ORIGINAL RESEARCH ARTICLE
Early recurrence after primary TURBT for non-muscle-invasive bladder cancer in Iceland: the critical role of surgical quality
Oddur Björnssona,b, Guðmundur Geirssonc, Árni Stefán Leifssonc, Sigfús Þór Nikulássond and Sigurður Guðjónssona,c
aFaculty of Medicine, University of Iceland, Reykjavík, Iceland; bDepartment of Urology, Sahlgrenska University Hospital, Gothenburg, Sweden; cDepartment of Urology, Landspitali University Hospital, Reykjavik, Iceland; dDepartment of Pathology, Landspitali University Hospital, Reykjavik, Iceland
ABSTRACT
Introduction: Transurethral resection of bladder tumor (TURBT) is the standard treatment for non-muscle-invasive bladder cancer (NMIBC), but early recurrences remain frequent. The objective of this study was to standardize TURBT management at our institution through the implementation of a treatment protocol designed to reduce early recurrence after primary TURBT.
Material and methods: All patients with newly diagnosed NMIBC who underwent primary TURBT at Landspítali University Hospital between 2013–2015 (control) and 2017–2019 (intervention) were included. The treatment protocol restricted procedures to four surgeons, mandated blue-light cystoscopy, routine postoperative bladder irrigation, and guideline-based instillation therapy. The primary endpoint was recurrence rate at first follow-up cystoscopy; secondary endpoints included adherence to protocol measures and recurrence-free survival.
Results: A total of 133 control and 138 intervention patients were included. Baseline characteristics were comparable. After implementation, bladder irrigation increased from 46 to 90%, blue-light cystoscopy was used in 59% of intervention cases, and instillation therapy rates rose (Mitomycin-C: 0–49% in intermediate-risk; Bacillus Calmette-Guérin (BCG):35–63% in high-risk patients). Early recurrence rates remained comparable (15% vs. 16%). Early recurrence varied markedly between surgeons (11–40%) and correlated strongly with detrusor muscle presence. No significant difference in recurrence-free survival was observed, though a trend toward lower recurrence was seen in the intervention group after adjusting for risk factors.
Conclusion: Implementation of a standardized TURBT protocol improved adherence to recommended measures but did not reduce early recurrence. Marked inter-surgeon variability highlights surgical quality as the key determinant of early recurrence in non-muscle-invasive bladder cancer.
KEYWORDS: nonmuscle invasive bladder cancer; early recurrence; surgical quality
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 131–137. https://doi.org/10.2340/sju.v61.45711.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 24 November 2025; Accepted: 16 February 2026; Published: 17 April 2026
CONTACT: Oddur Björnsson oddur.bjornsson@vgregion.se Department of Urology, Sahlgrenska University Hospital, Bruna Stråket 11B, 413 46, Gothenburg, Sweden
Competing interests and funding: The authors report no conflict of interest.
Introduction
Transurethral resection of bladder tumor (TURBT) is the standard treatment for non-muscle-invasive bladder cancer (NMIBC). The disease is characterized by a high risk of recurrence, often detected at the first follow-up cystoscopy. Clinical guidelines recommend several adjunctive measures to TURBT to reduce the risk of recurrence after surgery, such as adjuvant intravesical instillation therapy in selected patients [1]. Other measures shown to decrease recurrence rates after TURBT include the use of blue-light cystoscopy, postoperative continuous bladder irrigation, and improved surgical quality through the implementation of quality performance indicators [2–7]. Studies have also demonstrated that improving perioperative management and establishing standardized treatment protocols for TURBT can significantly reduce early recurrence rates [8, 9].
The aim of this study was to standardize treatment for newly diagnosed NMIBC at our institution through the introduction of a structured protocol for primary TURBT, with the goal of reducing early recurrence rates after surgery.
Material and methods
Study design and patients
This was a cohort study of all patients with newly diagnosed NMIBC who underwent primary TURBT at Landspítali University Hospital, Reykjavík. The control group consisted of patients treated between January 2013 and December 2015, and the intervention group comprised those treated between January 2017 and December 2019. The treatment protocol was introduced in 2016; patients treated during that year were not included. Clinical data were obtained retrospectively for the control group and prospectively for the intervention group.
Treatment protocol
The standardized treatment protocol implemented in 2016 included:
Restricting primary TURBTs to four designated surgeons.
Routine use of hexaminolevulinate (Hexvix) blue-light cystoscopy.
Postoperative continuous bladder irrigation with saline for ≥ 4 hours.
Guideline-based adjuvant intravesical instillation:
Low risk: No adjuvant instillation.
Intermediate risk: Mitomycin-C weekly × 6.
High risk: BCG induction and maintenance instillation therapy.
Mandatory re-resection for T1 tumors within 6–8 weeks.
Pathological examination
All specimens were retrospectively reviewed by an experienced pathologist and staged according to the 2017 TNM classification and graded by both the WHO 1973 and the WHO 2004/2016 systems. The presence of detrusor muscle was recorded.
Endpoints
The primary endpoint was early recurrence, defined as tumor recurrence at first follow-up cystoscopy (or after re-resection, if performed). Secondary endpoints included adherence to protocol measures and recurrence-free survival.
Statistical analysis
Categorical variables were compared using Fisher’s exact and chi-squared tests. Recurrence-free survival was analyzed with the Kaplan–Meier method and multivariable Cox regression analysis to estimate hazard ratios (HRs). Statistical significance was defined as p < 0.05. Analyses were performed using R software.
Results
Patient characteristics
A total of 271 patients were included in the study, with 133 patients in the control group (2013–2015) and 138 in the intervention group (2017–2019). Baseline characteristics are summarized in Table 1. The two groups were broadly comparable with respect to age, gender distribution, and tumor stage and grade. A modest imbalance was observed in tumor multiplicity: multiple tumors were more frequent in the intervention group (38% vs. 23%), while solitary tumors were correspondingly more common in the control cohort. The distribution of tumor grade and stage did not differ significantly between groups.
| Variable | Control group (2013–2015) | Intervention group (2017–2019) | P-value |
| No. of patients | 133 | 138 | 0.11 |
| Male | 114 (86%) | 107 (78%) | |
| Female | 19 (14%) | 31 (22%) | |
| Age (years) | 0.43 | ||
| Median (IQR) | 71 (64–79) | 69 (62–77) | |
| Presenting symptomsa | 0.08 | ||
| Hematuria | 110 (83%) | 101 (73%) | |
| LUTS | 19 (14%) | 13 (9%) | |
| Other symptoms | 1 (1%) | 2 (1%) | |
| Incidental | 10 (8%) | 22 (16%) | |
| Tumor stage (pT) | 0.73 | ||
| Ta | 97 (73%) | 106 (77%) | |
| Tis | 4 (3%) | 3 (2%) | |
| T1 | 32 (24%) | 29 (21%) | |
| Ta/T1 tumors with concomitant CIS | 8 | 11 | |
| Tumor grade (WHO 1973) | 0.19 | ||
| G1 | 44 (33%) | 32 (23%) | |
| G2 | 40 (30%) | 48 (35%) | |
| G3 | 49 (37%) | 58 (42%) | |
| Tumor grade (WHO 2004/2016) | 0.66 | ||
| PUNLMP | 1 (1%) | 1 (1%) | |
| Low grade | 63 (47%) | 57 (41%) | |
| High grade | 69 (52%) | 80 (58%) | |
| Number of tumors | 0.02* | ||
| 1 | 103 (77%) | 85 (62%) | |
| 2 | 15 (11%) | 28 (20%) | |
| ≥ 3 | 15 (11%) | 24 (17%) | |
| NA | 0 (0%) | 1 (1%) | |
| Size of tumor | <0.01* | ||
| < 3 cm | 40 (30%) | 32 (23%) | |
| ≥ 3 cm | 20 (15%) | 70 (51%) | |
| NA | 73 (55%) | 36 (26%) | |
| Patients are stratified by study groups. Data are shown as number of patients (%) unless otherwise indicated. NMIBC: non-muscle-invasive bladder cancer; TURBT: transurethral resection of bladder tumor; IQR: Interquartile range; LUTS: Lower urinary tract symptoms; CIS: carcinoma in situ; PUNLMP: Papillary urothelial neoplasm of low malignant potential. aPatients presenting with more than one symptom are counted more than once. *Indicates statistical significance. |
|||
Implementation of the standardized protocol
As Table 2 illustrates, after the introduction of the standardized protocol, adherence to recommended perioperative measures improved. Blue-light cystoscopy was used in 59% of cases in the intervention group, whereas it had not been available during the earlier period. Postoperative continuous bladder irrigation was also more frequently applied (90% vs. 46%). Guideline-based adjuvant therapy increased notably: mitomycin-C was administered to 49% of intermediate-risk patients in the intervention group compared with none in the control period, and BCG instillation was given to 63% of high-risk patients compared with 35% previously. In the intervention period, 79% of primary TURBTs were performed by the four designated surgeons, compared with a much more heterogeneous distribution in the control group. Despite these improvements, the proportion of resections with detrusor muscle present in the pathological specimen did not increase significantly between the two periods (Table 2).
| Variable | Control group (2013–2015) | Intervention group (2017–2019) | P-value |
| Total number of surgeons | 10 | 11 | |
| Number of surgeons performing over 10 operations | 8 | 4 | |
| Detrusor muscle in the specimen | 98 (74%) | 93 (67%) | 0.31 |
| T1 tumors only | 25/32 (78%) | 20/29 (69%) | 0.6 |
| Blue light cystoscopy (Hexvix) | 0 (0%) | 81 (59%) | < 0.01* |
| Postoperative continuous bladder irrigation | 61 (46%) | 122/136 (90%) | < 0.01* |
| Re-resection within 6–8 weeks of primary TURBT | |||
| T1 tumors | 21/32 (66%) | 19/29 (66%) | 1 |
| TaG3 | 5/24 (21%) | 6/30 (20%) | 0.95 |
| Adjuvant BCG instillation | |||
| T1 tumors | 14/32 (44%) | 20/29 (69%) | 0.1 |
| High-risk tumors | 21/60 (35%) | 40/63 (63%) | < 0.01* |
| Adjuvant Mitomycin instillation | |||
| Intermediate-risk tumors | 0/38 (0%) | 24/49 (49%) | < 0.01* |
| High-risk tumors | 0/56 (0%) | 3/62 (5%) | 0.04 |
| Recurrence at first follow-up cystoscopy | 18/122 (15%) | 21/128 (16%) | 0.85 |
| Data are shown as the number of patients (%) unless otherwise indicated. TURBT: transurethral resection of bladder tumor. *Indicates statistical significance. |
|||
Early recurrence rate
At the first follow-up cystoscopy, early recurrence was observed in 15% of patients in the control group and 16% in the intervention group, a difference that was not statistically significant. Importantly, recurrence rates displayed marked heterogeneity between surgeons, especially in the intervention period, ranging from 11 to 40%. A higher early recurrence rate was strongly correlated with the absence of detrusor muscle in the resection specimen (Table 3).
| Variable | Control group (2013–2015) | ||||||
| Urologist nr. 1 | Urologist nr. 2 | Urologist nr. 3 | Urologist nr. 4 | Other urologists | Total | P-value | |
| Number of operations performed | 0 | 20 | 19 | 10 | 84 | 133 | |
| Multifocal tumors | - | 5/20 = 25% | 3/19 =16% | 1/10 = 10% | 21/84 = 25% | 30 (23%) | 0.71 |
| High risk tumors | - | 8/20 = 40% | 5/19 = 26% | 3/10 = 30% | 40/84 = 48% | 56 (42%) | 0.61 |
| Detrusor in specimen | - | 7/20 = 35%* | 15/19 = 79% | 7/10 = 70% | 69/84 = 82% | 98 (74%) | < 0.01* |
| Recurrence at first follow-up cystoscopy** | - | 4/18 = 22% | 2/18 = 11% | 2/10 = 20% | 10/76 = 13% | 18/122 = 15% | 0.67 |
| Recurrence at first follow-up cystoscopy or residual tumor at second look TURBT** | - | 5/18 = 28% | 4/18 = 22% | 3/10 = 30% | 16/76 = 21% | 28/122 = 23% | 0.82 |
| Variable | Intervention group (2017–2019) | ||||||
| Urologist nr. 1 | Urologist nr. 2 | Urologist nr. 3 | Urologist nr. 4 | Other urologists | Total | P-value | |
| Number of operations performed | 53 | 21 | 21 | 14 | 29 | 138 | |
| Multifocal tumors | 23/53 = 43% | 6/21 = 29% | 7/21 = 33% | 3/14 = 21% | 13/29 = 45% | 52 (38%) | 0.43 |
| High-risk tumors | 27/53 = 51% | 11/21 = 52% | 9/21 = 43% | 8/14 = 57% | 8/29 = 28% | 62 (45%) | 0.28 |
| Use of Hexvix blue-light cystoscopy | 47/53 = 89% | 3/21 = 14%* | 9/21 = 43% | 10/14 = 71% | 12/29 = 41% | 81 (59%) | < 0.01* |
| Use of postoperative continuous bladder irrigation | 53/53 = 100%* | 16/21 = 76% | 17/21 = 81% | 13/14 = 93% | 23/29 = 79% | 122 (88%) | < 0.01* |
| Use of BCG instillation for high-risk tumors | 16/27 = 59% | 7/11 = 63% | 8/9 = 89% | 5/8 = 63% | 4/8 = 50% | 40/62 = 65% | 0.50 |
| Detrusor in specimen | 41/53 = 77% | 4/21 = 19%* | 18/21 = 86% | 8/14 = 57% | 22/29 = 76% | 93 (67%) | < 0.01* |
| Recurrence at first follow-up cystoscpy** | 5/47 = 11% | 8/20 = 40%* | 3/21 = 14% | 3/14 = 21% | 2/26 = 8% | 21/128 = 16% | < 0.01* |
| Recurrence at first follow-up cystoscopy or residual tumor at re-resection** | 7/47 = 15% | 9/20 = 45%* | 4/21 = 19% | 6/14 = 43% | 3/26 = 12% | 31/128 = 24% | < 0.01* |
| Data are shown as number of patients (%) unless otherwise indicated. *Indicates statistical significance, found with pairwise comparison. **Patients who had no follow-up cystoscopy were excluded. |
|||||||
Recurrence-free survival
Median follow-up was 34.3 months in the control group and 36.8 months in the intervention group. Kaplan–Meier analysis revealed no significant difference in recurrence-free survival between the two groups (Figure 1). At 1 year, recurrence-free survival was 71% in the control group and 74% in the intervention group. When all patients were analyzed together, 1-year recurrence-free survival varied significantly between surgeons, ranging from 51 to 83% (p < 0.001) (Figure 2). No statistical difference was found in progression to muscle-invasive disease between the study groups during follow-up. Five year progresson-free survival in the control group and intervention group was 87 and 95%, respectively (p = 0.1).
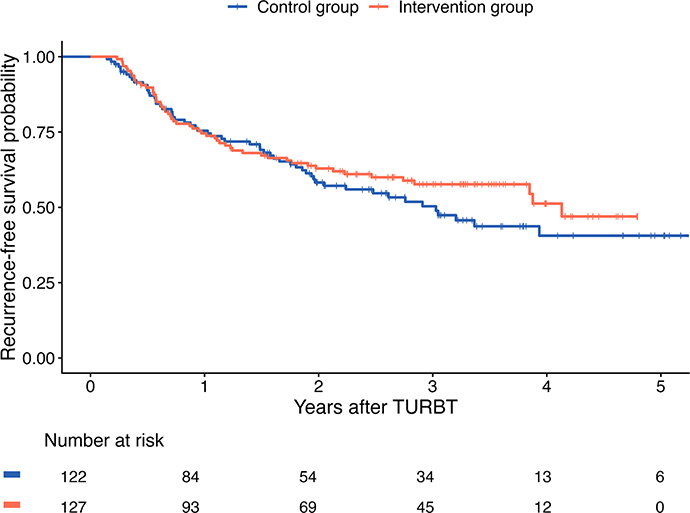
Figure 1. Kaplan–Meier plot of recurrence-free survival in patients who underwent primary TURBT for newly diagnosed NMIBC during the study period, stratified by study group. NMIBC: non-muscle-invasive bladder cancer; TURBT: transurethral resection of bladder tumor.
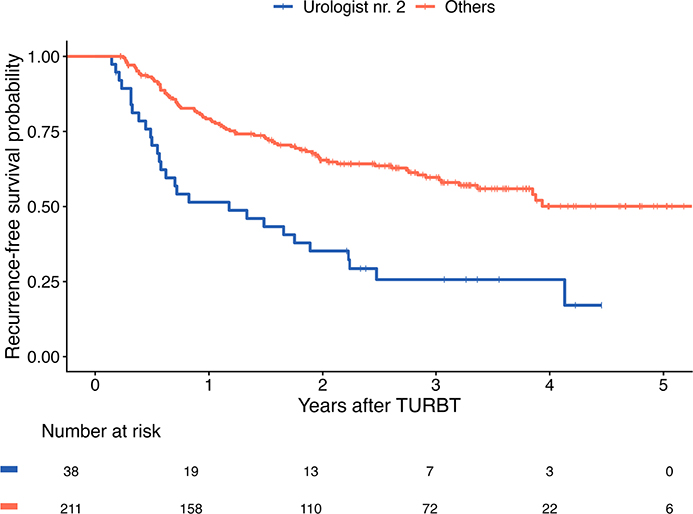
Figure 2. Kaplan–Meier plot of recurrence-free survival in patients who underwent primary TURBT for newly diagnosed NMIBC during the study period when combining the control and intervention groups and stratified by operating surgeon. NMIBC: non-muscle-invasive bladder cancer; TURBT: transurethral resection of bladder tumor.
Multivariable Cox regression analysis, adjusting for group, tumor grade, stage, multiplicity, and operating surgeon, confirmed the absence of a statistically significant difference in recurrence-free survival between the intervention and control groups. Patients with carcinoma in situ (CIS) had a markedly higher recurrence risk compared with those with Ta tumors, while patients with a single tumor had a lower recurrence risk compared with those with multiple tumors. The surgeon remained a highly significant predictor of recurrence, with HRs ranging from 0.26 to 0.36 depending on the surgeon (Table 4).
| Variable | HR (95% CI) | P-value | |
| Group | |||
| Control (Reference) | |||
| Intervention | 0.64 (0.40–1.01) | 0.054 | |
| Grade (WHO 1973) | |||
| G1 (Reference) | |||
| G2 | 1.40 (0.84–2.36) | 0.200 | |
| G3 | 1.01 (0.58–1.77) | 0.969 | |
| T stage | |||
| Ta (Reference) | |||
| Tis | 3.76 (1.51–9.35) | 0.004* | |
| T1 | 1.24 (0.75–2.04) | 0.397 | |
| Multiplicity | |||
| Single (Reference) | |||
| Multiple | 1.97 (1.29–2.99) | 0.002* | |
| Surgeon | |||
| Surgeon 2 (Reference) | |||
| Surgeon 1 | 0.32 (0.17–0.61) | < 0.001* | |
| Surgeon 3 | 0.33 (0.18–0.61) | < 0.001* | |
| Surgeon 4 | 0.36 (0.17–0.75) | 0.007* | |
| Others | 0.26 (0.15–0.44) | < 0.001* | |
| HR: Hazard ratio; CI: Confidence interval. *Indicates statistical significance. |
|||
Discussion
In this study, implementation of a standardized treatment protocol for patients with newly diagnosed NMIBC undergoing primary TURBT did not result in a reduction in early recurrence rates. Despite substantial improvements in adherence to perioperative and adjuvant measures – including increased use of blue-light cystoscopy, postoperative bladder irrigation, and guideline-based adjuvant instillation therapy – the early recurrence rate remained essentially unchanged between the intervention and control groups. However, multivariable Cox regression analysis revealed a trend toward lower recurrence in the intervention group after adjusting for tumor stage, grade, multiplicity, and operating surgeon, approaching statistical significance (HR = 0.64; 95% CI [confidence interval], 0.40–1.01; p = 0.054). With a larger cohort, this difference might have reached statistical significance, suggesting that improved perioperative and adjuvant management did contribute to better outcomes. Notably, the greatest difference in recurrence-free survival between the groups emerged after 1 year from the primary TURBT (Figure 1). This delayed effect suggests that the measures improving recurrence-free survival were those with a more prolonged effect, such as adjuvant instillation therapy, rather than surgical factors, which would be expected to influence outcomes earlier.
Supporting this interpretation is the lack of increased detrusor muscle in resection specimens after implementation of the protocol. The presence of detrusor muscle has repeatedly been validated as a surrogate marker of TURBT quality, strongly associated with reduced risk of early recurrence and dependent on operator skill and experience [6, 10, 11]. In this study, the lack of improvement in this indicator suggests that surgical quality did not improve substantially, providing a plausible explanation for the lack of effect on early recurrence. Equally important was the striking variability observed between surgeons, both in recurrence rates and in the presence of detrusor muscle. Such differences cannot be explained by tumor characteristics, which were comparable across patients treated by different surgeons (Table 3). Instead, they underscore the central role of the surgeon in determining outcomes. Because adherence to the treatment protocol varied among surgeons, we performed a sensitivity analysis excluding urologist nr. 2 from the intervention group. This analysis did not reveal any differences between the groups regarding early recurrence rates or recurrence-free survival. Previous multicenter studies, including the landmark analysis by Brausi et al. and more recently the RESECT trial, have similarly shown wide inter-institutional and inter-surgeon differences in recurrence that exceed what could reasonably be attributed to biological tumor variation [12, 13]. Our findings reinforce this message, that limiting TURBT to a small group of surgeons did not eliminate variability, and one surgeon in particular had significantly poorer outcomes. These results suggest that focusing solely on structural changes, such as restricting the number of operators, may be insufficient unless paired with systematic training, feedback, and quality assurance.
Another factor that warrants consideration is the modest imbalance in tumor multiplicity between the two groups. Patients in the intervention cohort more frequently presented with multiple tumors compared with the control cohort (38% vs. 23%). Multiplicity is a well-established predictor of recurrence, as reflected in our multivariable regression analysis, and this imbalance may have diluted the apparent effect of improved perioperative measures [1, 14]. Although not statistically significant, it raises the possibility that the intervention group was inherently predisposed to a higher risk of recurrence, thereby limiting the measurable benefit of protocol implementation.
Although single early instillation (SEI) of intravesical chemotherapy is recommended in international guidelines and has demonstrated efficacy in lowering recurrence risk, we opted instead for postoperative continuous bladder irrigation [1, 3]. This choice was motivated by evidence indicating non-inferiority of postoperative bladder irrigation compared with SEI in terms of recurrence prevention, along with its higher feasibility, better adherence in routine practice, and lower risk of complications [15–17]. Therefore, our protocol prioritized an intervention that was both effective and more broadly applicable across the patient cohort. The expanded use of blue-light cystoscopy and postoperative bladder irrigation during the intervention period also warrants consideration. Both measures have demonstrated benefit in controlled settings, but their effect size may be modest in real-world practice and highly dependent on the underlying quality of the resection [7, 18, 19]. For instance, blue-light cystoscopy can enhance tumor detection but cannot ensure adequate resection depth or completeness. Similarly, postoperative bladder irrigation likely reduces reimplantation of tumor cells but cannot mitigate the consequences of incomplete resection. Therefore, the unchanged recurrence rate in our cohort is not necessarily evidence against these interventions but rather an indication that they function best as adjuncts to technically proficient TURBT.
Although several studies have shown that interventions such as blue-light cystoscopy, postoperative bladder irrigation, and adjuvant intravesical therapy can reduce early recurrence rates, our real-world implementation likely did not replicate the conditions required to achieve those effects [7, 9, 19–21]. The reasons are probably multifactorial. First, adherence to the protocol, while improved, was incomplete – blue-light cystoscopy was used in 59% of cases, and use of adjuvant instillation therapy remained lower than expected – thereby diluting potential benefits. Second, surgical quality did not improve, as reflected by stable detrusor muscle rates and persistent inter-surgeon variability, both of which can overshadow the incremental effect of adjunctive measures. The four designated surgeons assigned to perform TURBTs in the intervention group compleded 79% of the procedures, which was slightly lower than anticipated. This was primarily due to some patients requiring subacute surgery for hematuria, in which case the procedure was performed by the on-call urologist. In addition, a proportion of operations took place during vacation preiods when one or more of the designated surgeons were unavailable. Finally, the study’s relatively modest cohort size limited the ability to detect small differences.
The results of this study should be interpreted in light of several limitations. The retrospective collection of data in the control group may have introduced information bias, particularly for clinical parameters such as tumor size and multifocality. The relatively modest cohort size, although comprising all newly diagnosed NMIBC cases at a national referral center in Iceland during the study periods, limited the statistical power to detect subtle effects of the intervention. In addition, as noted earlier, adherence to the treatment protocol, while improved, did not reach the level anticipated. Nevertheless, the consistency of our findings with large international studies supports the central conclusion that surgical quality remains the key determinant of patient outcomes.
Looking forward, efforts to reduce early recurrence after TURBT should prioritize structured surgical training and robust quality monitoring. Dedicated teaching programs and the use of quality indicators, such as the presence of detrusor muscle, should be embedded into routine practice. Moreover, feedback systems allowing surgeons to benchmark outcomes against peers could foster improvements. While perioperative measures and guideline-based intravesical therapy remain essential components of NMIBC management, they cannot substitute for meticulous surgery. Future research should explore interventions specifically designed to enhance surgical quality, including simulation-based training, standardized operative checklists, and real-time feedback mechanisms.
In conclusion, this study demonstrates that implementation of a standardized treatment protocol improved adherence to key perioperative measures but did not result in a reduction in early recurrence after TURBT for patients with newly diagnosed non-muscle-invasive bladder cancer. The persistence of marked surgeon-dependent variability highlights that surgical quality remains the most critical determinant of early recurrence and overall patient outcome. Meaningful progress in reducing recurrence rates will therefore require not only consistent adherence to evidence-based perioperative and adjuvant measures but also systematic efforts to strengthen surgical training, performance evaluation, and continuous quality improvement.
References
[1] Babjuk M, Burger M, Capoun O, et al. European Association of Urology guidelines on non-muscle-invasive bladder cancer (Ta, T1, and Carcinoma in Situ). Eur Urol. 2022;81(1):75–94. https://doi.org/10.1016/j.eururo.2021.08.010
[2] Chou R, Selph S, Buckley DI, et al. Comparative effectiveness of fluorescent versus white light cystoscopy for initial diagnosis or surveillance of bladder cancer on clinical outcomes: systematic review and meta-analysis. J Urol. 2017;197(3 Pt 1):548–558. https://doi.org/10.1016/j.juro.2016.10.061
[3] Gudjónsson S, Adell L, Merdasa F, et al. Should all patients with non-muscle-invasive bladder cancer receive early intravesical chemotherapy after transurethral resection? The results of a prospective randomised multicentre study. Eur Urol. 2009;55(4):773–780. https://doi.org/10.1016/j.eururo.2009.01.006
[4] Li M, Toniolo J, Nandurkar R, et al. Continuous bladder irrigation after transurethral resection of non-muscle invasive bladder cancer for prevention of tumour recurrence: a systematic review. ANZ J Surg. 2021;91(12):2592–2598. https://doi.org/10.1111/ans.16740
[5] Mariappan P, Johnston A, Trail M, et al. Achieving benchmarks for national quality indicators reduces recurrence and progression in non-muscle-invasive bladder cancer. Eur Urol Oncol. 2024;7(6):1327–1337. https://doi.org/10.1016/j.euo.2024.01.012
[6] Mariappan P, Zachou A, Grigor KM. Detrusor muscle in the first, apparently complete transurethral resection of bladder tumour specimen is a surrogate marker of resection quality, predicts risk of early recurrence, and is dependent on operator experience. Eur Urol. 2010;57(5):843–849. https://doi.org/10.1016/j.eururo.2009.05.047
[7] Onishi T, Sugino Y, Shibahara T, et al. Randomized controlled study of the efficacy and safety of continuous saline bladder irrigation after transurethral resection for the treatment of non-muscle-invasive bladder cancer. BJU Int. 2017;119(2):276–282. https://doi.org/10.1111/bju.13599
[8] Brausi M, Gavioli M, Peracchia G, et al. Dedicated teaching programs (DTP) can improve the quality of TUR of non muscle invasive bladder tumors (NMIBT): experience of a single institution. Eur Urol Suppl. 2008;7:180. https://doi.org/10.1016/S1569-9056(08)60433-9
[9] Sörenby A, Baseckas G, Bendahl PO, et al. Reducing recurrence in non-muscle-invasive bladder cancer by systematically implementing guideline-based recommendations: effect of a prospective intervention in primary bladder cancer patients. Scand J Urol. 2019;53(2–3):109–115. https://doi.org/10.1080/21681805.2019.1604568
[10] Kassem A, Assem A, Sharawy A, et al. The effect of surgeon experience on the recurrence of non-muscle invasive bladder cancer (NMIBC), following transurethral resection of the bladder tumor (TURBT): a double blinded prospective randomized study. Asian Pac J Cancer Prev. 2025;26(5):1767–1771. https://doi.org/10.31557/APJCP.2025.26.5.1767
[11] Rouprêt M, Yates DR, Varinot J, et al. The presence of detrusor muscle in the pathological specimen after transurethral resection of primary pT1 bladder tumors and its relationship to operator experience. Can J Urol. 2012;19(5):6459–6464.
[12] Brausi M, Collette L, Kurth K, et al. Variability in the recurrence rate at first follow-up cystoscopy after TUR in stage Ta T1 transitional cell carcinoma of the bladder: a combined analysis of seven EORTC studies. Eur Urol. 2002;41(5):523–531. https://doi.org/10.1016/S0302-2838(02)00068-4
[13] Gaba F, Gallagher KM, Bhatt NR, et al. Tumour factors and the variation in non-muscle invasive bladder cancer recurrence after transurethral resection surgery between sites: results from the RESECT study. J Clin Oncol. 2023;41(16_Suppl):4510. https://doi.org/10.1200/JCO.2023.41.16_suppl.4510
[14] Sylvester RJ, van der Meijden AP, Oosterlinck W, et al. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur Urol. 2006;49(3):466–477; discussion 75–77. https://doi.org/10.1016/j.eururo.2005.12.031
[15] Bani-Hani M, Abdel Majid A, Al-Zubi MT, et al. Continuous saline bladder irrigation in reducing recurrence and progression when compared to immediate mitomycin- C instillation post- resection of bladder tumor: a short communication. Asian Pac J Cancer Prev. 2022;23(1):171–175. https://doi.org/10.31557/APJCP.2022.23.1.171
[16] Bijalwan P, Pooleri GK, Thomas A. Comparison of sterile water irrigation versus intravesical mitomycin C in preventing recurrence of nonmuscle invasive bladder cancer after transurethral resection. Indian J Urol. 2017;33(2):144–148. https://doi.org/10.4103/iju.IJU_371_16
[17] Wishahi M, Nour H, Elesaily K, et al. Efficacy of continuous bladder irrigation with saline after transurethral resection of nonmuscle-invasive bladder cancer stage Ta T1 to prevent cancer recurrence and progression in comparison with a single immediate instillation of mitomycin C chemotherapeutic. Egypt J Surg. 2021;40(1):278–283.
[18] Mariappan P, Rai B, El-Mokadem I, et al. Real-life experience: early recurrence with hexvix photodynamic diagnosis–assisted transurethral resection of bladder tumour vs good-quality white light TURBT in new non–muscle-invasive bladder cancer. Urology. 2015;86(2):327–331. https://doi.org/10.1016/j.urology.2015.04.015
[19] Stenzl A, Burger M, Fradet Y, et al. Hexaminolevulinate guided fluorescence cystoscopy reduces recurrence in patients with nonmuscle invasive bladder cancer. J Urol. 2010;184(5):1907–1913. https://doi.org/10.1016/j.juro.2010.06.148
[20] Huncharek M, McGarry R, Kupelnick B. Impact of intravesical chemotherapy on recurrence rate of recurrent superficial transitional cell carcinoma of the bladder: results of a meta-analysis. Anticancer Res. 2001;21(1b):765–769.
[21] Malmström PU, Sylvester RJ, Crawford DE, et al. An individual patient data meta-analysis of the long-term outcome of randomised studies comparing intravesical mitomycin C versus bacillus Calmette-Guérin for non-muscle-invasive bladder cancer. Eur Urol. 2009;56(2):247–256. https://doi.org/10.1016/j.eururo.2009.04.038