ORIGINAL RESEARCH ARTICLE
Lower urinary tract symptoms in adults treated for vesico-ureteral reflux in childhood, compared to healthy controls
Thora S. Olafsdottira,b,c, Sofia Tjernlundb, Göran Läckgrenb, Nele Brusselaersd and Tryggve Nevéusc
aLandspitali University Hospital, Reykjavik, Iceland; bSection of Pediatric Urology, University Childrens Hospital, Uppsala, Sweden; cDepartment of Women’s and Children’s Health, Uppsala University, Uppsala, Sweden; dDepartment of Family Medicine and Population Health, Global Health Institute, University of Antwerp, Antwerp, Belgium
ABSTRACT
Objective: To compare lower urinary tract (LUT) symptoms in young adults with previous vesico-ureteral reflux (VUR) and healthy controls. VUR is associated with LUT dysfunction in childhood but less is known regarding long-term prognosis.
Methods: A questionnaire was sent to 483 patients treated for VUR between 1993 and 2003 and a control group of 640 healthy young adults. Presence of LUT symptoms was compared by multivariable logistic regression, expressed as odds ratios (OR) and 95% confidence intervals (CI), adjusting for age, sex and body mass index.
Results: The study group response rate was 45.8% versus 41.9% in the controls. There was a female predominance in both groups (77.9% and 74.3%). Overall, 45.7% of the VUR group and 36.6% of the controls reported current LUT symptoms, with unspecified LUT symptoms being significantly more common (OR = 3.86, 95% CI 1.33–11.20), especially in individuals receiving single treatment, and women without a pregnancy history. A larger proportion of those with VUR reported childhood incontinence than controls (20.4% vs. 5.6%, OR = 3.86, 95% CI 1.53–9.79). There were no significant differences in pyelonephritis during adulthood, yet individuals in the VUR group tended to have a higher micturition frequency than controls.
Conclusion: Bladder dysfunction related to VUR during early childhood seems to improve with age, yet voiding difficulties and a higher micturition frequency appeared to remain more common in adults, predominantly females, with previous reflux than in healthy controls.
KEYWORDS: Vesicoureteral reflux; LUT symptoms; NASHA-dx copolymer; long-term follow up study; enuresis; incontinence; pyelonephritis
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 8–14. https://doi.org/10.2340/sju.v60.45318.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 8 October 2025; Accepted: 18 December 2025; Published: 14 January 2026
CONTACT: Thora S. Olafsdottir thora.sif.olafsdottir@kbh.uu.se Department of Women’s and Childrens Health, Uppsala University, Akademiska Sjukhuset 75185 Uppsala, Sweden
Supplemental data for this article can be accessed online at https://doi.org/10.2340/sju.v61.45318
Competing interests and funding: H.R.H. Crown Princess Lovisa Society for Child Care (Dnr: 2022-00711).
Gillbergska Stiftelsen.
Study sponsors had no involvement in the study.
Introduction
Vesico-ureteral reflux (VUR) is a common urinary tract malformation with potential serious long-term consequences such as renal scarring, hypertension and, rarely, end-stage renal disease. The condition allows for retrograde flow of urine from the bladder to the kidneys with consequent pressure increase in the pelvis and calyces, which can damage the renal parenchyma in combination with bacteria from the bladder [1, 2].
Primary reflux implies an absence of the physiological sphincter created by the oblique insertion of the ureter through the bladder wall that prevents retrograde urine flow. Secondary reflux, on the other hand, is caused by increased bladder pressure that overrides the physiological sphincter mechanism in conditions such as neurogenic bladder or posterior urethral valves.
It is difficult to know the exact prevalence of VUR, as many children can be symptom-free and thus never diagnosed, but studies have estimated the prevalence from 0.4% to 1.8% in the general population [3] whereas in young children presenting with urinary tract infection (UTI) it can be as high as 30–50% [4].
The severity of VUR is graded from one to five according to the International Reflux Study Committee standards/system [5]. It is well established that grade I–II VUR does not have serious consequences and is thus not an indication for active treatment [6] while grade III–V is associated with recurrent UTIs and reflux nephropathy [7].
The main objective of management of VUR grade III–V is preservation of renal function. Historically, most patients were treated with open bladder surgery, that is, ureteral reimplantation, but with increasing evidence there has been a clear trend toward a more conservative approach ranging from watchful waiting through antibiotic prophylaxis to endoscopic treatment with sub-ureteric bulking agents [8]. There is a lack of high quality evidence supporting current guidelines since most studies are retrospective in nature [8].
Using the terminology recommended by the International Children’s Continence Society (ICCS) [9] lower urinary tract (LUT) symptoms are either storage symptoms, voiding symptoms or other symptoms. The most common LUT symptoms in children are the storage symptoms urgency, holding maneuvers, enuresis and daytime incontinence [10].
The relationship between LUT dysfunction and VUR is complicated and not yet fully understood. Some studies have shown cystometric abnormalities such as detrusor overactivity or detrusor-sphincter discoordination in over half of infants with VUR [11–13], while other authors have shown equal prevalence of detrusor overactivity in children with and without VUR [14]. A balanced conclusion is that LUT dysfunction is indeed overrepresented in infants and young children with VUR but that the causal relationship, if any, is unclear. There seem to be two patterns of LUT symptoms prevailing in children with VUR. Either a small, uncompliant and overactive bladder or a large bladder with incomplete emptying and discoordinated voiding. The former is more common in boys and the latter in girls. This might be a partial explanation to why girls with VUR have greater risk for recurrent UTIs and acquired renal scarring than boys [10].
Nevertheless, there is little knowledge about the long-term risks for continued LUT disturbance in adulthood [12]. Thus, the aim of this study was to investigate the prevalence of LUT symptoms in adulthood in previously treated VUR patients, compared to a healthy control population.
Subjects and methods
This is a sub-study of a large cohort of 483 consecutive patients treated for VUR grade III-IV between 1 May 1993 and 30 April 2003 at a tertiary pediatric clinic [15, 16] serving a population of approximately 3 million inhabitants. All patients in this cohort were diagnosed with dilating VUR (grade III-IV) after pyelonephritis, defined as positive culture, high fever and high CRP. Exclusion criteria were neurogenic bladder, VUR grade V and urinary tract malformations such as posterior urethral valves. VUR grade V was excluded due to differences in treatment regimens at that time compared to treatment for grade III-IV. The work-up was performed in pediatric departments in local hospitals under the guidance of the tertiary clinic and included ultrasound, renal scintigraphy and VCUG (most often after recurrent febrile UTI). After the diagnosis of dilating VUR was made they were given long-term antibiotic prophylaxis for at least 1 year (median 24 months) and a new investigation with VCUG, ultrasound and renal scintigraphy was performed. If the child had persistent VUR of grade III–IV she or he was referred to the tertiary hospital for endoscopic treatment with sub-ureteric injection of dextranomer/hyaluronic acid (Deflux®). The cohort thus consists of endoscopically treated, persistent VUR patients, after at least 1 year of conservative management. Due to the regional organization we do not have information on the patients with spontaneous resolution of VUR.
The present investigation is part of a larger study of the cohort’s overall health and gut microbiome status. At the time of participation in this study all subjects were > 18 years of age.
The cohort received a non-validated questionnaire asking about LUT symptoms, and other concurrent disorders. Aside from this study, the questionnaire was designed to be used in other substudies mentioned above as well, with an assortment of questions of which only some were used in this substudy (see Appendix A). The questionnaire was assembled from a priori study protocols previously approved by the Swedish Ethics Authority [15]. Questions regarding the LUT symptoms were from an original follow-up protocol from the start of the cohort which the families, and patients, depending on their age, had answered previously (See Appendix A).
An invitation to participate along with the questionnaire and informed consent was sent via mail to all 483 patients. After two mail contacts, non-responders received phone calls and then finally extra reminders were sent out with text messages. No reimbursement was offered to participants (see Figure 1).
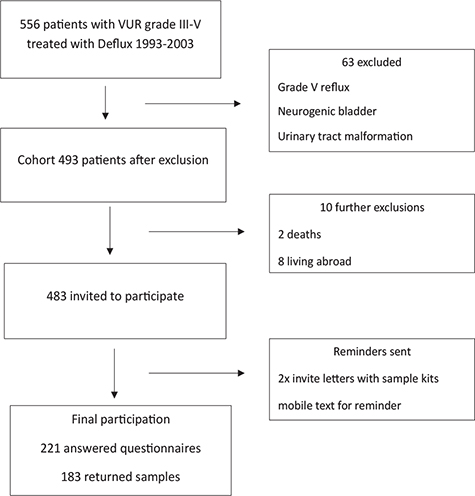
Figure 1. Inclusion process in VUR Cohort.
The control group consisted of 640 students at Uppsala University. They received a modified version of the questionnaire with focus only on bladder symptoms. The total number of students that was reached out to in these groups is estimated to 640 people.
To ensure anonymity, given the sensitive nature of some of the questions, participants only reported their age in intervals. Exclusion criteria in the control group were malformations of the urinary tract that could affect bladder function. Conditions such as duplex ureters were not considered exclusion criteria in either group.
Lower urinary tract outcome variables
We assessed different LUT symptoms and categorized them by timing of symptoms, type of symptoms and severity. Pyelonephritis was defined as febrile, culture-positive UTI, subcategorized as < 15 years, ≥ 15 years, overall and during pregnancy, and considered the most severe outcome. Childhood incontinence was defined as involuntary wetting at day or night and was considered the most easily remembered symptom of LUT disturbance during childhood. Among current LUT symptoms high micturition frequency was defined as voiding more than 8 times daily and low micturition frequency under 4 times daily (frequencies between 4 and 8 being considered normal). The following current storage symptoms were assessed: urgency, daytime incontinence and enuresis. Urgency was defined as having to hurry to the toilet. The questionnaire also gave the option to state that there were unspecified LUT problems. Presence of UTI both febrile and non-febrile were reported for the last year, and during pregnancy. Data regarding UTIs were self-reported in the control group and extracted from The Swedish Patient Registry in the VUR group. This was mainly for practical reasons since it would have been more difficult to recruit that many controls if we had to ask for permission to go through their patient files. All variables were dichotomized as yes or no. A composite outcome was constructed combining current unspecified LUT problems and storage symptoms (urgency, daytime incontinence and enuresis).
Statistical analyses were performed using Stata v.14 (Stata Corporation). Multivariable logistic regression was used to adjust for confounding by age, sex and body mass index (BMI) and results presented as odds ratios (OR) and 95% confidence intervals (CI). Assuming a prevalence of urinary incontinence of 25% in the general population [17, 18] and that we do not wish to miss a true increased prevalence among the test population of 15%, with a power of 0.8 and a significance level of 95%, at least 165 subjects are needed in each group.
The study was approved by the Swedish ethical authority (reg no 2020-00542) and performed in accordance with the Helsinki protocol.
Results
Among the former VUR patients, 221 answers were received, yielding a response of 45.8%. Of these subjects, 77.8% were female (Table 1). The majority was treated successfully with single endoscopic Deflux injection treatment (64.3%), 21.3% with repeated endoscopic Deflux-injection treatment and 14.5% went to open surgery after failed treatment. A non-responder analysis was performed among the former VUR patients regarding sex, reflux grade and time at first diagnosis. In women 54.9% responded compared to 28.8% of the men. There was no difference in reflux grade in women but there was a significantly higher reflux grade in the men who participated compared to those who did not. No difference was found in time to diagnosis and treatment between the responders and non-responders.
| Controls | Vesico-ureteral reflux | |
| N (%) | N (%) | |
| Total | 268 (100) | 221 (100) |
| Sex | ||
| Men | 68 (25.4) | 49 (22.2) |
| Women | 199 (74.3) | 172 (77.8) |
| Other | 1 (0.4) | 0 (0.0) |
| Women with prior pregnancy | 12 (6.0) | 59 (34.3) |
| Age, in years | ||
| 18–22 | 184 (68.7) | 14 (6.3) |
| 23–27 | 70 (26.1) | 53 (24.0) |
| ≥ 28 | 14 (5.2) | 154 (69.7) |
| Body mass index, in kg/m2 | ||
| < 20.0 | 38 (14.2) | 34 (15.4) |
| 20.0–24.9 | 184 (68.7) | 100 (45.3) |
| ≥ 25.0 | 42 (15.7) | 80 (36.3) |
| Missing | 4 (1.5) | 7 (3.2) |
| Treatment | ||
| Single treatment* | - | 142 (64.3) |
| Open bladder surgery after one or more treatments | - | 32 (14.5) |
| Re-retreatment** | - | 47 (21.3) |
| *single treatment = one injection of Deflux. **retreatment = second or third injection of Deflux after failed first treatment. |
||
Among the participating controls, one person had received endoscopic treatment for VUR in childhood and was therefore excluded from the analysis, resulting in 268 participants (41.9%), of which 74.3% were female. One person identified themselves as ‘other’ and was excluded from statistical analyses regarding biological sex. The controls were younger than the VUR group (68.7% vs. 6.3% younger than 20 years), and a higher proportion in the VUR group had a BMI above 25 (26.3% vs. 15.7%). The proportion of previous pregnancies was 6.0% in the control group (Table 1).
Pyelonephritis
As described above, all reflux patients had a history of pyelonephritis in infancy and their VUR grade ranged from III to IV, since patients with grade V were not included in the study. Thirteen (4.9%) of the control subjects had had a pyelonephritis in childhood (Tables 2, 3).
| Control group | VUR group | VUR group | |||||||
| Total | Male | Female | Total | Male | Female | Single treatment | Open bladder surgery | Retreatment | |
| N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | |
| Total N | 268 | 68 | 199 | 221 | 49 | 172 | 142 | 32 | 47 |
| LUT symptoms | |||||||||
| Pyelonephritis | |||||||||
| < 15 years | 13 (4.9) | 2 (2.9) | 11 (5.5) | 221 (100.0) | 49 (100.0) | 172 (100.0) | 142 (100.0) | 32 (100.0) | 47 (100.0) |
| ≥ 15 years | 21 (7.8) | 2 (2.9) | 19 (9.6) | 35 (15.8) | 2 (4.1) | 33 (19.2) | 23 (16.2) | 6 (18.8) | 6 (12.8) |
| Any | 27 (10.1) | 3 (4.4) | 24 (12.1) | 221 (100.0) | 49 (100.0) | 172 (100.0) | 142 (100.0) | 32 (100.0) | 47 (100.0) |
| Urinary tract infection last year* | 28 (10.5) | 2 (2.9) | 25 (12.6) | 15 (6.8) | 1 (2.0) | 14 (8.1) | 9 (6.3) | 3 (9.4) | 3 (6.4) |
| Childhood incontinence/enuresis | 15 (5.6) | 4 (5.9) | 11 (5.5) | 45 (20.4) | 13 (26.5) | 32 (18.6) | 28 (19.7) | 6 (18.8) | 11 (23.4) |
| Micturition frequency (daily) | |||||||||
| Low (< 4) | 35 (13.1) | 8 (11.8) | 27 (13.6) | 21 (9.5) | 9 (18.4) | 12 (7.0) | 8 (5.6) | 6 (18.8) | 7 (14.9) |
| Normal (4–8) | 222 (82.8) | 60 (88.2) | 161 (80.9) | 172 (77.8) | 39 (79.6) | 133 (77.3) | 113 (79.6) | 24 (75.0) | 35 (74.5) |
| High (> 8) | 11 (4.1) | 0 (0.0) | 11 (5.5) | 28 (12.7) | 1 (2.0) | 27 (15.7) | 21 (14.8) | 2 (6.3) | 5 (10.6) |
| Current LUT symptoms | |||||||||
| Unspecified LUT problems | 12 (4.5) | 3 (4.4) | 9 (4.5) | 36 (16.3) | 2 (4.1) | 34 (19.8) | 21 (14.8) | 5 (15.6) | 10 (21.3) |
| Urgency | 73 (27.2) | 11 (16.2) | 61 (30.7) | 84 (38.0) | 10 (20.4) | 74 (43.0) | 56 (39.4) | 13 (40.6) | 15 (31.9) |
| Daytime incontinence | 4 (1.5) | 2 (2.9) | 2 (1.0) | 16 (7.2) | 0 (0.0) | 16 (9.3) | 8 (5.6) | 4 (12.5) | 4 (8.5) |
| Current enuresis | 3 (1.1) | 0 (0.0) | 3 (1.5) | 3 (1.4) | 1 (2.0) | 2 (1.2) | 2 (1.4) | 0 (0.0) | 1 (2.1) |
| At least 1 | 98 (36.6) | 15 (22.1) | 82 (41.2) | 101 (45.7) | 11 (22.5) | 90 (52.3) | 67 (47.1) | 16 (50.0) | 18 (38.3) |
| During prior pregnancy | |||||||||
| Urinary tract infection | - | - | 1 (8.3) | - | - | 18 (30.5) | 9 (21.4) | 3 (50.0) | 6 (54.6) |
| Pyelonephritis | - | - | 1 (8.3) | - | - | 1 (1.7) | 1 (2.4) | 0 (0.0) | 0 (0.0) |
| *included both febrile and non-febrile infections. | |||||||||
The proportion of pyelonephritis in adulthood (after 15 years) was 15.8 % in the VUR group, while 7.8 % among controls. In the VUR group, pyelonephritis in adulthood was most common in women (19.2% vs. 4.1% in men); and in the subgroup receiving open bladder surgery (18.8%) (Table 2). The prevalence was higher among those with a previous pregnancy (25.4% compared to 15.9%) (Table 3). However, after adjustment for age, sex and BMI the odds of pyelonephritis in adulthood did not differ between those with VUR and the controls (OR = 0.94, 95% CI: 0.37–2.38) (Table 4). UTIs during the last year were more common among controls (10.5% vs. 6.8% in VUR group); resulting in an OR of 0.80 (0.29–2.22) for VUR compared to controls (Table 4).
Childhood LUT symptoms
Childhood enuresis and incontinence were clearly more frequent in the VUR group (20.4%) than the controls (5.6%), with limited differences between the VUR-treatment groups (Table 2). After adjustments for age, sex and BMI the odds were strongly increased in the VUR group compared to the controls (OR = 3.86, 95% CI 1.53–9.79) (Table 4).
Micturition frequency
Micturition frequency was normal in 77.8% of the VUR group and 82.8% of the controls. Low micturition frequency was more common in controls (13.1% vs. 9.5% in the VUR group); while high micturition frequency was more commonly reported among the VUR group (12.7% vs. 4.1% among controls). In the VUR group, high micturition frequency was particularly common in women (15.7% vs. 2.0% in men); and those receiving single treatment (14.8% vs. 6.3% with open bladder surgery) (Table 2). High micturition frequency was more common in women with pregnancy history than those without, both in the VUR group and the controls (Table 3). After adjustments, low micturition frequency was less common in the VUR group (OR = 0.83), while high micturition frequency was more common (OR = 1.92), yet this did not reach statistical significance (Table 4).
Other current LUT symptoms
Overall, 45.7% of the VUR group reported current LUT symptoms, compared to 36.6% of the controls. Unspecified LUT problems (16.3%), urgency (38.0%) and daytime incontinence (7.2%) were more common in the VUR group than in controls (4.5%, 27.3% and 1.5%, respectively) (Table 2). There was no difference in enuresis (1.1–1.4% in both groups) (Table 2, 4). In the VUR group, unspecified LUT problems were more common in women (19.8%) than men (4.1%); and in those with re-treatment (21.3% compared to 14.8% after single treatment). Urgency was twice as common in women than men in both the VUR group and controls and most common after open bladder surgery (40.6%). After adjustments, unspecified LUT problems were clearly more common in the VUR group than the controls (OR = 3.86, 95% CI 1.33–11.20), particularly in the group receiving single treatment and women (overall and without a pregnancy history) (Table 4). The more specific LUT symptoms were not significantly more common in the VUR than the controls, with the composite outcome showing an OR = 1.32 (95% CI 0.75–2.35).
Pregnancy history
Among the VUR group, the differences between women with and without a pregnancy history were limited, with only pyelonephritis being more commonly reported (25.4% vs. 15.9%) (Table 3). In the controls, pyelonephritis, unspecified LUT problems and daytime incontinence were more common among those with a pregnancy history than those without (Table 3). UTI during pregnancy was reported by 30.5% of the VUR group, and 8.3% of the controls, while only 1 case with VUR and 1 control reported a pyelonephritis during pregnancy. Power was too limited to compare these risks with regression modeling.
Discussion
This is one of the first studies to compare LUT symptoms in adulthood in previously treated VUR patients to healthy young adults. Our results were reassuring, with our main findings that LUT symptoms in childhood linked to VUR generally disappear in adulthood. As previously reported by Sillen et al. and Läckgren et al. [19, 20] females seem to have greater LUT problems than men even if no statistical difference was achieved in our study.
It is worth mentioning that current indications for endoscopic injection treatment of VUR have become stricter than before and repeat VCUG is only indicated in repeat UTIs [21]. In our study, subjects with previous VUR had more LUT problems suggesting detrusor overactivity, such as incontinence, urgency or high micturition frequency than the control group. There were concerns regarding confounding factors since the control group differed in age, BMI and pregnancy rates. All differences in current symptoms except unspecified micturition problems disappeared when correcting for potential confounders such as age, sex, BMI and previous pregnancy. Unspecified micturition symptoms were more common in females previously treated for VUR compared to men in same group. One clear difference between the groups, however, was that subjects with previous VUR had higher risk for incontinence during childhood. Our interpretation of these results is that the bladder dysfunction that is often observed in children with VUR seems to slowly improve with age even though it does not do so immediately after correction of the reflux.
Also, although subjects with previous VUR had a higher risk for pyelonephritis in adulthood, this difference disappeared when adjusted for age, sex and BMI. This is also reassuring. It would be interesting to elucidate with urodynamic studies which LUT pattern (detrusor overactivity or incomplete emptying) predominates in those who still did have UTIs in adulthood, but that is beyond the scope of this article.
Our findings of a handful of adults with current enuresis, both among the previous VUR patients and the controls, agrees with current knowledge about enuresis prevalence in adults [22]. This underlines the fact that enuresis is not just a pediatric condition that always remits spontaneously before adulthood. Enuresis, although somatically benign, is not a trivial disorder, especially not among adults among whom it may severely inhibit social well-being and quality of life. But our data suggest there is no relationship between VUR and enuresis.
Only a few other studies have been performed addressing the long-term prognosis of reflux-associated LUT dysfunction, but the Swedish reflux study group has recently published long-term follow-up data, which are largely in agreement with our results [19]. They found that the prevalence of bladder and bowel dysfunction decreased with age; at the end of the study the prevalence was 37% and dropped to 23% at 10 year follow-up. Interestingly, at the 10 year follow-up LUT symptoms were mainly present in girls whereas at the end of the study the sex distribution was even. The same trend was also observed in the proportion of UTIs, which supports the theory that the LUT dysfunction correlates with the development of UTI in adolescence but in infancy other factors are crucial for UTI risk.
Limitations of this study include its retrospective nature and design with questionnaires, contributing to both selection and recall bias. The response proportion is just under 50%, although if we exclude the men, the proportion goes up to just under 60%. The lower response rate of men may limit the generalizability of our results. For ethical reasons, we could not look into the patient files of subjects not responding to the questionnaires and check if there were any healthcare appointments due to UTIs and/or LUT problems in this subgroup. The age distribution and pregnancy history differed between the previous VUR patients and the controls, with the controls being younger and having had fewer pregnancies but we compensated for this in the analyses. The questions posed in the questionnaire were not part of validated packages but chosen to conform to previous studies in the same cohort in hope of increasing participation and to fit into the requirements of concurrent and planned studies related to the patients’ microbiome and general health. In retrospect, we would have chosen slightly different and more specific questions regarding micturition habits and LUT symptoms. Specifically, we would have avoided asking for unspecified bladder problems without further clarifications. Compromises were made in formulating certain questions for controls to avoid sensitive issues such as daytime incontinence and certain issues such as abortions were avoided completely in order not to diminish response rate.
Furthermore, no objective bladder function data or urodynamics such as voided volumes, residual urine and flowmetry, were included since this was a part of a larger project in which it was not possible to include such data. This would make an interesting follow-up study.
Conclusion
VUR in infancy and early childhood is linked to symptoms of LUT dysfunction in later childhood but these problems tend to disappear before adulthood. It seems that females previously treated for VUR in childhood are more prone to LUT symptoms compared to males. These results provide general reassurance but at the same time warrant continued awareness of LUT symptoms in adulthood especially in females. Enuresis among adults is not rare.
Acknowledgments
Anna Bohlin and Birgitta Karanikas for assistance regarding sending out invites to VUR group and contacting nonresponders by telephone and text messages.
Ethical approval
The study was approved by the Regional Ethical Committée Dnr 2020-00542.
Declaration of Generative AI and AI-assisted technologies in the writing process
The authors did not use generative AI or AI-assisted technologies in the development of this manuscript.
References
[1] Stokland E, Hellström M, Jacobsson B, et al. Renal damage one year after first urinary tract infection: role of dimercaptosuccinic acid scintigraphy. J Pediatr. 1996;129(6):815–820. https://doi.org/10.1016/S0022-3476(96)70024-0
[2] Goren MR, Ceyhan E, Ozer C, et al. The impact of reflux pressure on renal scarring in children with sterile vesicoureteral reflux. J Pediatr Urol. 2023;19(1):130.e1–130.e5. https://doi.org/10.1016/j.jpurol.2022.09.016
[3] Sargent MA. What is the normal prevalence of vesicoureteral reflux? Pediatr Radiol. 2000;30(9):587–593. https://doi.org/10.1007/s002470000263
[4] Hannula A, Venhola M, Renko M, et al. Vesicoureteral reflux in children with suspected and proven urinary tract infection. Pediatr Nephrol. 2010;25(8):1463–1469. https://doi.org/10.1007/s00467-010-1542-x
[5] Lebowitz RL, Olbing H, Parkkulainen KV, et al. International system of radiographic grading of vesicoureteric reflux. International reflux study in children. Pediatr Radiol. 1985;15(2):105–109. https://doi.org/10.1007/BF02388714
[6] Elder JS, Peters CA, Arant BS, et al. Pediatric Vesicoureteral Reflux Guidelines Panel summary report on the management of primary vesicoureteral reflux in children. J Urol. 1997;157(5):1846–1851. https://doi.org/10.1016/S0022-5347(01)64882-1
[7] Swerkersson S, Jodal U, Sixt R, et al. Relationship among vesicoureteral reflux, urinary tract infection and renal damage in children. J Urol. 2007;178(2):647–651. https://doi.org/10.1016/j.juro.2007.04.004
[8] Gnech M, ’T Hoen L, Zachou A, et al. Update and summary of the European Association of Urology/European Society of Paediatric Urology Paediatric Guidelines on vesicoureteral reflux in children. Eur Urol. 2024;85(5):433-442.
[9] Austin PF, Bauer SB, Bower W, et al. The standardization of terminology of lower urinary tract function in children and adolescents: update report from the Standardization Committee of the International Children’s Continence Society. J Urol. 2014;191(6):1863.e13–1865.e13. https://doi.org/10.1016/j.juro.2014.01.110
[10] Sillén U. Vesicoureteral reflux in infants. Pediatr Nephrol. 1999;13(4):355–361. https://doi.org/10.1007/s004670050625
[11] Karami H, Razi A, Mazloomfard MM, et al. Is there any role for urodynamic study in children with high-grade vesicoureteral reflux? Urology. 2012;79(4):888–891. https://doi.org/10.1016/j.urology.2011.06.057
[12] Koff SA, Wagner TT, Jayanthi VR. The relationship among dysfunctional elimination syndromes, primary vesicoureteral reflux and urinary tract infections in children. J Urol. 1998;160(3 Part 2):1019–1022. https://doi.org/10.1016/S0022-5347(01)62686-7
[13] Sillen U, Bachelard M, Hansson S, et al. Video cystometric recording of dilating reflux in infancy. J Urol. 1996;155(5):1711–1715. https://doi.org/10.1016/S0022-5347(01)66179-2
[14] Taskinen S, Rintala R. Post-pyelonephritic renal scars are not associated with high voiding pressures. J Pediatr Urol. 2007;3(1):40–44. https://doi.org/10.1016/j.jpurol.2006.01.015
[15] Stenberg A, Läckgren G. Treatment of vesicoureteral reflux in children using stabilized non-animal hyaluronic acid/dextranomer gel (NASHA/DX): a long-term observational study. J Pediatr Urol. 2007;3(2):80–85. https://doi.org/10.1016/j.jpurol.2006.08.001
[16] Stenbäck A, Olafsdottir T, Sköldenberg E, et al. Proprietary non-animal stabilized hyaluronic acid/dextranomer gel (NASHA/Dx) for endoscopic treatment of grade IV vesicoureteral reflux: Long-term observational study. J Pediatr Urol. 2020;16(3):328.e1–328.e9. https://doi.org/10.1016/j.jpurol.2020.04.008
[17] Karasu AFG, Cetin C, Pasin Ö, et al. Prevalence of urinary incontinence and anal incontinence: an internet-based cross-sectional study of female Turkish University students. Int Urogynecol J. 2023; 34(9):2309–2315. https://doi.org/10.1007/s00192-023-05573-7
[18] Osama M, Shaukat S, Javed M, et al. Prevalence of urinary incontinence among young female adults. JPMA J Pak Med Assoc. 2022; 72(11):2283–2287.
[19] Sillen U, Hansson S, Jernmark S, et al. Longitudinal follow-up of bladder function in children who participated in the Swedish Reflux Trial. J Pediatr Urol. 2024;20(1):118–126. https://doi.org/10.1016/j.jpurol.2023.08.024
[20] Läckgren G, Cooper CS, Neveus T, et al. Management of vesicoureteral reflux: what have we learned over the last 20 years? Front Pediatr. 2021;9:650326. https://doi.org/10.3389/fped.2021.650326
[21] Tekgül S, Riedmiller H, Hoebeke P, et al. EAU Guidelines on vesicoureteral reflux in children. Eur Urol. 2012;62(3):534–542. https://doi.org/10.1016/j.eururo.2012.05.059
[22] Akhavizadegan H, Locke JA, Stothers L, et al. A comprehensive review of adult enuresis. Can Urol Assoc J. 2019;13(8):282–287. https://doi.org/10.5489/cuaj.5485