ORIGINAL RESEARCH ARTICLE
Introduction of a modified surgical approach for ProACT balloon placement: a retrospective cohort study
Jakob Klint Axelsena,b, Lars Boesena,b  , Rasmus Bisbjerga
, Rasmus Bisbjerga  , Nis Nørgaarda
, Nis Nørgaarda  , Helene Reif Andersena,b and Mikkel Fodea,b,
, Helene Reif Andersena,b and Mikkel Fodea,b, 
aDepartment of Urology, Copenhagen University Hospital, Herlev and Gentofte Hospital, Herlev, Denmark; bDepartment of Clinical Medicine, University of Copenhagen, Copenhagen, Denmark
ABSTRACT
Objective: To compare adjustable continence balloons (ProACT) implanted above the pelvic floor with or without subsequent placement of a third balloon (AP) with a modified technique placing balloons below the pelvic floor (BP).
Materials and methods: We retrospectively compared 38 men who underwent the BP procedure and 38 men who received the AP technique. Primary outcome was continence (≤1 pad/day or <8 ml/24 h leakage). Secondary outcomes included operative time, complications, number of balloon filling visits, and satisfaction.
Results: Median age was 75.5 years (BP) and 70.5 years (AP) (p < 0.0001). Groups were comparable in preoperative incontinence severity. Median surgical time was 15 min (BP) versus 20 min (AP) (p = 0.034). Early retention occurred in 18.4% (BP) versus 0% (AP) (p = 0.012). A third balloon was inserted in 39.4% of AP patients, and median number of fillings (6 vs. 10, p = 0.0006) was lower in BP group. Late complications consisting of migration, erosion, and balloon puncture were similar (23.7% BP vs. 36.8% AP, p = 0.32). Continence was achieved in 52.6% (BP) and 57.9% (AP) (p = 0.64), and satisfaction was reported by 71.1% (BP) and 57.9% (AP) (p = 0.34). On multivariate analyses, surgical method did not predict outcomes, and Cox regression shoved that BP was associated with a non-significant hazard of revision (HR: 1.60; 95% confidence interval [CI]: 0.53–4.85; p = 0.40).
Conclusions: ProACT placement below the pelvic floor yielded similar continence but reduced operative time and postoperative visits, at the cost of more early retention.
KEYWORDS: Urinary incontinence; prostatectomy; minimally invasive surgical procedures; ProACT; prostheses and implants
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 38–43. https://doi.org/10.2340/sju.v61.45579.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 3 September 2025; Accepted: 30 January 2026; Published: 9 March 2026
CONTACT: Mikkel Fode Mikkel.mejlgaard.fode@regionh.dk Department of Urology, Copenhagen University Hospital, Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark
Introduction
Stress urinary incontinence following RARP and TUR-P is a well-documented side effect. Following radical prostatectomy, rates range from 1% to 87%, depending on definition, whereas incontinence after TUR-P is reported at around 8% [1, 2]. The pathophysiology includes iatrogenic injury to the internal urethral sphincter complex alongside injury to periprostatic neurovascular structures [3, 4]. Additionally, patient-related factors such as advanced age, elevated body mass index, and interindividual anatomical variability, particularly in prostate size and the configuration of adjacent pelvic tissues, have been associated with increased risk of incontinence after radical prostatectomy [2]. For some, incontinence is transient [5], while in others it persists, which impacts quality of life and self-image [6].
Over the past two decades, several minimally invasive surgical therapies (MISTs) for benign prostatic obstruction have been introduced with the aim of reducing perioperative morbidity while preserving urinary continence. These include transurethral microwave thermotherapy (TUMT), transurethral needle ablation (TUNA), prostatic urethral lift (UroLift), prostatic arterial embolization, and convective water vapor therapy (Rezūm) [7]. In general, MISTs are considered to carry a low risk of stress urinary incontinence, likely due to reduced mechanical disruption of the bladder neck and sphincteric structures. Notably, TUMT appears to be associated with a particularly low risk of postoperative stress urinary incontinence, despite inducing substantial thermal effects within the prostate and being applicable even in patients with large prostate volumes [8].
For patients who do develop persistent stress urinary incontinence, several surgical incontinence treatments are available. This includes male slings, artificial urinary sphincters (AUS), and adjustable continence balloons (ProACT). Although various surgical techniques are available, none guarantee full continence, and complication rates and treatment failures remain a concern [9, 10]. This highlights the continued need for technical innovation and refinement in the management of male stress urinary incontinence.
This article will focus on ProACT balloons, described in the literature as a minimally invasive technique with moderate efficacy [11]. At the Department of Urology, Herlev and Gentofte Hospital, the conventional ProACT procedure consists of initial placement of two balloons, with the option of a third in cases of insufficient clinical effect. In 2023, a modified implantation technique was introduced with the aim of optimizing outcomes and reducing patient burden. Placing the balloons beneath the pelvic floor rather than above it was theorized to improve continence outcomes due to a more targeted position along the urethra rather than at the bladder neck. It was further hypothesized that this positioning would allow for the use of smaller balloon volumes, thereby reducing the need for repeated postoperative filling sessions. To our knowledge, this represents the first clinical application of this modified technique. The aim of the present study was to assess the effectiveness and safety of placing ProACT balloons below the pelvic floor, compared to the standard technique with placement above the pelvic floor.
Materials and methods
This retrospective cohort study was conducted at the Department of Urology, Herlev and Gentofte Hospital, Denmark. We included patients from the Capital Region of Denmark and Region Zealand who had undergone ProACT placement and had no prior surgical treatment for stress urinary incontinence. All eligible patients who underwent the modified procedure with balloon placement below the pelvic floor (BP group) from its initiation in September 2023 to February 2025 were included. Matching was performed chronologically with a control group (AP group) comprised of an equal number of patients who had undergone the original technique with balloon placement above the pelvic floor immediately preceding the introduction of the modified approach. The same selection criteria for ProACT implantation were applied for both the BP and AP methods, and allocation was determined solely by the time of surgery.
The preoperative workup included a physical examination, a pressure–flow study performed to exclude urge incontinence, and flexible cystoscopy to assess the anatomical conditions and to rule out bladder pathology. Patients provided a 24-h diaper test and reported their use of continence pads. All procedures were performed under general anesthesia by three experienced surgeons. Small perineal incisions were made on either side of the midline. The bladder was filled with 50 ml of contrast, and ultrasound- and fluoroscopy-guided trocar placement was used to insert the balloons on either side of the urethra. In the AP group, 5 ml of Carbocain 0.5% was deposited posterolateral to the bladder neck above the pelvic floor, while in the BP group, the anesthetic was administered lateral to the urethra, beneath the pelvic floor. Balloons were inserted at the site of local anesthesia and initially filled with 1 ml contrast. Tubing was tunneled to subcutaneously placed ports in the posterior scrotum. All incisions were closed with Vicryl Rapid 4-0. A final urethrography was performed to confirm balloon position. Perioperative antibiotic prophylaxis included vancomycin (1 g i.v.) and hexamycin (5 mg/kg, max. 400 mg i.v.). The balloons remained untouched for 4 weeks to allow tissue ingrowth. Patients then attended ProACT nurse-led adjustment weekly visits, during which approximately 1 ml per visit was added to each balloon until adequate continence and/or a maximum of 8 ml per balloon. Patients submitted 24-h pad-weight tests and reported their current pad use at their visits, and continence outcomes were based on the latest available results.
Data collection was performed retrospectively using electronic patient charts in April 2025. The primary endpoint was continence, defined as a maximum of one pad per day or a daily leakage of less than 8 ml [12]. Secondary endpoints included reduction in daily pad use and urinary leakage as measured by a 24-h pad test, operative time, intra- and perioperative complications, cases of urinary retention, number of visits for balloon filling, final balloon volume, late complications in the form of migration, erosion, and balloon puncture, revision surgery, device explantations, and patient-reported satisfaction. Patient satisfaction was dichotomized and assessed during clinical follow-up through direct questioning by nursing staff, thus not including the responsible surgeon.
In cases where the clinical effect following initial ProACT implantation above the pelvic floor is deemed insufficient, it is institutional practice to insert a third balloon. Accordingly, outcome comparisons were performed after the third balloon insertion in the AP group for those patients in whom it was required, as this reflects the definitive treatment achievable with that technique. Third balloon placements were not included in the revision count. Due to anatomical constraints, placement of a third balloon is not technically feasible in patients with a prior insertion of two balloons below the pelvic floor.
Statistical analyses were conducted using the Wilcoxon Two-Sample Test for continuous variables and Fisher’s exact test for categorical variables. Multivariate logistic regression was used to adjust for preoperative urine loss, patient age, and follow-up. Cox regression was used to estimate hazard ratios for complications needing surgical intervention. A p-value < 0.05 was considered statistically significant. All statistical tests were performed using SAS Enterprise Guide version 8.4. Missing data were handled using SAS default procedures. Descriptive analyses were based on available cases. For continence outcomes, patients were classified as continent if either daily pad use or urine loss was available and indicated continence; patients with both parameters missing were classified as not continent. Regression analyses were conducted using complete-case analysis, excluding observations with missing values in any variable included in the model. No imputation of missing data was performed. This approach was chosen to maintain transparency and avoid assumptions regarding the mechanism of missingness.
The study was registered and approved by the Regional Center for Register Research of the Capital Region of Denmark according to Danish law (journal number R-23071694). The manuscript was prepared according to the STROBE statement.
Results
During the study period, 38 men were treated with the modified implantation technique (BP group). The comparison group comprised 38 patients who had most recently undergone the original implantation technique (AP group). Thus, a total of 76 patients were included. The median age at surgery was 75.5 years (range 64–80) in the BP group and 70.5 years (range 42–77) in the AP group (p < 0.0001). In the AP group, 35 men were incontinent due to RARP and 3 due to TURP, while the corresponding numbers in the BP group were 37 and 1 (p = 0.62). No patients had undergone previous pelvic radiation therapy. In all included patients, more than 1.5 years had elapsed between the initial prostate surgery and the ProACT implantation. The surgeon’s contribution was comparable between the BP and AP groups with only 4/76 procedures performed by a different surgeon across the two techniques. Preoperatively, complete data on daily pad use and urine loss were available for 34 patients in the BP group and 31 patients in the AP group. In the BP group, pad use data were missing for four patients, while in the AP group, urine loss data were missing for one patient and pad use data for six patients.
The median preoperative urine loss by 24-h diaper test was 298.5 ml (range 60–1,565) in the BP group and 375 ml (range 19.5–1,550) in the AP group (p = 0.92). The median number of pads used per day preoperatively was 5 (range 1–12) in the BP group and 5 (range 1–16) in the AP group (p = 0.66). Baseline characteristics and urinary function are summarized in Table 1.
The median surgical time was 15 min (range 9–24) in the BP group and 20 min (range 13–26) in the AP group (p = 0.034). No intra- or perioperative complications were recorded in either group, and all patients were discharged the same day. The median follow-up time was 9.9 months (range 2–19.1) in the BP group and 24.9 months (range 2.9–51.3) in the AP group (p ≤ 0.0001). Postoperative urinary retention within the first 24 h of the procedure occurred in 7/38 (18.4%) patients in the BP group and in 0/38 (0%) patients in the AP group (p = 0.012). All cases of urinary retention were managed with an indwelling catheter, which was subsequently removed either at the patient’s general practitioner or during an outpatient visit, with catheterization lasting 4–14 days. One catheterized patient developed a febrile urinary tract infection, initially suspected to be an abscess. Intravenous antibiotics were given, and the patient was discharged on oral therapy after the abscess was ruled out. One patient in the BP group developed urinary retention after the initial balloon filling requiring catheterization. Removal of 1 ml from each balloon resolved the issue, and no further episodes occurred despite subsequent fillings.
Due to insufficient effect, a third balloon was inserted in 15/38 (39.4%) patients in the AP group. The mean balloon volume compressing the urethra at the time of data collection was 9.5 ml in the BP group and 17.1 ml combined in the AP group (p < 0.0001). This was achieved after a median of six filling visits in the BP group (range 1–17) and 10 filling visits in the AP group (range 4–22) (p = 0.0006).
The incidence of late complications was 23.7% (9/38) in the BP group and 36.8% (14/38) in the AP group (p = 0.32). Specifically, migration was observed in 3/38 (7.9%) patients in the BP group and 6/38 (15.8%) patients in the AP group (p = 0.48). Erosion was observed in 4/38 (10.5%) patients in the BP group and 2/38 (5.3%) in the AP group (p = 0.68). Balloon puncture was observed in 2/38 (5.3%) patients in the BP group and 6/38 (15.8%) patients in the AP group (p = 0.26). Revision surgery was performed in eight patients (21.1%) in the BP group and 10 patients (26.3%) in the AP group (p = 0.79). In the BP group, explantation was performed in eight patients (21.1%). In four cases, new balloons were reinserted. In the AP group, nine patients underwent explantation, and all received new balloon insertions (p = 0.78 for comparison of explantation rate). Postoperative complications and device-related outcomes are summarized in Table 2.
Postoperatively, complete data were available for 30 patients in the BP group and 27 patients in the AP group. In the BP group, urine loss data were missing for four patients and pad use data for four patients, whereas in the AP group urine loss data were missing for two patients, pad use data for five patients, and both measurements for four patients. The median postoperative urine loss by 24-h diaper test was 20 ml (range 0–875) in the BP group and 16 ml (range 0–1550) in the AP group (p = 0.47) representing a >50% improvement in 78.3% (95% confidence interval [CI]: 66.7–87.3) in the BP group and in 69.7% (95% CI: 51.3–84.4) in the AP group (p = 0.15). The median postoperative pad use was 1 pad per day (range 0–7) in the BP group and 2 pads per day (range 0–10) in the AP group (p = 0.206). Continence was achieved by 20/38 (52.6%) patients in the BP group and 22/38 (57.9%) patients in the AP group (p = 0.64), and satisfaction with the outcome of the procedure was reported by 27/38 (71.1%) patients in the BP group and 22/38 (57.9%) patients in the AP group (p = 0.338) (Figure 1).
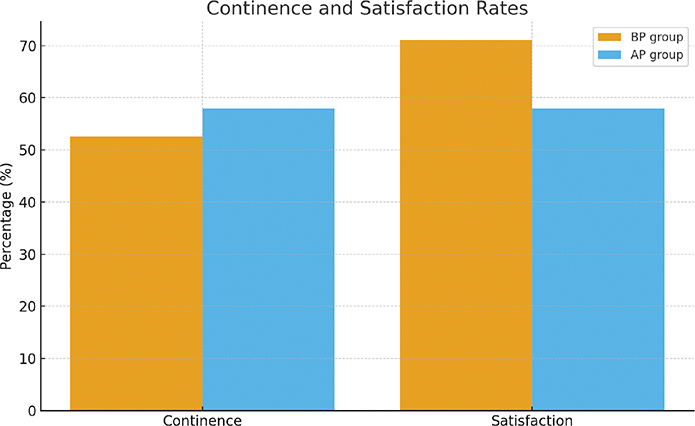
Figure 1. Continence and patient satisfaction rates after ProACT implantation with the original (AP) and modified (BP) technique. There were no statistically significant differences for either continence (p = 0.6445) or satisfaction (p = 0.338).
On multivariate analysis adjusting for preoperative urine loss and age, the surgical method (BP vs. AP group) was not a significant predictor of postoperative urine loss (p = 0.13), postoperative pad use (p = 0.16), continence (p = 0.90), or patient satisfaction (p = 0.39). Meanwhile, preoperative urine loss predicted both postoperative urine loss, postoperative pad use, and patient satisfaction. For each additional 100 ml of preoperative urine loss, postoperative urine loss increased by 20 ml (95% CI: 5–35, p = 0.01) and pad use increased by an average of 0.13 pads (95% CI: 0.02–0.25, p = 0.028), while the odds of satisfaction decreased by about 12% (OR 0.88, 95% CI: 0.78–0.99, p = 0.035). Using Cox regression adjusting for age, BP was associated with a higher but non-significant hazard of revision (HR: 1.60; 95% CI: 0.53–4.85; p = 0.40).
Discussion
ProACT represents a minimally invasive and adjustable treatment for male stress urinary incontinence. It is designed to combine procedural simplicity with long-term symptom relief. The principle is to identify the optimal balloon volume through a stepwise adjustment process until continence is achieved. With passive compression that does not require manual activation, the system offers a solution that does not rely on patient dexterity or daily interaction. The procedure, however, comes with a clinical caveat. According to current European guidelines, ProACT is considered effective in the treatment of post-prostatectomy incontinence, but patients should be informed that, although cure can be achieved, there is a high risk of complications, mechanical failure, and the potential need for explantation. The device should be implanted only in expert centers, and its use is not advised in men with a history of pelvic radiotherapy [10]. As a result, the method occupies an uncertain position in clinical practice. In this context, technical refinement becomes clinically relevant, both to optimize outcomes and to help guide its appropriate use in selected patient populations. The present study therefore set out to explore whether a simple shift in anatomical placement could influence the clinical trajectory of ProACT.
Across both groups continence status improved following ProACT implantation, with a majority of patients reporting satisfaction after the procedure. However, complications occurred in a notable proportion of cases and – although these are managed relatively easily – this highlights the continued need to weigh risks against benefits. Despite achieving similar continence outcomes, the two surgical groups differed notably in operative characteristics. In the AP group, more than one in three patients required the insertion of a third balloon as part of the final treatment strategy. Nonetheless, the BP group achieved comparable continence outcomes while requiring significantly lower total balloon volumes and fewer postoperative filling visits. The absence of third-balloon placement, along with the reduced follow-up intensity, reflects a practical advantage of the BP approach by sparing patients additional procedures and reducing disruption to daily life, while also lowering demands on healthcare resources. The median surgical time was also significantly shorter in the BP group, but with limited clinical meaning with a median decrease of just 5 min.
A notable difference between the groups was the incidence of early postoperative urinary retention. Within the first 24 h following surgery, urinary retention occurred in 18.4% in the BP group, compared to none in the AP group. All cases were managed with temporary catheterization, and subsequent treatment proceeded as intended. The higher incidence of retention may reflect the more immediate urethral compression associated with the BP technique. Although this side effect was transient and did not lead to long-term complications, it represents a potential drawback of the modified approach. The overall rate of late complications did not differ significantly between the groups, and a Cox regression analysis for complications requiring revision surgery did not indicate a significant difference between the groups (p = 0.4). The overall continence outcomes observed in this study reflect patterns seen in previously published experience from this department by Kjær et al. [13]. Comparable results were reported in the 2024 systematic review by Tricard et al. [11], suggesting a consistent level of clinical effectiveness for ProACT. Similarly, the complication rates reported in earlier studies were in line with our findings [9, 11].
The results may be carefully compared with those of other established surgical treatments for male stress urinary incontinence reported in previous trials. In a randomized controlled trial, both the AUS and male sling procedures were shown to improve continence in men after prostate surgery, with the AUS demonstrating superior outcomes after the first year [14]. AUS is regarded as the gold standard, particularly for patients with more severe incontinence. Reported continence outcomes are consistently high, with pooled dry rates approaching 80% [9]. However, this level of efficacy comes with drawbacks, including a more complex surgical procedure, the requirement for lifelong manual activation, a reoperation rate of approximately 50% within 10 years, and a relatively high device cost [2]. These factors limit its applicability for certain patient groups and underscore the need for alternative treatment options. Male slings represent one such category. However, both sling systems and ProACT are limited by reduced effectiveness in patients with a history of pelvic radiation and are generally considered most suitable for men with mild to moderate incontinence [11, 15]. Both fixed and adjustable slings have been used in the treatment of post-prostatectomy incontinence. Adjustable systems may offer higher objective cure rates, but this potential benefit may come at the cost of increased complication and explantation rates [15]. Among these, the Adjustable Transobturator Male System has shown favorable outcomes. A systematic review and meta-analysis reported superior results compared to ProACT, with dryness rates of 68% [16]. The limited availability of robust comparative data remains a central barrier to establishing a meaningful surgical hierarchy for the treatment of male incontinence. Ideally, such a hierarchy would be informed by multicenter, randomized trials with standardized surgical indications and uniform definitions of continence and treatment success. In the case of ProACT, the existing evidence is primarily based on single-center, retrospective cohort studies with methodological limitations, including lack of randomization [11].
In the present study, patients were generally satisfied with the outcome, and in case of indication for explantation, the balloon is easily removed under local anesthetics in a outpatient setting. Further, the device does not preclude subsequent continence procedures. Although a clearly superior ProACT implantation technique was not identified, the results suggest that the BP approach may be more efficient, although it was associated with a higher rate of perioperative urinary retention. Meanwhile, several factors should be considered when interpreting the results of this study. Most notably, follow-up time was substantially longer in the AP group, reflecting its earlier adoption. This temporal imbalance increases the likelihood of detecting late complications or revisions in that group and limits the strength of direct comparisons. Furthermore, the BP group represents the initial phase of a newly introduced surgical approach. Although the surgeons were highly experienced with the AP technique, a learning curve may still have influenced early outcomes, particularly in relation to complications or revision rates. Furthermore, the relatively small group sizes may have reduced the ability to detect subtle but potentially meaningful differences, and some findings may not have reached statistical significance due to limited statistical power. In addition, the absence of detailed baseline characteristics such as comorbidity burden, smoking status, and body mass index limits the ability to fully account for potential confounding between the groups. Regarding the results, the use of a direct question rather than a standardized questionnaire may have biased outcomes in a positive direction. However, this effect is likely minimal because follow-up was conducted by nurses who were not involved in the surgery. Finally, the external validity of the findings is limited by the retrospective single-center design. While these limitations call for caution in interpretation, the observed outcomes can still be meaningfully situated within the broader context of existing evidence.
To determine whether the differences are clinically meaningful and sustained over time and to better define ProACT’s position relative to other surgical options, prospective randomized studies with extended follow-up are warranted.
References
- [1] Porto JG, Bhatia AM, Bhat A, et al. Evaluating transurethral resection of the prostate over twenty years: a systematic review and meta-analysis of randomized clinical trials. World J Urol. 2024;42:639. https://doi.org/10.1007/S00345-024-05332-3
- [2] Gacci M, De Nunzio C, Sakalis V, et al. Latest evidence on post-prostatectomy urinary incontinence. J Clin Med. 2023 Feb 2;12(3):1190. https://doi.org/10.3390/JCM12031190
- [3] Giannantoni A, Mearini E, Zucchi A, et al. Bladder and urethral sphincter function after radical retropubic prostatectomy: a prospective long-term study. Eur Urol. 2008;54:657–664. https://doi.org/10.1016/j.eururo.2007.10.054
- [4] Lardas M, Grivas N, Debray TPA, et al. Patient- and tumour-related prognostic factors for urinary incontinence after radical prostatectomy for nonmetastatic prostate cancer: a systematic review and meta-analysis. Eur Urol Focus. 2022;8:674–689. https://doi.org/10.1016/j.euf.2021.04.020
- [5] Canning A, Raison N, Aydin A, et al. A systematic review of treatment options for post-prostatectomy incontinence. World J Urol. 2022;40:2617–2626. https://doi.org/10.1007/S00345-022-04146-5
- [6] Green R. Experiences and management of urinary incontinence following treatment for prostate cancer: disrupted embodied practices and adapting to maintain masculinity. Health (London) 2024;28:489–506. https://doi.org/10.1177/13634593231185266
- [7] Cornu J-N, Zantek P, Burtt G, et al. Minimally invasive treatments for benign prostatic obstruction: a systematic review and network meta-analysis. Eur Urol. 2023;83:534–547. https://doi.org/10.1016/j.eururo.2023.02.028
- [8] Winck-Flyvholm L, Fode M, Marsh A, et al. Transurethral microwave thermotherapy with the CoreTherm®Concept in men with prostates larger than 100 grams – a consecutive case series. Scand J Urol. 2025;60:23–28. https://doi.org/10.2340/sju.v60.42784
- [9] Averbeck MA, Woodhouse C, Comiter C, et al. Surgical treatment of post-prostatectomy stress urinary incontinence in adult men: report from the 6th International Consultation on Incontinence. Neurourol Urodyn. 2019;38:398–406. https://doi.org/10.1002/NAU.23845
- [10] Gacci M, Sakalis VI, Karavitakis M, et al. European Association of Urology Guidelines on Male Urinary Incontinence. Eur Urol. 2022;82:387–398. https://doi.org/10.1016/j.eururo.2022.05.012
- [11] Tricard T, Song QX, Munier P, et al. Adjustable continence therapy (proACT) for the treatment of male stress urinary incontinence post-prostatectomy: a systematic review and meta-analysis (2023 update). World J Urol. 2023;41:1793–1802. https://doi.org/10.1007/S00345-023-04452-6
- [12] Moore K, Allen M, Voaklander DC. Pad tests and self-reports of continence in men awaiting radical prostatectomy: establishing baseline norms for males. Neurourol Urodyn. 2004;23:623–626. https://doi.org/10.1002/NAU.20067
- [13] Kjær L, Fode M, Norgaard N, et al. Adjustable continence balloons: clinical results of a new minimally invasive treatment for male urinary incontinence. Scand J Urol Nephrol. 2012;46:196–200. https://doi.org/10.3109/00365599.2012.660986
- [14] Abrams P, Constable LD, Cooper D, et al. Outcomes of a noninferiority randomised controlled trial of surgery for men with urodynamic stress incontinence after prostate surgery (MASTER)[Formula presented]. Eur Urol. 2021;79:812–823. https://doi.org/10.1016/j.eururo.2021.01.024
- [15] Meisterhofer K, Herzog S, Strini KA, et al. Male slings for postprostatectomy incontinence: a systematic review and meta-analysis. Eur Urol Focus. 2020;6:575–592. https://doi.org/10.1016/j.euf.2019.01.008
- [16] Angulo JC, Schönburg S, Giammò A, et al. Systematic review and meta-analysis comparing Adjustable Transobturator Male System (ATOMS) and Adjustable Continence Therapy (ProACT) for male stress incontinence. PLoS One. 2019;14. https://doi.org/10.1371/JOURNAL.PONE.0225762