ORIGINAL RESEARCH ARTICLE
Risk of infectious complications after transperineal prostate biopsy compared to transrectal biopsy: nationwide population-based cohort study in Sweden
Jesper Bonnedahla  , Karl-Johan Lundströma, Erik Lampab, David Robinsonc
, Karl-Johan Lundströma, Erik Lampab, David Robinsonc  , Johan Stranned,e
, Johan Stranned,e  , Stefan Carlssonf
, Stefan Carlssonf  , Magnus Wageniusg, Adam Linderg, Joakim Örtegrenh
, Magnus Wageniusg, Adam Linderg, Joakim Örtegrenh  , Anders Andreassond,e, Martin Holmbomi
, Anders Andreassond,e, Martin Holmbomi  , Pär Stattinj
, Pär Stattinj  and Johan Styrkea
and Johan Styrkea 
aDepartment of Diagnostics and Intervention, Urology and Andrology, Umeå University, Umeå, Sweden; bEpistat AB, Uppsala, Sweden; cDepartment of Urology, Ryhov Hospital, Jönköping, Sweden; dDepartment of Urology, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden; eDepartment of Urology, Region Västra Götaland, Sahlgrenska University Hospital, Gothenburg, Sweden; fDepartment of Molecular Medicine and Surgery, Section of Urology, Karolinska Institutet, Stockholm, Sweden; gDepartment of Clinical Sciences, Division of Infection Medicine, Lund University, Lund, Sweden; hDepartment of Surgery, Region Kronoberg; Department of Urology, Institute of Clinical Science, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden; iDepartment of Urology and Department of Clinical and Experimental Medicine, Linköping University, Linköping, Sweden; jDepartment of Surgical Sciences, Uppsala University, Uppsala, Sweden
ABSTRACT
Objective: Prostate biopsy is associated with a risk of significant infectious complications including sepsis. We investigated the risk of infections after transrectal (TR) biopsy compared to transperineal (TP) biopsy.
Materials and methods: Men who had undergone prostate biopsy and diagnosed with prostate cancer were identified in the National Prostate Cancer Register (NPCR) of Sweden. Linkage with Swedish health care registers provided information on hospitalization, antibiotic prescriptions and comorbidities. Rate ratios for hospitalization, for infections, after TR and TP biopsies over day 1–7, 1–14, and 1–30 were estimated with Poisson regression. Filled prescriptions for urinary tract related antibiotics were also assessed.
Results: Thirty-one thousand two hundred twenty-two men underwent biopsy between 1 January 2020 and 31 December 2023. 87% underwent TR and 13% TP biopsy.
Hospitalization occurred in 0.6% of men (n = 24) after TP biopsy and 2.0% (n = 548) after TR biopsy. Rate ratios for hospitalization in the TR group compared to TP were 8.0 (95% confidence interval [CI]: 4.0–16.2) for day 1–7, 6.2 (3.2–11.9) for day 1–14, and 4.1 (2.4–6.8) for day 1–30. Filled antibiotic prescriptions were found for 4.5% of men (n = 187) after TP biopsy and 6.9% (n = 1,883) after TR biopsy. For antibiotic prescriptions, the rate ratios were 2.3 (1.8–2.9) for day 1–7 as well as day 1–14, and 1.6 (1.3–1.9) for day 1–30.
Conclusions: A transrectal prostate biopsy was associated with a significantly higher risk of post-biopsy infectious complications compared to transperineal biopsy. These findings support the use of transperineal biopsy.
KEYWORDS: Urological diagnostic techniques; needle biopsies; infections; risk factors; prostatic neoplasms
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 44–50. https://doi.org/10.2340/sju.v61.45537.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 20 September 2025; Accepted: 13 January 2026; Published: 17 March 2026
CONTACT: Johan Styrke johan.styrke@umu.se Institutionen för diagnostik och intervention, enheten för urologi, Umeå universitet, 951 87 Umeå
Supplemental data for this article can be accessed online at https://doi.org/10.2340/sju.v61.45537
Competing interests and funding: The study had no financial support. The authors declare no financial conflict of interest. Johan Styrke and Martin Holmbom are members of the national guidelines group for antibiotic prophylaxis in urology.
Introduction
Prostate cancer is typically diagnosed via prostate biopsy with an ensuing risk of infection, ranging from urinary tract infection (UTI) to sepsis [1]. Several risk factors for post-biopsy infections have been identified, including antibiotic-resistant rectal flora, diabetes, prior UTI or prostatitis, previous antibiotic use, and high comorbidity [2–4]. Given the increase in post-biopsy infections over time [5, 6], efforts to minimize infectious complications are of critical importance.
One proposed strategy to reduce infection risk is the use of the transperineal (TP) route to the prostate. In multiple studies, including systematic reviews and meta-analyses, TP biopsy has been associated with lower incidence of infectious complications, including sepsis, while maintaining equivalent cancer detection rates [7–10]. Accordingly, the EAU now recommends TP biopsy as the preferred method [11]. Nonetheless, high-quality evidence from randomized controlled trials (RCTs) is limited. In 2024, two RCTs did not demonstrate statistically significant differences in infection rates between the two approaches [12, 13].
Assessing the optimal biopsy method for minimizing infection risk remains essential, and data from national healthcare registers can provide valuable insights to complement findings from RCTs. In Sweden, the adoption of TP biopsy varies between clinics: some use it exclusively, other clinics use it alongside transrectal (TR) biopsy, and some do not use it at all.
The primary aim of this study was to compare hospital admission for infection-related diagnoses 30 days after prostate biopsy – between the TR and TP biopsy in a nationwide population-based cohort. Secondary aims included evaluating antibiotic prescriptions and hospital length of stay.
Methods
Study population and setting
In this population-based cohort study data from the National Prostate Cancer Register (NPCR) of Sweden, which captures 98% of all prostate cancer cases compared to the mandatory Swedish National Cancer Register, was used [14, 15]. The overarching goal of NPCR is to ensure equal and high standard of care to all men with prostate cancer [16].
Using Sweden’s unique personal identification number, NPCR has been linked to several other national healthcare registers within the Prostate Cancer data Base Sweden (PCBase) RAPID, as previously described [17]. PCBase RAPID served as the database for this study, registers within the database used in this study are described next.
From the National Patient Register, which contains diagnostic and procedural codes for both inpatient and outpatient care [18], data on discharge diagnoses, hospital stays, biopsy date and type, urinary retention, and extended-spectrum beta-lactamase (ESBL) status were retrieved. ICD codes were used to calculate the Charlson Comorbidity Index [19]. Medication data were retrieved from the Prescribed Drug Register, which contains information on all filled prescriptions in Sweden [20], using Anatomical Therapeutic Chemical (ATC) codes. A Drug Comorbidity Index was constructed based on this data [21]. Cancer characteristics, including tumor stage, prostate-specific antigen (PSA) level, prostate volume, and number of biopsy cores taken, were obtained from NPCR. Finally, mortality data were collected from the Cause of Death Register, which records the date of death and underlying and immediate causes of death [22].
Inclusion criteria
All men in PCBase who had undergone a TR or TP prostate biopsy (classification of health care interventions [KKÅ]: TKE10, TKE13, TKE20, TKE23 in the Patient Register) and had a corresponding biopsy date recorded in NPCR between 1 January 2020 and 31 December 2023 were included in the cohort (Figure 1). This time frame was selected as TP biopsies were first recorded in the Patient Register from 1 January 2020 [23].
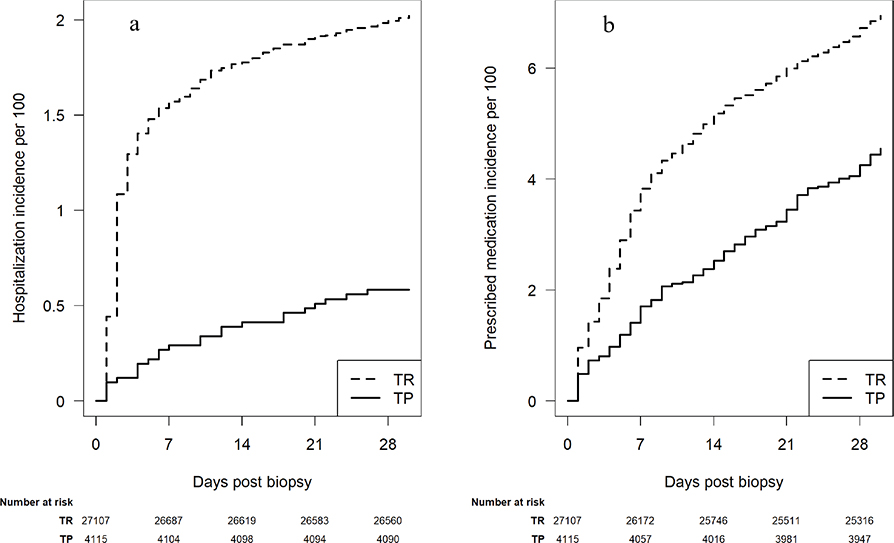
Figure 1. (a) Cumulative incidence of hospitalization after transperineal and transrectal prostate biopsy. (b) Cumulative incidence of filled prescription of antibiotics for urinary tract infection after transperineal and transrectal prostate biopsy. Data are extracted from the National Prostate Cancer Register of Sweden between 01 January 2020 and 31 December 2023.
TP: transperineal; TR: transrectal.
Exclusion criteria
In case of more than one hospital admission or more than one antibiotic prescription occurred during the same period after biopsy, only the first event was included in the analysis. If a man had both a hospitalization for infection and a post-biopsy antibiotic prescription, the events were treated as separate outcomes and included in their respective analyses. Furthermore, the same man could only be included in the study once, the first registered biopsy, even if the man underwent biopsies multiple times.
Main outcome measures
Hospitalisation for post-biopsy infection was defined as hospital admission within 4 weeks following biopsy (days 1–30) with a registered discharge ICD code related to UTI or sepsis in the National Patient Register (see Supplementary Table 1 for ICD codes used).
Secondary outcome measures
Antibiotic prescription after biopsy was defined as a registered filled prescription for UTI-related antibiotics between the day after biopsy and 1-month post-biopsy (days 1–30) in the Prescribed Drug Register. Prescriptions filled on the same day as the biopsy were excluded (see Supplementary Table 2 for ATC codes used).
Length of hospital stay was defined as number of days in hospital. If admission and discharge occurred on the same day, the length of stay was set to 0.5 days.
Covariates
All covariates, including their data sources, units, and definitions, are listed in Supplementary Table 3.
Statistical methods
Cumulative incidences were calculated. Rate ratios and risk differences for TR versus TP biopsies over day 1–7, 1–14, 1–30, 8–14, 15–21, and 22–28 were estimated using Poisson regression. Covariates used for adjustment are listed in Supplementary Table 3. Length of hospital stay was analysed using the Kaplan–Meier estimator, with the number of days until discharge as the outcome variable. Analyses were conducted on complete cases without imputation. Statistical analysis was performed using R (version 4.4.2).
Ethics
The study was approved by the Swedish Ethical Review Authority (Dnr 2021-06128-01).
Results
Between 1 January 2020 and 31 December 2023 31,222 men who underwent a prostate biopsy resulting in a prostate cancer diagnosis had been registered in NPCR. All men had a corresponding ICD-10 code for biopsy in the National Patient Register. Of these, 87% (27,107) underwent a TR biopsy and 13% (4,115) a TP biopsy.
Baseline characteristics for men who underwent TP and TR biopsy are presented in Table 1. The median age was 71 years, and median number of biopsy cores were six in the TP group and eight in the TR group. The prostate volume was similar (median 40 ml in the TP group and 41 ml in the TR group) but the proportion of ≥T3 tumors was lower in the TP group (8.3%) compared to the TR group (16%). Comorbidity was similar in the groups; however, antibiotic prescriptions within 2 weeks prior to biopsy were less common before TP (3.7%) than TR (6.9%).
| Characteristic | TP, N = 4,1151 | TR, N = 27,1071 |
| Age (years) | 70 (64–76) | 71 (65–77) |
| Year | ||
| 2020 | 195 (4.7%) | 5,781 (21%) |
| 2021 | 542 (13%) | 6,958 (26%) |
| 2022 | 1,112 (27%) | 8,013 (30%) |
| 2023 | 2,266 (55%) | 6,355 (23%) |
| PSA (ug/L) | 7 (5–12) | 8 (5–17) |
| Missing | 25 | 57 |
| Prostate volume (ml) | 40 (30–55) | 41 (31–55) |
| Missing | 206 | 1,332 |
| MRI performed | 1,707 (95%) | 13,661 (68%) |
| Missing | 2,311 | 7,029 |
| Number of biopsies | 6.0 (4–10) | 8.0 (5–12) |
| Missing | 3 | 165 |
| T-stage | ||
| <=T2 | 3,622 (88%) | 21,826 (81%) |
| >=T3 | 340 (8.3%) | 4,317 (16%) |
| Tx | 148 (3.6%) | 957 (3.5%) |
| Missing | 5 | 7 |
| ESBL | 5 (0.1%) | 20 (<0.1%) |
| History of Urinary retention | 45 (1.1%) | 358 (1.3%) |
| Diabetes mellitus with insulin | 143 (3.5%) | 1,053 (3.9%) |
| Diabetes mellitus without insulin | 543 (13%) | 3,646 (13%) |
| SGLT2 inhibitor without other DM medications | 54 (1.3%) | 222 (0.8%) |
| Benign prostate hyperplasia | 294 (7.1%) | 2,303 (8.5%) |
| Immunosuppressants (excluding corticosteroids) | 57 (1.4%) | 224 (0.8%) |
| Corticosteroids | 335 (8.1%) | 2,144 (7.9%) |
| Previous antibiotics ≥2 weeks before the biopsy | 976 (24%) | 5,887 (22%) |
| Antibiotic prescription <2 weeks before the biopsy |
154 (3.7%) | 1,866 (6.9%) |
| Drug comorbidity index | 0.66 (0.07–1.64) | 0.70 (0.08–1.72) |
| Charlson comorbidity index | ||
| 0 | 2,603 (63%) | 17,024 (63%) |
| 1 | 510 (12%) | 3,761 (14%) |
| 2+ | 1,002 (24%) | 6,322 (23%) |
| 1Median (IQR); n (%). IQR: interquartile range; PSA: prostate-specific antigen; MRI: Magnetic Resonance Imaging; ESBL: extended spectrum beta lactamases; SGLT2: Sodium-glucose cotransporter-2; DM: diabetes mellitus; TP: transperineal; TR: transrectal.. |
||
Hospitalized due to an infectious complication within 30 days after biopsy occurred in 0.6% (n = 24) of the men in the TP group, compared to 2.0% (n = 548) in the TR group (Table 2). The risk of hospitalization increased rapidly during the first days following TR biopsy, whereas the TP group showed a slower and more linear increase over the 30-day period (Figure 1a). This pattern was reflected in a stable risk difference from day 7 to 30 (Figure 2a). Hospitalization rate ratios peaked immediately after biopsy and declined rapidly thereafter. Significant differences favoring TP biopsy were observed only during the first week. Although no statistically significant differences were seen on days 8–14, 15–21, or 22–30, the overall differences remained significant across all follow-up periods (7, 14, and 30 days) (Table 3, Figure 2b).
| Outcome | TP, 13%1 | TR, 87%1 |
| Hospitalization within 30 days post biopsy | 0.6% (24) | 2.0% (548) |
| Antibiotics within 30 days post biopsy | 4.5% (187) | 6.9% (1,883) |
| 1% (n). TP: transperineal; TR: transrectal. |
||
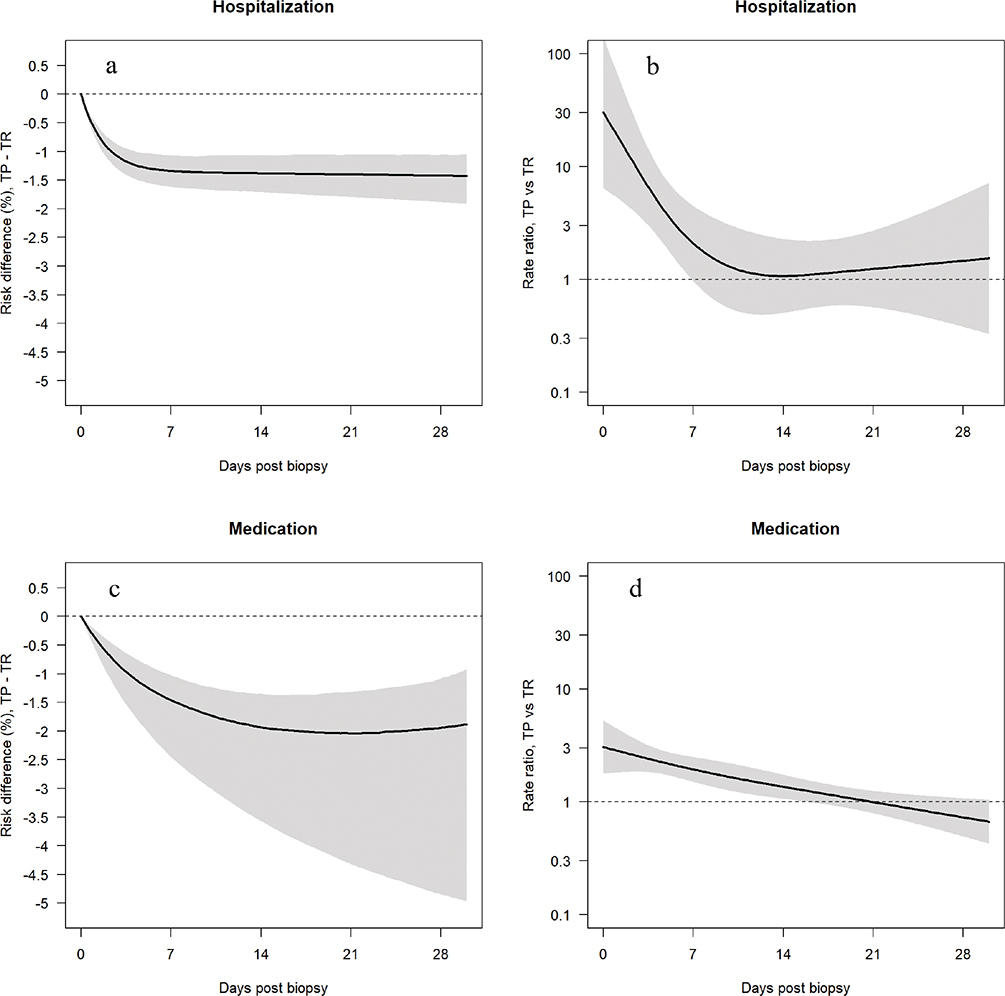
Figure 2. (a) Risk difference (solid line) for Transperineal versus Transrectal prostate biopsy regarding hospitalization. (b) Risk ratio (solid line) for Transperineal versus Transrectal prostate biopsy regarding hospitalization. (c) Risk difference (solid line) for Transperineal versus Transrectal prostate biopsy regarding filled prescription of antibiotics for urinary tract infection. (d) Risk ratio (solid line) for transperineal versus transrectal prostate biopsy regarding filled prescription of antibiotics for urinary tract infection. Confidence intervals are marked as shaded areas. Data are extracted from the National Prostate Cancer Register of Sweden between 01 January 2020 and 31 December 2023.
TP: transperineal; TR: transrectal.
The percentage of men with filled antibiotic prescriptions were 4.5% (n = 187) after TP biopsies and 6.9% (n = 1,883) after TR biopsies, respectively (Table 2). Differences in filled antibiotic prescriptions are illustrated in Figure 2b. The risk difference gradually increased until day 21 and then declined slightly (Figure 2c). Rate ratios significantly favored the TP biopsy for the time intervals 1–7, 1–14, 1–28, and 8–14 days (Table 3, Figure 2d).
The median length of hospital stay was 3 days for both groups.
Discussion
In this large nationwide population-based cohort study, men who underwent TR prostate biopsy had a four-fold higher incidence of hospital admission for infection-related complications compared to men who underwent TP biopsy. The rate ratio was particularly elevated for hospitalization during the first week following biopsy in favor of TP biopsies. After that, hospitalization occurred at similar pace in both groups and the rate ratios were non-significant during week 2, 3 and 4. Fillings for UTI-related infection was 50% higher after TR biopsy compared to TP biopsy with a similar pattern as hospitalization over the first 4 weeks. This pattern with the most marked differences in infection occurrence during the first week in favor of TP followed by similar curves during week 2–4 suggests that the mechanisms of infection might be different between the methods but also that infections are a problem in both groups, that is, not only in the TR group.
The strengths of this study include the completeness of register data for men diagnosed with prostate cancer in an entire nation and the high internal validity of the infection-related outcomes, especially hospitalization, which is not indicated if the patients are not clinically assessed as having a potentially severe infection. However, there are limitations. There were no available data on bacterial cultures in either of the groups so it cannot be excluded that some of the prescribed antibiotics might have been done based on inflammation and local symptoms rather than bacterial infections. Furthermore, men with negative biopsy results were not included, as only men with a prostate cancer diagnosis are registered in NPCR. However, a study from southern Sweden found no significant differences in the risk of infection between men with and without prostate cancer [24]. Data on administered prophylactic antibiotics are not available in Swedish healthcare registers, which may introduce confounding if prophylaxis varied within biopsy groups. Regarding filled prescribed antibiotics, day 0 was not included in the analysis because some clinics prescribe empiric short term treatment for 3–5 days with prophylactic purpose mainly in patients with risk factors for infection after TR biopsy (usually 20–25% of patients), therefore, some prescriptions for infectious symptoms may have been missed if they were conducted on day 0. Finally, we used a broad range of discharge and antibiotic codes to define hospitalization and UTI-related prescriptions, which may have led to misclassification if infections unrelated to the biopsy were included, this would however impact both TP and TR biopsy equally.
Our findings support previous evidence that TP biopsy carries a lower risk of infection compared to TR biopsy. A 2019 meta-analysis found a significantly lower risk of post-biopsy fever after TP (rate ratio 0.26, 95% confidence interval [CI]: 0.14–0.48) based on 1,618 men in four RCTs and three cohort studies [8]. Similarly, in an analysis of seven RCTs including 1,330 men a risk ratio of 0.55 (95% CI: 0.33–0.92) was found for a composite outcome of fever, sepsis, and symptomatic UTI in favor of TP biopsy. While this analysis of hospitalization yielded a non-significant rate ratio of 0.38 (95% CI: 0.09–1.61), a subsequent meta-analysis incorporating an additional RCT reported a pooled rate ratio of 2.48 (95% CI: 1.47–4.2) for infectious complications when comparing TR with TP biopsy [11, 25].
In contrast, two recent RCTs published in 2024 found no statistically significant differences in infection rates between the two biopsy techniques [12, 13]. In one of these, 658 men were randomized to either TR biopsy with targeted prophylaxis (based on rectal cultures) or TP biopsy without prophylaxis. Four patients in the TR group experienced infectious complications within 1 week, compared to none in the TP group (p = 0.06) [12]. The other study reported an odds ratio of 1.06 (95% CI: 0.43–2.65) for a 30-day composite outcome of suspected or confirmed UTI when comparing TR and TP biopsies in 718 randomized patients [13]. Their TR group received augmented prophylaxis with oral ciprofloxacin and trimethoprim–sulfamethoxazole before and 12 h after the biopsy, without rectal swabs or iodine–povidone preparation, whereas the TP group received no prophylaxis.
Our retrospective analysis of population-based data yielded results comparable to those of Mian et al., whose pragmatic trial included a broad patient population regardless of Magnetic Resonance Imaging (MRI) use or biopsy history. In Mian’s study, hospitalization rates were 0.6% in the TR group and 0.3% in the TP group; antibiotic prescription rates were 1.7% and 1.4%, respectively, at 30-day follow-up. By comparison, our study found higher infection rates: hospitalization occurred in 2.0% (TR) versus 0.6% (TP), and antibiotic prescriptions were filled in 6.9% (TR) versus 4.5% (TP). The most striking discrepancy lies in the higher infection burden in the TR group. Several factors may explain this difference. Firstly, the median age in our cohort was 71 years versus 65 years in Mian’s study, and the prevalence of diabetes was lower in the latter, indicating a selection of younger, healthier patients in the RCT. Secondly, in Sweden, a single-dose ciprofloxacin is typically used as prophylaxis for TR biopsy in patients without clear infection risk factors (e.g. diabetes, urinary retention, or catheterization) and an extended prophylaxis for higher-risk groups [26]. However, an increasing ciprofloxacin resistance, currently estimated at 11–14% in Sweden [27], necessitates use of better prophylactic strategies, such as augmented regimens or resistance screening. The absence of widespread implementation of such methods may contribute to the relatively high rate of post-biopsy infections.
If TR biopsies continue to be used, improvements in prophylactic strategies needs to be implemented. In 2023, the Swedish Urology Association issued new national guidelines recommending oral ciprofloxacin or trimethoprim–sulfamethoxazole before and in the evening after biopsy in patients without risk factors, and augmented prophylaxis (including oral and intravenous regimens) in high-risk individuals [28]. These recommendations aim to address the increasing issue of ciprofloxacin resistance. Both EAU and Swedish national guidelines also recommend rectal preparation with iodine–povidone prior to TR biopsy, although this was not routine during the study period [11, 29].
Prostate biopsy techniques are rapidly evolving. Broader implementation of iodine-povidone, targeted prophylaxis, and comprehensive patient risk factor assessment are likely to reduce the number of post-TR biopsy UTI, thereby decreasing the difference in this aspect between the two methods. A study by Berg et al. did however show significant higher rates of infectious complications, for TR compared to TP biopsies, when using targeted prophylaxis and comprehensive patient risk factor assessment, indicating that this may not be enough to get comparable rates of infectious complications [30].
Regardless of difference in complication rates, TP biopsy may outperform TR biopsy in other aspects as well. A RCT by Bryant et al. published in 2025 that included 1,126 men had a statistically significant higher detection rate for clinically significant prostate cancer in the TP group (60%) versus the TR group (54%) [31]. Furthermore, the study did not show any statistical differences for non-infectious complications but found TR biopsies less time consuming and more often less painful, indicating that TR may be beneficiary in other aspects.
The study by Bryant et al. found similar rates of post-biopsy hospital admission for infections as this study, where 1.6% in the TR and 0.4% in the TP biopsy group was hospitalized within 35 days. A difference between the studies was the number of biopsies performed, which varied between the TR and TP approaches in the present cohort. A higher median number of biopsies was taken in the TR group than in the TR group, suggesting that systematic biopsies are more frequently conducted with the TR technique. This may affect our results to some extent, but as the prespecified number of biopsies were equal in both groups in the article of Bryant et al., and the observed rates of infection-related hospitalizations were similar to our study, this difference is likely to be of limited importance.
Further studies are warranted to monitor infection complications and to evaluate different prophylactic regimens for TR biopsy, ideally with standardized registration of prophylaxis in registers such as the NPCR. A randomized trial comparing TR and TP biopsy in the Swedish healthcare setting could be considered, particularly in the light of conflicting evidence. It would also be interesting to look closer into subgroups of patients of different age with or without risk factors for infection to elaborate if certain groups would benefit more from TP biopsies. In addition, large cohort studies are needed to explore risk factors for infection following TP biopsy specifically.
Conclusion
This nationwide cohort study found that transrectal prostate biopsy was associated with a significantly higher risk of post-biopsy infectious complications, both in terms of hospital admission and antibiotic prescription compared to transperineal biopsy.
Acknowledgments
This project was made possible by the continuous work of NPCR steering group: Elin Axen, Johan Styrke, Johan Stranne, Jon Kindblom, Camilla Thellenberg, Andreas Josefsson, Ingrida Verbiene, Hampus Nugin, Stefan Carlsson, Anna Kristiansen, Mats Andén, Thomas Jiborn, Per Fransson, Eva Johansson, Magnus Törnblom, Viktoria Gaspar, Fredrik Jäderling, Elin Trägårdh, Erik Thimansson, Marie Hjälm Eriksson, Lotta Renström Koskela, Jonas Hugosson, Ola Bratt, Maria Nyberg, Fredrik Sandin, Marie Brus, Lena Pettersson, Petrus Stensson, Anna Hedström, Nina Hageman, Maria Moutran, Hans Joelsson and Gert Malmberg.
Chat GPT-4o was used for language review.
References
[1] Borghesi M, Ahmed H, Nam R, et al. Complications after systematic, random, and image-guided prostate biopsy. Eur Urol. 2017;71(3):353–365. https://doi.org/10.1016/j.eururo.2016.08.004
[2] Liss MA, Ehdaie B, Loeb S, et al. An update of the American Urological Association white paper on the prevention and treatment of the more common complications related to prostate biopsy. J Urol. 2017;198(2):329–334. https://doi.org/10.1016/j.juro.2017.01.103
[3] Roberts MJ, Bennett HY, Harris PN, et al. Prostate biopsy-related infection: a systematic review of risk factors, prevention strategies, and management approaches. Urology. 2017;104:11–21. https://doi.org/10.1016/j.urology.2016.12.011
[4] Örtegren J, Kohestani K, Elvstam O, et al. Risk factors for infection after transrectal prostate biopsy: a population-based register study. Eur Urol Open Sci. 2024;67:1–6. https://doi.org/10.1016/j.euros.2024.06.015
[5] Ljungquist O, Persmark A, Grabe M, et al. Increasing rates of urinary and bloodstream infections following transrectal prostate biopsy in South Sweden. BJU Int. 2022;130(4):478–485. https://doi.org/10.1111/bju.15713
[6] Lundström KJ, Drevin L, Carlsson S, et al. Nationwide population based study of infections after transrectal ultrasound guided prostate biopsy. J Urol. 2014;192(4):1116–1122. https://doi.org/10.1016/j.juro.2014.04.098
[7] Bennett HY, Roberts MJ, Doi SA, et al. The global burden of major infectious complications following prostate biopsy. Epidemiol Infect. 2016;144(8):1784–1791. https://doi.org/10.1017/S0950268815002885
[8] Xiang J, Yan H, Li J, et al. Transperineal versus transrectal prostate biopsy in the diagnosis of prostate cancer: a systematic review and meta-analysis. World J Surg Oncol. 2019;17(1):31. https://doi.org/10.1186/s12957-019-1573-0
[9] Newman TH, Stroman L, Hadjipavlou M, et al. EXIT from Transrectal prostate biopsies (TREXIT): sepsis rates of transrectal biopsy with rectal swab culture guided antimicrobials versus freehand transperineal biopsy. Prostate Cancer Prostatic Dis. 2022;25(2):283–287. https://doi.org/10.1038/s41391-021-00438-w
[10] Stangl-Kremser J, Ramaswamy A, Hu JC. Transperineal vs. transrectal biopsy to reduce post interventional sepsis. Curr Opin Urol. 2023;33(3):193–199. https://doi.org/10.1097/MOU.0000000000001083
[11] EAU Guidelines. Edn. presented at the EAU Annual Congress Milan 2023. ISBN 978-94-92671-19-6.
[12] Hu JC, Assel M, Allaf ME, et al. Transperineal versus transrectal magnetic resonance imaging-targeted and systematic prostate biopsy to prevent infectious complications: the PREVENT randomized trial. Eur Urol. 2024:S0302-2838(23)03342-0. https://doi:10.1016/j.eururo.2023.12.015
[13] Mian BM, Feustel PJ, Aziz A, et al. Complications following transrectal and transperineal prostate biopsy: results of the ProBE-PC randomized clinical trial. J Urol. 2024;211(2):205–213. https://doi.org/10.1097/JU.0000000000003788
[14] Tomic K, Berglund A, Robinson D, et al. Capture rate and representativity of The National Prostate Cancer Register of Sweden. Acta Oncol. 2015;54(2):158–163. https://doi.org/10.3109/0284186X.2014.939299
[15] Barlow L, Westergren K, Holmberg L, et al. The completeness of the Swedish Cancer Register: a sample survey for year 1998. Acta Oncol. 2009;48(1):27–33. https://doi.org/10.1080/02841860802247664
[16] Stattin P. How to improve cancer care by use of guidelines and quality registers. Scand J Urol. 2024;59:190–192. https://doi.org/10.2340/sju.v59.42272
[17] Van Hemelrijck M, Garmo H, Wigertz A, et al. Cohort profile update: the National Prostate Cancer Register of Sweden and prostate cancer data base – a refined prostate cancer trajectory. Int J Epidemiol. 2016;45(1):73–82. https://doi.org/10.1093/ije/dyv305
[18] Ludvigsson JF, Andersson E, Ekbom A, et al. External review and validation of the Swedish national inpatient register. BMC Public Health. 2011;11:450. https://doi.org/10.1186/1471-2458-11-450
[19] Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. https://doi.org/10.1016/0021-9681(87)90171-8
[20] Wettermark B, Hammar N, Fored CM, et al. The new Swedish Prescribed Drug Register – opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol Drug Saf. 2007;16(7):726–735. https://doi.org/10.1002/pds.1294
[21] Gedeborg R, Sund M, Lambe M, et al. An aggregated comorbidity measure based on history of filled drug prescriptions: development and evaluation in two separate cohorts. Epidemiology. 2021;32(4):607–615. https://doi.org/10.1097/EDE.0000000000001358
[22] Brooke HL, Talbäck M, Hörnblad J, et al. The Swedish cause of death register. Eur J Epidemiol. 2017;32(9):765–773. https://doi.org/10.1007/s10654-017-0316-1
[23] Classification of Surgical Procedures 1997 (KKÅ) [Internet]. The National Board of Health and Welfare; 2004 [cited 2023 Oct 23]. Available from:https://www.socialstyrelsen.se/globalassets/sharepoint-dokument/artikelkatalog/klassifikationer-och-koder/kkakum2005-2020.pdf
[24] Forsvall A, Jönsson H, Wagenius M, et al. Rate and characteristics of infection after transrectal prostate biopsy: a retrospective observational study. Scand J Urol. 2021;55(4):317–323. https://doi.org/10.1080/21681805.2021.1933169
[25] Lam W, Wong AHG, Chun S, et al. P0999 – prostate cancer detection, tolerability and safety of transperineal prostate biopsy under local-anaesthesia versus standard transrectal biopsy in biopsy-naïve men: a pragmatic, parallel group, randomized-controlled study. Eur Urol. 2021;79(S1):S1405. ISSN 0302-2838. https://doi.org/10.1016/S0302-2838(21)01372-5
[26] Styrke J, Resare S, Lundström KJ, et al. Current routines for antibiotic prophylaxis prior to transrectal prostate biopsy: a national survey to all urology clinics in Sweden. F1000Res. 2020;9:58. https://doi.org/10.12688/f1000research.19260.1
[27] Swedres-Svarm. 2023. Sales of antibiotics and occurrence of antibiotic resistance in Sweden. Solna/Uppsala. ISSN 2001-7901 Available at: www.sva.se/swedres-svarm/
[28] Antibiotic prophylaxis in urological surgery [Internet] Swedish Urology Association; 2023 [cited 2025 Jun 15]. Available from: https://strama-nationell.infosynk.se/category/profylax Accessed 2024-04-24
[29] Bratt O, Carlsson S, Fransson P, et al. Swedish National Prostate Cancer Guidelines Group. The Swedish national guidelines on prostate cancer, part 1: early detection, diagnostics, staging, patient support and primary management of non-metastatic disease. Scand J Urol. 2022;56(4):265–273. https://doi.org/10.1080/21681805.2022.2094462
[30] Berg S, Tully KH, Hoffmann V, et al. Assessment of complications after transperineal and transrectal prostate biopsy using a risk-stratified pathway identifying patients at risk for post-biopsy infections. Scand J Urol. 2023;57(1–6):41–46. https://doi.org/10.1080/21681805.2023.2168049
[31] Bryant RJ, Marian IR, Williams R, et al; TRANSLATE Trial Study Group. Local anaesthetic transperineal biopsy versus transrectal prostate biopsy in prostate cancer detection (TRANSLATE): a multicentre, randomised, controlled trial. Lancet Oncol. 2025;26(5):583–595.