ORIGINAL RESEARCH ARTICLE
Thermal imaging in the diagnosis of acute testicular pain – results of a feasibility pilot study
Lasse Hirvonena,b  , Konrad Sopylloc, Juhani Rantakallioc, Ilkka Paasoc and Mika Raitanenb,c
, Konrad Sopylloc, Juhani Rantakallioc, Ilkka Paasoc and Mika Raitanenb,c
aDepartment of Urology, The Wellbeing Services County of Satakunta, Satakunta Central Hospital, Pori, Finland; bFaculty of Medicine and Health Technology, University of Tampere, Tampere, Finland; cDepartment of Urology, The Wellbeing Services County of South Ostrobothnia, Seinäjoki, Finland
ABSTRACT
Objective: This study aimed to evaluate the feasibility of thermal imaging for the diagnosis of acute testicular pain. Pain is usually caused by infection, inflammation or torsion of the testis or testicular appendage. Diagnosis is based on examination, laboratory tests, and Doppler ultrasound (DU), which is investigator dependent and may cause delays. Thermal imaging is a fast and noninvasive method for measuring surface temperature.
Materials and methods: Our 3-month pilot study investigated the feasibility of thermal imaging for the diagnosis of acute testicular pain. Eighteen consecutive patients were examined using a thermal camera.
Results: Two patients had testicular torsion (group 1), two had torsion of the testicular appendage (group 2), four had bacterial epididymitis (group 3), and 10 had inflammatory epididymitis (group 4). The mean ± standard deviation (SD) temperature differences between symptomatic and a reference asymptomatic testicles were −0.65 ± 0.57°C (range, −1.1 to −0.2°C), −0.15 ± 0.21°C (range, −0.3 to 0.0°C), +0.65 ± 0.25°C (range, +0.4 to +1.0°C), and +0.39 ± 0.87°C (range, −1.0 to +1.8°C) in groups 1, 2, 3, and 4, respectively.
Conclusions: Thermal imaging is a feasible, noninvasive method for evaluating acute testicular pain. It may serve as a rapid diagnostic tool, but its clinical value must be confirmed in large prospective trials.
KEYWORDS: Testicular torsion; torsion of testicular appendix; acute testicular pain; thermal imaging
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 80–85. https://doi.org/10.2340/sju.v61.45714.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 24 November 2025; Accepted: 19 February 2026; Published: 26 March 2026
CONTACT: Lasse Hirvonen lasse.hirvonen@tuni.fi Faculty of Medicine and Health Technology, University of Tampere, Kalevantie 4, 33014 Tampere
Competing interests and funding: None.
The author (LH) received 3,500€ in pre-tax funding from the Wellbeing Services County of Satakunta specifically for the preparation and submission of this manuscript. Thermidas provided thermal camera for 3-month study period free of charge, whereas no additional financial support was received.
Introduction
The most common causes of acute testicular pain are infections or inflammations of the testis/ or epididymis and torsion of either the testicular appendage or of the testis itself. Testicular torsion is a rare but urgent urological condition that requires immediate surgical intervention. The incidence of testicular torsion is approximately 3.8–4.5/100,000 males [1, 2], with peak occurrences at 1 month and 12 years of age [2]. The diagnosis of testicular torsion is primarily clinical, based on history and physical examination. Urinalysis may assist in excluding alternative diagnoses but is not essential, while DU is the key imaging modality when available. The detection of testicular torsion using DU is based on decreased vascularity of the affected testis and characteristic spermatic cord morphology. DU has been shown to detect a proven rotated spermatic cord 78.6–100% of the time [3–6]. However, the detected intratesticular hypoperfusion may be as low as 76% [3]. The other drawbacks of DU are time delays [7] and the requirement of an experienced radiologist [3]. The testicular salvage rate is high within 6 h of symptom onset but decreases progressively thereafter. Reducing any unnecessary delays between symptom onset and operative intervention is a key factor in successful treatment [8–11].
Thermal imaging, also known as thermography, is a technique that uses infrared cameras to measure surface temperatures. In contrast to DU, it is easy, inexpensive, and noninvasive to perform. The main advantage, however, is that it takes only a few minutes to obtain the results. Testicles as paired organs close to each other are an ideal target for thermal imaging enabling a temperature comparison between symptomatic and asymptomatic side. Various causes of acute testicular pain may have different thermal properties, at least in theory. In testicular torsion, blood flow to the affected testicle may be reduced or absent, which would be reflected as a decreased temperature on thermal imaging, whereas in epididymitis, blood flow is usually increased, which may lead to increased temperature. The torsion of the testicular appendage, a small rudiment attached to the surface of the testis, may be thermally neutral. The aim of the study was to investigate the feasibility of thermal imaging for the diagnosis of acute testicular pain.
Methods
A prospective 3-month long pilot study on the feasibility of thermal imaging for the diagnosis of acute testicular pain was conducted at Seinäjoki Central Hospital, Finland, starting in October and ending in December 2023. The executive board of the hospital evaluated the ethical aspects of the study and approved it on September 28th, 2023. A total of 18 patients with acute testicular pain were examined via thermal imaging. The only exclusion criteria were undescended testis and having only a single testis. Patients, and in the case of minors, their parents, were given verbal and written information about the study, and signed informed consent was obtained from the patient or parent before the examination.
The patients with acute testicular pain were assorted by triage and either a urologist or a urology resident examined the patient and performed the thermal imaging. The patients stood allowing the scrotum to hang freely. To avoid the thermal effect of the penis on the skin of the scrotum, the penis was taped upward against the abdomen for 10 min before imaging. The distance from the camera to the scrotum was approximately 20 cm and duration of the imaging was approximately 2 min. The patients were diagnosed as usual using clinical evaluation and laboratory testing, including urine analysis and DU as needed. Thermal imaging results had no effect on decision making, as the analysis of temperatures was performed several weeks later. The core temperature of both testicles was measured by a collaboration of urologist and thermal imaging specialist from Thermidas (Thermidas Ltd, Tampere, Finland) who were blinded to the other clinical data, such as the DU results and final diagnosis.
Thermidas’ ThIR-T560 handheld thermal camera system was used (Figure 1). Thermidas’ infrared (IR) thermal imaging system is the first CE-approved medical device IR-thermal imaging system in the world. The analysis of the thermal images and temperatures in Celsius were measured by using a Thermidas Remote Imager SW on a laptop. The technical specifications of the camera are presented in Table 1.

Figure 1. Thermidas ThIR-T560 Handheld IR Imaging System.
Due to small sample size, especially because of the low number of patients with testicular torsion, formal statistical analyses were not justified. In the larger oncoming studies, McNemar’s test may be used to test for changes in sensitivity and specificity at different temperature difference cut off values and Receiver operating characteristic (ROC) curve to illustrate the effect.
Results
A total of 18 patients were included in the study. The patients’ age ranged from 4 to 73 years, with a mean ± standard deviation (SD) of 27.4 ± 21.5 years. The time from onset of testicular pain to presentation at the emergency department ranged from 3 to 72 h, with a mean ± SD of 33.7 ± 27.0 h. Eight (44%) patients had right-sided symptoms and 10 (56%) had left-sided symptoms. Among the patients, two (11%) had testicular torsion (group 1), two (11%) had torsion of the testicular appendage (group 2), four (22%) had epididymitis caused by chlamydia or other bacteria in the urine culture (group 3), and 10 (56%) had inflammatory epididymitis with negative urine culture and chlamydia test results (group 4). The mean ± SD temperature differences between symptomatic and a reference asymptomatic testicles were −0.65 ± 0.57°C (range, −1.1 to −0.2°C), −0.15 ± 0.21°C (range, −0.3 to 0.0°C), +0.65 ± 0.25°C (range, +0.4 to +1.0°C), and +0.39 ± 0.87°C (range, −1.0 to +1.8°C) in groups 1, 2, 3, and 4, respectively (Table 2, Figure 2). Both patients with testicular torsion had decreased blood flow on DU and decreased temperature on thermal imaging. A thermal image of the first patient with testicular torsion is shown in Figure 3.
| Diagnosis | Mean age (min–max) | Mean time delay in hours (min-max) | Symptomatic side | Mean temperature (°C) right (min-max) | Mean temperature (°C) left (min-max) | Mean temperature (°C) difference | Doppler ultrasound* |
| Testis Torsion | 13.5 (13–14) | 5 (3–7) | 31.25 (29.8–32.7) | 31.7 (29.6–33.8) | −0.65 (−1.1 to −0.2) | ||
| 1 | 14 | 3 | Right | 32.7 | 33.8 | −1.1 | 1 |
| 2 | 13 | 7 | Left | 29.8 | 29.6 | −0.2 | 1 |
| Torsion of appendage testis | 34 (10–58) | 26 (4–48) | 31.75 (31.2–32.3) | 31.9 (31.5–32.3) | −0.15 (−0.3 to 0) | ||
| 3 | 58 | 4 | Right | 32.3 | 32.3 | 0 | 3 |
| 4 | 10 | 48 | Right | 31.2 | 31.5 | −0.3 | 3 |
| Epididymitis (bacterial) | 48.8 (23–73) | 40.3 (3–72) | 34.1 (32.3–35.5) | 33.9 (32.7–35.1) | 0.7 (0.4–1.0) | ||
| 5 | 71 | 72 | Left | 32.3 | 32.9 | 0.6 | 2 |
| 6 | 73 | 72 | Right | 33.7 | 32.7 | 1 | 2 |
| 7 | 28 | 3 | Right | 35.5 | 34.9 | 0.6 | 2 |
| 8 | 23 | 14 | Left | 34.7 | 35.1 | 0.4 | 2 |
| Epididymitis (inflammatory) | 20.3 (4–46) | 38.2 (17–72) | 33.6 (30.9–35.7) | 34.3 (31.2–36.0) | 0.4 (−1.0 to +1.8) | ||
| 9 | 27 | 24 | Right | 30.9 | 31.2 | −0.3 | 4 |
| 10 | 6 | 19 | Left | 35.7 | 35.1 | −0.6 | 2 |
| 11 | 32 | 24 | Right | 31.9 | 32.9 | −1 | 2 |
| 12 | 35 | 72 | Left | 34.7 | 36 | 1.3 | 2 |
| 13 | 15 | 72 | Left | 33.6 | 34.5 | 0.9 | 2 |
| 14 | 10 | 24 | Left | 33 | 34.8 | 1.8 | 2 |
| 15 | 46 | 72 | Left | 34.2 | 35 | 0.8 | 2 |
| 16 | 15 | 17 | Left | 33.4 | 34 | 0.6 | 2 |
| 17 | 13 | 38 | Left | 34.1 | 34.3 | 0.2 | 4 |
| 18 | 4 | 21 | Right | 34.9 | 34.7 | 0.2 | 3 |
| *1 = Decreased blood flow; 2 = Increased blood flow; 3 = Symmetrical blood flow; 4 = Not done. | |||||||
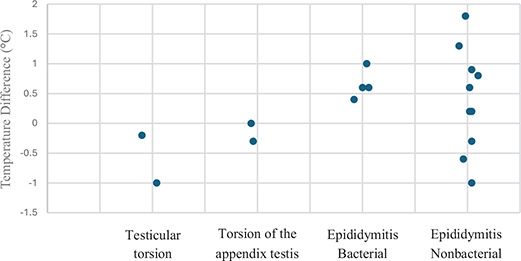
Figure 2. Temperature difference between symptomatic and asymptomatic testicle according to cause of the pain.
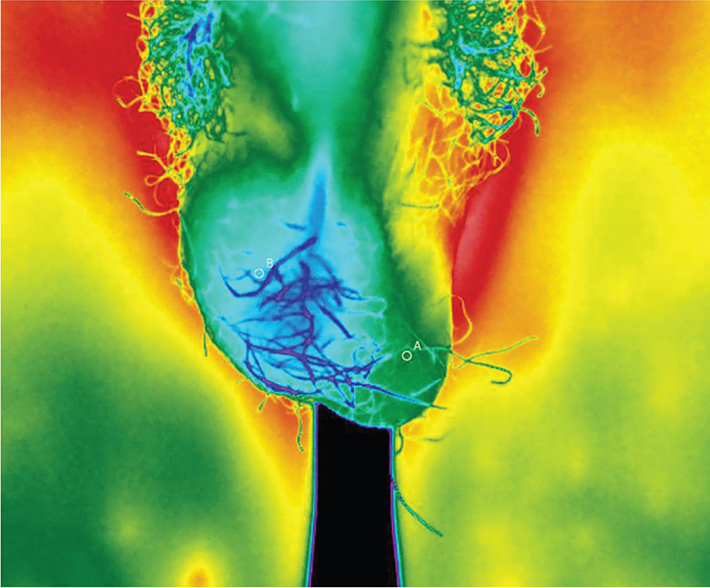
Figure 3. Thermal imaging at the diagnosis of testicular torsion on the right side. Clear asymmetry can be seen between the colder right side and the warmer left side. The temperature difference was 1.1°C.
Discussion
Clinical findings among patients with acute testicular pain include many unspecific and overlapping symptoms that limit the interpretation [12]. Moreover, physical examination is subjective and the reliability of DU is investigator dependent. The risk for testicular loss progressively increases beyond 6 h after symptom onset. Early salvage rates in testicular torsion repair between 0 and 6 h from the onset of symptoms are as high as 97.2%, yet salvage rates of 7.4% even after 48 h have been reported [13]. This might be due to the variance in the degree of testicular torsion [14]. Testicular torsion leads primarily to an orchiectomy on average in 39% of cases, and further loss occurs over time. After 1 year, the rate of testicular loss increases to 50% [15]. Chan et al. documented a median time delay of 48 min for DU [7], and Wright and Wright reported a delay of 119 min [16], significantly increasing the time from the emergency department to the operating room. Thus, there is a need for easy and fast objective tests to identify patients with testicular pain who would benefit from immediate surgical intervention.
There are few animal studies using thermal imaging for the diagnosis of testicular ischemia. In 2008, Capraro et al. reported a 1.7°C mean temperature decrease 2 h after 720 degrees of experimental torsion in anesthetized sheep [17]. More recently, Brooks et al. published a study in which a smartphone thermal camera identified all randomly ligated spermatic cords in 30 canines. The temperature differences between testicles 11 min after ligation ranged from 0.7°C to 3.7°C, with an average difference of 1.7°C lower in the ischemic testicle [18]. To date, the use of thermal imaging for the diagnosis of testicular torsion in humans has not been investigated. Thermal imaging has some clinical use, for example, in the surveillance of Charcot´s foot [19]. In addition, it has been studied for the detection of varicocele progression [20]. Thermal imaging is easy to perform and can be performed early in the emergency department, ideally immediately after triage. The result of thermal imaging, that is, the temperature difference between testicles, is consequently quickly available, reducing the time spent in the emergency department.
In our study, the first patient with testicular torsion was a 14-year-old boy who came to the ED after experiencing severe pain in the right testicle for 3 h with no urinary symptoms. At the time of medical investigation, the pain in the right testicle had become milder but increased again while waiting for DU. DU revealed that blood circulation was reduced in both the right testicle and epididymis. Blood and urine tests were normal. Thermal imaging revealed a surface temperature of 32.7°C on the right side of the scrotum and 33.8°C on the left side, with a 1.1°C difference (Figure 3). Immediate surgery was performed and testicular torsion on the right side was confirmed. Blood flow to the testis recovered with the correction of torsion. Bilateral orchiopexy was performed. The patient was discharged from the hospital free of symptoms on the first postoperative day. At 5 weeks, the patient returned to the control visit asymptomatic. DU showed symmetrical vascularity. The difference in the testicular temperature was 0.2°C, with 33.2°C for the right side and 33.0°C for the left side (Figure 4). In another patient with testicular torsion, torsion resolved while waiting for the operation, which may have influenced the temperature difference (−0.2°C), as thermal imaging was performed about 30 min after DU but about 60 min before the operation.
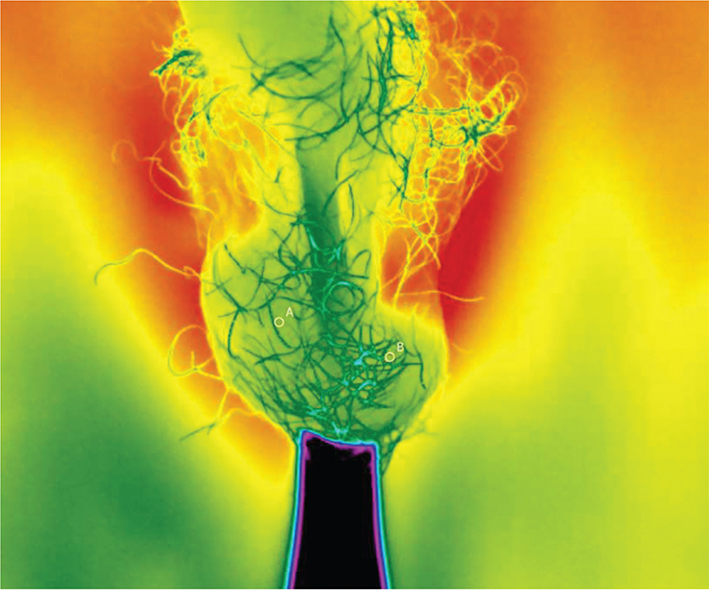
Figure 4. The same patient at 5 weeks after the operation. A symmetrical finding in the thermal imaging with only 0.2°C difference in the temperature between the right and the left testicles.
Very small sample size is the main limitation of this study. DU was neither performed on every patient nor was the sequence of thermal imaging and DU standardized. In addition, body and room temperature, lighting, penis and scrotal hair may interfere with measurement of the scrotal temperature. To minimize some of these factors, penis was taped upward before taking the thermal image. Image analysis was performed collaboratively by an experienced thermal imaging specialist from Thermidas and a urologist. The diagnostic property of thermal imaging in patients with acute testicular pain is based on temperature difference between left and right sides and therefore possible interference of external factors on this thermal difference may be limited. However, to the best of our knowledge, this is the first time ever that thermal imaging has been used in the evaluation of acute testicular pain in humans, and furthermore, testicular torsion has been documented with this technique. The temperature data were analyzed several weeks after a 3-month study period in a blinded manner to the clinical information such as DU result and final diagnosis, which we found important.
According to our results, thermal imaging is a feasible method in patients with acute testicular pain. In the future large prospective studies are needed to demonstrate accurate sensitivity and specificity of thermal imaging and to determine clinically significant temperature difference as there is lack of a predefined temperature threshold for temperature difference between sides. In addition, further research on the role of thermal imaging in epididymitis could improve our knowledge of temperature profiles between testicular torsion, torsion of the testicular appendage and infections. If diagnostic value of thermal imaging will be confirmed in subsequent studies, the diagnostic algorithm for testicular torsion in the future could include immediate thermal imaging and, in the case of significantly decreased temperature on the symptomatic side, immediate surgical intervention. In the case of no difference or increased temperature, further diagnostic procedures would be performed, including DU and laboratory tests. This may minimize the time delay, enabling more testicles to be saved. Furthermore, thermal properties of testicular tumors and, for example, Peyronie’s disease could be studied as potential urological applications in the future.
Conclusions
Thermal imaging is a feasible and noninvasive diagnostic tool in the evaluation of patients with acute testicular pain. However, further research with a larger sample size is needed to investigate the diagnostic value of thermal imaging. Due to this a prospective multi-center study is starting in Finland including all five university hospitals (Helsinki, Kuopio, Oulu, Tampere and Turku) and three large central hospitals (Jyväskylä, Pori and Seinäjoki) (ClinicalTrialGov ID: NCT07324590).
Availability of data and materials
The data are not publicly available due to patient confidentiality. However, it is possible to request the data from the author, pending patient consent.
Ethics approval and consent to participate
The executive board of Seinäjoki Central Hospital approved this pilot study on September 28th 2023. Patients and in case of a minor, also their parents were provided written and verbal information about the study, and informed consent was signed before the examination. The diagnosis of testicular torsion was made as usual, and thermal imaging had neither diagnostic nor therapeutic effects. Thermal images used in this publication are anonymous and published with permission from the patient and his parent.
Consent for publication
Written and signed consent to publish the case and pictures was obtained from the mother of the minor patient. In addition, the patient was provided both written and verbal information, and permission to use anonymous pictures was obtained.
Authors’ contributions statements
Lasse Hirvonen wrote the manuscript and prepared the article for review. Mika Raitanen created the project, recruited patients, provided figures and reviewed the manuscript. Konrad Sopyllo and Juhani Rantakallio recruited patients. Ilkka Paaso reviewed the thermal images.
References
- [1] Mansbach JM, Forbes P, Peters C. Testicular torsion and risk factors for orchiectomy. Arch Pediatr Adolesc Med. 2005;159:1167–1171. https://doi.org/10.1001/archpedi.159.12.1167
- [2] Zhao LC, Lautz TB, Meeks JJ, Maizels M. Pediatric testicular torsion epidemiology using a national database: incidence, risk of orchiectomy and possible measures toward improving the quality of care. J Urol 2011;186:2009–13. https://doi.org/10.1016/j.juro.2011.07.024
- [3] Kalfa N, Veyrac C, Lopez M, et al. Multicenter assessment of ultrasound of the spermatic cord in children with acute scrotum. J Urol. 2007;177:297–301; discussion 301. https://doi.org/10.1016/j.juro.2006.08.128
- [4] Altinkilic B, Pilatz A, Weidner W. Detection of normal intratesticular perfusion using color coded duplex sonography obviates need for scrotal exploration in patients with suspected testicular torsion. J Urol. 2013;189:1853–1858. https://doi.org/10.1016/j.juro.2012.11.166
- [5] Pepe P, Panella P, Pennisi M, et al. Does color Doppler sonography improve the clinical assessment of patients with acute scrotum? Eur J Radiol. 2006;60:120–124. https://doi.org/10.1016/j.ejrad.2006.04.016
- [6] Nussbaum Blask AR, Bulas D, Shalaby-Rana E, et al. Color Doppler sonography and scintigraphy of the testis: a prospective, comparative analysis in children with acute scrotal pain. Pediatr Emerg Care. 2002;18:67–71. https://doi.org/10.1097/00006565-200204000-00001
- [7] Chan EP, Wang PZT, Myslik F, et al. Identifying systems delays in assessment, diagnosis, and operative management for testicular torsion in a single-payer health-care system. J Pediatr Urol. 2019;15:251.e1–251.e7. https://doi.org/10.1016/j.jpurol.2019.03.017
- [8] Romao RLP, Anderson KH, MacLellan D, et al. Point-of-care influences orchiectomy rates in pediatric patients with testicular torsion. J Pediatr Urol. 2019;15:367.e1–367.e7. https://doi.org/10.1016/j.jpurol.2019.04.014
- [9] Ramachandra P, Palazzi KL, Holmes NM, et al. Factors influencing rate of testicular salvage in acute testicular torsion at a tertiary pediatric center. West J Emerg Med. 2015;16:190–194. https://doi.org/10.5811/westjem.2014.11.22495
- [10] Even L, Abbo O, Le Mandat A, et al. [Testicular torsion in children: Factors influencing delayed treatment and orchiectomy rate]. Arch Pediatr Organe Off Soc Francaise Pediatr. 2013;20:364–368. https://doi.org/10.1016/j.arcped.2013.01.008
- [11] Leape LL. Torsion of the testis. Invitation to error. JAMA. 1967;200: 669–672.
- [12] Abbas TO, Abdelkareem M, Alhadi A, et al. Suspected testicular torsion in children: diagnostic dilemma and recommendation for a lower threshold for initiation of surgical exploration. Res Rep Urol. 2018;10:241–249. https://doi.org/10.2147/RRU.S186112
- [13] Mellick LB, Sinex JE, Gibson RW, et al. A systematic review of testicle survival time after a torsion event. Pediatr Emerg Care. 2019;35: 821–825. https://doi.org/10.1097/PEC.0000000000001287
- [14] Bandarkar AN, Blask AR. Testicular torsion with preserved flow: key sonographic features and value-added approach to diagnosis. Pediatr Radiol. 2018;48:735–744. https://doi.org/10.1007/s00247-018-4093-0
- [15] MacDonald C, Kronfli R, Carachi R, et al. A systematic review and meta-analysis revealing realistic outcomes following paediatric torsion of testes. J Pediatr Urol. 2018;14:503–509. https://doi.org/10.1016/j.jpurol.2018.09.017
- [16] Wright HG, Wright HJ. Ultrasound use in suspected testicular torsion: an association with delay to theatre and increased intraoperative finding of non-viable testicle. N Z Med J. 2021;134:50–55.
- [17] Capraro GA, Coughlin BF, Mader TJ, et al. Testicular cooling associated with testicular torsion and its detection by infrared thermography: an experimental study in sheep. J Urol. 2008;180:2688–2693. https://doi.org/10.1016/j.juro.2008.08.004
- [18] Brooks JP, Brooks JM, Seals T. Smartphone thermal imaging in the detection of testicular ischemia. Urology. 2021;157:233–238. https://doi.org/10.1016/j.urology.2021.02.031
- [19] van Netten JJ, van Baal JG, Liu C, et al. Infrared thermal imaging for automated detection of diabetic foot complications. J Diabetes Sci Technol. 2013;7:1122–1129. https://doi.org/10.1177/193229681300700504
- [20] Kulis T, Knezevic M, Karlovic K, et al. Infrared digital thermography of scrotum in early selection of progressive varicocele. Med Hypotheses. 2013;81:544–546. https://doi.org/10.1016/j.mehy.2013.06.028