ORIGINAL RESEARCH ARTICLE
In pathological T2-prostate cancers a positive surgical margin is associated with adverse psychological outcomes
Rasmus Nilssona  , Tor Åge Myklebustb,c, Thomas Næss-Andresend, Tomm Bernkleve,f, Hege Kerstene,g and Erik Skaaheim Haugh,i,j
, Tor Åge Myklebustb,c, Thomas Næss-Andresend, Tomm Bernkleve,f, Hege Kerstene,g and Erik Skaaheim Haugh,i,j
aDepartment of Urology, Telemark Hospital Trust, Skien, Norway; bDepartment of Registration, Cancer Registry Norway, Norwegian Institute of Public Health, Oslo, Norway; cDepartment of Research and Innovation, Møre and Romsdal Hospital Trust, Ålesund, Norway; dDepartment of Surgery, Division of Urology, Vestre Viken Hospital Trust, Drammen, Norway; eInstitute of Clinical Medicine, Faculty of Medicine, University of Oslo, Oslo, Norway; fDepartment of Research and Innovation, Vestfold Hospital Trust, Tønsberg, Norway; gDepartment of Research, Telemark Hospital Trust, Skien, Norway; hDepartment of Urology, Vestfold Hospital Trust, Tønsberg, Norway; iInstitute of Informatics and Cancer Research, Oslo University Hospital, Oslo, Norway; jDepartment of Clinical Medicine, University of Bergen, Bergen, Norway
ABSTRACT
Objective: A positive surgical margin (PSM) after radical prostatectomy (RP) is considered an adverse surgical feature, but the clinical implications have been debated. Further motive to aim for negative margins (NSM) may be patient anxiety due to PSM, but studies on psychological consequences are lacking. We explored associations between psychological factors and PSM on a consecutive group of men after RP.
Material and methods: Men with suspected prostate cancer were invited to a study on psychological factors. Patient reported outcome measurement questionnaires were collected before diagnosis, and at 6, 12 and 24 months from those who underwent RP. Multivariable mixed models and post hoc pairwise comparisons were used to explore associations between PSM and psychological outcomes.
Results: In total, 387 men had RP and 94 (24%) had PSM. General psychological outcomes were not associated with margin status. At 12 months, men with PSM considered their personal risk of recurrence 6% (30% vs. 24%) points higher and had clinical fear of recurrence (FoR) 14% (47% vs. 33%) more often than men with NSM. In adjusted and stratified analyses, men with pathological (p-)T2-stage and NSM had less FoR than the rest of the sample. In pT3-disease, PSM had no additional impact on FoR. A limitation was the small size of the pT2 group with PSM and the lack of information on how patients were informed.
Conclusions: After RP, PSM was associated with higher FoR, particularly in men with pT2-cancer. Improved counselling and further research into causes and mitigation of FoR, is warranted.
KEYWORDS: Anxiety; fear of recurrence; prostate cancer; radical prostatectomy; surgical margins
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 15–21. https://doi.org/10.2340/sju.v61.45306.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 7 July 2025; Accepted: 18 December 2025; Published: 20 January 2026
CONTACT: Rasmus Nilsson Rasmus.Nilsson@sthf.no Department of Urology, Telemark Hospital Trust, P.O. Box 2900 Kjørbekk, 3710 Skien, Norway
Supplemental data for this article can be accessed online at https://doi.org/10.2340/sju.v61.45306
Competing interests and funding: The authors declare no conflicts of interest or any financial or personal connections related to this study. The study was partly funded by a donation to Telemark Hospital Trust from Tordis Elisabeth Dahl.
Introduction
The incidence of positive surgical margins (PSM) after radical prostatectomy (RP) varies from 6.5 to 32% in contemporary series and depends on histopathological factors, surgical technique and surgeon’s experience [1, 2]. Although PSM is considered an adverse surgical feature, the clinical significance of PSM has been debated [2]. PSM leads to an increased risk of biochemical recurrence (BCR) and salvage radiotherapy [2, 3]. However, regarding clinical progression and prostate cancer (PCa) specific mortality, the impact of PSM seems to diminish after adjusting for more important prognostic factors [3–5]. Nerve-sparing surgery leads to better functional outcomes after RP but increases the risk of PSM [6, 7]. During RP, it is therefore important to find the right balance between functional outcomes and risk of a PSM [7, 8].
Albeit the clinical implications of PSMs might be limited, avoiding anxiety triggered by PSM may be an additional motive to strive for negative surgical margins (NSM). However, studies on the psychological consequences of PSM are scarce. A previous study showed that patients with PSM experienced greater fear of recurrence (FoR) than patients with NSM, and that this difference increased over time [9]. Similarly, in a recent cross-sectional study from our research group, we found that PSM was significantly associated with high FoR [10].
Considering the unclear clinical consequences of PSM and the trade-off with functional outcomes, a deeper understanding of the psychological impact of PSM is important when counseling patients before and after RP. The primary aim of this study was to investigate the associations between margin status and psychological outcomes after RP. The secondary aim was to explore associations between margins and health-related quality of life (HRQoL) and adverse effects.
Material and methods
Design and participants
This prospective observational study was conducted at three community hospitals in the South-Eastern part of Norway. From 2016 to 2021, all men referred with a suspicion of PCa were asked to participate in a larger study on psychological factors in PCa. After consenting, participants filled out a baseline questionnaire prior to prostate biopsy. The study had a real-life design and the normal patient work-flow was not changed because of the study (Supplements F1). In accordance with the routine of the three centers, patients were allocated to an appropriate treatment through shared decision making and in this manuscript we focused on those who underwent immediate (<3 months) RP [11]. During a follow-up visit 6–8 weeks after surgery, these men received information about the pathological examination and their current PSA-levels, and a PSM was reported if malignant cells were in contact with the inked surface of the prostate. At follow-up, information to patients was provided non-standardized at the discretion of the visiting urologist. Likewise, the interval of further clinical follow-up visits and PSA-measurements were not standardized. Additional questionnaires were sent out per mail at 6, 12 and 24 months postoperatively.
Scales and variables
General psychological outcome included the emotional domain score of the EORTC Core Quality of Life questionnaire (EORTC QLQ-C30) version 3.0, fatigue levels measured with the Fatigue Questionnaire, levels of anxiety and depression measured with the Hopkins Symptom Checklist (HSCL-)5 and sleeping problems measured with questions from the population-based Health Survey of Nord-Trøndelag County (HUNT)- surveys [12–15].
Fear of recurrence, defined as ‘fear, worry, or concern relating to the possibility that cancer will come back or progress’, was measured with the Concerns About Recurrence Questionnaire (CARQ-) 5 [16, 17]. CARQ wars originally developed for breast cancer patients and has previously been translated from Danish to Norwegian. Limited validation on PCa-patients has been performed previously and revealed good psychometric properties [10]. The first three items of CARQ-5 assess the frequency, intrusiveness and degree of distress caused by FoR while the fourth and fifth ask the participant to estimate their personal risk of recurrence [17] (Supplements F2). CARQ-items 1–4 produce a sum-score (fear of recurrence-score, 0–40 points) and has a cut-off of 12 points to define clinical fear of recurrence, a level that is considered to have a significant impact on the quality of life of an individual [17]. In this study, self-estimated risk of recurrence (from 0 to 100%) refers to the fourth CARQ-item.
Educational level and comorbidities were measured with HUNT-items [15]. The EuroQoL-5D-5L (VAS-scale) and EORTC QLQ-C30 (global.QoL) were used as measures for general and cancer-specific health-related quality of life (HRQoL) and the Expanded Prostate Index Composite Short Form (EPIC-26) for RP-related adverse effects [12, 18, 19].
Relevant clinical data
Relevant cancer-related variables were captured from medical records. Persistent PSA was defined as a Prostate-specific antigen (PSA) ≥ 0.2 µg/L at first clinical follow-up, BCR as after having two or more postoperative PSA-values ≥ 0.2 µg/L and rising (after having undetectable levels at first follow-up). Pathological T-stage was dichotomized into pT2 and pT3, no participants with pT4 were identified. Treatment failure was defined as having recurrent or persistent PSA or having received postoperative oncological treatment).
Statistical analyses
Descriptive statistics are presented as the frequency and percentage for categorical variables, and as the mean and standard deviation (SD) for continuous variables. Univariate comparisons were made using Chi-square tests for categorical variables and independent sample t-test for continuous variables, Mann–Whitney U-test in case of skewed distribution. Linear mixed models were estimated to model the longitudinal structure of the data, and to adjust for confounders. The models included margin status, follow-up time, and the following a priori selected potential confounders; age, ISUP-score, PSA-level at diagnosis (iPSA), nerve-sparing dissection, pathological T-stage, and lymph node invasion. The models also included interaction effects between margin status and follow-up time. Likelihood ratio tests were used to test for the presence of three-way interactions (margin status, follow-up time and covariate of interest). If there were significant interaction effects models were also estimated stratified by levels of the covariate of interest. Predicted outcomes were calculated from the estimated models, for each combination of margin status and follow-up time, fixing the other covariates to their respective means in the total sample. Results were plotted with corresponding 95% confidence intervals (CI). Analyses were performed using Stata v.18.0.
Ethics
The Regional Committee for Medical and Health Science Research of South-East Norway approved the study (#2016/925). All invited patients received written information and were included after giving informed consent.
Results
Patient characteristics
In the main study, 2,162 (84%) of the 2,562 invited men accepted the invitation. At the time of data collection, 387 men receiving RP had reached the 24 months follow-up and out of those, 96% returned at least one follow-up questionnaire (87%, 86% and 82% at 6, 12 and 24 months, respectively). An attrition analysis showed that the 17 (4%) men not returning any follow-up questionnaire were younger (62.6 years vs. 66.8 years, p = 0.007), had higher iPSA (15.4 µg/L vs. 9.8 µg/L, p = 0.003), poorer emotional domain scores in the EORTC (80 vs. 89, p = 0.020) and more often sleep problems (44% vs. 21 % p = 0.031) compared to the 370 (96%) men returning at least one follow-up questionnaire (Supplements T1).
A total of 94 (24%) men had PSM. Compared to those with NSM, men with PSM less often had nerve-sparing dissection and higher iPSA, ISUP-grades and more advanced pathological stage. During the 24-month follow-up, they more often had persistent PSA, BCR and additional oncological treatment. There were no differences in age, education, Body Mass Index (BMI) or comorbidities between men with PSM and NSM (Table 1).
| Variables | Positive surgical margin N = 94 | Negative surgical margin N = 293 | p-value |
| Age (years) at diagnosis, mean (SD) | 67.4 (6.2) | 66.4 (6.4) | 0.09 |
| Level of education, N (%) ≤12 years >12 years |
52 (57) 40 (43) |
182 (63) 109 (37) |
0.3 |
| BMI (kg/m²), mean (SD) | 27.1 (3.5) | 27.0 (3.4) | 0.4 |
| Comorbidities, N (%) 0–1 2 or more |
73 (78) 21 (22) |
222 (76) 71 (24) |
0.7 |
| iPSA (µg/L), mean (SD) | 12.9 (10.8) | 9.2 (6.0) | <0.01 |
| ISUP on prostate biopsy, N (%) 1 2 3 4 5 |
7 (8) 13 (14) 38 (40) 22 (23) 14 (15) |
15 (5) 90 (31) 102 (35) 55 (19) 31 (10) |
0.029 |
| Uni- or bilateral nerve-sparing surgery, N (%) | 40 (43) | 180 (61) | 0.001 |
| Pathological stage, N (%) pT2 pT3 |
28 (30) 66 (70) |
149 (51) 144 (49) |
<0.001 |
| Pathological lymph node invasion, N (%) | 17 (18) | 18 (6) | <0.001 |
| Persistent PSA, N (%) | 17 (18) | 23 (8) | 0.005 |
| Biochemical recurrence*, N (%) | 19 (20) | 24 (8) | 0.001 |
| Postoperative radiotherapy*, N (%) | 28 (30) | 33 (11) | <0.001 |
| Hormonal treatment*, N (%) | 7 (7) | 10 (3) | 0.097 |
| *occurring anytime during the 24-month follow-up. ISUP: International Society of Urological Pathology score; iPSA: PSA-value at diagnosis; SD: Standard deviation. Statistically significant differences (p < 0.05) are highlighted with bold typeface. |
|||
Psychological outcomes
There were no differences in the emotional domain of EORTC, fatigue, sleep problems or levels of general anxiety and depression between PSM and NSM at any time points (Table 2). All the defined FoR-outcomes were higher among men with PSM, although not always reaching statistical significance (Table 2 and Supplements T2).
In this study, interaction effects were only present for pathological T-stage (Supplements T3). When stratifying for pT-stage, men with pT2 and NSM generally had lower scores on all FoR-outcomes and estimated their likelihood of future recurrence 11.7% – 15.5% lower than men with pT2 and PSM (Table 3). In pT3, we found no significant differences in FoR-scores between PSM and NSM (Table 3).
There were no differences in HRQoL or adverse effects between PSM and NSM. (Supplements T4).
Mixed models analyses
The mixed models-analyses showed that there were no differences in the emotional domain of EORTC QLQ-C30, fatigue, general anxiety and depression or sleep problems between men with PSMs and NSMs (Supplement F3). Figure 1 shows the results for FoR-score (A–C), clinical FoR (D–F) and self-estimated risk of recurrence (G–I), and Table 4 the post hoc pairwise comparisons between PSM and NSM.
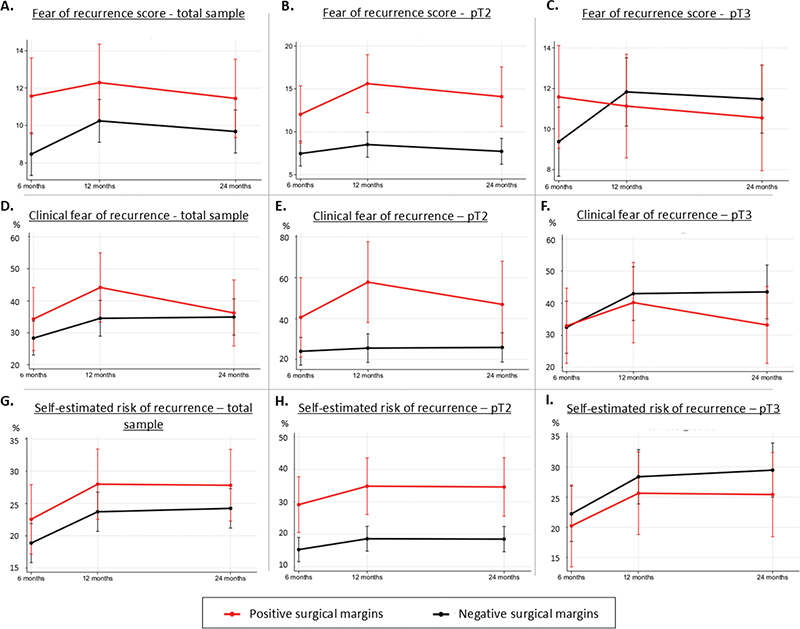
Figure 1. Mixed models-analyses of fear of recurrence-outcomes for the total sample, and stratified for pathological stage. Curves are adjusted for age, ISUP-score, iPSA, nerve-sparing dissection, pT (total sample only) and lymph node invasion. pT=Pathological T-stage.
Men with PSMs had higher FoR-score than NSM, but statistical significance was reached only at 6 months (11.6 points vs. 8.5 points) (Figure 1A, Table 4). Likewise, men with PSM more often had clinical FoR and a higher self-estimated risk of recurrence, but statistical significance was not reached (Figure 1D and 1G). When stratifying for pT-stage, pT2-patients with PSM had higher total FoR-scores (15.6 points vs. 8.5 points, at 12 months), more often clinical FoR (57.8% vs. 25.5%, at 12 months) and a higher self-estimated risk of recurrence (34.8% vs. 18.6%, at 24 months) than pT2-patients with NSM (Figure 1B, E and H, Table 4). In pT3-patients, there were no statistically significant differences in neither of the FoR-outcomes (Figure 1C, F and I).
To investigate whether treatment failure might be mediating the effect of PSM, we added treatment failure variables to the list of covariates in the mixed model analyses. Results changed minimally (Supplements F4). We also performed sensitivity analyses where we sequentially removed patients with treatment failure at each timepoint. Even though the level of FoR seemed lower for patients without treatment failure, differences between PSM and NSM remained (Supplements F5).
Discussion
This study aimed to assess the psychological consequences of having PSM after RP. The analyses showed that men with a PSM neither experienced more emotional distress, fatigue, general anxiety, depression nor sleeping problems. However, men with a PSM had higher FoR, and men with pT2-disease and NSM were less bothered with FoR compared to the total sample.
Contrary to what might be expected, men with a PSM did not have inferior HRQoL-scores or more adverse effects, which is in line with a previous Dutch study [20]. However, even though FoR has been reported as one of the most bothersome consequences of cancer, ordinary instruments measuring HRQoL and general psychological outcome do not seem to capture these specific concerns [21]. FoR seems to be a particular construct separate from ordinary psychological distress, which is confirmed by the lack of correlation with general levels of anxiety and depression in our study [22].
Hong et al. studied the impact of PSM after RP [9], and in line with our results found no differences in general HRQoL-scores or adverse effects, but higher FoR among men with a PSM. Compared to our cohort, their study contained younger patients with less aggressive disease. Hong did not stratify for pathological T-stage but instead adjusted for it in their analysis. Hence, they can only estimate a mean effect of T-stage, while we show that PSM was associated with FoR mostly in pT2-patients.
FoR-scores were quite high for the total sample, except for pT2 with NSM. In total, about one third had clinical FoR, which is comparable with previous studies on both PCa and other cancer forms [23]. A previous cross-sectional study from our research group found that, after adjustment, a PSM doubled the odds of having clinical FoR (OR 2.02, 1.18–3.45), which in this study was comparable only with results of pT2-patients [10]. In pT3, differences in FoR outcomes between PSM and NSM were small. This is an interesting finding, given the greater prognostic effect of PSM in pT3-patients than in pT2-patients. In fact, studies have shown that PSM has an effect on the risk of dying of PCa only when other adverse pathological features, such as pT3, are present [5]. One possible explanation could be that patients in this group perceive that the presence of capsular invasion overshadows the effect of margin.
After sensitivity analyses and adjustment for treatment failure, there was little change in FoR outcome, which means that margin status was associated with FoR independently of whether patients have received postoperative radiation therapy. This indicates a need to be aware that FoR is an issue for men with PSM, regardless of whether they have experienced treatment failure.
The self-estimated risk of recurrence was 15–35%. What kind of recurrence is not specified in the questionnaire, and we do not know if patients visualize this as for instance a rising PSA or a future progression to metastatic disease. In a Swedish registered-based study, the risk of experiencing a BCR within 15 years after RP was 30–46% for intermediate and high-risk cancer, while the risk of dying of PCa after a BCR was 4–9% 10 years after RP [24]. At the same time, another study found that men with PSM had a Hazard Ratio (HR) of 1.75 for developing BCR compared to men with NSM, while pT-stage was a more powerful predictor for BCR (HR: 2.06 for pT3a and 3.26 for T3b, vs. pT2) [25]. Furthermore, a multicenter study found that only among those with adverse clinical or pathological features (including pT3) was PSM associated with mortality [5]. Considering this, the distinct difference in FoR between the pT2-NSM and pT2-PSM patients in our study seems unjustified, which could be due to patients’ overestimation of the prognostic impact of PSM. Previous studies have indeed revealed that men with localized PCa often have misconceptions about their risks and proposed an increased focus on education to mitigate inaccuracies and concerns [26, 27]. In this observational study, patients were informed by their treating urologist during outpatient visits. We do not know how complexities regarding the consequences of PSM were explained, but the wordings at the first follow-up will clearly lay the foundation of the patient’s understanding of his situation, which again affect fears and concerns.
Strengths and limitations
This study’s strengths include the population-based setting, the detailed longitudinal clinical data, the use of validated questionnaires and adjustments for confounding factors. The high inclusion rate and low attrition at follow-up were important. The few (4%) men who refrained from returning any follow-up questionnaire were younger and had higher psychological distress. iPSA was higher in non-repondents while worse clinical characteristics were not confirmed by other factors, that is, ISUP-score or pT-stage. It is well known that anxious participants have higher drop-out rates in studies, and this may have led to an underestimation of psychological distress at follow-up. However, given the low numbers of drop-outs, and the lack of differences in key clinical factors in the attrition analysis, we don’t believe that these biases would change our overall findings.
Although models were adjusted for several covariates, we cannot rule out important unmeasured confounding. For instance, the characteristics of the PSM (i.e. length, location and multifocality) have prognostic implications, but we here treat PSM as a dichotomous variable, which could induce unmeasured bias. When analyzing subgroups, statistical power may have been somewhat low, especially in pT2 disease and PSM (N = 28). A larger sample size could reduce concerns about precision of estimates and the stability of the observed associations. One should interpret findings in light of this and further confirmatory research is required.
Although previous psychometric testing of CARQ on a PCa-sample revealed good performance, no testing has been done to establish a cut-off for clinical FoR in PCa [10]. However, the questions are general in character and previous studies found little difference in FoR-prevalence between breast cancer and PCa [23]. There is therefore little reason to believe that cut-off levels differ radically between these types of cancer and that a slightly different cut-off would change the overall conclusion of this article. Although there is a clear absolute difference in FoR-score between pT2-PSM and pT2-NSM, the clinical significance of this difference could be debated. It is, however, larger than the minimal important difference defined by a commonly used distribution-based method (0.5 SD of FoR-score in T2 at 6 months = 4.9) [28]. Although the other instruments were well validated, earlier adaptations of questionnaires in Nordic PCa-patients have revealed potential validity concerns for patients whose native language is non-Nordic [29].
The pragmatical study design may have induced several biases. Information about PSM and indication for follow-up and adjuvant treatment could differ between clinicians and centers. The first follow-up visit after 6–8 weeks is core to how patients perceive their situation and that postoperative information about the clinical consequences of PSM was not standardized, is a major source of bias and unmeasured confounding. Unbalanced or incorrect information at this timepoint could through an anchoring effect put the patient on the ‘wrong track’ mentally, influencing later questionnaire measurements. Moreover, although Norwegian health authorities impose shared decision making in PCa, we know that only three out of four men want shared decision making, and that this wish is met in only four out of five men [30]. Furthermore, patients might find information from other sources, such as media or social acquaintances. How the use of standardized and quality-assured information affects psychological reactions after RP is unknown, and future interventional studies would be informative. Moreover, apart from the first postoperative visit, the timing of later PSA-measurements was not standardized, and we do not know how this aligned with the collection of questionnaires, which is a weakness. Furthermore, the first postoperative assessment was at 6 months after surgery, meaning that several months had passed after patients had been informed about the pathological result. A measurement of the acute reaction to this information is lacking and is important to include in future studies.
Conclusion
This study found that a PSM was not associated with overall psychological distress, except for higher levels of FoR in men with pathological T2-cancer. This finding contrasts with the clinical impact of margin status and suggests that some patients may overestimate their actual risk. This implies a need for improved patient education on the true prognostic implications. Future studies should explore underlying drivers of FoR, and examine whether standardized, quality-assured information could alleviate unnecessary concerns.
References
- [1] Novara G, Ficarra V, Mocellin S, et al. Systematic review and meta-analysis of studies reporting oncologic outcome after robot-assisted radical prostatectomy. Eur Urol. 2012;62(3):382–404. https://doi.org/10.1016/j.eururo.2012.05.047
- [2] Yossepowitch O, Briganti A, Eastham JA, et al. Positive surgical margins after radical prostatectomy: a systematic review and contemporary update. Eur Urol. 2014;65(2):303–313. https://doi.org/10.1016/j.eururo.2013.07.039
- [3] Mauermann J, Fradet V, Lacombe L, et al. The impact of solitary and multiple positive surgical margins on hard clinical end points in 1712 adjuvant treatment-naive pT2-4 N0 radical prostatectomy patients. Eur Urol. 2013;64(1):19–25. https://doi.org/10.1016/j.eururo.2012.08.002
- [4] Mithal P, Howard LE, Aronson WJ, et al. Positive surgical margins in radical prostatectomy patients do not predict long-term oncological outcomes: results from the Shared Equal Access Regional Cancer Hospital (SEARCH) cohort. BJU Int. 2016;117(2):244–248. https://doi.org/10.1111/bju.13181
- [5] Pellegrino F, Falagario UG, Knipper S, et al. Assessing the impact of positive surgical margins on mortality in patients who underwent robotic radical prostatectomy: 20 years’ report from the EAU Robotic Urology Section Scientific Working Group. Eur Urol Oncol. 2024;7(4):888–896. https://doi.org/10.1016/j.euo.2023.11.021
- [6] Auffenberg GB, Qi J, Dunn RL, et al. Evaluation of patient- and surgeon-specific variations in patient-reported urinary outcomes 3 months after radical prostatectomy from a statewide improvement collaborative. JAMA Surg. 2021;156(3):e206359. https://doi.org/10.1001/jamasurg.2020.6359
- [7] Vis AN, van den Bergh RCN, van der Poel HG, et al. Selection of patients for nerve sparing surgery in robot-assisted radical prostatectomy. BJUI Compass. 2022;3(1):6–18. https://doi.org/10.1002/bco2.115
- [8] Moris L, Gandaglia G, Vilaseca A, et al. Evaluation of oncological outcomes and data quality in studies assessing nerve-sparing versus non-nerve-sparing radical prostatectomy in nonmetastatic prostate cancer: a systematic review. Eur Urol Focus. 2022;8(3):690–700. https://doi.org/10.1016/j.euf.2021.05.009
- [9] Hong YM, Hu JC, Paciorek AT, et al. Impact of radical prostatectomy positive surgical margins on fear of cancer recurrence: results from CaPSURE. Urol Oncol. 2010;28(3):268–273. https://doi.org/10.1016/j.urolonc.2008.07.004
- [10] Nilsson R, Næss-Andresen TF, Myklebust T, et al. Fear of recurrence in prostate cancer patients: a cross-sectional study after radical prostatectomy or active surveillance. Eur Urol Open Sci. 2021;25:44–51. https://doi.org/10.1016/j.euros.2021.01.002
- [11] Norwegian Health Directorate. Nasjonalt handlingsprogram med retningslinjer for diagnostikk, behandling og oppfølging av prostatakreft. Oslo: Helsedirektoratet (last updated 02.10.2024, read 07.07.2025) Available from: www.helsedirektoratet.no/retningslinjer/prostatakreft-handlingsprogram
- [12] Aaronson NK, Ahmedzai S, Bergman B, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365–376. https://doi.org/10.1093/jnci/85.5.365
- [13] Chalder T, Berelowitz G, Pawlikowska T, et al. Development of a fatigue scale. J Psychosom Res. 1993;37(2):147–153. https://doi.org/10.1016/0022-3999(93)90081-P
- [14] Tambs K, Moum T. How well can a few questionnaire items indicate anxiety and depression? Acta Psychiatr Scand. 1993;87(5):364–367. https://doi.org/10.1111/j.1600-0447.1993.tb03388.x
- [15] Langhammer A, Bratberg G, Heggland J, et al. Cohort profile: the HUNT Study, Norway. Int J Epidemiol. 2012;42(4):968–977. https://doi.org/10.1093/ije/dys095
- [16] Lebel S, Ozakinci G, Humphris G, et al. From normal response to clinical problem: definition and clinical features of fear of cancer recurrence. Support Care Cancer. 2016;24(8):3265–3268. https://doi.org/10.1007/s00520-016-3272-5
- [17] Thewes B, Zachariae R, Christensen S, et al. The Concerns About Recurrence Questionnaire: validation of a brief measure of fear of cancer recurrence amongst Danish and Australian breast cancer survivors. J Cancer Surviv. 2015;9(1):68–79. https://doi.org/10.1007/s11764-014-0383-1
- [18] Group TE. EuroQol – a new facility for the measurement of health-related quality of life. Health Policy (Amsterdam, Netherlands). 1990;16(3):199–208. https://doi.org/10.1016/0168-8510(90)90421-9
- [19] Szymanski KM, Wei JT, Dunn RL, et al. Development and validation of an abbreviated version of the expanded prostate cancer index composite instrument for measuring health-related quality of life among prostate cancer survivors. Urology. 2010;76(5):1245–1250. https://doi.org/10.1016/j.urology.2010.01.027
- [20] Richters A, Derks J, Husson O, et al. Effect of surgical margin status after radical prostatectomy on health-related quality of life and illness perception in patients with prostate cancer. Urol Oncol. 2015;33(1):16.e9–16.e15. https://doi.org/10.1016/j.urolonc.2014.10.006
- [21] Baker F, Denniston M, Smith T, et al. Adult cancer survivors: how are they faring? Cancer. 2005;104(11 Suppl):2565–2576. https://doi.org/10.1002/cncr.21488
- [22] Simard S, Savard J, Ivers H. Fear of cancer recurrence: specific profiles and nature of intrusive thoughts. J Cancer Surviv. 2010;4(4):361–371. https://doi.org/10.1007/s11764-010-0136-8
- [23] Luigjes-Huizer YL, Tauber NM, Humphris G, et al. What is the prevalence of fear of cancer recurrence in cancer survivors and patients? A systematic review and individual participant data meta-analysis. Psycho-oncology. 2022;31(6):879–892. https://doi.org/10.1002/pon.5921
- [24] Falagario UG, Abbadi A, Remmers S, et al. Biochemical recurrence and risk of mortality following radiotherapy or radical prostatectomy. JAMA Netw Open. 2023;6(9):e2332900. https://doi.org/10.1001/jamanetworkopen.2023.32900
- [25] Pompe RS, Bandini M, Preisser F, et al. Contemporary approach to predict early biochemical recurrence after radical prostatectomy: update of the Walz nomogram. Prostate Cancer Prostatic Dis. 2018;21(3):386–393. https://doi.org/10.1038/s41391-018-0033-1
- [26] Kendel F, Helbig L, Neumann K, et al. Patients’ perceptions of mortality risk for localized prostate cancer vary markedly depending on their treatment strategy. Int J Cancer. 2016;139(4):749–753. https://doi.org/10.1002/ijc.30123
- [27] van Stam MA, van der Poel HG, van der Voort van Zyp JRN, et al. The accuracy of patients’ perceptions of the risks associated with localised prostate cancer treatments. BJU Int. 2018;121(3):405–414. https://doi.org/10.1111/bju.14034
- [28] Revicki D, Hays RD, Cella D, et al. Recommended methods for determining responsiveness and minimally important differences for patient-reported outcomes. J Clin Epidemiol. 2008;61(2):102–109. https://doi.org/10.1016/j.jclinepi.2007.03.012
- [29] Ettala O, Bergroth R, Riikonen J, et al. Cross-cultural adaptation and psychometric validation of the Finnish version of the Expanded Prostate Cancer Index Composite-26 for patients with localised prostate cancer. Scand J Urol. 2025;60:151–157. https://doi.org/10.2340/sju.v60.44371
- [30] Otrebski Nilsson M, Aas K, Myklebust T, et al. Do all prostate cancer patients want, and experience shared decision making prior to curative treatment? Scand J Urol. 2023;58:133–140. https://doi.org/10.2340/sju.v58.14730