ORIGINAL RESEARCH ARTICLE
Risk of Achilles tendon rupture following single-dose ciprofloxacin use in transrectal prostate biopsies
Michaela Runnäsa, Erik Lampab, Johan Styrkea and Karl-Johan Lundströma
aDepartment of Diagnostics and Intervention, Umeå University, Umeå, Sweden; bEpistat AB, Uppsala, Sweden
ABSTRACT
Purpose: To assess the risk of Achilles tendon rupture (ATR) following single-dose ciprofloxacin prophylaxis for transrectal prostate biopsy.
Methods: Using the Prostate Cancer data Base Sweden (PCBaSe 5.0), we analysed 44,959 prostate biopsy exposures versus 662,520 non-exposures to assess the risk of ATR. Prostate biopsy served as a proxy for quinolone use, with single-dose ciprofloxacin being the recommended and most extensively documented prophylaxis in Sweden for this procedure. The outcome was ATR in men who underwent a biopsy compared to those who did not.
Results: The incidence rate of ATR was 60.46 per 100,000 person-years in the no-biopsy group, compared to 62,77 per 100,000 person-years in the biopsy group. The multivariable analysis yielded a hazard ratio of 0.98 (95% confidence interval [CI]: 0.83–1.15).
Conclusions: Transrectal prostate biopsy was not associated with an elevated risk of Achilles tendon rupture. This provides some evidence against single-dose ciprofloxacin being a relevant risk factor for this outcome, but there is remaining uncertainty related to study limitations.
KEYWORDS: Prostate biopsy; Achilles tendon rupture; ciprofloxacin; prophylaxis; fluoroquinolones.
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 121–126. https://doi.org/10.2340/sju.v61.45649.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 13 September 2025; Accepted: 16 February 2026; Published: 16 April 2026
CONTACT: Karl-Johan Lundström, Michaela Runnäs karl-johan.lundstrom@umu.se, Michaela.runnas@regionjh.se Department of Diagnostics and Intervention, Umeå University, 901 87 Umeå
Introduction
Fluoroquinolones, particularly ciprofloxacin, are among the most commonly used antibiotic prophylaxis agents during transrectal prostate biopsies worldwide [1]. About 84% of clinics in Sweden utilise ciprofloxacin, with 66% employing a single-dose prophylaxis regimen [2]. In 2018, the European Medicines Agency (EMA) recommended restrictions on its prophylactic use due to reported risks of complications [3, 4], which subsequently has made fluoroquinolone use a controversy [5]. Among these complications, tendon-related disorders, such as Achilles tendon ruptures (ATR) and aortic aneurysms, have been noted [6–9]. One theory suggests that fluoroquinolone-associated adverse effects may result from direct collagen effects on the Achilles tendon [10, 11]. In one study performed in 2021, no associations were found between aortic aneurysms and fluoroquinolone given as short-term prophylaxis. They did, however, find an association of high-risk prostate cancer and aortic aneurysms, concluding that this might be attributed to increased radiology in that study group [12].
A 2019 meta-analysis highlighted an increased risk of tendon-related disorders, such as ruptures, in patients receiving fluoroquinolones. However, many studies did not consider dosage amount and length of treatment and instead focused on concomitant corticosteroid use and age of the patients. The analysis also suggested further investigation into separate risks associated with different fluoroquinolones [13]. A systematic review aimed at identifying predictors of ATR found limited evidence supporting the association between fluoroquinolones and ATR. Additionally, evidence linking corticosteroid therapy, increased age, obesity, renal disease and ATR was found to be scarce [14].
This study aims to assess the risk of ATR following a transrectal prostate biopsy as a proxy for the administration of ciprofloxacin as a single-dose prophylaxis regimen.
Materials and methods
Study design
A register-based cohort study was conducted using data from a defined national population, including over 99% of all prostate cancer cases in Sweden, along with five age and residency-matched controls without prostate cancer diagnosis [15–17].
Study population
Data collection was made utilising the Prostate Cancer data Base Sweden (PCBaSe 5.0), a comprehensive national register containing the National Prostate Cancer Register (NPCR), the Swedish Cancer Registry, the Cause of Death Registry, the Prescribed Drug Registry, the National Patient Registry, the Longitudinal integrated database for health insurance and labour market studies (LISA) and the Multi-Generation Registry, where individuals are identified by the unique Swedish Personal Identity Number (PIN). For a detailed database description, see van Heljmrick et al. [17]. Each patient with prostate cancer in the register is matched with five, prostate cancer free, randomly selected controls in the same age group and geographic location [16].
Inclusion and exclusion
Inclusion consisted of patients with or without a documented prostate cancer diagnosis prior to transrectal prostate biopsies. Follow-up ranged from June 1, 2006 until December 30, 2020. In 2006, the Prescribed Drug Registry was initiated, while the last update of PCBaSe was executed in 2020. Exclusion criteria were defined as individuals registered before June 1, 2006 or after December 30, 2020, previously biopsied individuals and individuals with dates of death prior to the inclusion date in the cohort. Patients were only included at their first prostate biopsy (Figure 1), which was also considered the index date. The age and region-matched controls had the same index date as their matched case. Patients who had a previous prescription of a fluoroquinolone in the past year were also excluded from the main analysis.
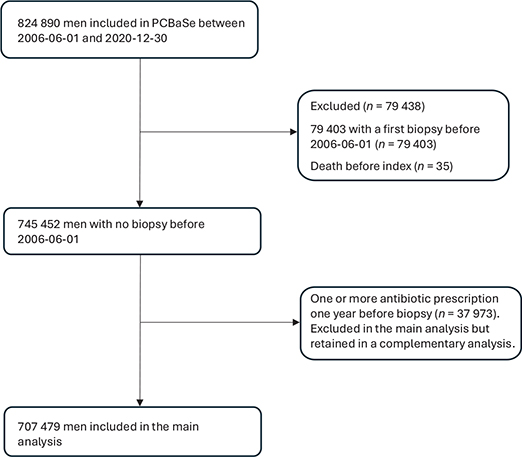
Figure 1. Inclusion and exclusion demonstrated as a flow chart, presenting the final inclusion.
Exposure
Patients after the index date (defined as above) having a surgical code indicating transrectal prostate biopsy were considered exposed. Prostate biopsy was chosen as the proxy since data on single-dose prophylaxis administration is not recorded in the prescribed drug register, as it is typically administered directly in clinics rather than prescribed [2]. Therefore, all patients undergoing biopsy were considered to have had single-dose ciprofloxacin exposure at the time of biopsy and were followed until ATR occurrence, death or December 31, 2020, whichever came first.
Baseline variables
Baseline variables included age, prior diagnosis of aortic aneurysm/dissection/arterial aneurysms (ICD-10 codes: I71-2), hypertension (ICD-10 codes: I10-I15), cardiovascular disease (ICD-codes: I20-I25), peripheral arterial disease (ICD10-codes: I70, I73), Charlson comorbidity index (CCI), income status (grouped into fourths), educational level (elementary school, high school, academic), corticosteroid therapy (ATC-code: H02AB01-18) within 90 days prior to the biopsy and prior injury related to trauma (ICD-10 codes: S42-43, S52-53, S56, S62-63, S66, S72-73, S76, S82, S86, S92-93, S96). The lookback period for the majority of the variables stretched to the beginning of the register; for CCI, the lookback period consisted of 10 years.
Outcomes
ATR was considered the outcome at 120 days after exposure. ATR was defined using the ICD-10 code S86.0, and the date of ATR was defined as the first occurrence of S86.0 in the National Patient Registry, including all subsequent visits in the hospital and specialised outpatient care [18]. The date of death was extracted from the Cause of Death Registry. If both the month and day of death were missing, they were imputed as June 30; if only the day was missing, it was imputed as the 15th. Death from any cause was considered a competing risk in all analyses.
Subgroup analysis
In the study population exposed to biopsy, we stratified the group in their respective prostate cancer risk group category to investigate whether this had any association with the incidence of ATR, in line with previous studies showing a possible association between high-risk prostate cancer and aortic aneurysms [12]. Prostate cancer risk was defined as low-risk (T1-T2a, Gleason score ≤ 6, PSA < 10 ng/ml), intermediate-risk (T2b-T2c, PSA 10–20 ng/ml, Gleason score 7 (4 + 3 or 3 + 4)), or high-risk (> T2 or PSA > 20 ng/ml or Gleason score > 7 or metastases) using a modified version of National Comprehensive Cancer Network (NCCN) classification [19].
Sensitivity and complementary analyses
A sensitivity analysis was made, including biopsied patients from departments known for having only ciprofloxacin as prophylaxis prior to transrectal prostate biopsies. A complementary analysis was made analysing patients with multiple prescriptions of fluoroquinolones within 1 year. This was to investigate whether there could be a dose–response relationship present. The information was apprehended from the Prescribed Drug Registry, where all prescriptions are imputed. We performed an additional complementary analysis calculating the incidence of ATR in the group with previous fluoroquinolone exposure in the past year compared with the unexposed group.
Statistical analysis
Men biopsied at the index date were classified as exposed at baseline, whereas men who did not undergo a biopsy at the index date were classified as unexposed. During follow-up, unexposed men who underwent a biopsy contributed additional person-time as exposed from the date of biopsy and onward. Thus, a single man could contribute person-time under both exposure categories. Cumulative incidence of ATRs was estimated using the Aalen-Johansen estimator, which is a multistate version of the Kaplan–Meier estimator, accounting for mortality incidence and, for men without a biopsy at baseline, the biopsy incidence. Hazard ratios (HR) and 95% confidence intervals (CI) were estimated using a Cox proportional hazards model, with exposure status included as a time-varying variable. Included in the model were variables assumed to influence the incidence of ATR: CCI (included as a continuous variable), age at baseline, a history of aortic dissection/arterial aneurysms, corticosteroid therapy, hypertension, peripheral artery disease, trauma, cardiovascular disease, household income in quarters and baseline calendar time. Baseline age and calendar time were modeled using restricted cubic splines with three knots placed at the 10th, 50th and 90th percentiles of each variable’s marginal distribution.
The model was further stratified by previous ATR, assuming that the baseline hazards for the two strata may differ, but the relative effect of the exposure is assumed to be the same. The proportional hazards assumption was assessed by testing the slope in a generalised linear regression of the scaled Schönfeld residuals on time [20]. All analyses were based on complete case data as the fraction of missing information was low.
Ethics
Individual pseudonymised patient data were analysed; no identifiable patient data were handled as patient identification was removed by Statistics Sweden during database assembly. Ethical approval was obtained from the Swedish Ethical Review Authority, Dnr 2023-02873-02.
Results
A total of 707,479 men were selected for the analyses, consisting of 44,959 patients exposed to biopsy and 662,520 not exposed to biopsy at baseline (Figure 1). Additionally, 24,833 in the no-biopsy group had their first biopsy during follow-up and were subsequently added to the biopsy group for further follow-up. A total of 104,270 died without being biopsied and 9,210 died after being biopsied. Overall, 2,251 cases of ATR were identified: 2,089 without prior biopsy, with an incidence rate of 60.46 per 100,000 person-years, and 162 with a prior biopsy, with an incidence rate of 62.77 per 100,000 person-years. Descriptive variables are shown in Table 1, and distributions among the variables were overall comparable. The total fraction of missing data was 2.8% (n = 19,913).
The cumulative incidence of ATR at 60 days in the unexposed and exposed groups was 16.72 (95% CI: 13.87–20.16) and 13.47 (95% CI: 7.01–25.08) per 100,000, respectively. At 120 days, the cumulative incidence was 27.10 (23.38–31.40) in the unexposed and 30.81 (19.87–47.76) in the exposed group per 100,000 (Figure 2). In the univariable Cox regression analysis, the HR of ATR when exposed to biopsy was 1.04 (95% CI: 0.96–1.22), and in the multivariable analysis, the HR was 0.98 (95% CI: 0.83–1.15) (Table 2). The assumption of proportional hazards was not rejected at conventional levels of statistical significance, Chi2 = 13.4 on 21 degrees of freedom, p = 0.895.
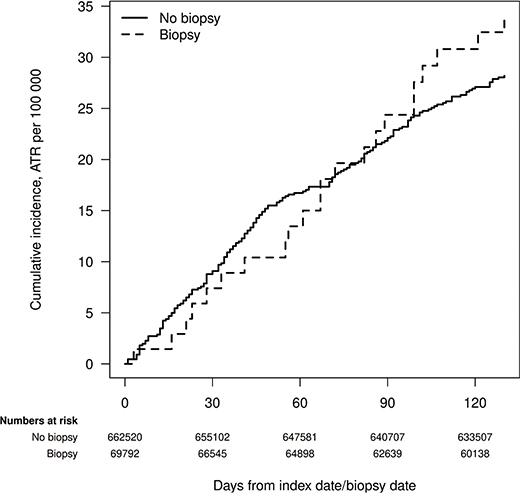
Figure 2. Cumulative incidence of Achilles tendon rupture in the subset of the exposed and the non-exposed groups with at most one antibiotic prescription during the last year. Individuals without a biposy at baseline who were biopsied during follow-up are part of the no-biopsy group until the date of their first biopsy in which they become part of the biopsy group, and their follow-up is reset to zero in the figure.
| Model | Exposure | HR (95% CI) |
| Univariable analysis | ||
| Exposure | ||
| No biopsy | Ref | |
| Biopsy | 1.04 (0.88–1.22) | |
| Multivariable analysis1 | ||
| Exposure | ||
| No biopsy | Ref | |
| Biopsy | 0.98 (0.83–1.15) | |
| Risk group at biopsy | ||
| No cancer | Ref | |
| Low | 0.84 (0.51–1.37) | |
| Intermediate | 1.12 (0.71–1.76) | |
| High | 0.61 (0.33–1.11) | |
| HR: hazard ratio; CI: confidence interval. 1Model adjusted for Charlson comorbidity index, age at baseline, aortic dissection/arterial aneurysms, hypertension, peripheral artery disease, trauma, cardiovascular disease, household income and baseline calendar time, and stratified on previous ATR. |
||
When comparing different risk groups in prostate cancer (low, intermediate and high risks) to the control group, there was a slight increase in the HR of ATR in the intermediate-risk group. In the multivariable analysis, there was a low HR of ATR in the high-risk group (Table 2).
As a sensitivity analysis, we investigated whether there were any differences in outcome in patients who had been biopsied at departments answering a questionnaire that they used ciprofloxacin as prophylaxis prior to transrectal prostate biopsies and found that the incidence numbers were equal and similar to the main analysis (data not shown). In the complementary analysis, the ATR was calculated for previous exposure with more than one prescription of fluoroquinolones to detect a possible dose–response relationship (Figure 3). There was no significant difference in ATR between the exposed and unexposed group, in the multiexposed group versus no exposure, the HR was 0.95 (95% CI: 0.75–1.19).
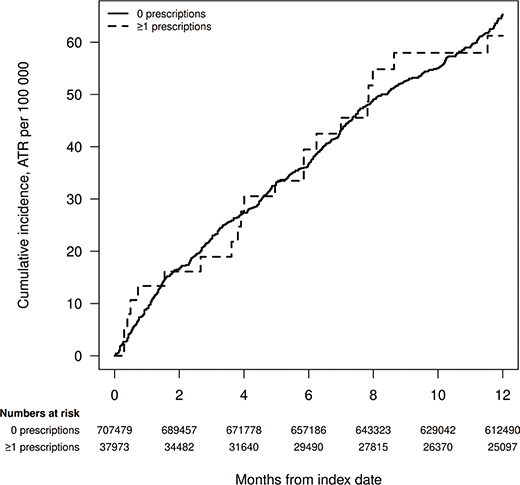
Figure 3. Complementary analysis, previous exposure within 1 year of biopsy. Cumulative incidence of Achilles tendon rupture in patients without or with one or multiple prescriptions (> 1) of fluoroquinolones during the past year.
Discussion
In summary, we did not find any clinicially significant difference in the incidence of ATR between men undergoing prostate biopsy and age-matched men from the general population. The precision of the estimates is good based on the large sample size, and no other major source of bias has been identified. Additionally, there was no significant difference in the incidence of ATR between biopsied patients exposed to none or multiple previous fluoroquinolone prescriptions prior to exposure; the authors are unaware of previous studies investigating this possible association.
Several studies have reported an association between fluoroquinolones and tendon-related disorders, but controversy remains. This, combined with other suspected associated adverse effects of fluoroquinolone use, led to the EMA issuing a ban on their prophylactic use [3]. However, fluoroquinolones are commonly used antibiotics in urological procedures, and most studies do not account for dosage or the specific fluoroquinolone used.
The incidence of ATR varies widely, ranging from 5.5 to 55.2 per 100,000 person-years, regardless of fluoroquinolone exposure [21–24]. Traumatic injuries in younger men and degenerative tendons in the elderly are the primary causes. In this study, the incidence of ATR was 60.46 and 62.77 per 100,000 person-years in the biopsy and no-biopsy group, respectively. Our study found similar incidence rates to previous studies, with a potential increase over the years, possibly due to greater participation in high-demand sports by the elderly [25]. The risk of ATR after administration of fluoroquinolones is still considered low although some studies indicate that the risk increases in combination with risk factors such as age, level of physical activity and concomitant corticosteroid use [9, 26–28]. The median age in our study population exceeds that of previous studies, which could also influence the incidence rates.
While some fluoroquinolones may increase the risk of ATR, others may have minimal significance and remain useful in specific situations. For example, one study found that the odds ratio of ATR in patients exceeding 60 years was 28.4 (95% CI: 7.0–115.3) for ofloxacin and 14.2 (95% CI: 1.6–128.6) for ciprofloxacin, with a dose-dependent relationship observed. However, the doses used in that study ranged from 750 to 1,250 mg administered daily over several consecutive days, and single-dose prophylaxis effect on ATR risk was not investigated [29]. One previous study investigated the risk of ATR within 30 days after fluoroquinolone treatment, with an HR of 1.06 (95% CI: 0.7–1.6) compared with no ciprofloxacin [30].
In our study, we specifically examined the association between ATR and single-dose prophylaxis with ciprofloxacin over a 120-day period after exposure. Baseline characteristics were similar between cases and controls, and there were no differences in ATR incidence rates between the biopsy and no-biopsy groups, even after adjustment. Another study also supported the notion that single-dose prophylaxis is associated with minimal risk of other collagen disorders [12].
We observed a numerically higher risk of ATR in the intermediate-risk prostate cancer group. The reason for this analysis is a previous study, suggesting that patients in a high-risk category would have an increased risk of aortic aneurysm. The reason for that difference is unclear, but it is possible that high-risk prostate cancer patients suffer from a higher level of anxiety and multiple diagnostic procedures within the months from diagnosis, thereby possibly reducing participation in high-demand sports or recreational activities.
Limitations
Using prostate biopsy as a proxy for exposure to ciprofloxacin was a study limitation, as other antibiotics may have been used. However, ciprofloxacin is the most used prophylaxis in Sweden, and most centres analysed typically use it, albeit with some variability. This could lead to under- or overestimation of ATR incidence, as approximately 16% of centres use trimethoprim-sulfametaxole prophylaxis [2]. However, using the questionnaire data, which was previously mentioned, to exclude these departments did not alter the results.
Reporting bias may also have affected our results, in absolute numbers, but national registers have shown reliability and high coverage [16, 17]. The mandatory nature of the national patient register and the conspicuous nature of ATRs mitigate this concern, ensuring comprehensive data coverage since ATR is managed in specialised care, even if in an outpatient setting. Thus, the absolute numbers could be lower than reported, but there is no reason to believe a systematic misclassification of ATR is done in the exposed versus unexposed group.
Strengths
One of the strengths of this study is the large study population, which exceeded 700,000 participants. The use of the personal identification number in Sweden, which is utilised in all interactions with authorities, allows for the tracking of individual patients over time and is an unique advantage in Swedish registers. The coverage of the registry has a high coverage when it comes to hospitals and specialised outpatient care [16, 17].
Conclusion
Transrectal prostate biopsy was not associated with an elevated risk of Achilles tendon rupture. This provides some evidence against single-dose ciprofloxacin being a relevant risk factor for this outcome, but there is remaining uncertainty related to study limitations.
Acknowledgements
This project was made possible by the continuous work of the National Prostate Cancer Register of Sweden (NPCR) steering group: Elin Axén, Johan Styrke, Åsa Jellvert, Andreas Josefsson, Camilla Thellenberg Karlsson, Hampus Nugin, Ingrida Verbiene, Stefan Carlsson, Anna Kristiansen, Mats Andén, Kimia Kohestani, Jon Kindblom, Thomas Jiborn, Viktoria Gaspar, Elin Trägårdh, Fredrik Jäderling, Gert Malmberg, Hans Joelsson, Lotta Renström Koskela, Magnus Törnblom, Marie Hjälm Eriksson, Erik Thimansson, Eva Johansson, Johan Stranne, Jonas Hugosson, Ola Bratt, Per Fransson, Petrus Stensson, Fredrik Sandin, Marie Brus, Lena Pettersson, Anna Hedström, Maria Moutran, Maria Nyberg and Nina Hageman.
References
- [1] Alidjanov JF, Cai T, Bartoletti R, et al. The negative aftermath of prostate biopsy: prophylaxis, complications and antimicrobial stewardship: results of the global prevalence study of infections in urology 2010–2019. World J Urol. 2021;39:3423–3432. https://doi.org/10.1007/s00345-021-03614-8
- [2] Styrke J, Resare S, Lundstrom KJ, et al. Current routines for antibiotic prophylaxis prior to transrectal prostate biopsy: a national survey to all urology clinics in Sweden. F1000Res. 2020;9:58. https://doi.org/10.12688/f1000research.19260.1
- [3] European Medicines Agency (2019). Quinolone- and fluoroquinolone-containing medicinal products. EMA/175398/2019. Available at: https://www.ema.europa.eu/en/medicines/human/referrals/quinolone-fluoroquinolone-containing-medicinal-products
- [4] European Medicines Agency (2018, October 5). Fluoroquinolone and quinolone antibiotics: PRAC recommends restrictions on use. EMA/668915/2018. Available at: https://www.ema.europa.eu/en/documents/press-release/fluoroquinolone-and-quinolone-antibiotics-prac-recommends-restrictions-use_en.pdf
- [5] Bonkat G, Pilatz A, Wagenlehner F. Time to adapt our practice? The European Commission has restricted the use of fluoroquinolones since March 2019. Eur Urol. 2019;76:273–275. https://doi.org/10.1016/j.eururo.2019.06.011
- [6] Lee CC, Lee MT, Chen YS, et al. Risk of aortic dissection and aortic aneurysm in patients taking oral fluoroquinolone. JAMA Intern Med. 2015;175:1839–1847. https://doi.org/10.1001/jamainternmed.2015.5389
- [7] Yu X, Jiang DS, Wang J, et al. Fluoroquinolone use and the risk of collagen-associated adverse events: a systematic review and meta-analysis. Drug Saf. 2019;42:1025–1033. https://doi.org/10.1007/s40264-019-00828-z
- [8] Pasternak B, Inghammar M, Svanstrom H. Fluoroquinolone use and risk of aortic aneurysm and dissection: nationwide cohort study. BMJ. 2018;360:k678. https://doi.org/10.1136/bmj.k678
- [9] Wise BL, Peloquin C, Choi H, et al. Impact of age, sex, obesity, and steroid use on quinolone-associated tendon disorders. Am J Med. 2012;125:1228. https://doi.org/10.1016/j.amjmed.2012.05.027
- [10] Chang HN, Pang JH, Chen CP, et al. The effect of aging on migration, proliferation, and collagen expression of tenocytes in response to ciprofloxacin. J Orthop Res. 2012;30:764–768. https://doi.org/10.1002/jor.21576
- [11] Corps AN, Harrall RL, Curry VA, et al. Ciprofloxacin enhances the stimulation of matrix metalloproteinase 3 expression by interleukin-1beta in human tendon-derived cells. A potential mechanism of fluoroquinolone-induced tendinopathy. Arthritis Rheum. 2002;46:3034–3040. https://doi.org/10.1002/art.10617
- [12] Lundstrom KJ, Garmo H, Gedeborg R, et al. Short-term ciprofloxacin prophylaxis for prostate biopsy and risk of aortic aneurysm. Nationwide, population-based cohort study. Scand J Urol. 2021; 55:221–226.
- [13] Alves C, Mendes D, Marques FB. Fluoroquinolones and the risk of tendon injury: a systematic review and meta-analysis. Eur J Clin Pharmacol. 2019;75:1431–1443. https://doi.org/10.1007/s00228-019-02713-1
- [14] Claessen FM, de Vos RJ, Reijman M, et al. Predictors of primary Achilles tendon ruptures. Sports Med. 2014;44:1241–1259. https://doi.org/10.1007/s40279-014-0200-z
- [15] Adolfsson J, Garmo H, Varenhorst E, et al. Clinical characteristics and primary treatment of prostate cancer in Sweden between 1996 and 2005. Scand J Urol Nephrol. 2007;41:456–477. https://doi.org/10.1080/00365590701673625
- [16] Ludvigsson JF, Andersson E, Ekbom A, et al. External review and validation of the Swedish National Inpatient Register. BMC Public Health. 2011;11:450. https://doi.org/10.1186/1471-2458-11-450
- [17] Van Hemelrijck M, Wigertz A, Sandin F, et al. Cohort profile: the National Prostate Cancer Register of Sweden and Prostate Cancer data Base Sweden 2.0. Int J Epidemiol. 2013;42:956–967.
- [18] Everhov AH, Frisell T, Osooli M, et al. Diagnostic accuracy in the Swedish national patient register: a review including diagnoses in the outpatient register. Eur J Epidemiol. 2025;40:359–369. https://doi.org/10.1007/s10654-025-01221-0
- [19] Mohler JL, Armstrong AJ, Bahnson RR, et al. Prostate Cancer, Version 1.2016. J Natl Compr Canc Netw. 2016;14:19–30. https://doi.org/10.6004/jnccn.2016.0004
- [20] Patricia M, Grambsch TMT. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika. 1994;81:515–526. https://doi.org/10.1093/biomet/81.3.515
- [21] Suchak AA, Bostick G, Reid D, et al. The incidence of Achilles tendon ruptures in Edmonton, Canada. Foot Ankle Int. 2005;26:932–936. https://doi.org/10.1177/107110070502601106
- [22] Leppilahti J, Puranen J, Orava S. Incidence of Achilles tendon rupture. Acta Orthop Scand. 1996;67:277–279.
- [23] Sheth U, Wasserstein D, Jenkinson R, et al. The epidemiology and trends in management of acute Achilles tendon ruptures in Ontario, Canada: a population-based study of 27 607 patients. Bone Joint J. 2017;99-B:78–86. https://doi.org/10.1302/0301-620X.99B1.BJJ-2016-0434.R1
- [24] Lantto I, Heikkinen J, Flinkkila T, et al. Epidemiology of Achilles tendon ruptures: increasing incidence over a 33-year period. Scand J Med Sci Sports. 2015;25:133–138.
- [25] Huttunen TT, Kannus P, Rolf C, et al. Acute achilles tendon ruptures: incidence of injury and surgery in Sweden between 2001 and 2012. Am J Sports Med. 2014;42:2419–2423. https://doi.org/10.1177/0363546514540599
- [26] Daneman N, Lu H, Redelmeier DA. Fluoroquinolones and collagen associated severe adverse events: a longitudinal cohort study. BMJ Open. 2015;5:e010077.
- [27] Khaliq Y, Zhanel GG. Fluoroquinolone-associated tendinopathy: a critical review of the literature. Clin Infect Dis. 2003;36:1404–1410. https://doi.org/10.1086/375078
- [28] Persson R, Jick S. Clinical implications of the association between fluoroquinolones and tendon rupture: the magnitude of the effect with and without corticosteroids. Br J Clin Pharmacol. 2019;85:949–959. https://doi.org/10.1111/bcp.13879
- [29] van der Linden PD, Sturkenboom MC, Herings RM, et al. Increased risk of achilles tendon rupture with quinolone antibacterial use, especially in elderly patients taking oral corticosteroids. Arch Intern Med. 2003;163:1801–1807. https://doi.org/10.1001/archinte.163.15.1801
- [30] Baik S, Lau J, Huser V, et al. Association between tendon ruptures and use of fluoroquinolone, and other oral antibiotics: a 10-year retrospective study of 1 million US senior Medicare beneficiaries. BMJ Open. 2020;10:e034844. https://doi.org/10.1136/bmjopen-2019-034844