ORIGINAL RESEARCH ARTICLE
Impact of asymptomatic bacteriuria on the outcomes and tolerability of Bacillus Calmette–Guérin immunotherapy
Antti Nummia, Pertti Nurminenb, Olli Kestib, Mikael Högermanb, Otto Ettalab, Peter J. Boströmb, Antti Kaipiac, Jukka Sairanena and Riikka Järvinena
aDepartment of Urology, Helsinki University Hospital, Helsinki, Finland; bDepartment of Urology, Turku University Hospital, Turku, Finland; cDepartment of Urology, Tampere University Hospital, Tampere, Finland
ABSTRACT
Objective: To determine whether asymptomatic bacteriuria (ABU) prior to Bacillus Calmette-Guérin (BCG) immunotherapy has an impact on the oncological results and overall tolerability of BCG treatment in patients with non-muscle-invasive bladder cancer (NMIBC).
Materials and methods: We analyzed retrospectively all patients who received ≥ 1 BCG instillations as treatment of NMIBC in Helsinki University Hospital and Turku University Hospital during 2009–2018. Patients submitted urine specimen 1–7 days prior to the initiation of BCG therapy. ABU was classified as having any positive urine culture but no dysuria or fever. Our primary endpoints were 5-yr recurrence-free survival and progression-free survival. Discontinuation rate of BCG therapy was a secondary endpoint.
Results: We identified 795 patients, of whom 154 (19%) had ABU prior to the first BCG instillation. The 5-yr recurrence-free survival rates in the uninfected and bacteriuric groups were 63% (95% confidence interval [CI]: 59–67%) vs. 69% (95% CI: 62–78%), respectively (hazard ratio [HR] 0.83, 95% CI 0.60–1.14). The 5-yr progression-free survival rates were 88% (95% CI: 86–90%) vs. 89% (95% CI: 84–94%), respectively (HR 0.86, 95% CI 0.50–1.49). The 3-yr discontinuation-free survival rates were 51% (95% CI: 47–54%) vs. 51% (95% CI: 44–60%), respectively (HR 0.98, 95% CI 0.75–1.28).
Conclusion: ABU did not significantly affect BCG immunotherapy outcomes. Intravesical BCG during ABU is safe, with similar discontinuation rates, indicating very similar treatment tolerability.
KEYWORDS: asymptomatic bacteriuria; Bacillus Calmette-Guérin; nonmuscle-invasive bladder cancer; tolerability; adverse effects; complications; BCG infection
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 148–155. https://doi.org/10.2340/sju.v61.45539.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 31 October 2025; Accepted: 21 January 2026; Published: 24 April 2026
CONTACT: Antti Nummi, MD antti.nummi@hus.fi Department of Urology, Helsinki University Hospital, Helsinki, Finland
Introduction
Intravesical immunotherapy with Bacillus Calmette-Guérin (BCG) remains the standard adjuvant treatment for high-risk non-muscle-invasive bladder cancer (NMIBC) following transurethral resection of bladder tumor (TURBT) [1, 2]. Instillation of BCG induces a strong cytokine-mediated immune reaction within the bladder wall. This response may also augment the innate immune response of the urothelial mucosa, resulting in the destruction of malignant urothelial cells [3, 4]. Looking at it through the lens of bladder biology, BCG immunotherapy can be seen as a purposeful urinary tract infection [5].
Similarly, uropathogen strains colonizing the bladder mucosa induce a modest inflammatory response and trigger the innate immune system [6]. It has been proposed that such microbial priming may modulate the mucosal immune environment, potentially enhancing the therapeutic response to BCG and contributing to antitumor activity [4, 7].
Asymptomatic bacteriuria (ABU) is common in patients with NMIBC and BCG administration during ABU is generally considered safe [8, 9]. Local and systemic adverse effects of BCGimmunotherapy are widely known and account for up to 29–53% of all treatment discontinuations in previous studies [10–12]. Our study group previously reported that ABU during BCG administration does not increase the risk of severe BCG adverse effects [13]. However, there are currently no published studies comparing the overall tolerability of BCG maintenance therapy between bacteriuric and uninfected patients.
The aim of this study was to determine whether ABU prior to BCG immunotherapy has an impact on the oncological results and overall tolerability of the BCG treatment.
Materials and methods
Data sources and patient identification
This study was conducted at the University Hospital of Helsinki and the University Hospital of Turku. We performed a retrospective review of institutional databases using the operation code for intravesical BCG instillation, including all patients who received at least one BCG instillation for the treatment of NMIBC between 2009 and 2018.
If BCG treatment was initiated before 2009, medical records were reviewed starting from the first BCG instillation within the regimen. For treatments that continued beyond 2018, records were reviewed up to the completion of the BCG maintenance.
Characterization of the patients
The following patient characteristics were retrospectively assessed: sex, age, patient comorbidities using Charlson comorbidity index, smoking status, immunosuppressive state defined as the presence of hematological disorder, human immunodeficiency virus (HIV) infection, history of splenectomy, systemic immunosuppressive medication, or systemic prednisolone use for any indication with a daily dose of ≥ 10 mg.
TURBT operation was performed on all patients before the first BCG instillation. Tumor characteristics were recorded according to Tumour, Node, Metastasis (TNM) and WHO 2004 classifications. Tumor size and multifocality based on cystoscopic assessment together with NMIBC risk group using EORTC risk group stratification were recorded.
Bacteriuria and BCG therapy
All patients submitted a clean-catch voided midstream urine specimen 1–7 days prior to the initiation of BCG therapy, collected according to standardized urine sampling protocols. Urine culture results were reported as a screening interpretation together with quantitative bacterial counts. We did not utilize specialized methods to identify the organisms characterizing the urine microbiome. Urine cultures were recorded as no growth or the presence of bacteriuria, classified as 1000–100,000 organisms per mL (including mixed flora). Urine culture results reported as urogenital normal flora were interpreted as contamination and were not included in the positive urine culture group. ABU was classified as having any positive urine culture and no severe dysuria or fever. Possible use of antibiotic prophylaxis was recorded.
Patients with negative urine culture prior to the first BCG administration were classified as uninfected. Patients with ABU prior to the first BCG administration were classified in bacteriuria group. After the induction course of BCG therapy consisting of 6 weekly BCG instillations, patients received BCG maintenance therapy for 1–3 years according to SWOG (Southwest Oncology Group) or monthly instillation regimens.
Discontinuation of BCG treatment
Patients were followed throughout and after BCG therapy with regular cystoscopies according to European Association of Urology (EAU) guidelines. The reason and date of discontinuation of BCG treatment were recorded. BCG failure was classified as discontinuation of BCG treatment due to recurrences or disease progression. Toxicity was classified as discontinuation due to local or general adverse effects without the usage of antituberculotic treatment. BCG infection was classified as discontinuation of BCG treatment due to BCG infectious complication requiring antituberculotic treatment. Compliance was classified as discontinuation due to patients own will without recurrence or BCG-related adverse effects reported. Reason for discontinuation was classified as other if the patient died due to non-bladder cancer-related reasons or was diagnosed with another severe disease or if the reason of discontinuation was unclear.
Oncological results
Recurrence during BCG treatment was defined as a biopsy-confirmed Ta or T1 tumors after 3 months of BCG treatment and within 3 months of the last BCG instillation. Carcinoma in situ tumors after 6 months of BCG treatment were considered as recurrences during BCG treatment. Recurrence after BCG treatment group consists of patients with biopsy-confirmed tumors after BCG treatment had ended (either finished or interrupted). Progression was defined as pT2 or higher disease or diagnosis of metastases.
Statistical analysis
Associations between presence or absence of bacteriuria at initial urine culture and explanatory patient, tumor specific and outcome of BCG treatment variables (age, sex, Charlson Comorbidity Index, immunosuppression, smoking, urine culture, tumor pattern, tumor type, tumor grade, tumor multifocality, tumor size and EORTC 2021 risk groups, number of instillations, number of discontinued BCG courses, and reasons for discontinuation, history of UTUC, recurrence, and progression during BCG treatment and after BCG treatment) were summarized with descriptive statistics and studied one by one with the Wilcoxon rank sum test for nonparametric continuous variables, Pearson’s chi-square test for categorical variables, and Fisher’s exact test for categorical variables with small sample sizes.
The Kaplan–Meier method was used to estimate time-to-event outcomes, such as recurrence-free survival, progression-free survival, and discontinuation-free survival in bacteriuric versus uninfected patients. Cox proportional hazards model and hazard ratio (HR) were calculated and reported for group comparisons and for the effect of explanatory variables (gender, Charlson Comorbidity Index, tumor pattern, tumor type, EORTC risk group, number of instillations, and positive urine culture prior to the first BCG instillation) to all. The statistical significance level was set at 0.05 in all tests (two tailed), and 95% confidence intervals (CIs) were calculated. The analyses were performed using RStudio (version 2024.04.2) based on R (version 4.4.1; RStudio, PBC, Boston, MA, USA).
Results
We identified a total of 816 patients, who received ≥ 1 BCG instillation as treatment of NMIBC during 2009–2018 in our hospitals. We excluded 21 patients due to lack of clinical data. Finally, 795 patients were eligible for further analysis.
Baseline characteristics and oncological details are presented in Table 1. Our cohort included 641 (81%) patients with negative urine culture and 153 (19%) patients with positive urine culture without symptoms of urinary tract infection (UTI) prior to the first BCG administration. Positive urine culture was significantly more common in female patients and in patients with higher median age and higher Charlson comorbidity index (p = 0.007, p = 0.006 and p = 0.007). There was no significant difference in immunosuppressive medication rate or smoking status between the groups. In addition, we found no significant difference in primary tumor size, multifocality, or tumor pattern between the groups. Positive urine culture was more common in patients with higher grade and more invasive tumors (p = 0.069 and p = 0.026). However, this was not reflected in the differences in EORTC risk categories between the groups (p = 0.8). Mixed flora was the most common finding in a positive urine culture, reported with 112 (73%) bacteriuric patients.
| Variable | Overall 795† | Uninfected 641 (81%)† | Bacteriuric 154 (19%)† | P-value‡ | |
| Median age | 73 (66, 79) | 72 (66, 78) | 75 (68, 81) | 0.006 | |
| Sex | Male | 662 (83%) | 545 (85%) | 117 (76%) | 0.007 |
| Female | 133 (17%) | 96 (15%) | 37 (24%) | ||
| Charlson comorbidity index | 2 | 385 (48%) | 328 (51%) | 57 (37%) | 0.007 |
| 3 | 199 (25%) | 153 (24%) | 46 (30%) | ||
| 4 or more | 211 (27%) | 160 (25%) | 51 (33%) | ||
| Immunosuppression | No | 764 (96%) | 613 (96%) | 151 (98%) | 0.164 |
| Yes | 31 (3.9%) | 28 (4.4%) | 3 (1.9%) | ||
| Immunosuppressive medication | 16 (2.0%) | 15 (2.3%) | 1 (0.6%) | ||
| Hematologic disease | 14 (1.8%) | 12 (1.9%) | 2 (1.3%) | ||
| Splenectomy | 1 (0.1%) | 1 (0.2%) | 0 | ||
| Smoking status | Never smoker | 225 (32%) | 186 (33%) | 39 (30%) | 0.324 |
| Ex smoker | 282 (41%) | 232 (41%) | 50 (38%) | ||
| Smoking | 186 (27%) | 144 (26%) | 42 (32%) | ||
| Unknown | 102 | 79 | 23 | ||
| Urine culture | No growth | 641 (81%) | 641 (100%) | - | |
| Mixed flora | 112 (14%) | - | 112 (73%) | ||
| 1000–100,000 CFU/mL | 5 (0.6%) | - | 5 (3.2%) | ||
| > 100,000 CFU/mL | 37 (4.7%) | - | 37 (24%) | ||
| Tumor pattern | Primary | 562 (71%) | 452 (71%) | 110 (71%) | 0.823 |
| Recurrent | 233 (29%) | 189 (29%) | 44 (29%) | ||
| Primary tumor type | Ta | 294 (37%) | 249 (39%) | 45 (29%) | 0.026 |
| Cis | 77 (9.7%) | 64 (10%) | 13 (8.5%) | ||
| T1 | 411 (52%) | 320 (50%) | 91 (59%) | ||
| T2–4 | 9 (1.1%) | 5 (0.8%) | 4 (2.6%) | ||
| Unknown | 4 | 3 | 1 | ||
| Primary tumor grade | PUNLPM | 9 (1.2%) | 8 (1.3%) | 1 (0.7%) | 0.069 |
| Low grade | 95 (12%) | 84 (13%) | 11 (7.2%) | ||
| High grade | 672 (87%) | 532 (85%) | 1140 (92%) | ||
| Unknown | 19 | 17 | 2 | ||
| Tumor multifocality | 1 | 430 (55%) | 343 (54%) | 87 (58%) | 0.777 |
| 2–7 | 334 (43%) | 273 (43%) | 61 (40%) | ||
| 8 or more | 20 (2.6%) | 17 (2.7%) | 3 (2.0%) | ||
| Unknown | 11 | 8 | 3 | ||
| Tumor size | < 1 cm | 54 (7.0%) | 47 (7.5%) | 7 (4.7%) | 0.166 |
| 1–3 cm | 445 (57%) | 365 (58%) | 80 (54%) | ||
| > 3 cm | 277 (36%) | 215 (34%) | 62 (42%) | ||
| Unknown | 19 | 14 | 5 | ||
| EORTC$ risk group | Low risk | 2 (0.3%) | 2 (0.3%) | 0 (0%) | 0.809 |
| Intermediate risk | 115 (15%) | 96 (15%) | 19 (12%) | ||
| High risk | 508 (64%) | 406 (64%) | 102 (67%) | ||
| Very high risk | 166 (21%) | 134 (21%) | 32 (21%) | ||
| †n (%), ‡Wilcoxon rank sum test, Pearson’s chi-squared test, $European Organisation for Research and Treatment of Cancer. BCG: Bacillus Calmette–Guérin; CFU: Colony-Forming Unit. | |||||
Outcomes of the BCG treatment are presented in Table 2. Median number of instillations given was 17 with no difference between the groups. BCG treatment was discontinued with 390 (49%) patients. There was no difference in the discontinuation rate between the groups, reported in 315 (49%) and 75 (49%) patients with negative and positive urine cultures, respectively (p = 0.9). Symptomatic urinary tract infection requiring hospital admission after any instillations during the BCG regimen was reported in 38 (4.8%) patients with no difference between the groups (p = 0.3). Recurrence during BCG treatment was reported in 128 (20%) patients with negative urine culture and in 25 (16%) patients with positive urine culture, respectively (p = 0.3). Recurrence after BCG treatment was reported in 117 (18%) and 24 (16%) patients with negative urine culture and positive urine culture, with no significant difference between the groups (p = 0.4). Disease progression during BCG treatment was reported in 34 (5.3%) patients with negative urine culture and in 5 (3.2%) patients with positive urine culture, respectively (p = 0.3). Progression after BCG treatment was reported in 63 (7.9%) patients with no difference between the groups (p > 0.9).
| Variable | Overall | Uninfected | Bacteriuric | P-value‡ | |
| n = 795† | n = 641† | n = 154† | |||
| Median number of instillations given | 17 (12, 21) | 17 (12, 21) | 17 (13, 21) | 0.604 | |
| Completion of BCG maintenance | Finished BCG | 403 (51%) | 324 (51%) | 79 (51%) | 0.985 |
| Discontinued BCG | 390 (49%) | 315 (49%) | 75 (49%) | ||
| Unknown | 2 | 2 | 0 | ||
| Reason for discontinuation | BCG failure | 89 (23%) | 72 (23%) | 17 (23%) | 0.349 |
| Toxicity | 195 (50%) | 160 (50%) | 35 (47%) | ||
| BCG infection or suspicion of BCG infection | 22 (5.6%) | 20 (6.3%) | 2 (2.7%) | ||
| Compliance | 21 (5.4%) | 14 (4.4%) | 7 (9.3%) | ||
| Other | 65 (17%) | 51 (16%) | 14 (19%) | ||
| Recurrence during BCG treatment | No | 642 (81%) | 513 (80%) | 129 (84%) | 0.291 |
| Low grade | 29 (3.6%) | 26 (4.1%) | 3 (1.9%) | ||
| High grade | 124 (16%) | 102 (16%) | 22 (14%) | ||
| Recurrence after BCG treatment | No | 654 (82%) | 524 (82%) | 130 (84%) | 0.436 |
| Low grade | 33 (4.2%) | 27 (4.2%) | 6 (3.9%) | ||
| High grade | 108 (14%) | 90 (14%) | 18 (12%) | ||
| Progression during BCG treatment | 39 (4.9%) | 34 (5.3%) | 5 (3.2%) | 0.288 | |
| Progression after BCG treatment | 63 (7.9%) | 51 (8.0%) | 12 (7.8%) | 0.946 | |
| Symptomatic UTI requiring hospital admission$ | 38 (4.8%) | 33 (5.1%) | 5 (3.2%) | 0.321 | |
| †n (%), ‡Wilcoxon rank sum test, Pearson’s chi-squared test, Fisher’s exact test, $during the whole BCG regimen. BCG: Bacillus Calmette–Guérin. | |||||
The Kaplan–Meier curves estimating recurrence-free survival (A) and progression-free survival (B) between the groups are presented in Figure 1 with median follow-up time of 66 months. Disease recurrence was reported in 298 (37%) patients in the whole cohort, of which 246 (38%) in the uninfected group and 49 (32%) in the bacteriuric group. A Cox regression analysis revealed a HR of 0.83 (95% CI: 0.60–1.14) in favor of the bacteriuric group. The Kaplan–Meier analysis indicated a 5-year recurrence-free survival (95% CI) in the uninfected vs. the bacteriuric group of 63% (59–67%) vs. 69% (62–78%).
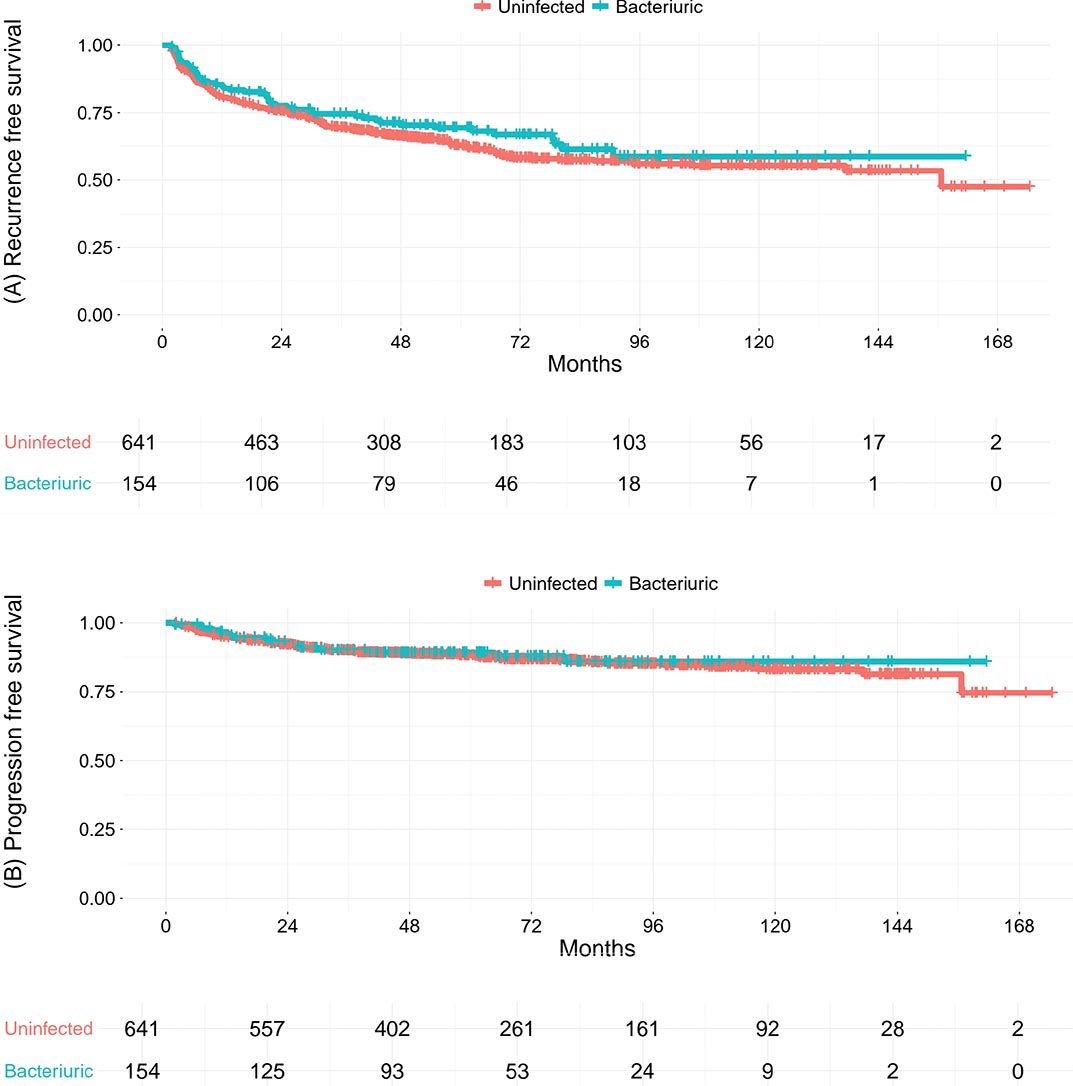
Figure 1. Kaplan–Meier curves for recurrence-free survival (A) and progression-free survival (B) in 795 patients with non–muscle-invasive bladder cancer undergoing intravesical Bacillus Calmette–Guérin (BCG) therapy, comparing bacteriuric (blue curve) and uninfected (red curve) patients prior to the first BCG instillation. The median follow-up time was 66 (IQR: 44–99) mo. IQR: interquartile range.
Disease progression was reported in 104 (13%) patients across the entire cohort, with 85 (13%) patients in the uninfected group and 17 (11%) in the bacteriuric group. A Cox regression analysis revealed a HR of 0.90 (95% CI: 0.53–1.50) in favor of the bacteriuric group. The Kaplan–Meier analysis demonstrated a 5-year progression-free survival (95% CI) in the uninfected vs. the bacteriuric group of 88% (86–90%) vs. 89% (84–94%).
Figure 2 presents a Kaplan–Meier curve estimating unplanned BCG treatment discontinuation-free survival. We found no difference in discontinuation rates between the groups. The 3-yr discontinuation-free survival rate was 51% (95% CI: 47–54%) in the uninfected and 51% (95% CI: 44–60%) in the bacteriuric group. As a result, no significant difference was observed in a Cox regression analysis with an HR of 0.98 (95% CI: 0.75–1.28) between the groups.
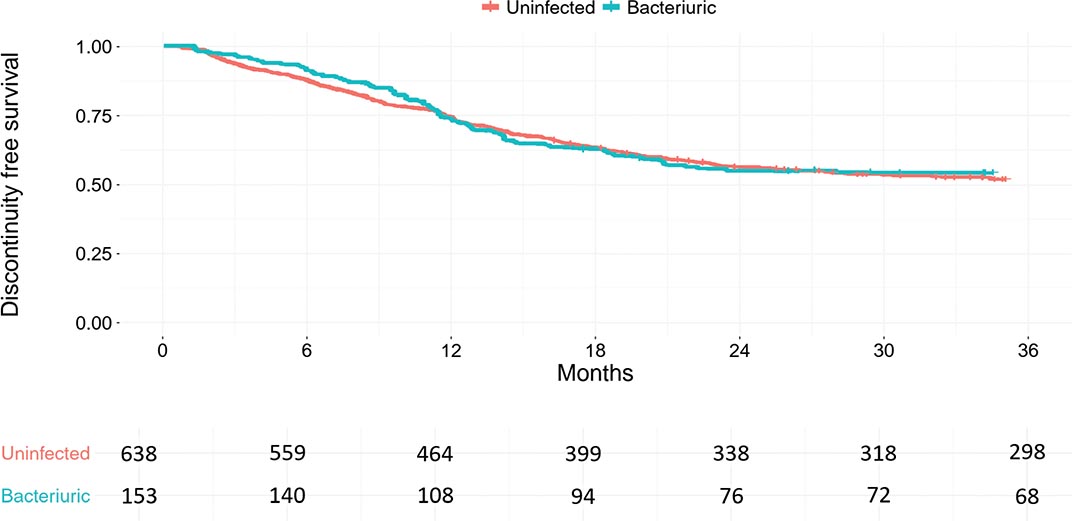
Figure 2. Kaplan–Meier curves for unplanned discontinuation of Bacillus Calmette–Guérin (BCG) therapy in 791 patients with non–muscle-invasive bladder cancer, comparing bacteriuric (blue curve) and uninfected (red curve) patients prior to the first BCG instillation. Four patients were excluded from the Kaplan–Meier analysis owing to missing data on treatment discontinuation status.
In Table 3, we present the Cox proportional hazards model assessing the risk factors for recurrences, disease progression, and discontinuation of BCG treatment. In this multivariate analysis, the result of urinary culture prior to the first BCG instillation did not significantly affect the rate of disease recurrence, progression, or treatment discontinuation. However, significant risk factors for disease recurrence rate were history of tumor recurrence with HR of 1.85 (95% CI: 1.35–2.54) compared with patients with primary tumors, primary carcinoma in situ with HR of 2.44 (95% CI: 1.57–3.79) compared with Ta tumors, and lower number of BCG instillations with HR of 0.94 (95% CI: 0.92–0.96). Significant risk factors for disease progression were CCI 4 or more with HR of 1.84 (95% CI: 1.14–2.97) compared with CCI 2, primary carcinoma in situ with HR of 2.94 (95% CI: 1.49–5.79) compared with Ta tumors, and high risk and very high risk EORTC risk groups with HRs of 2.29 and 4.06 (95% CI: 1.02–5.17 and 1.65–10.01) compared with low-intermediate risk groups. A higher number of BCG instillations was associated with a reduced risk of disease progression with HR of 0.91 (95% CI: 0.88–0.94).
| Variable | Recurrence | Progression | Discontinuation of BCG treatment |
| HR (95% CI)‡$ | HR (95% CI)‡$ | HR (95% CI)‡$ | |
| Gender | |||
| Female | ref | ref | ref |
| Male | 0.85 (0.62–1.15) | 0.63 (0.39–1.03) | 1.10 (0.82–1.48) |
| Charlson comorbidity index | |||
| 2 | ref | ref | ref |
| 3 | 1.00 (0.74–1.34) | 0.64 (0.36–1.16) | 1.02 (0.78–1.32) |
| 4 or more | 1.13 (0.83–1.52) | 1.84 (1.14–2.97) | 1.01 (0.78–1.33) |
| Tumor pattern | |||
| Primary | ref | ref | ref |
| Recurrent | 1.85 (1.35–2.54) | 1.13 (0.63–2.05) | 1.51 (1.14–2.01) |
| Primary tumor type | |||
| Ta | ref | ref | ref |
| Cis | 2.44 (1.57–3.79) | 2.94 (1.49–5.79) | 1.24 (0.82–1.86) |
| T1 | 1.13 (0.82–1.55) | 0.93 (0.52–1.65) | 0.92 (0.70–1.21) |
| EORTC risk group€ | |||
| Low-intermediate risk | ref | ref | ref |
| High risk | 1.10 (0.75–1.61) | 2.29 (1.02–5.17) | 1.60 (1.12–2.31) |
| Very high risk | 1.86 (1.19–2.91) | 4.06 (1.65–10.01) | 2.40 (1.57–3.67) |
| Number of instillations given | 0.94 (0.92–0.96) | 0.91 (0.88–0.94) | 0.79 (0.77–0.81) |
| Positive urine culture prior to the first BCG instillation | |||
| No | ref | ref | ref |
| Yes | 0.83 (0.60–1.14) | 0.86 (0.50–1.49) | 0.98 (0.75–1.28) |
| Median (IQR†) follow-up time is 66 (44–99) months. ref: Reference variable; †IQR: Interquartile range; ‡Cox proportional hazards model; $HR: Hazard ratio; CI: Confidence interval; €EORTC: European Organisation for Research and Treatment of Cancer; BCG: Bacillus Calmette–Guérin. | |||
Discussion
In this study, we demonstrated that ABU preceding BCG therapy does not have a significant impact on the oncological outcomes or the overall tolerability of the BCG treatment.
There are few previous studies comparing the efficacy of BCG immunotherapy with ABU and sterile urine. In 2011 and 2012, Herr et al. demonstrated that the oncological outcomes of BCG therapy were similar between patients with sterile urine and patients with significant single-organism bacteriuria detected in the pretreatment urine culture [8, 14]. However, in a more recent publication in 2020, which also included patients with lower bacterial burdens (including mixed flora), a significant difference in 3-year recurrence-free survival was found in favor of the bacteriuria group (83% vs. 65%, p = 0.001). The 3-year Recurrence-free survival (RFS) was superior in patients with the lowest but detectable (< 104 Colony-Forming Unit (CFU)/mL) bacterial burden [7]. Although the difference remains statistically insignificant in our study, we demonstrate a similar trend favoring the bacteriuria group in terms of 3-year RFS of 75% vs. 69% (HR 0.83, 95% CI: 0.60–1.14). In our study, most common finding in a positive urine culture was mixed flora, documented in 112 (73%) cases. These results are in line with the previous studies, supporting the hypothesis that the beneficial effect of BCG is most favorable in patients with unculturable organisms. However, this modest benefit did not translate into progression-free survival rates, as the results were similar between the bacteriuria and the uninfected groups (HR 0.86, 95% CI: 0.50–1.49) in our study.
There are currently no published studies comparing the overall tolerability of BCG therapy between bacteriuric and uninfected patients. Our study group has previously reported a discontinuation rate of (42–44%) with patients receiving BCG maintenance [12, 15]. In this study, we demonstrate a total discontinuation rate of 49% including also patients whose BCG therapy was discontinued during the BCG induction period. We found no difference in the discontinuation rates between the bacteriuria and the uninfected groups (p = 0.9). Toxicity was the most common reason for treatment discontinuation in both the bacteriuria and the uninfected groups, reported in similar rates 47 and 50% of discontinued patients, respectively (p = 0.3). These results suggest that even though uropathogen strains colonizing the bladder mucosa induce a modest inflammatory response, this does not enhance the irritative effects of BCG therapy. Decreased compliance, as a reason for discontinuation, was slightly more common in the bacteriuria group (9.3% vs. 4.4%), supporting the fact that ABU is more commonly found in elderly, comorbid patients. Overall, concurrent ABU does not seem to affect the general tolerability of BCG immunotherapy.
We report similar rates of ABU (19%) in patients undergoing BCG therapy as previously published [9, 16]. Positive urine culture was significantly more common in female and in patients with higher age and higher Charlson comorbidity index. All patients underwent transurethral procedures prior to the first BCG instillation. In cases of higher risk tumors, also a second-look resection was performed, increasing the risk of developing bacteriuria. This may partially explain why patients with more invasive and higher-grade tumors had increased rates of bacteriuria (p = 0.026 and p = 0.069). Although bacteriuria was associated with markers of tumor aggressiveness, this did not translate into inferior oncological outcomes, possibly reflecting immunomodulatory effects of bacterial exposure. Our multivariable analysis demonstrated only carcinoma in situ and high-very high EORTC risk groups as independent variables associated with both higher tumor recurrences and progressions. Charlson comorbidity index of 4 or higher was associated with higher progression rates and history of recurrent tumors with higher recurrence rates. Age, sex, and the presence of ABU were insignificant. ABU generally occurs more frequently in patients with poorer outcomes of BCG treatment, which may introduce a potential source of bias. Due to this, a multivariate analysis was performed, and the difference between the bacteriuria and uninfected groups did not emerge in our study.
The purpose of this study was to investigate the impact of ABU preceding BCG therapy on the outcomes and tolerability of the BCG treatment. Limitations of our study come with its retrospective setting. This study does not include a standardized form for patients to report milder adverse effects impacting quality of life during BCG treatment. As a result, overall tolerability is inferred from total discontinuation rates. In addition, mixed bacterial flora in urine cultures is commonly interpreted as contamination; however, in the context of BCG immunotherapy, bacterial presence may still influence the urothelial immune milieu. The authors acknowledge that the majority of positive urine cultures consisted of mixed flora, and such findings were included in the bacteriuria group, recognizing the inherent limitations of urine culture interpretation in retrospective analyses. Furthermore, individual data on the specific BCG strains (RIVM and Tice) were not available, but as BCG TICE represented less than 5% of all instillations, the effect of strain variation is likely negligible. Finally, a single urine culture prior to the initiation of BCG treatment may under- or overestimate the true number of patients with chronic ABU. Recently the long-standing assumption that the bladder mucosa is sterile has been questioned, highlighting the need for further investigation into the role of nonculturable microorganisms in modulating the immune response to BCG therapy.
Conclusion
ABU did not significantly impact the outcomes of BCG immunotherapy. Intravesical BCG administration in the presence of ABU is safe and does not compromise the overall tolerability of the treatment, as evidenced by comparable discontinuation rates. Future studies should also incorporate microbiome-based approaches to clarify how microbial communities modulate immune responses and clinical outcomes of BCG therapy.
References
- [1] Babjuk M, Böhle A, Burger M, et al. EAU guidelines on non–muscle-invasive urothelial carcinoma of the bladder: update 2016. Eur Urol. 2017;71(3):447–461. https://doi.org/10.1016/j.eururo.2016.05.041
- [2] Shelley MD, Kynaston H, Court J, et al. A systematic review of intravesical bacillus Calmette-Guérin plus transurethral resection vs transurethral resection alone in Ta and T1 bladder cancer. BJU Int. 2001;88(3): 209–216. https://doi.org/10.1046/j.1464-410x.2001.02306.x
- [3] Song J, Abraham SN. Innate and adaptive immune responses in the urinary tract. Eur J Clin Investig. 2008;38(s2):21–28. https://doi.org/10.1111/j.1365-2362.2008.02005.x
- [4] Ingersoll MA, Albert ML. From infection to immunotherapy: host immune responses to bacteria at the bladder mucosa. Mucosal Immunol. 2013;6(6):1041–1053. https://doi.org/10.1038/mi.2013.72
- [5] Morales A, Eidinger D, Bruce AW. Intracavitary Bacillus Calmette-guerin inthe treatment of superficial bladder tumors. J Urol. 1976;116(2): 180–182. https://doi.org/10.1016/S0022-5347(17)58737-6
- [6] Hernández JG, Sundén F, Connolly J, et al. Genetic control of the variable innate immune response to asymptomatic bacteriuria. PLoS One. 2011;6(11):e28289. https://doi.org/10.1371/journal.pone.0028289
- [7] Herr H. Does asymptomatic bacteriuria affect the response to intravesical bacillus Calmette–Guérin? Int J Urol. 2020;27(1):72–74. https://doi.org/10.1111/iju.14139
- [8] Herr HW. Intravesical Bacille Calmette-Guérin in patients with asymptomatic bacteriuria. JAMA. 2011;305(14):1413–1414. https://doi.org/10.1001/jama.2011.428
- [9] Herr HW. Cystoscopy and intravesical bacille Calmette–Guérin therapy in antibiotic-naïve patients with bladder cancer with asymptomatic bacteriuria: an update. Arab J Urol. 2016;14(2):75–77. https://doi.org/10.1016/j.aju.2016.03.002
- [10] Brausi M, Oddens J, Sylvester R, et al. Side effects of Bacillus Calmette-Guérin (BCG) in the treatment of intermediate- and high-risk Ta, T1 papillary carcinoma of the bladder: results of the EORTC Genito-Urinary Cancers Group randomised phase 3 study comparing one-third dose with full dose and 1 year with 3 years of maintenance BCG. Eur Urol. 2014;65(1):69–76. https://doi.org/10.1016/j.eururo.2013.07.021
- [11] van der Meijden APM, Sylvester RJ, Oosterlinck W, et al. Maintenance Bacillus Calmette-Guerin for Ta T1 bladder tumors is not associated with increased toxicity: results from a European Organisation for Research and Treatment of Cancer Genito-Urinary Group Phase III trial. Eur Urol. 2003;44(4):429–434. https://doi.org/10.1016/S0302-2838(03)00357-9
- [12] Nummi A, Järvinen R, Sairanen J, et al. A retrospective study on tolerability and complications of bacillus Calmette-Guérin (BCG) instillations for non-muscle-invasive bladder cancer. Scand J Urol. 2019;53(2–3): 116–122. https://doi.org/10.1080/21681805.2019.1609080
- [13] Nummi A, Nurminen P, Järvinen R, et al. Time to abandon routine urine screening prior bacillus Calmette-Guérin administration in asymptomatic patients. BJUI Compass. 2025;6(12):e70117. https://doi.org/10.1002/bco2.70117
- [14] Herr HW. Intravesical Bacillus Calmette-Guérin outcomes in patients with bladder cancer and asymptomatic bacteriuria. J Urol. 2012;187(2):435–437. https://doi.org/10.1016/j.juro.2011.10.032
- [15] Nurminen P, Nummi A, Kesti O, et al. Comparison of Bacillus Calmette-Guérin maintenance therapy with monthly instillations and the Southwest Oncology Group Protocol in the treatment of non-muscle-invasive bladder cancer. Eur Urol Focus. 2023;9(6): 1000–1007. https://doi.org/10.1016/j.euf.2023.04.012
- [16] Herr HW. Intravesical Bacille Calmette-Guérin eradicates bacteriuria in antibiotic-naïve bladder cancer patients. Eur Urol. 2013;63(5): 832–835. https://doi.org/10.1016/j.eururo.2012.08.013