ORIGINAL RESEARCH ARTICLE
Early experience of prostate artery embolization: a retrospective single-centre study
Emir Majbara,b  , Jakob Swanbergc
, Jakob Swanbergc  , Farhood Alamdaria
, Farhood Alamdaria  , Ralph Peekerd and Abbas Chabokb,e
, Ralph Peekerd and Abbas Chabokb,e 
aDepartment of Urology, Västmanland Hospital, Västerås, Sweden; bCenter for Clinical Research Västmanland, Uppsala University, Sweden; cDepartment of Radiology, Västmanland Hospital, Västerås, Sweden; dDepartment of Urology, Institute for Clinical Sciences, Sahlgrenska Academy, Gothenburg University, Sweden; eDivision of Surgery, Danderyd University Hospital, Stockholm, Sweden
ABSTRACT
Objective: The aim of this study was to evaluate the early experiences of prostate artery embolization (PAE) in patients with benign prostatic hyperplasia (BPH).
Material and methods: This retrospective study included all patients treated for BPH who were referred to the radiology department for PAE in Västmanland between 2018 and 2021. Data were collected on patient demographics, International Prostate Symptom Score (IPSS), prostate-specific antigen level, and peri- and post-procedure outcomes. Clinical success was defined as a decrease in the IPSS by ≤3 points or catheter freedom.
Results: A total of 96 patients (median age 74 years) underwent PAE. Before PAE, 65% of the patients were treated with α-blockers and 83% with 5α-reductase inhibitors. Clinical success was achieved in 60 patients (63%). Among catheterized patients, 33 (53%) achieved catheter freedom. In the non-catheterized group, 27 (79%) experienced clinical success. No significant differences in outcomes were observed based on the presence of a median lobe.
Conclusions: PAE appears to be a viable treatment option for BPH and may provide substantial symptom relief and catheter freedom for most patients. These findings suggest that PAE may be an effective alternative to more invasive procedures. Further research is needed to refine the patient selection criteria.
KEYWORDS: Prostate artery embolization; benign prostatic hyperplasia; lower urinary tract symptoms; International Prostate Symptom Score
Citation: Scandinavian Journal of Urology 2025, VOL. 60, 1–7. https://doi.org/10.2340/sju.v60.42495.
Copyright: © 2025 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 26 August 2024; Accepted: 11 November 2024; Published: 2 January 2025
CONTACT: Emir Majbar emir.majbar@regionvastmanland.se Urology department, Västmanlands Hospital Västerås, SE-723 35 Västerås, Sweden
Introduction
Lower urinary tract symptoms (LUTS) caused by benign prostatic hyperplasia (BPH) are frequently observed in older men and can affect up to 75% of men aged 50–70 years [1–3]. Untreated BPH can lead to various complications, such as acute and/or chronic urinary retention [3], bladder dysfunction, or kidney failure [4].
Various treatment options are available for BPH and range from medication such as 5α-reductase inhibitors and α-blockers [5] to minimally invasive procedures such as transurethral microwave thermotherapy (TUMT), transurethral needle ablation (TUNA), Rezumä (water vapour therapy), Urolift and laser treatments [6–8]. Additionally, adenoma enucleation involving en bloc removal of the prostatic adenoma to alleviate the obstruction caused by significant BPH [9] offers an alternative alongside transurethral resection of the prostate (TURP), which was introduced in the 1950s and is still considered the gold standard treatment for BPH [5, 10]. TURP is known to cause retrograde ejaculation and has risks for complications such as bleeding, infection, stricture formation, TURP syndrome and incontinence [10–12]. On the other hand, adenoma enucleation is considered to be the most invasive approach, and excessive bleeding, infection or bladder neck stenosis requiring reintervention are the primary concerns [13]. Considering that BPH is fundamentally a benign yet potentially serious condition and is not risk-free, it is imperative to explore treatment options that match the efficacy of TURP while striving to minimise any potential adverse effects or complications.
Selective arterial embolisation has been used for several other indications earlier [14, 15]. Prostate artery embolisation (PAE) is a minimally invasive interventional radiology technique for the treatment of BPH that was first described in 2010 [16]. PAE involves the selective embolisation of prostatic arteries using small embolic particles, which is thought to induce ischemia and subsequent shrinkage of prostatic tissue, thereby decreasing pressure on the urethra, mitigating urinary obstruction [17, 18].
The procedure time for PAE varies depending on the access route used. Gil et al. reported that the median procedure time was similar between trans-radial access (81 min) and transfemoral access (94 min) groups [19]. Patients typically experience significant symptom relief within a month post-procedure, with maximal reductions in the International Prostate Symptom Score (IPSS) observed at 6 months [20].
Prostate artery embolization was introduced in Helsingborg, Sweden, in 2015 and later in the Västmanland region in 2018. This retrospective single-centre study aims to evaluate our early experiences and clinical outcomes of PAE in patients with BPH in the Västmanland region, Sweden, focusing on symptom relief, catheter freedom and procedural safety.
Materials and methods
Population
This descriptive retrospective observational study was conducted at Central Hospital Västerås, Västmanland Region, in Sweden. All patients with BPH referred for PAE between 2018 and 2021 were included. The indication for PAE was either bothersome LUTS with a self-administered IPSS of ≥ 12 or urinary retention (using a permanent urinary catheter or intermittent). Patients were excluded if deemed unsuitable for PAE based on findings from pre-procedure computed tomography (CT) angiography (Figure 1).
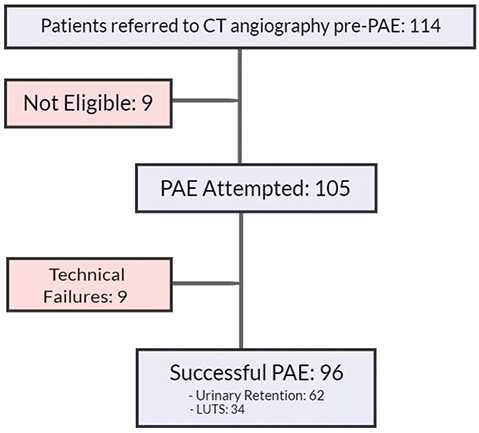
Figure 1. Patient population. Lower urinary tract symptoms. Urinary retention (patients with an indwelling catheter or using intermittent catheterisation).
A thorough review of medical records was conducted, and information on symptoms, IPSS, prostate-specific antigen (PSA) level, complications, length of hospital stay, and follow-up results for at least 2 years were recorded.
A total of 114 patients were referred for CT angiography as a preliminary assessment before the PAE procedure, and 96 of these patients successfully underwent the PAE procedure. However, nine patients were deemed ineligible for PAE after CT angiography, and another nine experienced technical failure during the procedure. Analysis of the reasons for PAE referral among the technically successful cases showed that 34 patients underwent the procedure because of bothersome LUTS and the other 62 had urinary retention.
Outcome definition
The achievement of technical success was defined as a successful embolisation of either one or both prostate arteries. Clinical success was defined at 2 years follow-up, the removal of the urinary catheter (‘catheter freedom’) with a post-void residual urine volume of less than 200 mL. For patients experiencing LUTS, a decrease of ³3 points for the IPSS (self-administered) as a minimal clinically important difference (MCID) [21], which indicates an improvement in the patient’s condition, was designated as a successful outcome.
Treatment routines
In Västerås, at the introduction of the PAE technique, patients were considered for the procedure if they had a prostate volume of over 40 mL, without an upper limit, and no diagnosis of prostate cancer. Additionally, a comprehensive workup is routinely performed for all patients considered for surgical treatment, including cystometry and uroflowmetry. The Bladder Outlet Obstruction Index (BOOI) was calculated using the formula BOOI = Pdet @ Qmax – 2Qmax. We also note that post-treatment cystometry is only performed in selected cases. PAE was performed in a day-care setting. Preoperative preparation included fasting for 2 h before treatment, implementation of antibiotic prophylaxis if a urine culture was positive, and placing a 14-Ch Foley catheter upon admission if it was not already in place. Anticoagulant therapy was generally not discontinued. Medications such as 5α-reductase inhibitors and α-blockers were discontinued.****
Preoperative CT angiography of the pelvis was performed on a Toshiba Aquilion One CT scanner (Otawara, Japan) equipped with a 16 cm-wide detector using a dedicated pre-PAE CT protocol in which 130 mL of contrast media (Omnipaque 350 mg I/mL) was injected into an antecubital vein at a rate of 7 mL/s followed by a 50 mL bolus of normal saline. CT scanning was triggered by bolus tracking at the level of the aortic bifurcation with a fixed delay of 7 s. Axial images with a slice thickness of 0.6 mm were obtained and reviewed in multiplanar reformation (MPR). The prostatic arterial anatomy and the degree of iliac arteriosclerosis and tortuosity were described, and the predicted eligibility for PAE was reported.
PAE technique
Most procedures were performed using a Siemens Artis Q Angiographic system (Erlangen, Germany). Left radial arterial access was used routinely. A 5-Fr radial sheath was inserted into the radial artery under local anaesthesia and ultrasound guidance. A 5-Fr 135 cm angiographic catheter was advanced under angiographic control into the main trunks of the internal iliac arteries. Zoomed-in angiographic runs were obtained as road maps for subsequent selective catheterisation of the prostatic arteries with a 2.0-Fr microcatheter. With super-selective contrast injections, cone beam CT images were produced to verify the contrast enhancement of the prostatic parenchyma. If present, extensive collaterals to adjacent pelvic organs were occluded with microcoils to minimize the risk of non-target embolisation. For embolisation, 300–500 mm acrylic gelatine microspheres (Merit Medical, South Jordan, UT, USA) were injected under fluoroscopic control until complete occlusion of the artery was obtained. The ‘PErFecTED technique’ was used when possible; this procedure involves embolisation first at the proximal and then at the distal segments of the prostate artery for a more effective reduction of blood flow [22]. A similar procedure was repeated on the contralateral side. Upon completion of the procedure, a compression device was applied at the puncture site.
Following the procedure, the puncture site and circulation in the arm were monitored for 2 h, during which patients were permitted to eat, drink, and move freely. Upon discharge, patients were reminded of the potential for post-embolisation pain, and appropriate pain relief was made available as needed. Catheter removal was typically scheduled 7 days after PAE.
Statistics
Descriptive data analyses were conducted using the R statistical programming language [23] and the following packages: flextable, gtsummary, and tidyverse [24–26].
Ethical considerations
This study adhered to the Helsinki Declaration and the General Data Protection Regulation (GDPR) guidelines, underwent an ethical review, and received approval from the Swedish Ethical Review.
Results
For the 96 patients treated with PAE, bilateral embolisation was achieved in 76% of patients, while 23% underwent unilateral embolisation. clinical features and outcomes were examined in relation to urinary catheter use before the treatment. The median age was 74 years. Patients who used a catheter tended to be older but not statistically significant (median 76 vs. 73 years, P = 0.4), and 65% of patients were on α-blockers and 83% on 5-a reductase inhibitors before undergoing PAE. Catheter users had a higher median PSA level (6.4 ng/mL) than non-users (4.0 ng/mL) (Table 1).
Clinical success, as defined above, was achieved in 60 patients (63%); 19 (20%) patients required further intervention; and 17 (17%) were lost to follow-up (Table 2). Within the group of patients with LUTS (non-catheter users), 27 (79%) showed a significant 65% decrease in the IPSS from 20 to 7, and improved quality of life (QoL) score from 4 to 2, which reflected a 50% improvement in 2 years. The median follow-up time was 25 months (IQR: 23.1–32.2 months) (Table 3).
| Variable | Clinical success (n = 60) | Further intervention (n = 19) | Lost to follow-up, (n = 17) | P* |
| Age, median (SD) | 74 (7) | 72 (8) | 76 (7) | 0.074* |
| α-blocker, frequency (%) | 41 (68) | 11 (58) | 10 (59) | 0.6** |
| 5α-reductase inhibitor, frequency (%) | 55 (92) | 10 (53) | 15 (88) | <0.001** |
| PSA, (µg/L) median (SD) | 5.2 (6.0) | 6.3 (5.5) | 5.3 (13.4) | 0.7* |
| Prostate volume (mL), median (SD) | 104 (43) | 100 (44) | 95 (70) | >0.9* |
| Median lobe | 0.6** | |||
| <10 mL, frequency (%) | 4 (6.7) | 1 (5.3) | 1 (6.7) | |
| 10–20 mL, frequency (%) | 26 (43) | 7 (37) | 8 (53) | |
| 20 mL, frequency (%) | 30 (50) | 11 (58) | 6 (40) | |
| BOOI, median (SD) | 61 (33) | 66 (43) | 74 (29) | 0.8* |
| BOOI: bladder outlet obstruction index; SD: Standard deviation. * Kruskal-Wallis test; **Chi-squared test.
A total of 33 of 62 (53%) patients on catheter treatment achieved catheter freedom. At the 24-month follow-up, they continued to enjoy ‘catheter freedom’ and had a median IPSS of 9 points. However, some patients required additional procedures or a repeat PAE, and, in one patient, an open adenoma enucleation (Millin) was necessary because of the large prostate volume (211 mL) (Table 4). The observed reduction in prostate volume after PAE varied significantly across patients, as indicated by the wide range of percentage reductions and standard deviations. At 3 months, the median reduction in prostate volume was 11% in the LUTS group and 27% in the urinary retention group. By 24 months, these reductions increased to 20% and decreased to 9%, respectively, with considerable variability (Tables 3 and 4). The pre-treatment medication profile revealed that most patients were on medication: 68% were taking an α-blocker, and 92% were on a 5α-reductase inhibitor, eight patients did not have pre-medication. Notably, the subgroup of patients who later required further intervention had a different pre-treatment medication profile and a slightly higher median PSA level, suggesting a possible influence of these factors on treatment outcomes (Table 2).
No direct major complications (Clavien-Dindo ≥3) were reported. However, three patients experienced haematuria following the PAE procedure, with cystoscopy revealing bladder cancer in two cases. Six patients presented directly to the urology department within the first 2 days post-PAE, reporting pain and irritative urinary symptoms, which were considered part of post-embolisation syndrome. These patients were managed with a 5-day course of prednisolone at 25 mg/day. DiscussionOur study demonstrated that PAE significantly reduces IPSS scores by a median of 7 points at 24 months post-procedure and achieves catheter freedom in 53% of patients. The procedure was performed efficiently using left radial arterial access. In our study, two primary outcome measures were used to assess the efficacy of PAE: a reduction in the IPSS by three points and the removal of the catheter (catheter freedom). These two measures, while related, reflect different aspects of patient improvement and highlight varying levels of clinical success. The reduction in the IPSS by three points, while widely used in studies as a measure of symptomatic relief, may be considered a weaker measure of success. This MCID does not always capture the full scope of patient improvement and can result in a higher reported success rate, as seen in our study, where 79% of patients achieved this threshold, but it is worth noting that this was achieved by a significant margin, with a median IPSS decrease of 10 points. However, this outcome primarily reflects symptomatic relief rather than significant functional improvements. In contrast, catheter removal (catheter freedom) is a stronger measure of clinical success, particularly in patients with urinary retention. Achieving catheter freedom indicates a meaningful functional improvement in the resolution of urinary retention. In our cohort, 53% of patients who required a catheter pre-PAE achieved catheter freedom at 24 months, reflecting a more substantial clinical outcome. In this group of patients undergoing PAE, a high percentage of patients had been on α-blockers and 5α-reductase inhibitors for a minimum of 6 months before the procedure; only eight patients did not have pre-medication. One may hypothesise that the additive effect of PAE combined with pre-PAE treatment using α-blockers and, more specifically, 5α-reductase inhibitors may improve the clinical success of the procedure. Cardarelli et al. examined the 12-month post-PAE outcomes in patients using 5α-reductase inhibitors during PAE and found that these drugs did not impact the outcomes [27]. By contrast, our findings hint at an additive benefit of PAE in those previously medicated with α-blockers and 5α-reductase inhibitors. This difference in findings raises the question as to whether there is a synergistic effect between PAE and pretreatment medications. If so, this suggests a potential combined advantage that merits further exploration. While no patient has started medication again in the available follow-up data of this group, we believe it is worth considering whether continuing medical treatment after PAE can decrease symptom recurrence and/or reduce the need for additional interventions, especially in fragile patients who are not surgical candidates. The patient group that required additional therapeutic interventions had a median prostate volume of 100 mL (SD = 44 mL). Since the European Association of Urology (EAU) guidelines recommend transurethral surgery for volumes <80 mL [28], an integrated approach involving PAE followed by TURP or other minimally invasive treatment may be considered for those with a larger prostate. However, this requires further validation before being recommended as an alternative to open adenoma enucleation. Bohlok et al. reported that transurethral enucleation of the prostate is a promising treatment option as an alternative to the open approach [29]. The PAE procedure does not require the interruption of anticoagulant treatment because the risk of peri-operative prostate bleeding is inherently not high, but this side effect is known to be a major concern with TURP, even with the latest advances in the techniques to mitigate the bleeding risk [30]. The reduction in prostate volume after PAE is thought to contribute to symptomatic relief by relieving pressure on the urethra [31, 32]. However, in our study, the extent of volume reduction did not always correlate directly with clinical success, as patients with substantial symptomatic improvement did not always show significant reductions in prostate size. This highlights the fact that PAE’s therapeutic effects may be more complex than simple volume reduction, potentially involving other mechanisms. Additionally, the variability in prostate volume reduction (e.g. from minimal changes to reductions as high as 43%) may reflect differences in individual patient anatomy, degree of atherosclerosis, or the technical challenges encountered during the procedure. This suggests that while prostate volume reduction can indicate PAE efficacy, it should be interpreted in the context of other clinical outcomes. Our data touch upon the potential influence of the median lobe on PAE outcomes. Contrary to the prevailing hypotheses, the presence or absence of a pronounced median lobe was not associated with poor clinical outcomes. Our findings appear to differ from those of Yu et al., who found a significant correlation between intravesical prostatic protrusion (IPP) and the outcome of PAE [33]. Although the study by Yu et al. suggests that the presence of the median lobe or IPP plays a negative role in PAE success, our data suggest a more nuanced perspective by implying that the influence of median lobe volume on PAE success may be more complex than previously thought. Differences in patient selection, treatment protocols, or follow-up durations between studies could account for these discrepancies. It is also worth mentioning that other minimally invasive treatment options are known to have a restricted effect on the median lobe [34]. In addition to reduction in prostate volume, the assessment of the IPSS yields additional insights. Notably, a considerable decrease in IPSS following PAE within the group experiencing clinical success signifies reduced symptoms. Nevertheless, there was a progressive increase in IPSS over time for some individuals, which implies the potential resurgence of symptoms or the requirement for supplementary interventions. This pattern is apparent in patients who subsequently underwent transurethral procedures, which underscores the effects of patient attributes and the potential advantages of taking an integrated approach in specific situations. Among the nine cases of technical failure, the primary cause was severe atherosclerosis, which complicated the catheterisation of the prostatic arteries. In retrospect, these patients could have been excluded during the pre-procedural CT angiography, as significant atherosclerosis and tortuosity were apparent but underestimated regarding procedural feasibility. Additionally, one case was due to high body mass index (BMI), which complicated imaging by requiring a higher radiation dose for sufficient imaging quality. Another failure occurred due to extreme arterial tortuosity, with no catheter long enough available at the time. These patients were excluded from the analysis, as they did not undergo a successful embolisation, and thus, an intention-to-treat approach was not applied. Including these patients would not provide insight into the therapeutic efficacy of PAE itself but rather into the feasibility of the procedure in patients with challenging profiles. These findings highlight the need for refining selection criteria during pre-procedural assessment and the steep learning curve associated with this technically demanding procedure. As operators gain more experience, better patient selection and procedural adjustments can help mitigate such challenges in the future. Upon contacting patients for follow-up, some were unreachable, while others declined, feeling well and seeing no need for further visits. Additionally, a few patients faced unrelated health issues, such as two who were diagnosed with bladder cancer and one with a brain tumour. Sadly, two patients passed away – one due to an accident and another from pneumonia 1.5 years post-PAE. These factors contributed to the challenge of maintaining consistent follow-up, which may have influenced the overall assessment of PAE outcomes. While our study shows significant symptom relief short to mid-term post-PAE, longer-term follow-up is necessary to determine the durability of these outcomes and to monitor for any late-onset complications. Strengths and limitationsThis study’s strengths include its single-centre design, which ensures consistency in procedural techniques and follow-up protocols, and radial artery access, which may reduce access-related complications. However, the study’s retrospective nature introduces potential biases, and the 18% loss to follow-up may limit the generalizability of our findings. Additionally, the absence of post-PAE cystometry data restricts our ability to assess changes in bladder dynamics following the procedure fully. Future prospective studies with standardized follow-up protocols are needed to validate these results and better understand the mechanisms underlying PAE’s therapeutic effects. Despite these limitations, this study provides valuable insights into our early experience with introducing a new treatment approach at a single centre. Initially, patients were admitted overnight for observation, but we transitioned to an outpatient model. Many patients had experienced prolonged periods of discomfort and faced long waiting times for surgery, which may have influenced the outcomes and interpretations of our patients. Nevertheless, integrating PAE into our treatment options offers a less invasive alternative that can help reduce surgical wait times and alleviate systemic pressures, particularly in the wake of the COVID-19 pandemic. As more data become available, we anticipate refining patient selection criteria to offer more tailored and effective treatment plans. Although the data for patients who were lost to follow-up initially provided some insights, the absence of subsequent data poses a challenge in drawing definite conclusions from this patient subset. ConclusionsPAE resulted in substantial reduction in IPSS in vast majority of patients with LUTS and catheter freedom in majority of patients who were catheter dependent. This outcome provides a stronger indication of PAE’s therapeutic impact. However, the data described here are preliminary findings and should be interpreted cautiously until more comprehensive research is conducted to confirm and build upon these observations. AcknowledgmentsWe extend our gratitude to the radiology and urology departments at Central Hospital Västerås for their support and contributions to this study. We also give special recognition to our statistician, Philippe Wagner, and our urology research nurse, Taraneh Nikberg, for their invaluable assistance and dedication to this project. Disclosure statementThe authors report no conflicts of interest. FundingThis study was supported by the Centre for Clinical Research (CKF) in Region Västmanland. ORCIDEmir Majbar Jakob Swanberg Farhood Alamdari Abbas Chabok References
| ||||
