ORIGINAL RESEARCH ARTICLE
Uptake of doublet and triplet therapy for men with de novo metastatic castration- sensitive prostate cancer. Population-based study
Paolo Zauritoa,b, Rolf Gedeborga, Hans Garmoa, Eugenio Ventimigliaa, Mats Alhberga, Pär Stattina and Marcus Westerberga
aDepartment of Surgical Sciences, Uppsala University, Uppsala, Sweden; bDivision of Experimental Oncology/Unit of Urology, Urological Research Institute, IRCCS Ospedale San Raffaele, Milan, Italy
ABSTRACT
Purpose: In randomised clinical trials, doublet and triplet therapy improved survival compared to standard androgen deprivation therapy (ADT) in men with de novo metastatic castration-sensitive prostate cancer (mCSPC). Guidelines recommend doublet therapy since 2020 and triplet therapy since 2022. The aim of this study was to assess the uptake of upfront doublet and triplet therapy at a population level and assess trends in survival for all men with mCSPC.
Methods: We included men registered with de novo mCSPC in 2016–2024 in the National Prostate Cancer Register of Sweden. We estimated the annual proportion of men with de novo mCSPC who upfront received doublet therapy (ADT plus androgen receptor pathway inhibitor [ARPI] or docetaxel) or triplet therapy (ADT plus docetaxel and ARPI). Kaplan–Meier curves were used to estimate 3-year overall survival.
Results: In 9294 men diagnosed with de novo mCSPC, the use of upfront doublet therapy increased from 19% in 2016 to 66% in 2024, and the use of triplet therapy rose from 4% in 2021 to 17% in 2024. Uptake was highest among men below age 65 years, of whom 46% received doublet and 48% received triplet therapy in 2024. Three-year survival increased from 51% (95% CI: 49–52%) in 2016–2018 to 61% (95% CI: 58–64%) in 2022–2024. Among men below age 65, survival increased from 69% (95% CI: 65–73) in 2019–2021 to 77% (95% CI: 71–84) in 2022–2024.
Conclusions: The uptake of doublet and triplet therapy increased substantially during the study period, in particular among men below age 65. In parallel, 3-year overall survival increased in all men diagnosed with de novo mCSPC. These data provide support for the benefit of upfront doublet or triplet therapy in clinical practice.
KEYWORDS: metastatic prostate cancer; doublet therapy; triplet therapy; androgen deprivation therapy; chemotherapy; androgen receptor pathway inhibitor
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 51–57. https://doi.org/10.2340/sju.v61.45652.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 29 October 2025; Accepted: 17 February 2026; Published: 17 March 2026
CONTACT: Marcus Westerberg, PhD marcus.westerberg@uu.se Department of Surgical Sciences, Uppsala University, SE-752 37, Uppsala, Sweden
Supplemental data for this article can be accessed online at https://doi.org/10.2340/sju.v61.45652
Competing interests and funding: The authors report no conflicts of interest.
This project was supported by The Swedish Research Council (2022-00544), The Swedish Cancer Society [19 00 30], Region Uppsala and Uppsala University. The sponsors had no involvement with the planning, execution or completion of the study.
Introduction
Doublet and triplet therapy – i.e. combinations of androgen deprivation therapy (ADT) with an androgen receptor pathway inhibitor (ARPI) and/or docetaxel – has been shown to increase survival compared to ADT only in men with de novo metastatic castration-sensitive prostate cancer (mCSPC) in randomized clinical trials (RCTs) [1–7].
Doublet therapy with ADT plus docetaxel has been recommended in clinical guidelines since 2016, ADT plus ARPI since 2020 and triplet therapy since 2022 [8–11]. Following these recommendations, the uptake of doublet therapy has increased in clinical practice in the United States and in Europe [12–14]. The increase in use of doublet therapy in 2017–2020 was paralleled by increased survival in all men with mCSPC in our previous study [12].
The aim of this study was to update our previous study of doublet therapy and to assess the uptake of triplet therapy among men with de novo mCSPC in Sweden and to describe time trends in survival.
Materials and methods
Data sources
The National Prostate Cancer Register (NPCR) of Sweden is a clinical cancer register with the aim to register data to ensure high-quality care for men with prostate cancer and adherence to national guidelines [15, 16]. NPCR captures virtually all men (> 98%) diagnosed with prostate cancer in Sweden compared to the Cancer Register, to which registration is mandated by law [17, 18]. In Prostate Cancer data Base Sweden (PCBase), NPCR has been linked to other health care registers and demographic databases, including The Patient Register, The Prescribed Drug Register and The Cause of Death Register [19]. In 2025, we enriched PCBase with data from health care Information Technology (IT) systems in all regions in Sweden, creating PCBase Xtend that holds longitudinal information on prostate-specific antigen (PSA), prostate biopsies, magnetic resonance imaging (MRI) of the prostate, ADT administered by healthcare providers and chemotherapy [20]. The Swedish Ethical Review Authority approved of this project. The study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline.
Study population
The study population consisted of men diagnosed with de novo mCSPC from 2016 to 2024 and registered in NPCR. The diagnosis was defined by the presence of skeletal or visceral metastases on radionuclide bone scan, computed tomography (CT), positron emission tomography (PET)/CT, or MRI of the skeleton. Data on emigration from Sweden were extracted from The Emigration Register.
Characterisation of prostate cancer at baseline
We extracted information on age, PSA at diagnosis, clinical T stage, Gleason score, number of bone metastases, presence of visceral metastases and use of PET/CT from NPCR.
Treatment
We extracted information on treatment with docetaxel within 6 months after diagnosis from NPCR. This information is available since March 2017. NPCR captured 70% of upfront docetaxel treatments after a diagnosis of mCSPC in a validation study of 500 men diagnosed in 2018–2022 [21]. We complemented this information with data on the use of docetaxel from regional health care IT systems (Supplementary Table 1). Slightly different time periods were covered in the regions since the implementation of new IT systems occurred at different calendar times, the extent of migration of data to new IT systems varied and because the regions delivered data at different calendar times to this project. The proportion of men covered by this complementary data collection was 71% in 2016, 90% in 2020–2021 and 13% in 2024 (Supplementary Table 2).
Use of ARPI was based on filled prescriptions for abiraterone (Anatomical Therapeutic Chemical code L02BX03), enzalutamide (L02BB04), apalutamide (L02BB05) and darolutamide (L02BB06) in the Prescribed Drug Register.
The start of treatment of an ARPI was defined at the date of a first filling of an ARPI prescription in the Prescribed Drug Register. The start of treatment with docetaxel was the date recorded in NPCR or in a regional health care IT system. Doublet therapy was defined as the initiation of an ARPI or docetaxel within 6 months from diagnosis, and triplet therapy (i.e. ADT, ARPI and docetaxel) was defined as the use of both an ARPI and docetaxel within 6 months from diagnosis.
Life expectancy
Health-adjusted life expectancy was calculated based on age and comorbidity [22]. Baseline comorbidity was quantified based on the combined information from a Multidimensional Diagnosis-based Comorbidity Index (MDCI) [23, 24] and a Drug Comorbidity Index (DCI) [25–27].
Mortality
Follow-up started at the date of diagnosis and ended on December 31, 2024 or at the date of death or migration out of Sweden, whichever event came first. The date of death was extracted from The Cause of Death Register.
Statistical analysis
We computed the annual proportion of men with de novo mCSPC who received doublet or triplet therapy. Crude overall survival was estimated by use of Kaplan–Meier analysis in three calendar periods (2016–2018, 2019–2021 and 2022–2024) and according to age at diagnosis (below 65, 65–79 and above 80 years). Median follow-up was estimated using a reverse Kaplan–Meier approach [28, 29].
To account for potential changes in baseline characteristics over time, we also estimated standardised survival curves using a parametric survival model adjusted for age, PSA, Gleason score, clinical T stage and comorbidity (DCI and MDCI) (see Supplementary Materials for details) [30]. All analyses were performed using R version 4.3.2.
Results
Characteristics of the study population
Between 2016 and 2024, 9294 men were diagnosed with de novo mCSPC, evenly distributed across three calendar periods (Table 1). Baseline characteristics remained stable over time, with a median age at diagnosis of 75 years (IQR 69–80) and virtually no changes in the distribution of clinical T stage or Gleason score. Median PSA at diagnosis decreased slightly from 104 ng/mL (IQR: 28–434) in 2016–2018 to 89 ng/mL (IQR: 25–354) in 2022–2024. PET/CT was used in 231 (8%) men in 2016–2018 and 516 (16%) men in 2022–2024.
Uptake of combination therapy
Use of upfront doublet or triplet therapy increased from 19% in 2016 to 66% in 2024 (Figure 1). Doublet therapy increased slightly from 45% in 2020 to 49% in 2024, with 48% receiving ADT + ARPI and only 1% receiving ADT + docetaxel. Triplet therapy reached 17% in 2024.
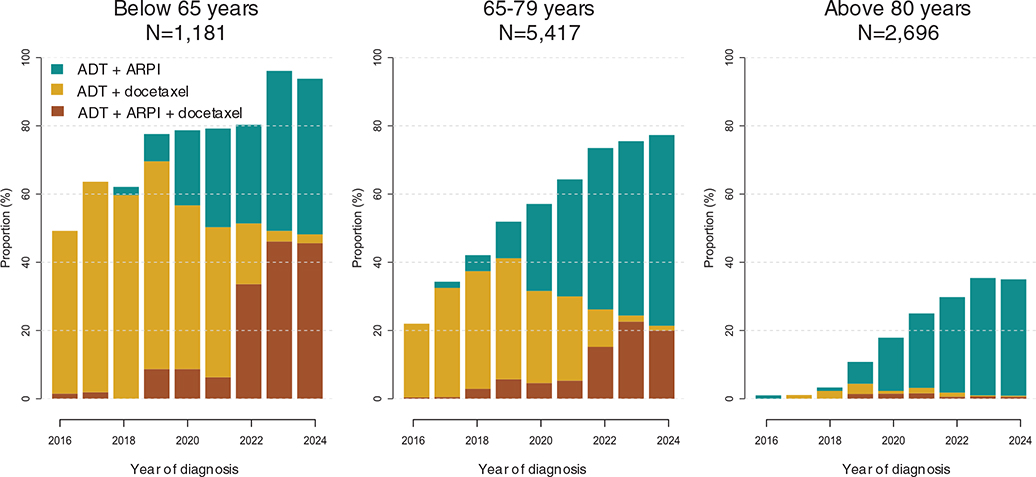
Figure 1. Uptake of doublet and triplet therapy in men with de novo metastatic castration-sensitive prostate cancer, 2016–2024.
In relation to age, doublet or triplet therapy was used in 94% of men below age 65 in 2024, 77% among men aged 65–79 and 35% among men above age 80.
Characteristics of men treated with triplet therapy
Men treated with triplet therapy were younger and had a more advanced disease, reflected by high PSA levels, clinical T stage and Gleason score (Table 2). In the oldest age group, less than 1% received triplet therapy.
Survival
Survival improved in all age groups during the entire study period (Figure 2, Supplementary Table 3). Among men < 65 years, 3-year survival increased from 69% (95% CI: 65–73) in 2019–2021 to 77% (95% CI: 71–84) in 2022–2024 and for men ≥ 80 years, survival increased from 40% (95% CI: 37–43) to 46% (95% CI: 40–52), whereas no increase was observed in men aged 65–79. Standardised survival curves showed the same patterns (Supplementary Figure 1, Supplementary Table 3).
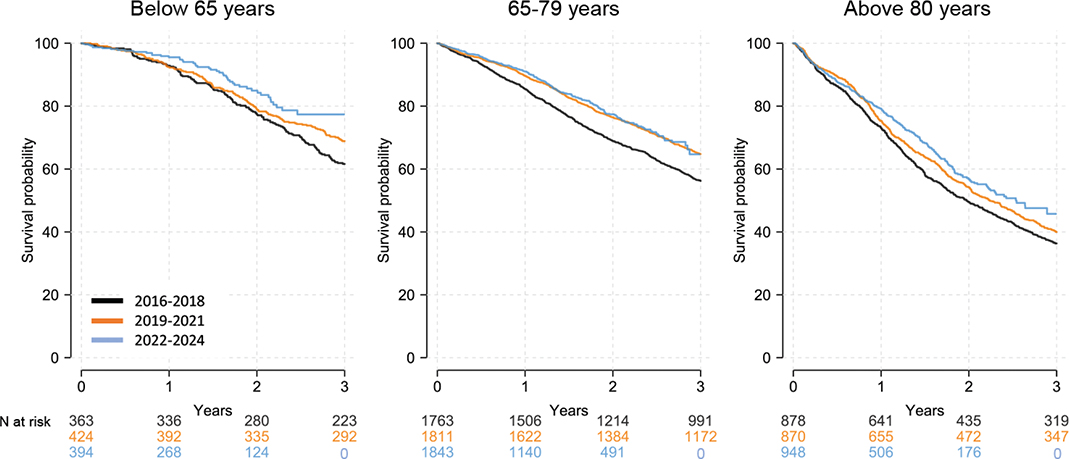
Figure 2. Standardised 3-year overall survival for men with de novo metastatic castration-sensitive prostate cancer diagnosed in 2016–2024. A total of 4687 (51%) men died for any cause during a median follow-up of 4.2 years (IQR: 2.2–6.3).
Discussion
Summary of findings
In this nationwide population-based study, use of upfront doublet or triplet therapy in men with de novo mCSPC increased substantially between 2016 and 2024. In 2024, approximately half of men received doublet therapy, and one fifth received triplet therapy. Triplet therapy essentially replaced ADT + docetaxel in the most recent years. The uptake of these therapies was particularly high in men below age 65. In 2024, nearly all these men received doublet or triplet therapy. The increased use of doublet and triplet therapy was paralleled by an increase in 3-year survival in all men with mCSPC.
Strength and limitations
This study has several strengths. NPCR captures nearly all men diagnosed with prostate cancer in Sweden and has been linked to other high-quality nationwide registers and health care IT systems in PCBase Xtend, providing comprehensive data on cancer characteristics, time-updated information on treatments, comorbidities and follow-up [15, 18, 31–33].
Some limitations should be noted. We did not have full coverage of upfront docetaxel during the study period, despite that we had access to data both from NPCR and regional health care IT systems. Use was particularly underestimated in 2023–2024. Men diagnosed before 2022 who received ADT, docetaxel and an ARPI within 6 months from diagnosis likely received docetaxel and ARPI in sequence.
Previous studies and interpretation
To our knowledge, this is the first population-based study to assess uptake of triplet therapy after its inclusion in clinical guidelines [8–10]. In a previous study with a similar design, we found an increased use of doublet therapy in Sweden up to 2020 [12]. Similar increases were also documented in the United States and Europe, with treatment intensification associated with improved survival [13, 14]. In this study, we observed a continued increase of doublet therapy – primarily ADT + ARPI – and triplet therapy largely replaced the use of ADT plus docetaxel.
Men who received triplet therapy were young and had disseminated disease. This is in accordance with Swedish guidelines that recommend triplet therapy for young and fit men with advanced disease, i.e. men with high-volume metastatic disease [10, 34].
RCTs have shown increased survival in men who received triplet therapy. For example, The PEACE-1 trial reported a 25% reduction in mortality at 4 years with ADT + docetaxel + abiraterone compared to ADT + docetaxel [5]. The ARASENS trial showed a 32% improvement at 3 years with ADT + docetaxel + darolutamide compared to ADT + docetaxel [6]. Participants in these trials were younger (median age 66–67) and had lower PSA levels (14–27 ng/mL) than men in our study (median age 75; PSA 89 ng/mL in 2022–2024). Similar differences apply to RCTs comparing doublet therapy with ADT only [2, 3].
The use of PET/CT doubled from 8 to 16% between 2016–2018 and 2022–2024. This may have led to an increase in men diagnosed with lower-volume metastatic prostate cancer since PET/CT has a higher sensitivity compared to conventional imaging [35]. Although tumor characteristics were stable over time and adjusted analyses showed similar improvements in survival as unadjusted analyses, this change in imaging modalities may still have contributed somewhat to the higher observed survival in the last calendar period (Will-Roger’s phenomenon). Nevertheless, we argue that the increased 3-year survival in men with de novo mCSPC observed during the study period is at least partly attributable to increased use of doublet and triplet therapy. In men below age 65, there was a gradual improvement between the three time periods, whereas for men aged 65–79 years, this improvement was only apparent between the two earliest time periods.
Conclusion
The uptake of doublet and triplet therapy among men with de novo mCSPC increased substantially from 2016 to 2024 in Sweden. In 2024, triplet therapy had largely replaced ADT + docetaxel, and use of ADT + ARPI continued to increase during the study period. The increased use of doublet and triplet therapy was paralleled by an increased 3-year survival. These data provide support for the benefit of upfront doublet or triplet therapy in clinical practice.
Acknowledgements
This project was made possible by the continuous work of the National Prostate Cancer Register of Sweden (NPCR) steering group: Elin Axén, Johan Styrke, Andreas Josefsson, Camilla Thellenberg, Hampus Nugin, Ingrida Verbiené, Stefan Carlsson, Anna Kristiansen, Mats Andén, Kimia Kohestani, Jon Kindblom, Thomas Jiborn, Olof Ståhl, Olof Akre, Eva Johansson, Magnus Törnblom, Fredrik Jäderling, Marie Hjälm-Eriksson, Lotta Renström Koskela, Erik Thimansson, Johan Stranne, Elin Trägårdh, Viktoria Gaspar, Fredrik Sandin, Petrus Stenson, Lena Pettersson, Mia Brus, Gustaf Hedström, Anna Hedström, Maria Moutran, Nina Hageman and Maria Nyberg and patient representatives Hans Joelsson and Gert Malmberg.
References
[1] Sathianathen NJ, Koschel S, Thangasamy IA, et al. Indirect comparisons of efficacy between combination approaches in metastatic hormone-sensitive prostate cancer: a systematic review and network meta-analysis. Eur Urol. 2020;77(3):365–372. https://doi.org/10.1016/j.eururo.2019.09.004
[2] James ND, de Bono JS, Spears MR, et al. Abiraterone for prostate cancer not previously treated with hormone therapy. N Engl J Med. 2017;377(4):338–351. https://doi.org/10.1056/NEJMoa1702900
[3] Fizazi K, Tran N, Fein L, et al. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N Engl J Med. 2017;377(4):352–360. https://doi.org/10.1056/NEJMoa1704174
[4] Vale CL, Burdett S, Rydzewska LHM, et al. Addition of docetaxel or bisphosphonates to standard of care in men with localised or metastatic, hormone-sensitive prostate cancer: a systematic review and meta-analyses of aggregate data. Lancet Oncol. 2016;17(2):243–256. https://doi.org/10.1016/S1470-2045(15)00489-1
[5] Fizazi K, Foulon S, Carles J, et al. Abiraterone plus prednisone added to androgen deprivation therapy and docetaxel in de novo metastatic castration-sensitive prostate cancer (PEACE-1): a multicentre, open-label, randomised, phase 3 study with a 2 × 2 factorial design. Lancet. 2022;399(10336):1695–1707. https://doi.org/10.1016/S0140-6736(22)00367-1
[6] Smith MR, Hussain M, Saad F, et al. Darolutamide and survival in metastatic, hormone-sensitive prostate cancer. N Engl J Med. 2022;386(12):1132–1142. https://doi.org/10.1056/NEJMoa2119115
[7] Jian T, Zhan Y, Hu K, et al. Systemic triplet therapy for metastatic hormone-sensitive prostate cancer: a systematic review and network meta-analysis. Front Pharmacol. 2022;13:955925. https://doi.org/10.3389/fphar.2022.955925
[8] Schaeffer EM, Srinivas S, Adra N, et al. NCCN guidelines® insights: prostate cancer, version 3.2024. J Natl Compr Canc Netw. 2024;22(3):140–150.
[9] Tilki D, van den Bergh RCN, Briers E, et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG guidelines on prostate cancer. Part II – 2024 update: treatment of relapsing and metastatic prostate cancer. Eur Urol. 2024;86(2):164–182. https://doi.org/10.1016/j.eururo.2024.04.010
[10] Bratt O, Carlsson S, Fransson P, et al. The Swedish national guidelines on prostate cancer, part 2: recurrent, metastatic and castration resistant disease. Scand J Urol. 2022;56(4):278–284. https://doi.org/10.1080/21681805.2022.2093396
[11] Cornford P, Bellmunt J, Bolla M, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part II: treatment of relapsing, metastatic, and castration-resistant prostate cancer. Eur Urol. 2017;71(4):630–642. https://doi.org/10.1016/j.eururo.2016.08.002
[12] Corsini C, Garmo H, Orrason AW, et al. Survival trend in individuals with de novo metastatic prostate cancer after the introduction of doublet therapy. JAMA Netw Open. 2023;6(10):e2336604. https://doi.org/10.1001/jamanetworkopen.2023.36604
[13] Schoen MW, Doherty J, Eaton D, et al. Treatment patterns and survival among veterans with de novo metastatic hormone-sensitive prostate cancer. JAMA Netw Open. 2025;8(5):e259433. https://doi.org/10.1001/jamanetworkopen.2025.9433
[14] Raval AD, Chen S, Littleton N, et al. Real-world use of androgen-deprivation therapy intensification for metastatic hormone-sensitive prostate cancer: a systematic review. BJU Int. 2025;135(3):408–421. https://doi.org/10.1111/bju.16577
[15] Stattin P. How to improve cancer care by use of guidelines and quality registers. Scand J Urol. 2024;59:190–192. https://doi.org/10.2340/sju.v59.42272
[16] Stranne J. 2023/2024 update of the national prostate cancer guidelines in Sweden. Scand J Urol. 2024;59:210–211. https://doi.org/10.2340/sju.v59.42656
[17] Tomic K, Berglund A, Robinson D, et al. Capture rate and representativity of The National Prostate Cancer Register of Sweden. Acta Oncol. 2015;54(2):158–163. https://doi.org/10.3109/0284186X.2014.939299
[18] Tomic K, Sandin F, Wigertz A, et al. Evaluation of data quality in the National Prostate Cancer Register of Sweden. Eur J Cancer. 2015;51(1):101–111. https://doi.org/10.1016/j.ejca.2014.10.025
[19] Van Hemelrijck M, Garmo H, Wigertz A, et al. Cohort profile update: The National Prostate Cancer Register of Sweden and prostate cancer data base – a refined prostate cancer trajectory. Int J Epidemiol. 2016;45(1):73–82. https://doi.org/10.1093/ije/dyv305
[20] Westerberg M, Holm L, Garmo H, et al. Cohort profile update: the National Prostate Cancer Register of Sweden and PCBase. Int J Epidemiol. 2025;54(5):dyaf172. https://doi.org/10.1093/ije/dyaf172
[21] Gedeborg R, Sandin F, Thellenberg-Karlsson C, et al. Uptake of doublet therapy for de novo metastatic castration sensitive prostate cancer: a population-based drug utilisation study in Sweden. Scand J Urol. 2023;58:9578. https://doi.org/10.2340/sju.v58.9572
[22] Ventimiglia E, Gedeborg R, Orrason AW, et al. A comparison of comorbidity indices and estimates of life expectancy for men with prostate cancer. Scand J Urol. 2025;60:105–107. https://doi.org/10.2340/sju.v60.43810
[23] Westerberg M, Irenaeus S, Garmo H, et al. Development and validation of a multi-dimensional diagnosis-based comorbidity index that improves prediction of death in men with prostate cancer: nationwide, population-based register study. PLoS One. 2024;19(1):e0296804. https://doi.org/10.1371/journal.pone.0296804
[24] Westerberg M, Garmo H, Robinson D, et al. Target trial emulation using new comorbidity indices provided risk estimates comparable to a randomized trial. J Clin Epidemiol. 2024;174:111504. https://doi.org/10.1016/j.jclinepi.2024.111504
[25] Gedeborg R, Sund M, Lambe M, et al. An aggregated comorbidity measure based on history of filled drug prescriptions: development and evaluation in two separate cohorts. Epidemiology. 2021;32(4): 607–615. https://doi.org/10.1097/EDE.0000000000001358
[26] Fallara G, Gedeborg R, Bill-Axelson A, et al. A drug comorbidity index to predict mortality in men with castration resistant prostate cancer. PLoS One. 2021;16(7):e0255239. https://doi.org/10.1371/journal.pone.0255239
[27] Gedeborg R, Garmo H, Robinson D, et al. Prescription-based prediction of baseline mortality risk among older men. PLoS One. 2020;15(10):e0241439. https://doi.org/10.1371/journal.pone.0241439
[28] Schemper M, Smith TL. A note on quantifying follow-up in studies of failure time. Control Clin Trials. 1996;17(4):343–346. https://doi.org/10.1016/0197-2456(96)00075-X
[29] Shuster JJ. Median follow-up in clinical trials. J Clin Oncol. 1991;9(1):191–192. https://doi.org/10.1200/JCO.1991.9.1.191
[30] Jackson CH. flexsurv: a platform for parametric survival modeling in R. J Stat Softw. 2016;70:i08. https://doi.org/10.18637/jss.v070.i08
[31] Ludvigsson JF, Andersson E, Ekbom A, et al. External review and validation of the Swedish national inpatient register. BMC Public Health. 2011;11:450. https://doi.org/10.1186/1471-2458-11-450
[32] Godtman R, Holmberg E, Stranne J, et al. High accuracy of Swedish death certificates in men participating in screening for prostate cancer: a comparative study of official death certificates with a cause of death committee using a standardized algorithm. Scand J Urol Nephrol. 2011;45(4):226–232. https://doi.org/10.3109/00365599.2011.559950
[33] Barlow L, Westergren K, Holmberg L, et al. The completeness of the Swedish Cancer Register: a sample survey for year 1998. Acta Oncol. 2009;48(1):27–33. https://doi.org/10.1080/02841860802247664
[34] Hussain M, Fizazi K, Shore ND, et al. Metastatic hormone-sensitive prostate cancer and combination treatment outcomes: a review. JAMA Oncol. 2024;10(6):807–820. https://doi.org/10.1001/jamaoncol.2024.0591
[35] Hofman MS, Lawrentschuk N, Francis RJ, et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet. 2020;395(10231): 1208–1216. https://doi.org/10.1016/S0140-6736(20)30314-7