ORIGINAL RESEARCH ARTICLE
Number of risk factors versus infection after transrectal prostate biopsy: a nationwide population-based study
Joakim Örtegrena,b  , Olof Elvstamc,d
, Olof Elvstamc,d  , Kimia Kohestanib,e
, Kimia Kohestanib,e  , Henrik Kjölhedeb,e
, Henrik Kjölhedeb,e  , Johan Styrkef
, Johan Styrkef  , Pär Statting
, Pär Statting  , Anders Berglundh,i and Ola Brattb,e
, Anders Berglundh,i and Ola Brattb,e 
aDepartment of Surgery, Region Kronoberg, Växjö, Sweden; bDepartment of Urology, Institute of Clinical Sciences, Sahlgrenska Academy, University of Göteborg, Göteborg, Sweden; cDepartment of Infectious Diseases, Region Kronoberg, Växjö, Sweden; dDepartment of Translational Medicine, Lund University, Malmö, Sweden; eDepartment of Urology, Sahlgrenska University Hospital, Göteborg, Sweden; fDepartment of Diagnostics and Intervention, Urology and Andrology, Umeå University, Umeå, Sweden; gDepartment of Urology, Institute of Surgical Science, University of Uppsala, Uppsala, Sweden; hEpiStat AB, Uppsala, Sweden; iDepartment of Clinical Neuroscience, Karolinska Institute, Stockholm, Sweden
ABSTRACT
Objective: It is unknown how risk factors for infection after transrectal prostate biopsy interact. We designed a study to evaluate this.
Methods: We identified biopsy procedures from 2006 to 2020 in the Swedish nationwide database PCBaSe. Primary outcome was post-biopsy infection, defined as a dispensed prescription of a urinary tract antibiotic and secondary outcome was inpatient care for infection both within 30 days. Risk factors were age, diabetes, medical treatment of lower urinary tract symptoms (LUTSs), prostate enlargement, immunosuppressives, corticosteroids, and defined antibiotic exposure during the past 1–12 months. When analysing risk in men with several risk factors clinically related factors were grouped as urinary tract infection (UTI)-antibiotics, treatment of LUTS, immunosuppressives including corticosteroids, and diabetes. Logistic regression was used to calculate odds ratios (ORs) with 95% confidence intervals (CI).
Results: A total of 139,056 transrectal prostate biopsy procedures were analysed. The grouped risk factors were significantly associated with post-biopsy infection (multivariable ORs: 1.22–1.72). Infection increased with number of risk factors; none: 4.0% (95% CI: 3.8–4.1), one: 6% (95% CI: 5.9–6.4), two: 10% (95% CI: 9.3–11), and three or four: 12% (95% CI: 9.8–14); inpatient care increased from 2.0% (95% CI: 1.9–2.1) to 3.1% (95% CI: 2.2–4.4).
Conclusion: Infection risk after transrectal prostate biopsy incrementally increases with the number of risk factors.
Clinical Implications: The transrectal biopsy route should be used with caution for patients with several risk factors for post-biopsy infections.
Patient Summary: Diabetes, urinary symptoms, previous urinary infection, and immune suppressing medication increase the risk of infection after a prostate biopsy through the rectum. Patients with many of these conditions have a particularly high risk.
What does the study add?: We used nationwide register data to estimate the infection risk after transrectal prostate biopsy by the number of these risk factors: diabetes, medical treatment of lower urinary tract symptoms, immunosuppressives including corticosteroids, and use of urinary tract antibiotics the past year. The risk incrementally increased from 4.0% in men with no risk factor to 12% in those with 3 or 4.
Take Home Message: Infection after transrectal prostate biopsy increases with number of risk factors: diabetes, medical treatment of lower urinary tract symptoms, immunosuppressives including corticosteroids, and use of urinary tract antibiotics the past year, from 4.0% (none) to 12% (3 or 4).
KEYWORDS: Prostate biopsy; transrectal; complication; risk factor; infection
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 99–105. https://doi.org/10.2340/sju.v61.45581.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 12 July 2025; Accepted: 21 January 2026; Published: 31 March 2026
CONTACT: Joakim Örtegren joakim.ortegren@gmail.com Växjö Regional Hospital, SE-35185 Växjö, Sweden
Supplemental data for this article can be accessed online at https://doi.org/10.2340/sju.v61.45581
Introduction
A prostate biopsy is a common diagnostic procedure in urology [1]. The transrectal route has long been standard of care [2], but this route is debated mainly owing to the risk for post-biopsy infection [3, 4]. European guidelines recommend the transperineal route since 2022 [5], but the American Urological Association (AUA) guidelines consider both the transrectal and transperineal routes ‘of choice’ [6]. Two randomised clinical trials showed no significant difference in post-biopsy infections between transperineal without and transrectal with antibiotic prophylaxis [7, 8]. Measures to reduce the risk of post-biopsy infection are relevant regardless of biopsy route [9].
Diabetes mellitus and previous urinary tract infection (UTI) have consistently been associated with the risk of infection after a transrectal prostate biopsy [10–12]. The importance of other potential risk factors is less clear. A study from a screening setting suggested that prostate enlargement is a risk factor [13]. In a previous register study, we found that medication for benign prostatic hyperplasia (BPH) and more than two previous negative urine culture, likely reflecting non-infectious lower urinary tract symptoms (LUTS), were both associated with post-biopsy infection [14]. It is thus not clear whether it is prostate enlargement as such or if it is the subsequent pathophysiological events that lead to LUTS that increase the risk of post-biopsy infection. There is also uncertainty about how different antibiotic exposures affect the risk of post-biopsy infection. In a previous study, we found that any kind of antibiotic treatment was associated with post-biopsy infection [14].
It is not known how the presence of several risk factors for infection after transrectal prostate biopsy affects the risk of infection. The primary aim of this study was therefore to explore the association between the number of risk factors and the risk of infection after transrectal prostate biopsy. We also aimed to explore whether prostate enlargement or LUTS, or both, increase the risk of infection, and how different antibiotic exposures affect this risk.
Material and methods
Study population
The National Prostate Cancer Register (NPCR) captures 98% of men with prostate cancer in in Sweden compared with the Cancer Register to which reporting is mandated by law [15]. The primary aim of NPCR is to assess the adherence to national guidelines [16–18]. Our study population was obtained from the Prostate Cancer data Base Sweden (PCBaSe), in which information from the NPCR has been enriched by data from other nationwide health care registers and demographic databases [19]. In addition to data on men with prostate cancer, PCBaSe includes data on five prostate cancer-free men, matched for year of birth and region of residence [20]. PCBaSe includes data on diagnosis and procedure codes for inpatient episodes and outpatient visits in secondary care from the National Patient Register, and data from the Swedish Prescribed Drug Register on all dispensed drug prescriptions from 2006 [21].
PCBaSe 5.0 and this specific study were both approved by the Swedish Ethical Review Authority.
Definition of exposure
Biopsy procedures from 2006 to 2019 were identified by the procedure code TKE00 (prostate needle biopsy). In 2020, codes for transrectal and transperineal biopsy were introduced [22] and from the same year, procedures coded TKE10 were included in the study. As the transperineal route was rarely used in Sweden before 2020, all biopsy procedures before 2020 were considered transrectal. Men in the matched prostate cancer-free population could have more than one registered biopsy procedure. To avoid double registration, we excluded men in the matched PCBaSe population who had a biopsy procedure with a benign result but were later diagnosed with prostate cancer. The number of procedures per individual is presented in Supplement Table S6. In our previous study, a history of one or several prostate biopsy procedures was not associated with infection [14], so this variable was not included in this study. A single dose of oral 750 mg ciprofloxacin was the most common prophylactic regimen in Sweden throughout the study period [23, 24]. The Swedish guidelines did not recommend any other prophylactic measure during the study period.
Outcome measures
The primary outcome was any post-biopsy infection within 30 days from biopsy, defined as a dispensed prescription of an antibiotic indicated for UTI 1–30 days after the biopsy in outpatient or inpatient care (a prescription on the day of the biopsy was regarded as prolonged prophylaxis). The secondary outcome was inpatient care 0–30 days after biopsy with a diagnosis compatible with infectious complication. The drug treatment and diagnosis codes used for these definitions are listed in Supplement Table S1 and Table S2.
Investigated potential risk factors
The following potential risk factors for infection were investigated: age, previous UTI, previous fluoroquinolone treatment, previous non-UTI antibiotic treatment, diabetes mellitus, immunosuppressive treatment, systemic corticosteroid treatment, prostate enlargement, and LUTS. To capture these factors, we used the following definitions.
A previous UTI was defined as a dispensed prescription from 4 weeks to 1 year before biopsy of either a fluoroquinolone or a non-fluoroquinolone UTI antibiotic (see Supplement Table S1 for the codes defining these antibiotics). A prescription of a UTI antibiotic within 4 weeks before biopsy may suggest that the patient was fragile or had a high risk for infection, and that the prescribed antibiotic was to provide periprocedural antibiotic prophylaxis or treatment rather than to treat a clinical UTI. We also analysed exposure to non-UTI antibiotics 4 weeks to 1 year before biopsy. The 4-week limit was also applied to previous non-UTI antibiotics for comparability. The 12-month limit was chosen in accordance with our previous study [14].
Diabetes mellitus was defined by ICD-10 code E11/E10 and/or a dispensed prescription of a diabetes drug within 1 year before biopsy.
To define LUTS, we used a dispensed prescription of treatment for BPH (LUTS/BPH) or for overactive bladder (LUTS/OAB) any time before or within 6 months after biopsy. Data on prostate volume were available only for men diagnosed with prostate cancer.
Systemic corticosteroid treatment was defined as ≥ 2 dispensed prescriptions within 7 months and immunosuppressive treatment as a dispensed prescription within 4 months, both before biopsy.
Statistical analysis
Univariable logistic regression was used to calculate odds ratios (ORs) for the outcomes with 95% confidence intervals (CIs). In the regression models, age was analysed as a continuous variable, whereas prostate volume was categorised as < 40, 40–79, and ≥ 80 cm3 because we assumed a non-linear association. The remaining variables were dichotomous. Diabetes mellitus, LUTS/OAB, LUTS/BPH, immunosuppressive treatment, and prostate volume are all associated with age and were therefore adjusted for age in a separate analysis. We adjusted for calendar year to avoid missing or misinterpreting factors with a changing prevalence over time. When we analysed the association between the number of risk factors and infection from the multivariable models, we merged closely related variables: non-fluoroquinolone antibiotic for UTI with fluoroquinolones, treatment for LUTS/OAB with treatment for LUTS/BPH, and immunosuppressives with corticosteroids. Prostate volume was not included in the multivariable model because of a strong association with BPH treatment and weak, non-consistent univariable association with the outcomes. Age was not included because a clinically insignificant association with the outcome on univariable analysis. Non-UTI antibiotics were not included in the analysis of the association between the number of risk factors and the absolute risk of infection, because of lacking association with inpatient care and poor biological underpinning as a causal risk factor.
To exclude that the association between LUTS treatment and infection was caused by the treatment as such, we performed a sensitivity analysis including only men who started LUTS treatment after the biopsy.
Although LUTS/BPH and LUTS/OAB both had a clinically relevant association with post-biopsy infection, their association with inpatient care was weak. Because of this and because a directly causal relation between treatment for LUTS and infection is unlikely, we analysed the cumulative effect of grouped risk factors both with and without LUTS to find out whether the addition of LUTS makes a clinically relevant difference.
As the outcome measure was assessed indirectly with antibiotics prescription, we assessed the ‘background prescription’ of antibiotics and of inpatient care in the studied population the same calendar month one year before the prostate biopsy.
The analyses were done with R version 4.3.2. The manuscript adheres to the STROBE checklist for observational studies.
Results
The study included 139,056 prostate biopsy procedures, out of which 64,217 led to a prostate cancer diagnosis and 74,839 did not (Figure 1). Baseline characteristics are presented in Table 1. A total of 6,608 (4.8%) of the biopsy procedures resulted in a post-biopsy infection and 2,979 (2.1%) in an inpatient care episode for an infection within 30 days (Supplement Figure 2). The proportion of post-biopsy infection increased from 3.4% in 2006 to 5.9% in 2020; the corresponding increase of infections leading to inpatient care was from 1.0% to 2.7% (Supplement Figure 1).
| Investigated potential risk factors | Number (%) | ||||
| Age (years) | < 60 | 12,321 (19.2) | |||
| 60–69 | 31,380 (48.9) | ||||
| 70–79 | 16,901 (26.3) | ||||
| ≥ 80 | 3,458 (5.4) | ||||
| Unknown | 157 (0.2) | ||||
| Diabetes mellitus | 13,566 (9.8) | ||||
| Previous UTI-antibiotics | 11,036 (7.9) | ||||
| Previous fluoroquinolone | 15,304 (11) | ||||
| Previous non-UTI antibiotic | 20,316 (15) | ||||
| Immunosuppressive treatment | 1,734 (1.3) | ||||
| Systemic corticosteroids | 4,323 (3.1) | ||||
| LUTS/BPH treatment* | 41,931 (30) | ||||
| LUTS/OAB treatment* | 7,921 (5.7) | ||||
| Prostate volume** | < 40 ml | 30,807 | |||
| 40–79.9 ml | 20,971 | ||||
| > 80 ml | 3,009 | ||||
| Unknown | 9,430 | ||||
| *Including treatment started within 6 months after the biopsy. **Information available only for those diagnosed with prostate cancer. LUTS: lower urinary tract symptoms; OAB: overactive bladder; UTI: urinary tract infection; BPH: benign prostatic hyperplasia. |
|||||
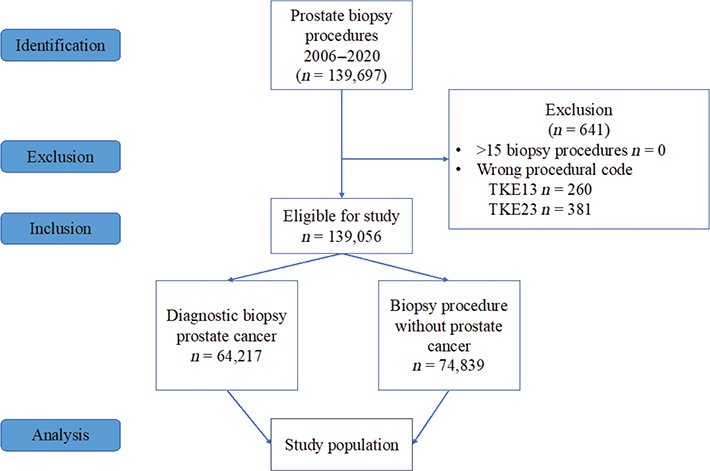
Figure 1. STROBE flow chart of study population from PCBaSe 5.0 included 2006–2020.
The variables for the analysis of the association between the number of risk factors and infection were based on the results of the univariable and multivariable analyses. The analysis including diabetes mellitus, previous UTI, and immunosuppressive treatment showed that 4.0% (95% CI: 3.8–4.1%) of men with none of these risk factors had a post-biopsy infection. The proportion with infection incrementally increased with the number of risk factors up to 12% (95% CI: 9.8–14) in men with three risk factors (Table 2). The proportion of procedures that led to inpatient care increased from 2.0% (95% CI: 1.9–2.1%) in men with no risk factor to 3.1% (95% CI: 2.2–4.4 %) in those with three factors (Table 2). The analysis with LUTS treatment as a fourth risk factor yielded similar results (Supplement Table S3).
| Risk factor | Any post-biopsy infection | Inpatient care for post-biopsy infection | |||
| Absolute risk (%) | 95% CI | Absolute risk (%) | 95% CI | ||
| 0 | 4.0 | 3.8–4.1 | 2.0 | 1.9–2.1 | |
| 1 | 6.1 | 5.9–6.4 | 2.5 | 2.3–2.6 | |
| 2 | 10.0 | 9.3–10.7 | 3.2 | 2.8–3.7 | |
| 3 | 11.7 | 9.8–13.9 | 3.1 | 2.2–4.4 | |
| UTI: urinary tract infection; CI: confidence interval. The here analysed risk factors were diabetes mellitus and grouped: immunosuppressive treatment and/or corticosteroid and non-fluoroquinolones UTI and/or fluoroquinolones. An analysis with LUTS as a fourth risk factor is presented in Supplement Table S3. |
|||||
LUTS/BPH and LUTS/OAB treatment were in univariable analysis both significantly associated with post-biopsy infection (ORs 1.43 and 1.61) and inpatient care (ORs 1.13 and 1.28), (Table 3), and so was the merged variable LUTS treatment in multivariable analysis (OR 1.34 for any post-biopsy infection and OR 1.11 for inpatient care) (Table 4). The sensitivity analysis of LUTS medication started after the biopsy included 30,067 men with LUTS/BPH medication and 4,475 men with LUTS/OAB medication; it gave gave identical results as the main analysis of the primary outcome: LUTS/BPH 5.9% versus 5.9%; LUTS/OAB 7.2% versus 7.2%.
| Any post-biopsy infection | Inpatient care for post-biopsy infection | ||||||
| OR | 95% CI | P-value | OR | 95% CI | P-value | ||
| Age at biopsy | 1.01 | 1.00–1.01 | < 0.001 | 0.99 | 0.99–1.00 | 0.019 | |
| Age at biopsy, groups | |||||||
| <60 years | 1.00 | reference | 1.0 | reference | |||
| 60–69 | 1.12 | 0.92–1.09 | 0.961 | 0.96 | 0.85–1.09 | 0.517 | |
| 70–79 | 1.16 | 1.03–1.22 | 0.012 | 0.93 | 0.82–1.05 | 0.229 | |
| 80+ | 1.16 | 1.04–1.29 | 0.006 | 0.85 | 0.73–0.99 | 0.043 | |
| Diabetes mellitus | 1.56 | 1.45–1.67 | < 0.001 | 1.54 | 1.39–1.71 | < 0.001 | |
| Previous non-fluoroquinolone UTI antibiotic | 2.26 | 2.11–2.42 | < 0.001 | 1.30 | 1.15–1.47 | < 0.001 | |
| Previous fluoroquinolone | 1.69 | 1.58–1.81 | < 0.001 | 1.09 | 0.97–1.22 | 0.137 | |
| Previous non-UTI antibiotic | 1.35 | 1.26–1.44 | < 0.001 | 1.12 | 1.01–1.23 | 0.029 | |
| Immunosuppressive treatment | 1.38 | 1.13–1.66 | 0.001 | 1.48 | 1.11–1.92 | 0.005 | |
| Systemic corticosteroids | 1.51 | 1.33–1.70 | < 0.001 | 1.38 | 1.14–1.64 | < 0.001 | |
| LUTS/BPH treatment | 1.43 | 1.36–1.50 | < 0.001 | 1.13 | 1.05–1.22 | 0.001 | |
| LUTS/OAB treatment | 1.61 | 1.47–1.76 | < 0.001 | 1.28 | 1.11–1.47 | < 0.001 | |
| Prostate volume* | |||||||
| < 40 ml | 1.00 | reference | 1.00 | reference | |||
| 40–79.9 ml | 1.16 | 1.07–1.26 | < 0.001 | 1.24 | 1.10 | < 0.001 | |
| ≥ 80 ml | 1.15 | 0.97–1.36 | 0.110 | 0.84 | 0.62 | 0.213 | |
| *Analysis restricted to biopsy procedures that led to a prostate cancer diagnosis in men with a registered prostate volume. UTI: urinary tract infection; OR: odds ratio; CI: confidence interval; LUTS: lower urinary tract symptoms; BPH: benign prostatic hyperplasia; OAB: overactive bladder. |
|||||||
The analyses of prostate volume gave inconsistent results: A moderate prostate enlargement (40–79 cm3) was significantly associated with both outcome measures on univariable analysis (ORs 1.16 and 1.24), but there was no significant association between a larger prostate (≥ 80 cm3) and any of the outcomes (OR 1.15 for any post-biopsy infection and OR 0.84 for in-patient care (Table 3). Adjustment for age and calendar year did not change the results (Supplement Tables S4 and S5).
Previous antibiotic treatment was associated with post-biopsy infection on univariable analysis (Table 3): non-fluoroquinolone UTI antibiotics (OR: 2.26), fluoroquinolones (OR: 1.69), and non-UTI antibiotics (OR 1.35), but only non-fluoroquinolone UTI antibiotics and non-UTI were significantly associated with inpatient care. In multivariable analysis (Table 4), the merged group UTI antibiotics was strongly associated with any post-biopsy infection (OR: 1.72) but not with inpatient care (OR 1.10). Non-UTI antibiotics had lower multivariable ORs for both outcome measures (ORs 1.22 and 1.07, Table 4). One year before the prostate biopsy 0.56% of the men had a dispensed antibiotics prescription and 0.09% had an inpatient care episode over a 30-day period.
Discussion
In this large nationwide, population-based register study, the risk of an infection after transrectal prostate biopsy increased with the number of risk factors (diabetes mellitus, previous UTI, immunosuppressive treatment, and LUTS), from 4% in men with none of these to 12% in men with three or four risk factors.
A secondary aim was to investigate how prostate enlargement and LUTS affect the risk of post-biopsy infection. Both prostate enlargement and BPH treatment have been reported to increase the risk of infection after transrectal prostate biopsy [11, 13]. Likely pathophysiological mechanisms are residual urine, bacteriuria, and bacteria in the prostate. In this study, treatment for OAB was more strongly associated with infection than treatment for BPH. This, together with the inconsistent and weak association between prostate volume and infection, suggests that the pathophysiological changes associated with LUTS are the causal link to infection, and that a prostatic enlargement per se does not increase the risk of infection.
Another secondary aim was to clarify different antibiotic exposures as risk factors. Both a previous fluoroquinolone treatment and a non-fluoroquinolone UTI treatment increased the risk of an infectious complication, but only the latter increased the risk of inpatient care. Fluoroquinolone treatment may lead to microbial resistance in the rectum [25] and thereby increase the risk of infection if a fluoroquinolone is used as prophylaxis [26], which was common practice in Sweden during the study period [14, 24]. Our observation that the ORs were consistently higher for previous non-fluoroquinolone UTI treatment than for previous fluoroquinolone treatment, suggests that a previous UTI may be a more important risk factor for post-biopsy infection than fluoroquinolone resistance, although a recent study from Sweden suggests the opposite [27].
Importantly, whereas microbial contamination from the rectal flora is overcome by using the transperineal biopsy route, the pathophysiological mechanisms behind the increased risks in men with a previous UTI and changes with LUTS are probably not.
A previous non-UTI antibiotic treatment was associated with both the primary and the secondary outcome. Although it cannot be excluded that this is related to changes in the gut bacteriome’s antibiotic resistance pattern, it may also be explained by that men with previous non-UTI antibiotic treatment have a general propensity to seek medical care and thereby are more likely to receive antibiotic treatment for unspecific symptoms after a biopsy. We did, therefore, not include non-UTI antibiotic treatment as a risk factor in the analysis of how the presence of several risk factors affect the absolute risk of post-biopsy infection.
Immunosuppressive treatment, including corticosteroids, is associated with a generally increased risk of infections and infectious complications [28]. A previous Swedish register study, with a design like ours, reported a similar OR for inpatient care [11].
Age has previously been positively associated with inpatient care for infection after prostate biopsy [29]. In our study, the association between age and post-biopsy infection was statistically significantly but the OR was only 1.01 and considered clinically insignificant. There was, surprisingly, a (similarly weak) negative association with inpatient care.
The risk of an infection after a transrectal prostate biopsy doubled between 2006 and 2020. The increase is likely caused by several different factors, necessitating a reassessment of prophylactic measures. Future research, and the development of strategies to prevent infectious complications, may lead to a choice between the transrectal and transperineal route being based on the individual patient´s risk factors.
The main strengths of our study include the population-based national cohort, the large number of biopsy procedures, and the accuracy of the Swedish registers. The rate of infection in this national study was similar to our previous regional study in which we validated the prescriptions and the diagnosis of admitted patients [14]. Weaknesses include the indirect definitions of previous UTI and post-biopsy infection by dispensed antibiotic prescriptions. The low ‘background prescription’ 1 year before biopsy does, however, suggest that misclassification does not much affect our results. Another weakness is that we could not evaluate the risk factors in a setting with routine rectal cleansing with povidone-iodine, as now recommended in the European guidelines [5], because this preventive measure was not used in Sweden during the study period. Finally, we had no data on specific antibiotic prophylactic measures, for example, men with a previous UTI or post-biopsy infection, and those with diabetes or immunosuppressive treatment, may have received prolonged antibiotic prophylaxis or targeted treatment.
Conclusion
An increasing number of the risk factors diabetes, medical treatment of lower urinary tract symptoms, immunosuppressives including corticosteroids, and use of urinary tract antibiotics the past year, results in an incrementally increased risk of infection after transrectal prostate biopsy. Our results suggest that LUTS increases the risk of infection but that prostate enlargement per se does not. If several risk factors are present, measures such as prolonged antibiotic prophylaxis and povidone-iodine should be considered or the transperineal route chosen.
Acknowledgements
This research was supported by grants from Cancerstiftelsen Kronoberg. It was made possible by the continuous work of the National Prostate Cancer Register of Sweden (NPCR) steering group: Elin Axén, Johan Styrke, Andreas Josefsson, Camilla Thellenberg, Hampus Nugin, Ingrida Verbiené, Stefan Carlsson, Anna Kristiansen, Mats Andén, Kimia Kohestani, Jon Kindblom, Thomas Jiborn, Olof Ståhl, Olof Akre, Eva Johansson, Magnus Törnblom, Fredrik Jäderling, Marie Hjälm-Eriksson, Lotta Renström Koskela, Erik Thimansson, Johan Stranne, Elin Trägårdh, Viktoria Gaspar, Fredrik Sandin, Petrus Stenson, Lena Pettersson, Mia Brus, Gustaf Hedström, Anna Hedström, Maria Moutran, Nina Hageman, and Maria Nyberg and patient representatives Hans Joelsson and Gert Malmberg.
References
[1] Loeb S, Vellekoop A, Ahmed HU, et al. Systematic review of complications of prostate biopsy. Eur Urol. 2013;64(6):876–892. https://doi.org/10.1016/j.eururo.2013.05.049
[2] Mottet N, Bellmunt J, Bolla M, et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2017;71(4):618–629. https://doi.org/10.1016/j.eururo.2016.08.003
[3] Grummet J, Gorin MA, Popert R, et al. ‘TREXIT 2020’: why the time to abandon transrectal prostate biopsy starts now. Prostate Cancer Prostatic Dis. 2020;23(1):62–65. https://doi.org/10.1038/s41391-020-0204-8
[4] Mian BM. Prostate biopsy: hyperbole and misrepresentation versus scientific evidence and equipoise. Eur Urol. 2024;85(2):99–100. https://doi.org/10.1016/j.eururo.2023.05.039
[5] EAU Guidelines. Edn. presented at the EAU Annual Congress Milan. 2023 [cited 2024 Jul 18]. Available from: http://uroweb.org/guidelines/compilations-of-all-guidelines/.
[6] Wei JT, Barocas D, Carlsson S, et al. Early detection of prostate cancer: AUA/SUO Guideline Part II: considerations for a prostate biopsy. J Urol. 2023;210(1):54–63. https://doi.org/10.1097/JU.0000000000003492
[7] Hu JC, Assel M, Allaf ME, et al. Transperineal versus transrectal magnetic resonance imaging–targeted and systematic prostate biopsy to prevent infectious complications: the PREVENT randomized trial. Eur Urol. 2024;86(1):61–68. https://doi.org/10.1016/j.eururo.2024.04.014
[8] Mian BM, Feustel PJ, Aziz A, et al. Complications following transrectal and transperineal prostate biopsy: results of the ProBE-PC randomized clinical trial. J Urol. 2023;211(2):205–213. https://doi.org/10.1097/JU.0000000000003788
[9] Pradere B, Veeratterapillay R, Dimitropoulos K, et al. Nonantibiotic strategies for the prevention of infectious complications following prostate biopsy: a systematic review and meta-analysis. J Urol. 2021;205(3):653–663. https://doi.org/10.1097/JU.0000000000001399
[10] Lei H, Dong X, Li L, et al. Retrospective study of the etiology and risk factors of systemic inflammatory response syndrome after systematic transrectal ultrasound-guided prostate biopsy. Infect Drug Resist. 2020;13:3187–3193. https://doi.org/10.2147/IDR.S256548
[11] Lundstrom KJ, Drevin L, Carlsson S, et al. Nationwide population based study of infections after transrectal ultrasound guided prostate biopsy. J Urol. 2014;192(4):1116–1122. https://doi.org/10.1016/j.juro.2014.04.098
[12] Roberts MJ, Bennett HY, Harris PN, et al. Prostate biopsy-related infection: a systematic review of risk factors, prevention strategies, and management approaches. Urology. 2017;104:11–21. https://doi.org/10.1016/j.urology.2016.12.011
[13] Loeb S, van den Heuvel S, Zhu X, et al. Infectious complications and hospital admissions after prostate biopsy in a European randomized trial. Eur Urol. 2012;61(6):1110–1114. https://doi.org/10.1016/j.eururo.2011.12.058
[14] Örtegren J, Kohestani K, Elvstam O, et al. Risk factors for infection after transrectal prostate biopsy: a population-based register study. Eur Urol Open Sci. 2024;67:1–6. https://doi.org/10.1016/j.euros.2024.06.015
[15] Van Hemelrijck M, Garmo H, Wigertz A, et al. Cohort profile update: the National Prostate Cancer Register of Sweden and Prostate Cancer Data Base – a refined prostate cancer trajectory. Int J Epidemiol. 2016;45(1):73–82. https://doi.org/10.1093/ije/dyv305
[16] Bratt O, Carlsson S, Fransson P, et al. The Swedish national guidelines on prostate cancer, part 1: early detection, diagnostics, staging, patient support and primary management of non-metastatic disease. Scand J Urol. 2022;56(4):265–273. https://doi.org/10.1080/21681805.2022.2094462
[17] Bratt O, Carlsson S, Fransson P, et al. The Swedish national guidelines on prostate cancer, part 2: recurrent, metastatic and castration resistant disease. Scand J Urol. 2022;56(4):278–284. https://doi.org/10.1080/21681805.2022.2093396
[18] Stattin P. How to survey adherence to guidelines by use of clinical cancer registers. Scand J Urol. 2022;56(4):285–286. https://doi.org/10.1080/21681805.2022.2107069
[19] Hagel E, Garmo H, Bill-Axelson A, et al. PCBaSe Sweden: a register-based resource for prostate cancer research. Scand J Urol. 2009;43(5):342–349. https://doi.org/10.3109/00365590903024577
[20] Van Hemelrijck M, Wigertz A, Sandin F, et al. Cohort profile: the National Prostate Cancer Register of Sweden and Prostate Cancer Data Base Sweden 2.0. Int J Epidemiol. 2013;42(4):956–967. https://doi.org/10.1093/ije/dys068
[21] Wettermark B, Hammar N, Fored CM, et al. The new Swedish Prescribed Drug Register – opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol Drug Saf. 2007;16(7):726–735. https://doi.org/10.1002/pds.1294
[22] Classification of Procedural codes The National Board of Health and Welfare; [cited 2024 Apr 02]. Available from: https://www.socialstyrelsen.se/globalassets/sharepoint-dokument/artikelkatalog/klassifikationer-och-koder/kkakum2005-2020.pdf
[23] Carlsson S, Bratt O, Stattin P, et al. Current routines for transrectal ultrasound-guided prostate biopsy: a web-based survey by the Swedish Urology Network. Scand J Urol Nephrol. 2012;46(6): 405–410. https://doi.org/10.3109/00365599.2012.691111
[24] Styrke J, Resare S, Lundström K-J, et al. Current routines for antibiotic prophylaxis prior to transrectal prostate biopsy: a national survey to all urology clinics in Sweden. F1000Research. 2020;9:58. https://doi.org/10.12688/f1000research.19260.1
[25] Abughosh Z, Margolick J, Goldenberg SL, et al. A prospective randomized trial of povidone-iodine prophylactic cleansing of the rectum before transrectal ultrasound guided prostate biopsy. J Urol. 2013;189(4):1326–1331. https://doi.org/10.1016/j.juro.2012.09.121
[26] Cussans A, Somani BK, Basarab A, et al. The role of targeted prophylactic antimicrobial therapy before transrectal ultrasonography-guided prostate biopsy in reducing infection rates: a systematic review. BJU Int. 2016;117(5):725–731. https://doi.org/10.1111/bju.13402
[27] Holmbom M, Forsberg J, Fredrikson M, et al. Fluoroquinolone-resistant Escherichia coli among the rectal flora is the predominant risk factor for severe infection after transrectal ultrasound-guided prostate biopsy: a prospective observational study. Scand J Urol. 2023;58:32–37. https://doi.org/10.2340/sju.v58.11920
[28] Chastain DB, Spradlin M, Ahmad H, et al. Unintended consequences: risk of opportunistic infections associated with long-term glucocorticoid therapies in adults. Clin Infect Dis. 2024;78(4):e37–e56. https://doi.org/10.1093/cid/ciad474
[29] Anastasiadis E, van der Meulen J, Emberton M. Hospital admissions after transrectal ultrasound-guided biopsy of the prostate in men diagnosed with prostate cancer: a database analysis in England. Int J Urol. 2015;22(2):181–186. https://doi.org/10.1111/iju.12634