ORIGINAL RESEARCH ARTICLE
Consequences of different definitions of disease progression in observational studies of men with advanced prostate cancer
Eugenio Ventimigliaa, Marcus Westerberga, Paolo Zauritob, Tiago Bonde Mirandac, David Robinsonc, Rolf Gedeborga, Pär Stattina and Hans Garmoa
aDepartment of Surgical Sciences, Uppsala University, Uppsala, Sweden; bDivision of Experimental Oncology/Unit of Urology, Urological Research Institute, IRCCS Ospedale San Raffaele, Milan, Italy; cDepartment of Urology, Skåne University Hospital, Malmö, Sweden
ABSTRACT
Introduction and objectives: Definitions of prostate specific antigen progression for men with prostate cancer on androgen deprivation therapy (ADT) are mainly derived from randomised trials, and their applicability to the clinical practice remains uncertain. This study aimed to assess how different PSA-based definitions of progressions while on ADT affect estimates of progression, treatment initiation, and outcomes in men with prostate cancer.
Methods: Using data from the Prostate Cancer database of Sweden with extended treatments and endpoints data (PCBase Xtend), we identified 3718 men who initiated ADT between 2009 and 2022 and who had longitudinal PSA and treatment data. PSA progression was defined according to four modified guideline-based definitions ranging from the European Association of Urology (EAU) that has the most stringent criteria for progression to our previously used and less stringent definition (PCBase). We analysed cumulative incidence of PSA progression, treatment for castration resistant prostate cancer before and after PSA progression, and prostate cancer-specific mortality, accounting for competing risks.
Results: ADT was prescribed as the primary treatment in 52% of included men. The number of men with PSA progression ranged by definition from 1047 men (28%, EAU) to 2378 men (64%, PCBase) at 10 years after initiation of ADT. Earlier progression was observed with less stringent criteria, with a difference in median time to progression of 3 months (PCBase vs EAU). Despite variation in incidence proportion of PSA progression, the proportion of men treated within 5 years after progression was similar (45–52%), as was prostate cancer-specific mortality (26–27%) across definitions.
Conclusion: While definitions of PSA progression significantly impacted estimated incidence proportion of disease progression, they had limited influence on treatment initiation and long-term mortality. These findings suggest that in the clinical practice, decisions are guided by factors other than formal progression criteria. PSA-based definitions can be useful in observational studies if supported by sensitivity analyses.
KEYWORDS: castration resistant prostate cancer; disease progression; prostate specific antigen; prostate cancer mortality; androgen deprivation therapy
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 16–22. https://doi.org/10.2340/sju.v60.45401.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 20 September 2025; Accepted: 2 January 2026; Published: 04 February 2026
CONTACT: Eugenio Ventimiglia, MD, PhD eugenio.ventimiglia@gmail.com Department of Surgical Sciences, Uppsala University, SE-752 37, Uppsala, Sweden
Supplemental data for this article can be accessed online at https://doi.org/10.2340/sju.v61.45401
Introduction
During the past decades, several new treatments for men with prostate cancer who progress while on androgen deprivation therapy (ADT) have been introduced. Randomised clinical trials (RCTs) have shown that chemotherapy with docetaxel and treatment with androgen receptor pathway inhibitors (ARPIs) increase survival and improve quality of life for men with castration resistant prostate cancer (CRPC) [1, 2]. However, little is known if these improvements are achieved in clinical practice where men are often older and have more comorbidities than men in RCTs [3, 4].
Various definitions of CRPC in RCTs have been recommended by guideline groups. These definitions have been based on repeated measurements of PSA, levels of testosterone, and imaging [5–7]. However, little is known to what extent these variables are measured in clinical practice and to what extent they are used as a basis for decision-making. The aim of this study is to assess how different criteria of PSA progression impact estimates of incidence proportion, timing, and outcomes of disease progression during ADT in men with prostate cancer.
Materials and methods
Data sources
The National Prostate Cancer Register (NPCR) records 98% of all new prostate cancer cases in Sweden as compared to the Cancer Register, which is legally mandated for reporting. In the Prostate Cancer database Sweden (PCBase) NPCR have been enrichedwith information from additional health care registers, such as the Patient Register, Cause of Death Register, and the Prescribed Drug Register, using the Swedish personal identity number [8]. In addition, we have collected longitudinal data from health care IT systems in five regions in Sweden on serum levels of PSA, and use of ADT and chemotherapy distributed in-hospital. The study was approved by the Swedish Research Ethics Authority.
Study population
We identified men who had started ADT between May 7, 2009 and October 31, 2022. (Supplementary Table 1). We excluded men who had received adjuvant ADT in conjunction with radical radiotherapy and men who received ARPI or chemotherapy as the primary therapy; however, men who re-started ADT after that adjuvant ADT had been stopped were included. At least two PSA measurements within 270 days from the start of ADT were required for inclusion and follow-up started on the date of second PSA (Supplementary Figure 1).
Definitions of PSA progression indicative of disease progression to CRPC
Four definitions of PSA progression indicative of CRPC according to guideline recommendation were evaluated. Since imaging results were not available, we based our analysis on PSA levels only. PSA nadir was determined by observing PSA values starting from the last PSA before initiation of ADT. Castration status was based on the use of ADT since testosterone levels were unavailable. We considered a man to be castrated after ≥3 months of ADT during a 6-month period or on the date of bilateral orchidectomy. The date for PSA indicative of progression while on ADT was defined as the date for the last PSA needed to fulfil each definition.
Modified EAU definition
The definition in European Association of Urology (EAU) guidelines requires three consecutive PSA measurements in castrated men, with two consecutive increases of 50% or more, and a PSA value >2 ng/mL [6].
Modified Swedish definition
The Swedish National Guidelines require two consecutive PSA measurements at least 7 days apart in castrated men with an increase >1 ng/mL and PSA value >2 ng/mL [7].
Modified PCBase definition
This definition has previously been used in studies based on data from PCBase [9, 10] and requires either that the PSA is more than twice the PSA nadir or that PSA has increased ≥5 ng/mL compared to the previous measurement in castrated men.
Modified Prostate Cancer Clinical Trials Working Group 3 (PCWG3) definition
The definition from Prostate Cancer Clinical Trials Working Group 3 (PCWG3), also part of the guidelines of the American Urological Association, requires a >25% increase in PSA based on two consecutive measurements at least 1 week apart with PSA ≥2.0 ng/mL higher compared to nadir [11].
Treatment of CRPC
Start date of CRPC treatment was defined as the earliest date when treatment with ARPI or chemotherapy was initiated.
Chemotherapy
Chemotherapy with docetaxel (ATC code L01CD02) was identified in specific chemotherapy registers held at each region.
Androgen receptor pathway inhibitors
Filled prescriptions for ARPIs were retrieved in the Prescribed Drug Register using the ATC codes for abiraterone (L02BX03), enzalutamide (L02BB04), darolutamide (L02BB06), and apalutamide (L02BB05).
Estimates of life expectancy
Life expectancy was calculated based on age, a multidimensional diagnosis-based comorbidity index (MDCI) and a drug comorbidity index (DCI) [12]. The MDCI was computed using ICD-10 codes registered in the Patient Register during a 10-year look-back period preceding the start of follow-up [13, 14]. The DCI was calculated based on Anatomical Therapeutic Chemical (ATC) codes (five digits) identified in the Prescribed Drug Register from prescriptions filled during the 365-day period preceding the start of follow-up [15, 16].
Statistical analysis
For each of the definitions, follow-up started on the date of second PSA and ended on the date of PSA progression, date for start of CRPC treatment, date of death, or last date of follow-up (December 31, 2023, Supplementary Table 1), whichever came first.
We censored for reaching end of follow-up without the following competing events: PSA progression, CRPC treatment without prior PSA progression, and death without prior CRPC treatment or PSA progression. Stacked cumulative incidence proportions were estimated, accounting for competing events [9].
In the next step, men who fulfilled PSA progression criteria were followed from the date of progression to the date of death or end of follow-up (Supplementary Table 1), whichever came first.
To describe the proportion of men who received CRPC treatment within 2 years from the date of CRPC, we applied a kernel estimate function to smooth the proportions treated for CRPC in strata of age. Finally, we also estimated the cumulative incidence of prostate cancer death and compared populations selected with different definitions of PSA progression. December 31, 2023 was the last day of follow-up.
All analyses were performed using R version 4.3.2.
Results
Baseline characteristics
In the 3718 study men, the median (IQR) age was 78 (72–83) years and median (IQR) life expectancy was 8 (5–12) years (Table 1). 1922 (52%) men received primary ADT, and 1358 (37%) men received ADT following deferred treatment and 308 (11%) at progression after radical treatment.
PSA progression during ADT and treatment initiation without progression
Depending on the criteria for PSA progression used, the number of men who progressed during the 10-year follow-up ranged from 2378 using PCBase criteria to 1047 when using the more stringent EAU criteria (Figures 1 and 2). Conversely, the probability of CRPC treatment before definition of PSA progression was fulfilled was lowest for the PCBase definition (5%) and highest for the EAU definition (24%) (Figure 1). An earlier progression was detected using the PCBase and Swedish criteria compared with the EAU criteria, with a median difference of 3 months (Supplementary Table 3).
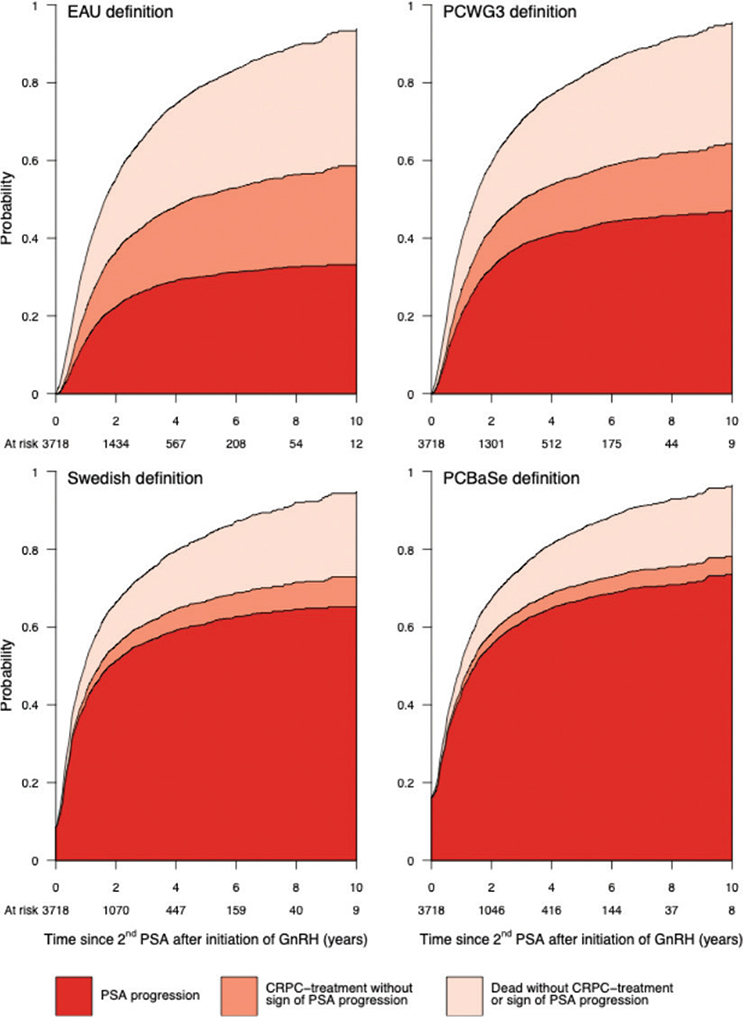
Figure 1. Cumulative incidence among 3,718 Swedish men diagnosed with prostate cancer and on androgen deprivation therapy of PSA progression, receipt of CRPC medication, and death, according to the definition by the European Association of Urology, Prostate Cancer Working Group 3 (PCWG3), Swedish guidelines and PCBase.
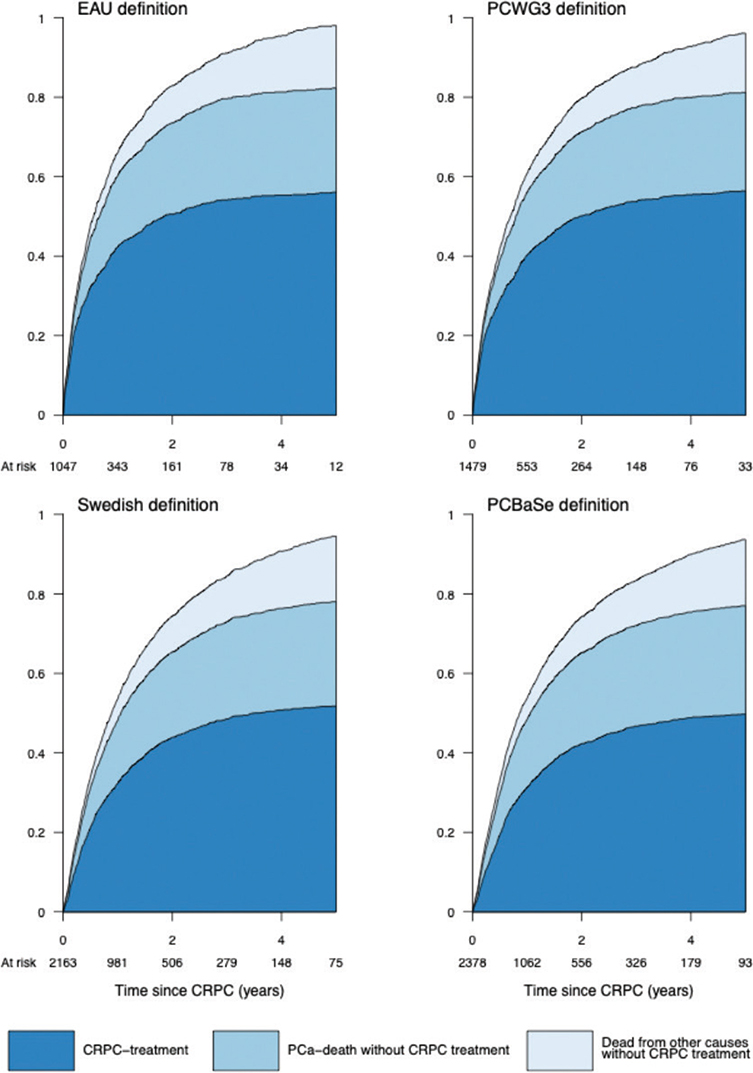
Figure 2. Cumulative incidence of receipt of treatment for castration resistant prostate cancer (CRPC), prostate cancer death without receiving CRPC treatment, and death from other causes in men with definition of PSA progression according to the European Association of Urology, Prostate Cancer Working Group 3 (PCWG3), Swedish guidelines, and PCBase.
CRPC treatment after PSA progression
Figure 2 depicts men who had a PSA progression according to different definitions. Despite a two-fold difference in the number of men who fulfilled the different CRPC criteria, the proportion of men who initiated CRPC treatment within 5 years after PSA progression was quite similar: 45% for the PCBase definition and 52% for the EAU definition. The proportion of men who died from prostate cancer without CRPC treatment was also similar, regardless of the definition (26–27%), as was the proportion of those who died from other causes (15–16%).
CRPC treatment in relation to age at disease progression
For all definitions of disease progression, the likelihood of CRPC treatment declined with older age (Figure 3). This decline was particularly pronounced for chemotherapy in men over 70. Among men over 75, the decrease became steeper, and for those over 80, a similar decline was also observed for ARPI use.
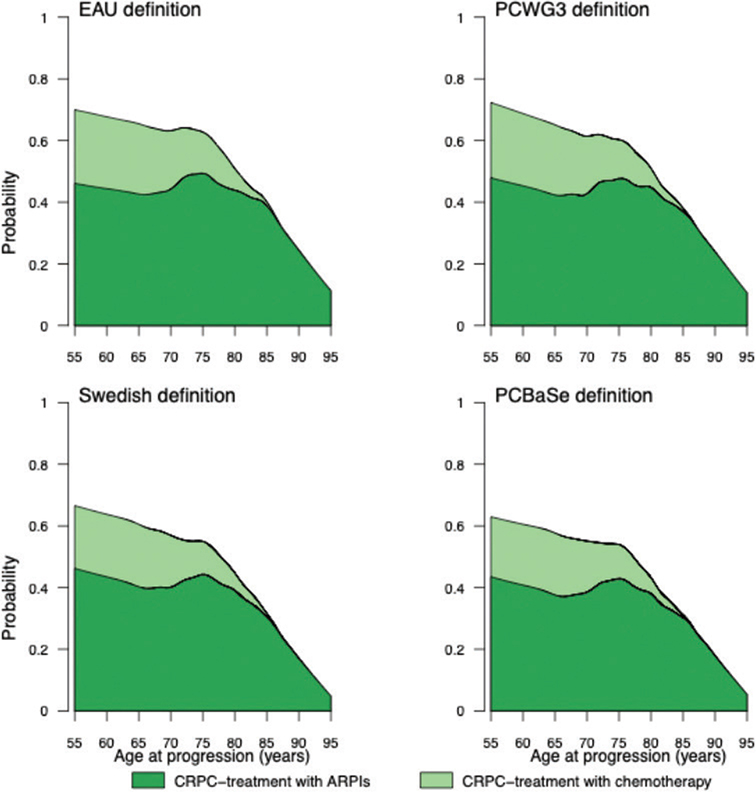
Figure 3. Probability of receipt of treatment for castration resistant prostate cancer (CRPC) according to age at PSA progression as defined by the European Association of Urology, Prostate Cancer Working Group 3 (PCWG3), Swedish guidelines, and PCBase.
Risk of death in relation to PSA progression and CRPC treatment
The cumulative incidence of prostate cancer death at 8 years of follow-up in men who started ARPIs was quite similar after 6 years of follow-up, irrespective of definition for progression (range 71–76%) (Figure 4). Similarly, prostate cancer death in men who started ARPI or chemotherapy was the same (89%), irrespective of criteria for progression (Figure 4).
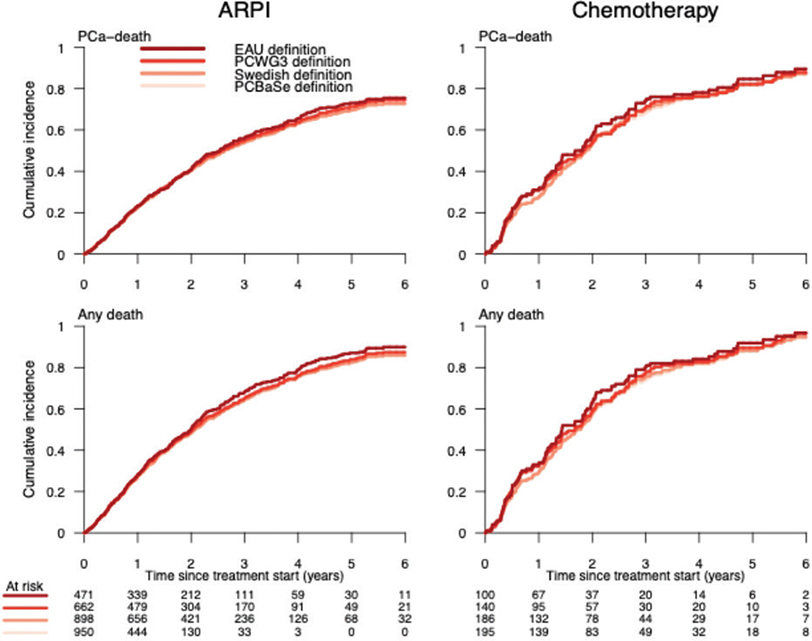
Figure 4. Cumulative incidence of death from prostate cancer and from all causes according to different PSA progression definitions since start of androgen receptor pathway inhibitors (ARPIs) or chemotherapy.
Discussion
In this population-based observational study, the proportion of men with prostate cancer on ADT with PSA progression varied 2-fold according to the stringency of the definition of PSA progression. For the less stringent definitions of PSA progression, the proportion of men who received CRPC treatment without fulfilling the definition was small whereas when a more stringent definition was applied, this proportion was substantially higher. In contrast, only small differences in the proportion of men who received CRPC treatment or died of prostate cancer were observed for the different definitions. Our study indicates that more stringent definitions of CRPC, mostly derived from clinical trials, are not always used in clinical practice where factors other than the definition of PSA progression seem to influence treatment decisions.
Proportion of men who progressed according to different definitions of PSA progression
The definitions of PSA progression assessed in this study – EAU, Swedish, PCBase, and PCWG3 – yielded very different numbers of men who progressed. When using PSA progression criteria and the start of CRPC treatment as a composite criterion, the PCBase definition identified a larger number of men as compared to the EAU definition. These discrepancies highlight the difficulty in applying criteria for progression derived from RCTs to clinical practice. The EAU definition’s stringent criteria appeared to be rarely used in clinical practice in Sweden, likely reflecting the clinicians’ will to initiate early treatment for progression. The mortality with the respective treatment was, however, largely comparable irrespective of definition, being only slightly higher using the EAU definition [17–20].
In a systematic review of adherence to recommendations in the EAU guidelines on CRPC treatment, adherence was low in all studies [21]. While rising PSA levels were frequently used as a diagnostic criterion, radiological progression was used less often [22].
Survival after start of CRPC treatment
The mortality following treatment with ARPI or chemotherapy was largely comparable irrespective of the definition of PSA progression used, with minimal differences at 6 years for men who started ARPIs (71–76%).
Strengths and limitations
The strengths of the study include the use of nationwide, population-based data with detailed longitudinal information on PSA levels, treatments, and outcomes. However, several limitations warrant consideration. The lack of data on imaging precludes the assessment of radiographic progression, a key component of CRPC in clinical trials. The lack of data on testosterone levels is another limitation. However, a recently published population-based study showed that only 1% of men on ADT had testosterone levels outside the castration range. Moreover, progression documented by imaging is usually anticipated by a PSA progression which likely triggered the choice to perform imaging [23].
Conclusion
In clinical practice, the choice of definition of PSA progression in men with prostate cancer on ADT had a strong impact on the proportion of treated men, whereas only minor differences in estimated time to progression and long-term mortality following treatment initiation were observed. The small differences observed in the timing of treatment and survival support the use of PSA progression-based definitions in observational studies, as long as they are complemented by appropriate sensitivity analyses, especially when the goal is to estimate the proportion of men living with CRPC.
References
[1] Francini E, Agarwal N, Castro E, et al. Intensification approaches and treatment sequencing in metastatic castration-resistant prostate cancer: a systematic review. Eur Urol. 2025;87:29–46. https://doi.org/10.1016/j.eururo.2024.09.008
[2] Riaz IB, Naqvi SAA, He H, et al. First-line systemic treatment options for metastatic castration-sensitive prostate cancer: a living systematic review and network meta-analysis. JAMA Oncol. 2023;9:635. https://doi.org/10.1001/jamaoncol.2022.7762
[3] Gillessen S, Bossi A, Davis ID, et al. Management of patients with advanced prostate cancer – metastatic and/or castration-resistant prostate cancer: report of the advanced prostate cancer consensus conference (APCCC) 2022. Eur J Cancer. 2023;185:178–215. https://doi.org/10.1016/j.ejca.2023.02.018
[4] Sayegh N, Swami U, Agarwal N. Recent advances in the management of metastatic prostate cancer. JCO Oncol Pract. 2022;18:45–55. https://doi.org/10.1200/OP.21.00206
[5] Stranne J. 2023/2024 update of the national prostate cancer guidelines in Sweden. Scand J Urol. 2024;59:210–1. https://doi.org/10.2340/sju.v59.42656
[6] EAU Prostate cancer guidelines 2024. https://uroweb.org/guidelines/prostate-cancer, [accessed Dec 31, 2025]
[7] Bratt O, Carlsson S, Fransson P, et al. The Swedish national guidelines on prostate cancer, part 2: recurrent, metastatic and castration resistant disease. Scand J Urol. 2022;56:278–84. https://doi.org/10.1080/21681805.2022.2093396
[8] Van Hemelrijck M, Wigertz A, Sandin F, et al. Cohort profile: the National Prostate Cancer Register of Sweden and Prostate Cancer data base Sweden 2.0. Int J Epidemiol. 2013;42:956–67. https://doi.org/10.1093/ije/dys068
[9] Khoshkar Y, Westerberg M, Adolfsson J, et al. Mortality in men with castration-resistant prostate cancer – a long-term follow-up of a population-based real-world cohort. BJUI Compass. 2022;3:173–83. https://doi.org/10.1002/bco2.116
[10] Ventimiglia E, Bill-Axelson A, Adolfsson J, et al. Modeling disease trajectories for castration-resistant prostate cancer using nationwide population-based data. Eur Urol Open Sci. 2022;44:46–51. https://doi.org/10.1016/j.euros.2022.07.010
[11] Scher HI, Morris MJ, Stadler WM, et al. Trial design and objectives for castration-resistant prostate cancer: updated recommendations from the prostate cancer clinical trials working group 3. J Clin Oncol. 2016;34:1402–18. https://doi.org/10.1200/JCO.2015.64.2702
[12] Westerberg M, Ahlberg M, Orrason AW, et al. Assessment of variability in life expectancy in older men by use of new comorbidity indices. A nationwide population-based study. Scand J Urol. 2024;59:207–9. https://doi.org/10.2340/sju.v59.42504
[13] Westerberg M, Garmo H, Robinson D, et al. Target trial emulation using new comorbidity indices provided risk estimates comparable to a randomized trial. J Clin Epidemiol. 2024;174:111504. https://doi.org/10.1016/j.jclinepi.2024.111504
[14] Westerberg M, Irenaeus S, Garmo H, et al. Development and validation of a multi-dimensional diagnosis-based comorbidity index that improves prediction of death in men with prostate cancer: nationwide, population-based register study. PLoS One. 2024;19:e0296804. https://doi.org/10.1371/journal.pone.0296804
[15] Gedeborg R, Sund M, Lambe M, et al. An aggregated comorbidity measure based on history of filled drug prescriptions: development and evaluation in two separate cohorts. Epidemiology. 2021;32:607–15. https://doi.org/10.1097/EDE.0000000000001358
[16] Gedeborg R, Garmo H, Robinson D, et al. Prescription-based prediction of baseline mortality risk among older men. PLoS One. 2020;15:e0241439. https://doi.org/10.1371/journal.pone.0241439
[17] Orrason AW, Scilipoti P, Garmo H, et al. Improved adherence to prostate cancer guidelines concomitant with public reporting. Nationwide population-based study. Scand J Urol. 2025;60:50–8. https://doi.org/10.2340/sju.v60.43107
[18] Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–12. https://doi.org/10.1056/NEJMoa040720
[19] Beer TM, Armstrong AJ, Rathkopf DE, et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424–33. https://doi.org/10.1056/NEJMoa1405095
[20] Ryan CJ, Smith MR, Fizazi K, et al. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2015;16:152–60. https://doi.org/10.1016/S1470-2045(14)71205-7
[21] Kirby M, Hirst C, Crawford ED. Characterising the castration- resistant prostate cancer population: a systematic review. Int J Clin Pract. 2011;65:1180–92. https://doi.org/10.1111/j.1742-1241.2011.02799.x
[22] Machidori A, Shiota M, Kobayashi S, et al. Prognostic significance of complete blood count parameters in castration-resistant prostate cancer patients treated with androgen receptor pathway inhibitors. Urol Oncol. 2021;39:365.e1–7. https://doi.org/10.1016/j.urolonc.2020.09.036
[23] Bonde Miranda, T. (2025). Hormonal Therapy, Progression to Castration-resistant Prostate Cancer and Survival (PhD dissertation, Linköping University Electronic Press). https://doi.org/10.3384/9789181180824