1097/GOX.0000000000005708
[17] van Isacker T, Barbier O, Traore A, et al. Forearm reconstruction with bone allograft following tumor excision: a series of 10 patients with a mean follow-up of 10 years. Orthop Traumatol Surg Res. 2011; 97(8): 793–799.
[18] Davis JA, Choo A, O’Connor DP, et al. Treatment of infected forearm nonunions with large complete segmental defects using bulk allograft and intramedullary fixation. J Hand Surg Am. 2016; 41(9): 881–887. https://doi.org/10.1016/j.jhsa.2016.05.021
[19] Taylor EM, Iorio ML. Surgeon-based 3D printing for microvascular bone flaps. J Reconstr Microsurg. 2017; 33(6): 441–445. https://doi.org/10.1055/s-0037-1600133
[20] Schmidt M, Holzbauer M, Froschauer SM. Metacarpal reconstruction with a medial femoral condyle flap based on a 3D-printed model: a case report. Case Reports Plast Surg Hand Surg. 2022; 9(1): 52–56. https://doi.org/10.1080/23320885.2022.2029453
[21] Guzzini M, Ciclamini D, Arioli L, et al. Correlation between risk factors and healing times in long bone nonunions treated with corticoperiosteal flap from the medial femoral condyle. J Reconstr Microsurg. 2023; 39(7): 502–507. https://doi.org/10.1055/a-2004-2364
[22] de Smet L. Treatment of non-union of forearm bones with a free vascularised corticoperiosteal flap from the medial femoral condyle. Acta Orthop Belg. 2009; 75(5): 611–615.
[23] Hussain A, Nema SK, Sharma D, et al. Does operative fixation of isolated fractures of ulna shaft results in different outcomes than non-operative management by long arm cast? J Clin Orthop Trauma. 2018; 9(Suppl 1): S86–S91. https://doi.org/10.1016/j.jcot.2017.12.004
[24] Banic A, Hertel R. Double vascularized fibulas for reconstruction of large tibial defects. J Reconstr Microsurg. 1993; 9(6): 421–428. https://doi.org/10.1055/s-2007-1006751
[25] Chauhan C, Howard A, Saleh M. Severely comminuted forearm fracture treated with acute shortening. Injury. 1995; 26(6): 415–416. https://doi.org/10.1016/0020-1383(95)00036-9
[26] Schnapp L, Colakoglu S, Couceiro J, et al. Forearm shortening impact on pronation and supination. J Hand Surg Asian Pac Vol. 2019; 24(3): 289–296. https://doi.org/10.1142/S242483551950036X
[27] Daugherty T, Sawyer J, Gillin T, et al. The effect of forearm shortening on finger flexion: a biomechanical study. J Hand Surg Am. 2025; 50(9):1126.e1-1126.e7.
[28] Hamada Y, Hibino N, Kobayashi A. Expanding the utility of modified vascularized femoral periosteal bone-flaps: an analysis of its form and a comparison with a conventional-bone-graft. J Clin Orthop Trauma. 2014; 5(1): 6–17. https://doi.org/10.1016/j.jcot.2014.01.002