ORIGINAL RESEARCH ARTICLE
Treatment with CernitinTM of induced urinary bladder inflammation downregulates relevant pro-inflammatory biomarkers – an experimental in vivo rodent model
Nishtman Dizeyia, Celine Augéb, Philippe Lluelb and Magnus Grabec 
aAB Cernelle, Ängelholm, Sweden; bUrosphere, Toulouse, France; cDepartment of Translational Medicine, Lund University, Malmö, Sweden
ABSTRACT
Objective: Chronic inflammation of the urinary bladder is associated with the bladder pain syndrome. The treatment alternatives in humans are far from satisfactory and need further attention. Well-established preclinical models have shown that pro-inflammatory cytokines contribute to the progress of the inflammatory response behind pain and hyperalgesia. Previously presented results indicate that treatment with CernitinTM pollen extracts active pharmaceutical ingredients (APIs) (Cernitin GBX and Cernitin T60) significantly alleviated pain in cyclophosphamide-induced urinary bladder inflammation in a rodent model through downregulation of PGD2 and cyclooxygenase-2 (COX-2) mediators when compared to the vehicle alone. The objective was to extend the original study by exploring the correlation between the two APIs and cytokines expression and to identify a possible biomarker pattern.
Material and method: The Olink® Target 48 Mouse Cytokine assay was conducted on the homogenised tissue extracts of the bladder wall with induced inflammation from a previous study to identify the potential impacts on protein biomarkers.
Results: The test revealed that treatment with the APIs significantly downregulated the cytokines interleukin (IL)-1α, IL-2, IL-4, IL-6, and with trend to significance the biomarkers IL-12 α, β, CCL4 and fibroblast growth factor 21 when administered in combination (GBX+T60) or each component alone, compared to vehicle controls.
Conclusion: This study identified seven cytokines that were significantly or markedly reduced. The results suggest that CernitinTM APIs impact a series of key pro-inflammatory biomarkers demonstrating an ability to restrain inflammation. Therefore, they warrant further investigation as potential therapeutic candidates.
KEYWORDS: Cytokines; inflammatory biomarkers; chronic cystitis; interstitial cystitis; bladder pain syndrome; animal model
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 58–63. https://doi.org/10.2340/sju.v61.45606.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 30 October 2025; Accepted: 5 February 2026; Published: 20 March 2026
CONTACT: Magnus Grabe magnus.grabe@med.lu.se Department of Translational Medicine, Lund University, Malmö, Sweden
Supplemental data for this article can be accessed online at https://doi.org/10.2340/sju.v61.45606
Competing interests and funding: The authors declare a potential conflict of interest and state it as below:
Dr. Dizeyi declares financial support from AB Cernelle for conducting and editing the present experiment.
M Grabe, M.D., Ph.D., urologist, has acted as external, independewnt scientific advisor to AB Cernelle in matters related to scientific analysis, research projects, and regulatory and educational purposes.
C Augé and Ph Lluel declare no conflict of interest for the present experiment. They conducted the previous studies at Urosphère, Toulouse, France.
AB Cernelle, Ängelholm, Sweden, is the owner and manufacturer of CernitinTM. The company has provided the APIs and given financial support for performing the test and necessary analysis within the frame of a research project aimed at better understanding the mechanisms of action of the medical product.
Introduction
Interstitial cystitis (IC) is a heterogeneous group of chronic bladder inflammations with no primary proven infectious genesis or other obvious underlying pathology, and is one of the important causes of chronic bladder pain syndrome (BPS) [1, 2]. The diagnosis of IC is based on clinical symptoms in association with typical cystoscopic findings and characteristic pathology at biopsy. The treatment of patients with BPS due to IC (IC/BPS) is challenging and must be individualised. It includes dietary advice, antihistaminic, analgesic and anti-inflammatory drugs, instillation into the bladder of defined medicinal products (e.g. glycosaminoglycan layer repair) and cystoscopic fulguration, all depending on the clinical situation [3]. Clinical studies are fastidious, and treatment alternatives have shown limited efficacy. Therefore, there is a critical need for new approaches and animal studies are essential tools to better understand the mechanisms of action and evaluate the efficacy of different treatment modalities.
Systemic administration of Cyclophosphamide (CYP) in a rodent model induces an inflammatory process mimicking human IC/BPS and has been performed by various research groups [4–6] including ours. The model is valuable for enhancing our understanding of the pathophysiological progression of the disease [7].
CernitinTM is a pollen extract including two well-defined active pharmaceutical ingrediencies (APIs) – the water‐soluble fraction T60 and the fat‐soluble fraction GBX (glycosides from pollen extract). CernitinTM has been used for decades as a phytotherapeutic alternative in the management of Benign Prostatic Hyperplasia (BPH) and of chronic non-bacterial prostatitis (CP) associated with chronic pelvic pain (CPP), resulting in the reduction of overall clinical symptom score, particularly the pain domain [8].
In previous in vitro studies, we demonstrated that the APIs of CernitinTM significantly alleviated pain scores in induced chronic prostatitis and in urinary bladder inflammation (cystitis) in rats by reducing the levels of cyclooxygenase-2 (COX-2) and monocyte chemoattractant protein (MCP-1) [6, 9]. Although knowledge about the mechanism of action remains limited, these results suggest that the chronic inflammatory process is a possible point of action of the medical product. These results inspired us to further extend the study and explore a panel of biomarkers that play a pivotal role in the inflammatory process and eventually identify a pattern for improved diagnosis.
Cytokines are categorised as either pro-inflammatory or anti-inflammatory cytokines, and the outcome of the inflammatory process depends on the balance between their relative effects under different microenvironments [10]. Cytokines are key factors in initiating, modulating and maintaining inflammation in damaged tissue, and indirectly cause pain by activating nociceptive sensory neurons [11]
The present study aimed at evaluating the effects of CernitinTM APIs on biomarkers including key inflammatory cytokines and interleukins (IL): IL-1α, IL-2, IL-4, IL-6, IL-12a,b, Chemokine CCL4, and fibroblast growth factor 21 (FGF21), and to discuss the findings in relation to other inflammatory diseases.
Material and methods
The materials utilised in this study consisted of bladder wall tissue homogenate extracted from adult female Sprague Dawley rats (8 weeks old; initial body weight 235 ± 35 g). These samples were derived from a previously performed experiment [6]. Briefly after CYP-induced inflammation, as described previously [4, 6], the animals were randomly allocated to six groups (5 animals per group). The solutions were given by gavage. Group 1 received the vehicle 1 (H2O); group 2 the vehicle 2 DMSO ( dimethyl sulfoxide)/corn oil; group 3 the active water-soluble fraction T60 (22.5 mg/kg) in H2O; group 4 the active fat-soluble fraction GBX (1.2375 mg/kg) in DMSO/corn oil; group 5 the combination T60+GBX (22.5 + 1.2375 mg/kg); and group 6: gabapentin (100 mg/kg) prepared in H2O.
The tissues were preserved in a freezer at –80°C, until use. The preparation of homogenates was performed according to the Olink® recommendations. Levels of proteins were determined with the Olink® Target 48 Mouse Cytokines (Olink® Bioscience, Uppsala, Sweden), which is a reagent kit measuring 43 well-established protein biomarkers relevant in inflammation in mouse (5/48 are internal controls), also shown suitable for rat proteins [https://olink.com/products/olink-target]. Biomarkers are listed in Supplement 1 (S1). At the same time, a Proximity Extension Assay (PEA) technology was used according to the manufacturer’s instructions. PEA means that a pair of oligonucleotide-labelled antibodies bind to their respective target protein. When the two antibodies are close to each other, a polymerase chain reaction (PCR) is initiated which is then quantified by real time PCR. Results were expressed as an absolute quantification pg/mL.
Statistical analysis
The student’s t-test was used to compare the means of two independent groups with equal variance and small sizes, expressed as mean ± SD. Due to small size in each group, an analysis of variance (ANOVA) was added to test any difference between the means of the independent groups. A p-value of < 0.05 was considered statistically significant and < 0.08 as a statistical trend. These are described as notable findings in the text.
Results
Of the 43 cytokines and chemokines studied, 7 showed statistically significant trends or trends toward statistically significant reduction compared with the controls/vehicles (Table 1). The other biomarkers were either of very low value or undetectable, and were therefore excluded from all analyses. In general, animals treated with GBX and the combination T60+GBX showed to be more effective than T60 alone in reducing the cytokines levels. Figure 1A–D shows that the cytokines IL-1α, IL-2, IL-4 and IL-6 were significantly downregulated in animals treated with the combination T60+GBX (p < 0.02, p < 0.05, p < 0.01 and p < 0.03, respectively) and there was a tendency to decrease the expression of IL-12a,b (p < 0.07) and CCL4 (p < 0.08) (Figure 1E). Treatment with GBX alone significantly decreased IL-1α (p < 0.03) and CCL4 (p < 0.01), while IL-4 (p < 0.08) and IL-6 (p < 0.07) showed a reduction trend compared to vehicle treated animals (Figure 1C, D). Interestingly, T60 treatment significantly decreased only the level of IL-12a,b (p < 0.05). Furthermore, gabapentin treatment significantly curbed IL-4 (p < 0.01) with a trend to suppress IL-1α (p < 0.03) and IL-2 (p < 0.08) as well. But no effects were observed on the rest of the biomarkers (Figure 1A–C). These two statistical tests gave a very close result profile, confirming the level of statistical value.
| Treatment | IL-1α | IL-2 | IL-4 | IL-6 | IL-12a,b | CCL4 | FGF21 |
| T60 | 0.18 0.38 |
0.15 0.32 |
0.1 0.16 |
0.27 0.54 |
0.05 0.06 |
0.25 0.50 |
0.33 0.51 |
| GBX | 0.03 0.06 |
0.38 0.77 |
0.08 0.22 |
0.07 0.14 |
0.14 0.30 |
0.01 0.03 |
0.05 0.1 |
| T60+GBX | 0.02 0.05 |
0.05 0.07 |
0.01 0.05 |
0.03 0.03 |
0.07 0.15 |
0.08 0.17 |
0.14 0.28 |
| Gabapentin | 0.03 0.02 |
0.08 0.08 |
0.01 0.02 |
NS NS |
NS NS |
NS NS |
NS NS |
| Values are means SD and are expressed in pg/mL, calculated with student t-test for independent groups (upper p-value) and ANOVA (lower p-value). Values equal or less than 0.05 (t-test) were considered as significant (green cells) and values equal or less than 0.08 as showing a trend towards significant (in text also called ‘notably’). | |||||||
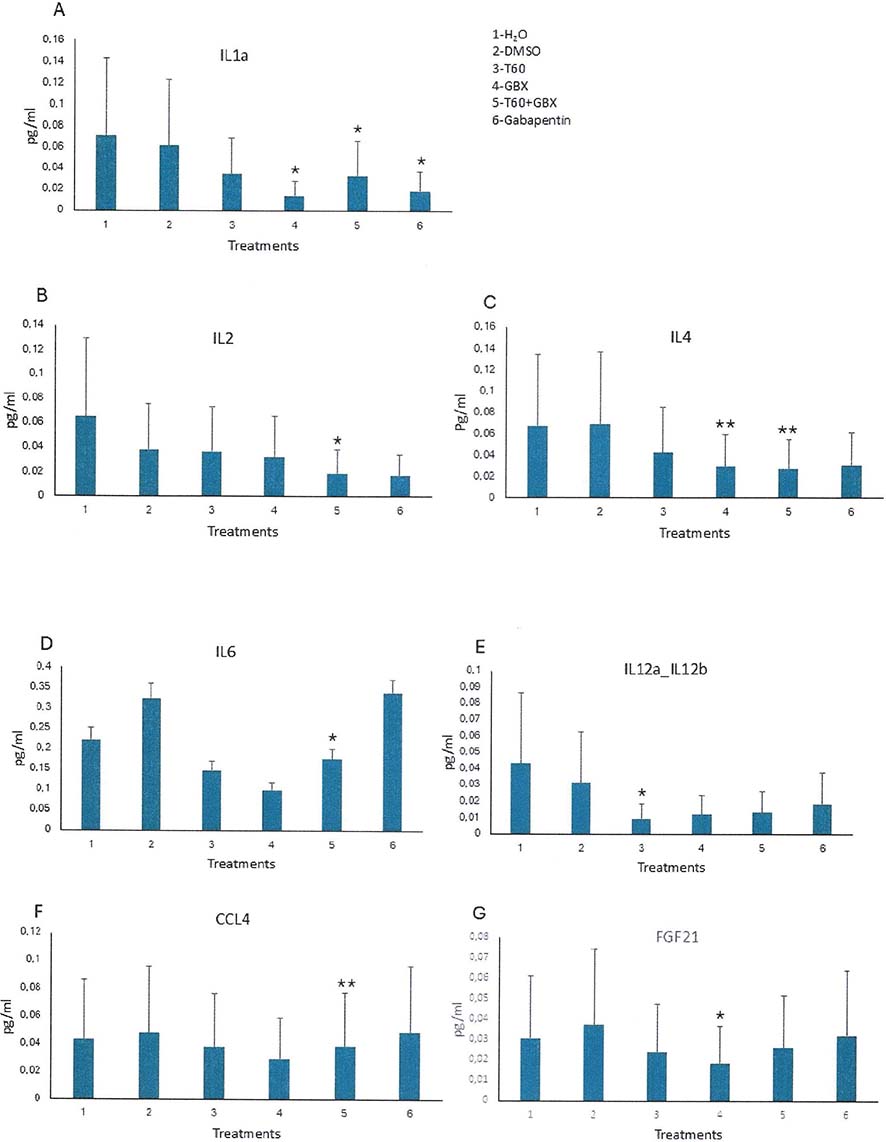
Figure 1. Graphs showing expression of cytokines in the induced urinary bladder inflammation as indicated for each biomarker. P values are presented in Table 1. Values are means SD and are expressed in pg/mL. *Is for p < 0.05 and ** for p < 0.01.
Discussion
In a previous in vivo study using the APIs (T60 and GBX) of the pollen extract medical product CernitinTM, we demonstrated an anti-nociceptive effect, characterised by a significant reduction in pain scores and a decrease in inflammatory COX-2 levels [6]. In continuation, the present study aims at assessing the impact of the same APIs on inflammatory biomarkers. A panel of 43 biomarkers (Olink®, Target 48-mouse) was used to measure the levels in the rat bladder tissue homogenate extraction.
Results revealed that there was a significant or a trend to significant association for seven biomarkers compared to vehicle-treated controls. These seven proteins have a variety of biological functions such as maintaining inflammation, growth factors, and chemo-attractants response (Table 2). It is likely that a single cytokine will not serve as an adequate biomarker for either diagnosis or disease severity. Instead, panels of inflammatory mediators may reveal more about the different pathways of inflammation. Current investigation has highlighted that there is a correlation between CernitinTM treatment and pain in animals, expressed in the significant decreased levels of several cytokines related to inflammation.
| Biomarker | Function | References |
| IL-1α | Ubiquitous and pivotal pro-inflammatory cytokine. IL-1α inhibitor used for treatment of various inflammations in clinical scenario | [12] |
| IL-2 | Pleiotropic cytokine which plays a crucial role in the immune system response and cancer. Proliferation of T cells | [13] |
| IL-4 | Involved in mast cells recruitment and histamine production. The inhibition of IL-4 signalling is being evaluated for atopic dermatitis | [14] |
| IL-6 | One of the most prominent pro-inflammatory cytokines | [10] |
| IL-12a,b | Depending on the context they have pro-inflammatory as well as anti-inflammatory functions. | [15] |
| CCL4 | A chemokine plays a pivotal role in the immune system by guiding the migration of immune cells to the site of inflammation | [16] |
| FGF21 | Improve tissue damage resulted from inflammatory and/or immune stress state, the so-called metabolic inflammation, e.g. Diabetes type 2 | [17] |
Cytokines and chemokines are the most studied biomarkers; they are widely used as predictors for diagnosis and treatments of many conditions including some cancers and chronic inflammatory diseases [10, 18, 19]. Although typically secreted by immune cells, cytokines could also be produced from epithelial cells and may locally modulate the inflammatory response [10]. The current report is based on the cytokines secreted locally from urothelial cells and potentially by immune cells prevailing in the disrupted bladder wall. Given the important role of cytokines in the inflammation process, investigations are dedicated to developing inhibition of pro-inflammatory cytokines as therapeutic option in acute and chronic bladder inflammation [10]. Numerous studies have shown that cytokines are markedly increased in both IC/BPS patients and in experimental models [20, 21]. Here, we have identified biomarkers that were downregulated after treatment with CernitinTM APIs, in line with earlier studies showing that CernitinTM induced anti-inflammatory effect in CP in rats mainly through inhibition of pro-inflammatory cytokines such as tumour necrosis factor‐α (TNF‐α), IL-6, and IL-1β. The result from the present study identified CernitinTM APIs combination to significantly reduce IL-1α and IL-6 level in rat bladder inflammation (Figure 1A, D). IL-1 inhibitors are used in the treatment of several inflammatory condition. Animal study models have demonstrated that blocking of IL-1α receptor antagonist, anakinra, reduces the pain response in the animals [22].
IL-6 and IL-8 are the cytokines mostly correlated to the urological diseases, such as BPH, prostate cancer, and IC [5, 12]. It is well established that IL-6 is a pro-inflammatory cytokine and mediates its action through activation of signal transducer and activator of transcription three pathway [23]. Zheng and co-workers have shown that intervention with IL-6 receptor antagonist (tocilizumab) led to reverse inflammation induced by hyaluronidase perfusion in a rat model [5]. Studies showing downregulation of IL-6 confirm the biomarker’s key role in the control of inflammation and how curbing it could lead to modulation of chronic inflammatory disease development and associated symptoms alleviation.
Furthermore, the APIs’ suppression of IL-4 expression is particularly interesting because studies have linked the overexpression of IL-4 with the activation of mast cells and production of histamine, that markedly aggravates the symptoms of IC/BPS in some patients [24]. Sugaya et al. have reported that patients with IC had some types of allergic or autoimmune disease which was associated with overproduction of IL-4 [25]. Besides its role in IC, IL-4 has also shown to play a central role in pathogenesis of atopic dermatitis [14]. Atopic dermatitis is a common chronic condition causing intense itching and persistent eczematous lesions. The first approved monoclonal antibody, dupilumab, which blocks IL-4Rα led to significant improvements in patient symptoms. This observation may suggest that the APIs could have induced the symptom relief, reported in previous report [6], either through altered mast cell proliferation by IL-4 downregulation or through inhibition of degranulation and histamine release from mast cells.
While the role of the five above-mentioned cytokines is well documented, the role of IL-2, IL-12a,b and FGF21 is less known in the context of bladder chronic inflammation. Results of in vivo studies have collectively shown that IL-2 stimulate proliferation, specifically proliferation of antigen-stimulated T cells and are involved in cancer immunology [13]. Reduced expression of IL-2 by CernitinTM may have occurred through inhibition of immune cell proliferation producing IL-2.
A hampering of IL-12a,b expression was demonstrated with T60 treatment only. This biomarker functions as a growth factor activating T lymphocytes and natural killer cells and induces the secretion of IFN-g from these cells [15]. IL-12 is a cytokine derived from dendritic cells and mast cells, among others [26]. Increased expression of IL-12 is observed in induced bladder inflammation in rat [20]. Our study detected a decreased levels of IL-12a,b which may indicate that an impact on the bladder through proliferation inhibition of immune cells. We have previously shown that CernitinTM APIs have an antiproliferative effect on BPH cell lines [9].
Interestingly, FGF21 exhibits significant decrease after GBX treatment. FGF21 is a hormone that expresses in several metabolically active organs and its correlation to the obesity induced inflammation has previously been reported [17]. However, its direct action in the bladder inflammation is not known.
Chemokines CCL2, CCL4, CCL5, CCL7 have been found significantly elevated in either urine or serum samples or bladder tissue in patients suffering of IC/BPS [21, 27]. Numerous studies have shown that CCL4 inhibitors significantly downregulated adhesion and inflammation proteins in various inflammatory diseases conditions, for example atherosclerosis [16]. However, CCL4 inhibition in bladder inflammation has not been studied and can become a target of interest for therapeutic intervention. Given its role in the decreasing level of CCL4, CernitinTM presents a novel finding that may have new treatment implications.
The specific role each of these cytokines plays in orchestrating the anti-inflammatory and pain action remains unclear. The results taken together suggest nonetheless that a characteristic pattern of several different cytokines might be identified and that eventually could improve the diagnostic and follow-up possibilities of IC/BPS.
Pain is a hallmark of inflammation, and inflammatory induced pain is a major clinical issue in many clinical disorders including both urological conditions IC/BPS and CP/CPP. Cyclooxygenase-2 (COX-2) is a key enzyme that synthesises prostaglandins PGE2 [28], which, in turn, sensitises peripheral nociceptive nerves and neuropathic pain through its receptors [29]. Acting on this level by reducing the COX-2 level by means of pro-inflammatory cytokine modulation, CernitinTM could hypothetically offer a protective role against bladder wall damage. Theoretically, this effect could also be the mechanism explaining the clinical improvement on both overall symptom score and, especially, the pain domain in CP. Combining this effect with the anti-proliferative effect on prostatic cell lines, as shown previously [9], a double impact on BPH could explain a score reduction in the urological domain as well.
Gabapentin, a pain relief medicine, was used in a comparative arm as an established drug for treatment of for example pelvic pain. Gabapentin was shown in this study to reduce levels of IL-1α, IL-2 and IL-4, which has also been shown in intestinal inflammation for IL-1β [30].
There are limitations to consider in this study. Firstly, the absence of homogenate tissue unexposed to CYP-inflammation induction as baseline controls. The reason being that the original study followed the validated model design and protocol [4, 6]. However, it is relevant to mention that in an early study during the development of the model, CYP-inflammation induced animal tissues were compared to a saline group. As for the biomarkers, the CYP-groups showed a significant elevation of IL-1β, IL-6, and MCP-1, not found in the saline group [31]. Secondly, detecting very low levels of the different cytokines is a challenge, considering also the biomarkers secretory dynamic and their short half-lives. By using the Olink® assay we were, nonetheless, able to detect seven interesting inflammatory biomarkers on which the APIs treatment impacted beneficially compared to control animals receiving the vehicles alone. Thirdly, the small number of animals in each group which, among others, is reflected by the wide variation in standard deviation and the observation of borderline statistical values for some of the cytokines. However, we found a consistency in the statistical analysis that confirmed the aim of the study to detect useful biomarkers for diagnosis and follow-up even in benign urological conditions and serves as an encouragement for further investigations in the same direction [32].
Conclusion
Correlation analysis between biomarker levels and CernitinTM APIs in animals with CYP-induced bladder inflammation showed that the combination of the fractions GBX and T60 was significantly or noticeably (trend to significance) associated with downregulation of several important inflammatory biomarkers including IL-1α, IL-2, IL-4, IL-6, IL-12a, IL-12b, CCL4, and FGF21. The fraction GBX was superior to the T60 fraction alone, while the combination of the two APIs gave additive effect. Results from the current data suggest that CernitinTM might be a candidate inhibitor of pro-inflammatory cytokines to relieve the inflammatory process and by extension the related pain symptoms. However, whether it can be translated to clinical use in patients suffering from IC/BPS remains to be further investigated.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethical statement
All experiments were performed according to the European Community Council Directive 2010/63/UE and the French Ministry for Agriculture, Agrifood and Forestry (Decree 2013-118). Experimental protocols were reviewed by CEEA-122 Ethical Committee for Protection of Animals used for Scientific Purposes and approved by French Ministry for National Education, Higher Education and Research under the number APAFIS #16506- 2018082411278474.
Authors’ contribution
ND and MG designed the experiments and supervised the studies. CA performed the in vivo experiments while ND performed the present analyses. ND and MG have written and edited the manuscript. PL provided clinical view to the manuscript. All authors critically reviewed and approved the final form of the manuscript.
References
[1] Jhang JF, Kuo HC. Pathomechanism of interstitial cystitis/bladder pain syndrome and mapping the heterogeneity of disease. Int Neurourol J. 2016;20:95–104. https://doi.org/10.5213/inj.1632712.356
[2] Neuhaus J, Berndt-Paetz M, Gonsior A. Biomarkers in the light of the etiopathology of IC/BPS. Diagnostics. 2021;11(12):2231. https://doi.org/10.3390/diagnostics11122231
[3] Quentin Clemens J, Erickson DR, Varela NP, et al. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome. J Urol. 2022;208(1):34–42. https://doi.org/10.1097/JU.0000000000002756
[4] Augé C, Gamé X, Vergnolle N, et al. Characterization and validation of a chronic model of cyclophosphamide-induced interstitial cystitis/bladder pain syndrome in rats. Front Pharmacol. 2020;11:1305. https://doi.org/10.3389/fphar.2020.01305
[5] Zheng Z, Zhang J, Zhang C, et al. The study on the function and cell source of interleukin-6 in interstitial cystitis/bladder painful syndrome rat model. Immun Inflamm Dis. 2021;9(4):1520–1528. https://doi.org/10.1002/iid3.505
[6] Augé C, Dizeyi N, Ramnemark L, et al. Experimental in vivo model to evaluate the impact of CernitinTM on pain response on induced chronic bladder inflammation. Scand J Urol. 2022;56(4):320–328. https://doi.org/10.1080/21681805.2022.2090602
[7] Tay C, Grundy L. Animal models of interstitial cystitis/bladder pain syndrome. Front Physiol. 2023;14:1232017. https://doi.org/10.3389/fphys.2023.1232017
[8] Wagenlehner FM, Schneider H, Ludwig M, et al. A pollen extract (Cernilton) in patients with inflammatory chronic prostatitis-chronic pelvic pain syndrome: a multicentre, randomised, prospective, double-blind, placebo-controlled phase 3 study. Eur Urol. 2009;56(3):544–551. https://doi.org/10.1016/j.eururo.2009.05.046
[9] Chabot S, Dizeyi N, Ramnemark L, et al. Impact of CernitinTM on induced chronic prostatitis in animal model for understanding management of lower urinary tract symptoms. Phytomedicine Plus. 2021;1:100057. https://doi.org/10.1016/j.phyplu.2021.100057
[10] Turner MD, Nedjai B, Hurst T, et al. Cytokines and chemokines: at the crossroads of cell signaling and inflammatory disease. Biochim Biophys Acta. 2014;1843(11):2563–2582. https://doi.org/10.1016/j.bbamcr.2014.05.014
[11] Zhang JM, An J. Cytokines, inflammation, and pain. Int Anesthesiol Clin. 2007;45(2):27–37. https://doi.org/10.1097/AIA.0b013e318034194e
[12] Yu WR, Jiang YH, Jhang JF, et al. Use of urinary cytokine and chemokine levels for identifying bladder conditions and predicting treatment outcomes in patients with interstitial cystitis/bladder pain syndrome. Biomedicines. 2022;10(5):1149. https://doi.org/10.3390/biomedicines10051149
[13] Abbas AK. The surprising story of IL-2: from experimental models to clinical application. Am J Pathol. 2020;190(9):1776–1781. https://doi.org/10.1016/j.ajpath.2020.05.007
[14] Torres T, Mendes-Bastos P, Cruz MJ, et al. Interleukin-4 and atopic dermatitis: why does it matter? A narrative review. Dermatol Ther. 2025;15(3):579–597. https://doi.org/10.1007/s13555-025-01352-y
[15] Ullrich KA-M, Schulze LL, Paap E-M, et al. Immunology of IL-12: an update on functional activities and implications for disease. EXCLI J. 2020;19:1563–1589. https://doi.org/10.17179/excli2020-3104
[16] Chang TT, Yang HY, Chen C, et al. CCL4 inhibition in atherosclerosis: effects of plaque stability, endothelial cell adhesiveness, and macrophages activation. Int J Mol Sci. 2020;21(18):6567. https://doi.org/10.3390/ijms21186567
[17] Yang M, Liu C, Jiang N, et al. Fibroblast growth factor 21 in metabolic syndrome. Front Endocrinol. 2023;14:121220426. https://doi.org/10.3389/fendo.2023.1220426
[18] Kureshi CT, Dougan SK. Cytokines in cancer. Cancer Cell. 2025;43(1):15–35. https://doi.org/10.1016/j.ccell.2024.11.011
[19] Wullt B, Butler DSC, Ambite I, et al. Immunomodulation – a molecular solution to treating patients with severe bladder pain syndrome? Eur Urol Open Sci. 2021:31:49–58. https://doi.org/10.1016/j.euros.2021.07.003
[20] Smaldone MC, Vodovotz Y, Tyagi Y, et al. Multiplex analysis of urinary cytokine levels in rat model of cyclophosphamide-induced cystitis. Urology. 2009;73(2):421–426. https://doi.org/10.1016/j.urology.2008.07.031
[21] Jiang YH, Jhang JF, Hsu YH, et al. Usefulness of urinary biomarkers for assessing bladder condition and histopathology in patients with interstitial cystitis/bladder pain syndrome. Int J Mol Sci. 2022;23(19):12044. https://doi.org/10.3390/ijms231912044
[22] Dinarello CA, Simon A, Van Der Meer JWM. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov 2012;11(8):633–652. https://doi.org/10.1038/nrd3800
[23] Dizeyi N, Mattisson IY, Ramnemark L, et al. The effects of Cernitin® on inflammatory parameters and benign prostatic hyperplasia: an in vitro study. Phytother Res. 2019;33(9):2457–2464. https://doi.org/10.1002/ptr.6438
[24] Sant GR, Kempuraj D, Marchand JE, et al. The mast cell in interstitial cystitis: role in pathophysiology and pathogenesis. Urology. 2007;69(4 Suppl):34–40. https://doi.org/10.1016/j.urology.2006.08.1109
[25] Sugaya K, Nishijima S, Yamada T, et al. Molecular analysis of adrenergic receptor genes and interleukin-4/interleukin-4 receptor genes in patients with interstitial cystitis. J Urol. 2002;168(6):2668–2671. https://doi.org/10.1016/S0022-5347(05)64241-3
[26] Tait Wojno ED, Hunter CA, Stumhofer JS. The immunobiology of the interleukin-12 family: room for discovery. Immunity. 2019;50(4):851–870. https://doi.org/10.1016/j.immuni.2019.03.011
[27] Lemmon B, Kyrgiou M, Mullins E, et al. Cytokines in bladder pain syndrome: a review of the literature. Int Urogynecol J. 2024;35(6):1119–1129. https://doi.org/10.1007/s00192-024-05778-4
[28] Zmigrodzka M, Rzepecka A, Krzyzowska M, et al. The cyclooxygenase-2/prostaglandin E 2 pathway and its role in the pathogenesis of human and dog hematological malignancies. J Physiol Pharmacol. 2018;69(5):653–661. https://doi.org/10.26402/jpp.2018.5.01
[29] Wang C, Li GW, Huang LY. Prostaglandin E2 potentiation of P2X3 receptor mediated currents in dorsal root ganglion neurons. Mol Pain. 2007;32:22. https://doi.org/10.1186/1744-8069-3-22
[30] De Brito TV, Dias Junior GJ, da Cruz Junior JS, et al. Gabapentin attenuates intestinal inflammation: role of PPAR-gamma receptor. Eur J Pharmacol. 2020;873:172974. https://doi.org/10.1016/j.ejphar.2020.172974
[31] Augé C, Chene G, Dubourdeau M, et al. Relevance of the cyclophosphamide-induced cystitis model for pharmacological studies targeting inflammation and pain of the bladder. Eur J Pharmacol. 2013;707:32–40. https://doi.org/10.1016/j.ejphar2013.03.008
[32] Akdere H, Blir B, Kuvan HC, et al. Interleukins in urological diseases. Discov Med. 2025;37:647–658. https://doi.org/10.24976/Discov.Med.202537195.56