ORIGINAL RESEARCH ARTICLE
Complications and clinical outcomes of retroperitoneal lymph node dissection for testicular cancer in a centralized population-based cohort in Sweden: insights from SWENOTECA
Anna Thora,b  , Anna Grenabo Bergdahlc,d
, Anna Grenabo Bergdahlc,d  , Armin Abnikib, Axel Gerdtssona,e,f
, Armin Abnikib, Axel Gerdtssona,e,f  , Ingrid Glimeliusg
, Ingrid Glimeliusg  , Martin Hellströmh, Anna K. Janssong
, Martin Hellströmh, Anna K. Janssong  , Berglind Johannsdottiri,j
, Berglind Johannsdottiri,j  , Torgrim Tandstadk,l
, Torgrim Tandstadk,l  , Gabriella Cohn-Cedermarki,j
, Gabriella Cohn-Cedermarki,j  , Anders Kjellmana,b
, Anders Kjellmana,b  and Per-Olof Lundgrena,b
and Per-Olof Lundgrena,b 
aDepartment of Clinical Science, Intervention and Technology, Division of Urology, Karolinska Institute, Stockholm, Sweden; bDepartment of Urology, Pelvic Cancer, Karolinska University Hospital, Stockholm, Sweden; cDepartment of Urology, Institute of Clinical Science, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden; dRegion Västra Götaland, Sahlgrenska University Hospital, Department of Urology, Gothenburg, Sweden; eDepartment of Urology, Skåne University Hospital, Malmö, Sweden; fInstitution of Translational Medicine, Lund University, Malmö, Sweden; gDepartment of Immunology, Genetics & Pathology, Cancer Precision Medicine, Uppsala University, Uppsala, Sweden; hDepartment of Diagnostics and Intervention, Oncology, Umeå University, Umeå, Sweden; iDepartment of Oncology – Pathology, Karolinska Institute, Stockholm, Sweden; jDepartment of Pelvic Cancer, Genitourinary Oncology Unit, Karolinska University Hospital, Stockholm, Sweden; kDepartment of Clinical and Molecular Medicine, Norwegian University of Science and Technology, Trondheim, Norway; lThe Cancer Clinic, St. Olavs University Hospital, Trondheim, Norway
ABSTRACT
Objective: Retroperitoneal lymph node dissection (RPLND) for testicular germ cell cancer is a complex procedure associated with postoperative complications and long-term morbidity, best performed by experienced surgeons at high-volume centers. This study evaluates surgical outcomes of RPLND in a centralized population-based cohort.
Methods: This is a retrospective analysis of a prospective multicenter cohort of all RPLNDs in Sweden between 2018 and 2022. 217 patients (175 nonseminomas and 42 seminomas) underwent unilateral or bilateral primary RPLND or post-chemotherapy RPLND. Primary outcomes were complications, loss of ejaculation, and histopathology.
Results: Intraoperative complications occurred in 8% of unilateral and 0% of bilateral templates in primary RPLND, and in 0 and 8% in post-chemotherapy RPLND, most commonly renal injury. Postoperative complications rate was significantly higher with bilateral templates in post-chemotherapy RPLND (49% vs 18%, p < 0.01). Clavien-Dindo ≥ IIIb complications occurred in 2 (primary) and 3% (post-chemotherapy), respectively. Loss of ejaculation was numerically more common after bilateral templates (primary: 60% vs 31%, p = 0.07; post-chemotherapy: 53% vs 38%, p = 0.09). Viable cancer was found in 95% of seminomas and 52% of nonseminomas for primary RPLND and in nonseminoma post-chemotherapy RPLND, 11% viable cancer, 50% teratoma, and 39% benign nodes. Robotic surgery did not increase complications or loss of ejaculation.
Conclusions: RPLND demonstrated low complication rates and rare serious events. Bilateral templates were associated with increased loss of ejaculation. Robotic surgery was safe, and prior chemotherapy did not preclude laparoscopy. Post-chemotherapy RPLND showed more teratoma and viable cancer, and fewer benign findings than previously reported.
KEYWORDS: Complications; germ cell cancer; metastatic; nonseminoma; retroperitoneal; robotic surgery; RPLND; seminoma; testicular
Citation: Scandinavian Journal of Urology 2026, VOL. 61, 112–120. https://doi.org/10.2340/sju.v61.45743.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Chirurgica Scandinavica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 23 September 2025; Accepted: 24 February 2026; Published: 13 April 2026
CONTACT: Anna Thor anna.thor@ki.se Department of Clinical Science, Intervention and Technology, Division of Urology, Karolinska Institute, 171 77 Stockholm, Sweden
Competing interests and funding: Funding from Karolinska University Hospital has supported Anna Thor’s research hours.
Introduction
Testicular germ cell cancer (TGCC) is the most common malignancy in younger males (ages 18–40) in Scandinavia and many western countries [1] but remains rare. Incidence is high in the Nordic countries, with 8.8/100,000 in Sweden and 11.7/100,000 in Norway, compared to 6.2/100,000 in Western Europe, with approximately 350 new Swedish cases annually and rising [2]. Ten-year cancer-specific survival after metastatic disease is high at 91% for Sweden and Norway [3]. Given the excellent prognosis and young age, reducing treatment-related morbidity is a priority.
Standard treatment for metastatic TGCCs is three cycles of bleomycin, etoposide, and cisplatin (BEP), per the Swedish and Norwegian Testicular Cancer Group (SWENOTECA) guidelines, published 2020 [4]. Retroperitoneal lymph node dissection (RPLND) is indicated for nonseminoma patients with a residual tumor in the retroperitoneal space measuring 10 mm in largest axial diameter following systemic therapy. For seminoma patients, RPLND is an option for primary treatment for clinical stage (CS) IIA and IIB, with up to two metastatic nodes not exceeding 30 mm in any axis. Results from this novel strategy have been published by our group [5], as well as from the PRIMETEST, SEMS, and CO-TRIMS trials [6–8], and cohorts from Indiana [9] and New York [10], showing low rates of complications and relapses. For CS IIA nonseminomas without elevated tumor markers, our guidelines also recommend RPLND, and this patient population has been evaluated in a previously published study from our group [11]. The aim of surgery over systemic agents, despite the very high cure rate, is to avoid the long-term adverse effects of platinum-based chemotherapy. Testicular cancer survivors face reduced life expectancy and significantly increased risks of cardiovascular disease and secondary malignancies [12–14].
In late 2017, Swedish health authorities centralized all RPLND procedures for TGCCs to two tertiary centers and introduced a weekly national consensus conference for case review. Since the 1980s, all TGCC cases have been recorded in a national registry, and from 2007 onward, SWENOTECA has maintained a prospective database on RPLND procedures. Prior data from this registry reported on intra- and postoperative complications and loss of antegrade ejaculation after RPLND [15], a condition regarded by many as one of the most significant iatrogenic sequalae. Since the study’s publication in 2019, robot-assisted RPLNDs in our centers have increased significantly. A recent systematic review reported that robot-assisted RPLND are associated with fewer postoperative complications [16]. Complication rates in experienced centers vary from 3.7 to 45% [15, 17–19], most commonly ileus (0.5–21%) [18, 20]. Extended resections are reported in 18–33% of RPLNDs, usually nephrectomy (4.6–14%) [17, 20, 21]. Intraoperative complications occur in 3.7–8% [15, 18, 19] frequently with ureteral injury. Loss of antegrade ejaculation affects 15–32% after unilateral and 21–75% after bilateral templates in post-chemotherapy patients [15, 17, 19, 22]. Histology after post-chemotherapy RPLND reveals necrosis/fibrosis in 28–55%, teratoma in 29–49%, and viable cancer in 10–32% [15, 17, 19, 20, 23, 24].
This study aimed to evaluate both functional and oncological outcomes in patients undergoing RPLND, including primary and post-chemotherapy cases. Functional outcomes comprised intra- and postoperative complications as well as loss of antegrade ejaculation, while oncological outcomes focused on histological findings, specifically the presence of viable cancer or teratoma.
Materials and methods
Patients
All TGCC patients undergoing RPLND were prospectively included in the Retrop registry. This report covers procedures between January 1, 2018 and December 31, 2022 at two Swedish centers: Karolinska University Hospital and Sahlgrenska University Hospital. Exclusions were lumpectomies, testicular stromal tumors, and recurrent tumors after prior RPLND. The cohort comprised 217 patients (175 nonseminomas and 42 seminomas), with all procedures performed by seven experienced surgeons.
Data collection was approved by the regional ethics committee.
Intervention
Figure 1 illustrates the SWENOTECA retroperitoneal templates [4]. Right-sided unilateral templates include areas 1, 2, 4, 5, 7, and 9 for right-sided testicular tumors; left-sided unilateral templates include areas 3, 6, 8, and 10 for left-sided testicular tumors. Both avoid crossing the midline aorta to preserve the nerves from the sympathetic trunk involved in ejaculation. Earlier guidelines included area 3 for right-sided templates and area 2 for left-sided templates; therefore, a fraction of patients in this cohort were operated accordingly [25]. Bilateral templates were employed for residual tumors ≥ 5 cm after completion of chemotherapy, or if visible metastasis is present on the contralateral template side of the testicular tumor. Suprahilar resection (areas 11 and 12) was performed if tumor extended cranially to the renal vessels on imaging.
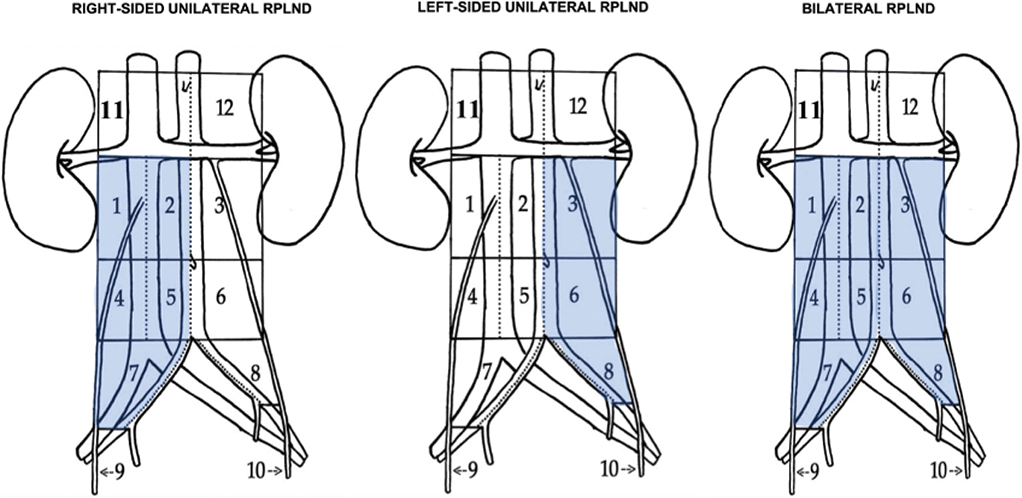
Figure 1. Retroperitoneal templates according to SWENOTECA guidelines [4] SWENOTECA: Swedish and Norwegian Testicular Cancer Group.
Follow-up
Each patient underwent postoperative follow-up with the treating urologist, followed by additional monitoring at an oncology department in accordance to SWENOTECA guidelines [4]. Last follow-up for data collection ranged from April to September 2024, unless the patient was diseased or lost to follow-up.
Data analysis
Clinical data were retrieved through the patient charts and the national registry, including CS (Royal Marsden), International Germ Cell Cancer Collaborative Group (IGCCCG) prognostic classification, age, chemotherapy, side, size and histology of tumor in orchiectomy specimen, serum biomarkers, robotic intervention, intraoperative bleeding, procedure duration, hospital stay, nerve-sparing, extended surgery, suprahilar resection, histology and number of resected retroperitoneal lymph nodes, as well as intra- and postoperative complications and persistent loss of antegrade ejaculation. Data on dissected areas and crossing of the aortic midline were collected. The most recent notation on antegrade ejaculation was recorded. Nerve-sparing was classified as partial (one sided in bilateral templates), full or none. Extended surgery was defined as procedures outside the retroperitoneal templates to achieve tumor-free margins. Intraoperative complications were defined as unforeseen events. Histology was categorized as benign (necrosis/fibrosis), teratoma, or viable cancer. Postoperative complications were recorded within 30 days using the Clavien-Dindo (CD) classification [26]. Chemotherapy was classified as none, adjuvant, standard, or intensified. One case report form per patient was created. The cohort was divided into primary RPLND and post-chemotherapy RPLND groups, with subanalyses conducted for robotic procedures and for post-chemotherapy nonseminoma patients.
Statistical analysis
Statistical analyses were conducted using STATA v 16.1 (StataCorp LCC, College Station, Tx, USA). Chi-squared tests assessed differences in proportions between groups. Mann–Whitney U tests compared continuous variables, allowing median comparisons. Quantile regression was utilized for a similar purpose as Mann–Whitney U, for non-normal distributions. Logistic regression evaluated the association between template type and the likelihood of loss of antegrade ejaculation, with results presented as odds ratios. A two-sided p-value of < 0.05 was considered statistically significant.
Results
Overall characteristics
The analysis included 217 patients (175 nonseminomas and 42 seminomas). Median follow-up was 43 months (interquartile range [IQR]: 29–57): 46 months for nonseminomas (IQR: 34–62) and 35 months for seminomas (IQR: 24–45). Primary (P) RPLND was performed in 61 patients (24 nonseminomas and 37 seminomas; Table 1). The post-chemotherapy (PC) group included 156 patients (5 seminomas and 151 nonseminomas; Table 2). Of these, 125 patients (58%) received BEP only, while 29 (14%) underwent intensified regimens: paclitaxel, ifosfamide, and cisplatin (TIP), gemcitabine, oxaliplatin, and paclitaxel (GOP), or high-dose chemotherapy with stem cell rescue.
| Post-chemotherapy group, n = 156 | Unilateral RPLND | Bilateral RPLND |
| Number of cases, n (%) | 76 (49) | 80 (51) |
| Age, years (IQR) | 30 (25–37) | 31 (25–39) |
| Histological classification, n (%) | ||
| Nonseminoma | 74/76 (97) | 77/80 (96) |
| Seminoma | 2/76 (3) | 3/80 (4) |
| Histology orchiectomy specimen, n (%) | ||
| Embryonal carcinoma | 9/46 (20) | 4/45 (9) |
| Yolk sac tumor | 1/46 (2) | 0 |
| Pure teratoma | 2/46 (4) | 4/45 (9) |
| Mixed nonseminomous tumor | 32/46 (70) | 23/45 (51) |
| Unknown (primary chemotherapy) | 2/46 (4) | 12/45 (27) |
| Extragonadal GCC | 0 | 2/45 (4) |
| Missing | 28/76 (37) | 32/80 (40) |
| Median size of tumor in orchiectomy specimen, mm (IQR) | 32 (25–50) | 35 (20–70) |
| Clinical stage according to Royal Marsden, modified*, n (%) | ||
| CS II | 55/72 (76) | 25/78 (32) |
| CS III | 4/72 (6) | 11/78 (14) |
| CS IV | 13/72 (18) | 42/78 (54) |
| Abdominal stage | ||
| A | 11/75 (15) | 4/80 (5) |
| B | 50/75 (67) | 30/80 (38) |
| C | 13/75 (17) | 22/80 (28) |
| D | 1/75 (1) | 24/80 (30) |
| Prognostic group according to International Germ Cell Cancer Collaborative Group (IGCCCG), n (%) | ||
| Good | 47/71 (66) | 26/75 (35) |
| Intermediate | 21/71 (30) | 22/75 (29) |
| Poor | 3/71 (4) | 27/75 (36) |
| Chemotherapy**, n (%) | ||
| Standard | 72/74 (97) | 53/80 (66) |
| Intensified | 2/74 (3) | 27/80 (34) |
| Marker positive at surgery, n (%) | 2/62 (3) | 19/69 (28) |
| Histological outcome nonseminomas, n = 151, n (%) | ||
| Benign/necrosis/fibrosis | 25/71(35) | 32/75 (43) |
| Viable nonseminoma other than teratoma | 5/71 (7) | 11/75 (15) |
| Teratoma | 41/71 (58) | 32/75 (43) |
| Histological outcome seminomas, n = 5, n (%) | ||
| Benign/necrosis/fibrosis | 0 | 2/3 (67) |
| Viable seminoma | 2/2 (100) | 1/3 (33) |
| No of resected lymph nodes, median, n (IQR) | 14 (10–18) | 23 (15–28) |
| Malignant lymph node yield, median, n (IQR) | 1 (0–2) | 0 (0–2) |
| Follow-up, median, months (IQR) | 48 (34–58) | 46 (32–64) |
| RPLND: retroperitoneal lymph node dissection; IQR: interquartile range; CS: clinical stage; GCC: germ cell cancer; SWENOTECA: Swedish and Norwegian Testicular Cancer Group. *A modified Royal Marsden clinical staging system was used, where stage D denotes metastases ≥10 cm. **Standard initial chemotherapy for metastatic nonseminoma according to the SWENOTECA protocol is three courses of BEP (bleomycin, etoposide, cisplatin). Intensified classification of treatment comprises stepwise addition of ifosfamide or paclitaxel or high-dose treatment including stem cell reconstitution. |
||
Robot-assisted surgery was used in 69 cases (32%) with a 10% conversion rate (7/69) to open surgery, of which four were post-chemotherapy. Conversion rates did not differ significantly between primary and post-chemotherapy patients (p = 0.4).
Nerve-sparing was applied in 66% (81/123) of unilateral and 4% (3/87) of bilateral templates. Extended surgery was performed in 3% (2/60) of P-RPLND and 11% (17/155) of PC-RPLND, most commonly nephrectomy. Suprahilar dissection occurred in 3% (2/60) of P-RPLND and 19% (30/154) of PC-RPLND.
Operating time
For robotic procedures, operating times did not differ significantly between primary and post-chemotherapy groups, for unilateral (median 178 minutes (IQR 155–260) vs 200 minutes (IQR 116–433, p = 0.8) or bilateral templates (median 235 minutes (IQR 215–490) vs 608 minutes (IQR 435–780, p = 0.6) (Tables 3 and 4). For open surgery, no significant difference was observed for unilateral templates (median 145 minutes (IQR 110–255) vs 160 minutes (IQR 119–216, p = 0.9), but bilateral templates required significant longer operating times in post-chemotherapy patients (median 174 minutes (IQR 153–196) vs 255 minutes (IQR 197–414, p = 0.03)).
| Primary group, n = 61 | ROBOTIC | OPEN | ||||
| Unilateral | Bilateral | Total | Unilateral | Bilateral | Total | |
| Number of cases, n (%) | 33 | 6 | 39 (64) | 18 | 4 | 22 (36) |
| Seminoma, n | 26 | 0 | 26 | 9 | 2 | 11 |
| Nonseminoma, n | 7 | 6 | 13 | 9 | 2 | 11 |
| Conversion laparoscopic to open, n (%) | 2 | 1 | 3 (8) | - | - | - |
| Right-sided unilateral template, n (%) | 13 (39) | - | - | 10 (56) | - | - |
| Left-sided unilateral template, n (%) | 20 (61) | - | - | 8 (44) | - | - |
| Operating time, median (IQR) | 178 (155–260) | 235 (215–490) | 210 (155–260) | 145 (110–255) | 174 (153–196) | 155 (76–245) |
| Intraoperative blood loss, median (IQR) | 50 (20–100) | 50 (50–150) | 50 (20–100) | 120 (100–250) | 210 (110–250) | 125 (100–250) |
| Postoperative nights, median (range) | 2 (1–16) | 3 (2–6) | 2 (1–16) | 6 (2–13) | 6 (6–7) | 6 (2–13) |
| Suprahilar dissection, n (%) | 0 | 1 (17) | 1 (3) | 1 (6) | 0 | 1 (5) |
| Extended surgery*, n (%) | 0 | 0 | 0 | 2 (11) | 0 | 2 (9) |
| Nerve-sparing technique, n (%) | 26 | 1 | 27 (68) | 9 | 0 | 9 (41) |
| Partial | 0 | 0 | 1 | - | 1 (5) | |
| Full | 26 | 1 | 27 (68) | 8 | - | 8 (36) |
| Intraoperative complication, n (%) | 2 | 0 | 2 (5) | 2 | 0 | 2 (9) |
| Postoperative complication, ≤ 30 days, n (%) | 8 | 3 | 11 (28) | 5 | 1 | 6 (27) |
| Postoperative complication, ≥ CDIIIb ≤ 30 days, n (%) | - | 0 | - | 1 | 0 | 1 (5) |
| Clavien-Dindo grade, < 30 days, n | ||||||
| I | 1 | 1 | 1 | 1 | ||
| II | 3 | 2 | 5 | 2 | 1 | 3 |
| IIIa | 4 | 4 | 1 | 1 | ||
| IIIb | ||||||
| IV | 1 | 1 | ||||
| V | ||||||
| Unspecified | 1 | 1 | ||||
| Loss of antegrade ejaculation, n (%) | 10 | 3 | 13 (33) | 5 | 3 | 8 (36) |
| Loss of ant ejaculation, nerve-sparing, n (%) | 9/26 | 1/1 | 10/27 (37) | 3/9 | 0 | 3/9 (33) |
| RPLND: retroperitoneal lymph node dissection; IQR: interquartile range; CD: Clavien-Dindo. *Extended surgery is defined as resection extending outside the template, or removal of organs other than lymph nodes. |
||||||
| Post-chemotherapy group, n = 156 | ROBOTIC | OPEN | ||||
| Unilateral | Bilateral | Total | Unilateral | Bilateral | Total | |
| Number of cases, n (%) | 28 | 2 | 30 (19) | 48 | 78 | 126 (81) |
| Seminoma, n | 2 | 0 | 2 | 0 | 3 | 3 |
| Nonseminoma, n | 26 | 2 | 28 | 48 | 75 | 123 |
| Conversion laparoscopic to open, n (%) | 4 | 0 | 4 (13) | - | - | - |
| Right-sided unilateral template, n (%) | 8 (29) | - | - | 18 | - | - |
| Left-sided unilateral template, n (%) | 20 (71) | - | - | 30 | - | - |
| Operating time, median (IQR) | 200 (116–433) | 608 (435–780) | 224 (121–435) | 160 (119–216) | 255 (197–414) | 215 (151–314) |
| Intraoperative blood loss, median (IQR) | 50 (25–150) | 75 (50–100) | 50 (25–106) | 200 (75–350) | 500 (200–1100) | 350 (150–750) |
| Postoperative nights, median (range) | 3 (1–7) | 4 (3–4) | 3 (1–7) | 6 (1–14) | 7 (2–32) | 6 (2–17 |
| Suprahilar dissection, n (%) | 1 | 0 | 1 (3) | 4 | 25 | 29 (23) |
| Extended surgery*, n (%) | 0 | 0 | 0 | 4 | 17 | 21 (17) |
| Nerve-sparing technique, n (%) | ||||||
| Partial | 3 | 1 | 4 (13) | 1 | 0 | 1 (0.0) |
| Full | 19 | 0 | 19 (63) | 23 | 1 | 24 (19) |
| Intraoperative complication, n (%) | 0 | 0 | 0 | 0 | 6 | 6 (5) |
| Postoperative complication, < 30 days, n (%) | 3 | 0 | 3 | 10 | 39 | 49 (39) |
| Postoperative complication, > CDIIIb < 30 days, n (%) | 0 | 0 | 0 | - | 4 | 4 (4) |
| Clavien-Dindo grade, < 30 days | ||||||
| I | 2 | 2 | 1 | 4 | 5 | |
| II | 1 | 1 | 6 | 16 | 22 | |
| IIIa | 1 | 14 | 15 | |||
| IIIb | 3 | 3 | ||||
| IV | 1 | 1 | ||||
| V | ||||||
| Unspecified | 2 | 1 | 3 | |||
| Loss of antegrade ejaculation, n (%) | 6 | 1 | 7 (23) | 20 | 30 | 50 (40) |
| Loss of ant ejaculation, nerve-sparing, n (%) | 3/22 | 1/1 | 4 (17) | 7/20 | 0 | 7/25 (28) |
| RPLND: retroperitoneal lymph node dissection; IQR: interquartile range; CD: Clavien-Dindo. *Extended surgery is defined as resection extending outside the template, or removal of organs other than lymph nodes. |
||||||
Intraoperative blood loss
For robotic procedures, no significant difference in blood loss was observed between primary and post-chemotherapy patients, for unilateral (median 50 mL (IQR 20–100) vs 50 (IQR 25–150, p = 0.1)) or bilateral templates (median 50 mL (50–150) vs 75 (IQR 50–100, p = 0.8)).
For open surgery, blood loss was comparable for unilateral templates (median 120 mL (IQR 100–250) vs 200 mL (IQR 75–350, p = 0.3)), while post-chemotherapy patients undergoing bilateral templates experienced greater blood loss, reaching statistical significance (median 210 mL (IQR 110–250) vs 500 mL (IQR 200–1100, p = 0.03)) (Tables 3 and 4).
Intraoperative complications
Intraoperative complications for P-RPLND occurred in 8% (4/50) of unilateral and 0% of bilateral templates. For PC-RPLND, rates were 0 and 8% (6/79), respectively. Intraoperative complications rates did not differ by surgical approach (P-RPLND 2 vs 2, p = 0.5; PC-RPLND 0 vs 6, p = 0.2) (Tables 3 and 4). When stratifying for nonseminoma PC-RPLND, the rate of intraoperative complications was 0% for unilateral and 8% (6/76) for bilateral templates. Renal injury was the most frequent event.
Postoperative complications
For P-RPLND, 30-day postoperative complications occurred in 26% (13/51) of unilateral and 40% (4/10) of bilateral templates (p = 0.2). Of these 17 patients, 1 (2%) experienced CD grade ≥ IIIb (Table 3). No significant difference was observed between robot-assisted and open P-RPLND (p = 1.0).
For PC-RPLND, complications were observed in 18% (13/74) of unilateral and 49% (39/79) of bilateral templates (p < 0.01). CD grade ≥ IIIb complications were 0% for unilateral and 5% (4/79) for bilateral templates (Table 4). Open surgery was associated with a higher complication rate than robot-assisted PC-RPLND (p < 0.01). In nonseminoma PC-RPLND, CD grade ≥ IIIb was 0% for unilateral and 10% (4/39) for bilateral templates.
No grade V complications occurred. Infection requiring antibiotics was the most frequent postoperative complication.
Length of hospital stay
Median hospital stay was 2 nights (IQR: 2–3) for robotic procedures and 6 nights (IQR: 5–8) for open surgery (p = 0.0), with no significant difference based on prior chemotherapy (robot- assisted p = 0.2; open p = 1.0).
For P-RPLND, intraoperative complications significantly prolonged median stay: robotic 6 nights (IQR: 5–6) vs 2 (IQR: 2–3) (p = 0.02) and open 11 nights (IQR: 8–13) vs 6 (IQR: 4–6) (p = 0.02) (Table 3). In open PC-RPLND, intraoperative complications increased median stay to 12 nights (IQR: 8–16) vs 6 (IQR: 5–8) (p < 0.01) (Table 4). No intraoperative complications occurred in robot-assisted PC-RPLND, precluding any comparison in that group.
Postoperative complications did not affect P-RPLND stay (robotic p = 0.2; open p = 0.2). In open PC-RPLND, postoperative complications increased stay to 8 nights (IQR: 6–9) vs 6 (IQR: 4–7) (p < 0.01), but no effect was observed in robot-assisted PC-RPLND (p = 0.2).
Loss of antegrade ejaculation
Data on lost antegrade ejaculation were available for 86% of patients (187/217). For P-RPLND, 36% (21/61) experienced loss. Rates were 31% (15/49) for unilateral and 60% (6/10) for bilateral templates. No significant difference was observed between robotic and open approaches (unilateral p = 0.8; bilateral p = 0.4) for P-RPLND. There was a significantly larger proportion of patients with lost antegrade ejaculation in left-sided templates (13/28) compared to right-sided templates (2/23), (p < 0.01, OR 9.2, 95% CI = 1.8–48).
In the post-chemotherapy group, 45% (57/128) reported lost antegrade ejaculation: 38% (26/69) for unilateral and 53% (31/59) for bilateral templates. Nerve-sparing significantly reduced anejaculation rates in unilateral templates (p = 0.02) but not in bilateral (p = 0.3). For nonseminoma PC-RPLND, rates were similar (unilateral 38%, bilateral 52%). Robotic surgery had no significant impact on preserved ejaculation in this group (unilateral p = 0.16; bilateral p = 0.9), and no difference was seen between right- and left-sided templates (p = 0.4).
Histological outcome
In seminoma P-RPLND, 95% (35/37) had viable cancer; the remaining two had benign nodes. Median lymph node yield was 11 (IQR: 8–17) with a median of 1 malignant node (IQR: 1–2) (Table 1). In nonseminoma P-RPLND, 52% (12/23) had viable cancer, 30% (7/23) teratoma, and 17% (2/23) benign nodes, with median yield 18 (IQR: 11–30) and 1 malignant node.
For nonseminoma PC-RPLND, the rate of viable cancer was 11% (16/145), teratoma 50% (72/145), and benign histology 39% (57/145) (Table 2). Among the five seminoma patients who underwent post-chemotherapy RPLND, three had viable cancer (60%).
Discussion
This population-based study provides a comprehensive analysis of all RPLNDs performed in 5 years, focusing on complications and the accuracy of patient selection. The cohort reflects a novel treatment strategy, particularly the use of primary RPLND for low-stage, low-volume disease, explaining shorter follow-up for seminoma patients. Robotic procedures, performed in about a third of cases, also influenced outcomes. Overall, intraoperative complications were low. For nonseminoma PC-RPLND, our 4% complication rate is comparable to 3.7% reported by Cary et al. in an 8-year analysis of a high-volume center [18]. Stratifying for CD grade ≥ IIIb, our postoperative complication rate remains modest, at 3% for post-chemotherapy patients. In a recent study from the UK, another centralized region, the rate of more severe complications in post-chemotherapy cases reached 7% [27].
Chemotherapy prior to RPLND did not significantly affect robotic outcomes in operating time, blood loss, or complications. Open bilateral PC-RPLND, however, was associated with higher blood loss and complications, likely reflecting more advanced disease. Chemotherapy did not significantly affect the robotic conversion rate, though our limited sample size of seven converted patients restricts definitive conclusions. In contrast, a recent systematic review by Garg et al. reported conversion rates of 2.2% for robotic P-RPLND and a notably higher 9% for PC-RPLND [16], suggesting that chemotherapy may impact conversion in other settings. The most common reasons for reduced surgical accessibility, which led to conversion from laparoscopic to open surgery, were either a high amount of intra-abdominal fat or densely adherent tissues following chemotherapy, which posed a risk of oncological compromise. Our findings further demonstrate that robotic RPLND was associated with shorter hospital stays compared to open surgery, irrespective of prior chemotherapy or template extension. While intraoperative complications notably extended hospital stays, postoperative complications did not significantly impact stay duration in primary cases. Overall, robotic procedures showed no increase in intra- or postoperative complications, likely due to selective application in cases with less advanced disease. We conclude that robot-assisted RPLND is safe with careful patient selection.
Aligning with previous knowledge, the rate of lost antegrade ejaculation in this cohort was markedly higher following bilateral templates, both pre- and post-chemotherapy. Importantly, nerve-sparing efforts in bilateral templates did not overall improve antegrade ejaculation outcomes although the sample size is small. Among patients who underwent unilateral P-RPLND, the incidence of lost antegrade ejaculation was approximately 10-fold higher in left-sided templates, a finding not observed in PC-RPLND cases. However, this subset of primary unilateral RPLNDs is small; therefore this finding must be interpreted with caution.
Comparing our results to previously reported data [15], histopathological outcomes after RPLND are comparable only for nonseminoma PC-RPLND, since most of the remaining patients in this cohort were treated based on novel indications. The rate of viable cancer remains steady at 11%, with a slight increase in teratoma rate (50% vs. 45%) and a slight decrease in benign findings (39% vs. 44%), though these differences are not statistically significant. The rate of lost antegrade ejaculation remains comparable to prior results, with 52% loss in bilateral templates (previously 59%) and 38% in unilateral templates (previously 32%). Severe complication rates (CD grade ≥ IIIb) remain unchanged, with 0% in unilateral templates (previously 2%) and 10% in bilateral templates (previously 8%). The most notable improvement observed is the reduction in intraoperative complications, with no complications recorded in unilateral templates (previously 4%) and a reduction from 14 to 8% in bilateral templates. This decline may be due to the increased use of minimally invasive surgical technique.
In this analysis, we have not reported data on relapses after RPLND. Relapses are particularly relevant for primary RPLNDs, as relapses after post-chemotherapy RPLND are very rare. The relapse data for primary RPLND in seminoma patients has been published previously by our group [5], showing a relapse rate of 10% among the first 62 patients operated between 2019 and 2022.
This study has several limitations, the most notable being the cohort’s heterogeneity and the sample size. Nevertheless, it represents a relatively large group for advanced TGCC and is population-based and centralized with respect to surgery.
In summary, this evaluation of surgery for advanced testicular cancer demonstrates a reduction in intraoperative complications alongside an increase in minimally invasive procedures, subsequently reducing hospital stays. The establishment of a national consensus conference, providing treatment recommendations for all cases considered for RPLND, appears to have improved patient selection and resulting in fewer benign outcomes. Reevaluation in 5 years could provide further insight into potential advancements or lack thereof.
Conclusions
In conclusion, intra- and postoperative complications following RPLND at our centers are low, with serious complications being rare. The rate of lost antegrade ejaculation increases with bilateral templates, consistent with our previous findings and other published data. The increasing use of robotic surgery has contributed to better outcomes, and prior chemotherapy did not pose a barrier to laparoscopic procedures. In PC-RPLND, we observed a trend toward increased rates of teratoma and viable cancer, with a corresponding decrease in benign histopathological findings. Overall, careful patient selection is crucial for RPLND, and close monitoring of outcomes remains essential.
Author contributions
Anna Thor had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Thor, Cohn-Cedermark, Kjellman, Lundgren.
Acquisition of data: Thor, Grenabo Bergdahl, Abniki, Gerdtsson.
Analysis and interpretation of data: Thor, Kjellman, Lundgren.
Drafting of the manuscript: Thor.
Critical revision of the manuscript for important intellectual content: Thor, Grenabo Bergdahl, Gerdtsson, Glimelius, Hellström, Jansson, Johannsdottir, Tandstad, Cohn-Cedermark, Kjellman, Lundgren.
Statistical analysis: Thor, Kjellman, Lundgren.
Obtaining funding: Thor, Kjellman.
Administrative, technical or material support: Thor, Kjellman, Lundgren.
Supervision: Cohn-Cedermark, Kjellman, Lundgren.
Other: none.
Disclosures
The authors report no financial relationships or activities relevant to this article.
References
- [1] Gurney JK, Florio AA, Znaor A, et al. International trends in the incidence of testicular cancer: lessons from 35 years and 41 countries. Eur Urol. 2019;76(5):615–623. https://doi.org/10.1016/j.eururo.2019.07.002
- [2] Socialstyrelsen. The National Board of Health and Welfare in Sweden [Internet]. 2024. Available from: https://www.socialstyrelsen.se/en/statistics-and-data/statistics/statistical-databases/
- [3] Olofsson SE, Tandstad T, Jerkeman M, et al. Population-based study of treatment guided by tumor marker decline in patients with metastatic nonseminomatous germ cell tumor: a report from the Swedish-Norwegian Testicular Cancer Group. J Clin Oncol. 2011;29(15):2032–2039. https://doi.org/10.1200/JCO.2010.29.1278
- [4] SWENOTECA. SWENOTECA X management program of testicular cancer [Internet]. 2020. Available from: www.swenoteca.org
- [5] Thor A, Negaard HFS, Grenabo Bergdahl A, et al. Primary retroperitoneal lymph node dissection as treatment for low-volume metastatic seminoma in a population-based cohort: the Swedish Norwegian Testicular Cancer Group Experience. Eur Urol Open Sci. 2024;65: 13–19. https://doi.org/10.1016/j.euros.2024.05.006
- [6] Hiester A, Che Y, Lusch A, et al. Phase 2 single-arm trial of primary retroperitoneal lymph node dissection in patients with seminomatous testicular germ cell tumors with clinical stage IIA/B (PRIMETEST). Eur Urol. 2023;84(1):25–31. https://doi.org/10.1016/j.eururo.2022.10.021
- [7] Daneshmand S, Cary C, Masterson T, et al. Surgery in early metastatic seminoma: a phase II trial of retroperitoneal lymph node dissection for testicular seminoma with limited retroperitoneal lymphadenopathy. J Clin Oncol. 2023;41(16):3009–3018. https://doi.org/10.1200/JCO.22.00624
- [8] Heidenreich A, Paffenholz P, Hartmann F, et al. Retroperitoneal lymph node dissection in clinical stage IIA/B metastatic seminoma: results of the COlogne Trial of Retroperitoneal Lymphadenectomy In Metastatic Seminoma (COTRIMS). Eur Urol Oncol. 2024;7(1):122–127. https://doi.org/10.1016/j.euo.2023.06.004
- [9] Tachibana I, Alabd A, Tong Y, et al. Primary retroperitoneal lymph node dissection for stage II seminoma: is surgery the new path forward? J Clin Oncol. 2023;41(23):3930–3938. https://doi.org/10.1200/JCO.22.01822
- [10] Matulewicz RS, Benfante N, Funt SA, et al. Primary retroperitoneal lymph node dissection for seminoma metastatic to the retroperitoneum. J Urol. 2024;211(1):80–89. https://doi.org/10.1097/JU.0000000000003697
- [11] Gerdtsson A, Negaard HFS, Almås B, et al. Initial surveillance in men with marker negative clinical stage IIA non-seminomatous germ cell tumours. BJU Int. 2024;133(6):717–724. https://doi.org/10.1111/bju.16289
- [12] Haugnes HS, Bosl GJ, Boer H, et al. Long-term and late effects of germ cell testicular cancer treatment and implications for follow-up. J Clin Oncol. 2012;30(30):3752–3763. https://doi.org/10.1200/JCO.2012.43.4431
- [13] Hellesnes R, Kvammen Ø, Myklebust T, et al. Continuing increased risk of second cancer in long-term testicular cancer survivors after treatment in the cisplatin era. Int J Cancer. 2020;147(1):21–32. https://doi.org/10.1002/ijc.32704
- [14] Kvammen Ø, Myklebust T, Solberg A, et al. Long-term relative survival after diagnosis of testicular germ cell tumor. Cancer Epidemiol Biomarkers Prev. 2016;25(5):773–779. https://doi.org/10.1158/1055-9965.EPI-15-1153
- [15] Gerdtsson A, Håkansson U, Törnblom M, et al. Surgical complications in postchemotherapy retroperitoneal lymph node dissection for nonseminoma germ cell tumour: a population-based study from the Swedish Norwegian Testicular Cancer Group. Eur Urol Oncol. 2020;3(3):382–389. https://doi.org/10.1016/j.euo.2019.08.002
- [16] Garg H, Mansour AM, Psutka SP, et al. Robot-assisted retroperitoneal lymph node dissection: a systematic review of perioperative outcomes. BJU Int. 2023;132(1):9–30. https://doi.org/10.1111/bju.15986
- [17] Heidenreich A, Pfister D, Witthuhn R, et al. Postchemotherapy retroperitoneal lymph node dissection in advanced testicular cancer: radical or modified template resection. Eur Urol. 2009;55(1):217–224. https://doi.org/10.1016/j.eururo.2008.09.027
- [18] Cary C, Masterson TA, Bihrle R, et al. Contemporary trends in postchemotherapy retroperitoneal lymph node dissection: additional procedures and perioperative complications. Urol Oncol. 2015;33(9):389.e15–e21. https://doi.org/10.1016/j.urolonc.2014.07.013
- [19] Subramanian VS, Nguyen CT, Stephenson AJ, et al. Complications of open primary and post-chemotherapy retroperitoneal lymph node dissection for testicular cancer. Urol Oncol. 2010;28(5):504–509. https://doi.org/10.1016/j.urolonc.2008.10.026
- [20] Djaladat H, Nichols C, Daneshmand S. Adjuvant surgery in testicular cancer patients undergoing postchemotherapy retroperitoneal lymph node dissection. Ann Surg Oncol. 2012;19(7):2388–2393. https://doi.org/10.1245/s10434-012-2284-8
- [21] Mosharafa AA, Foster RS, Koch MO, et al. Complications of post-chemotherapy retroperitoneal lymph node dissection for testis cancer. J Urol. 2004;171(5):1839–1841. https://doi.org/10.1097/01.ju.0000120141.89737.90
- [22] Pettus JA, Carver BS, Masterson T, et al. Preservation of ejaculation in patients undergoing nerve-sparing postchemotherapy retroperitoneal lymph node dissection for metastatic testicular cancer. Urology. 2009;73(2):328–331; discussion 31–32. https://doi.org/10.1016/j.urology.2008.08.501
- [23] Wells H, Hayes MC, O’Brien T, et al. Contemporary retroperitoneal lymph node dissection (RPLND) for testis cancer in the UK – a national study. BJU Int. 2017;119(1):91–99. https://doi.org/10.1111/bju.13569
- [24] Carver BS, Serio AM, Bajorin D, et al. Improved clinical outcome in recent years for men with metastatic nonseminomatous germ cell tumors. J Clin Oncol. 2007;25(35):5603–5608. https://doi.org/10.1200/JCO.2007.13.6283
- [25] Gerdtsson A, Thor A, Grenabo Bergdahl A, et al. Unilateral or bilateral retroperitoneal lymph node dissection in nonseminoma patients with postchemotherapy residual tumour? Results from RETROP, a Population-based Mapping Study by the Swedish Norwegian Testicular Cancer Group. Eur Urol Oncol. 2022;5(2):235–243. https://doi.org/10.1016/j.euo.2021.02.002
- [26] Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–196. https://doi.org/10.1097/SLA.0b013e3181b13ca2
- [27] Pearce AK, Manson-Bahr D, Reid A, et al. Outcomes of postchemotherapy retroperitoneal lymph node dissection from a high-volume UK centre compared with a national data set. Eur Urol Open Sci. 2021;33:83–88. https://doi.org/10.1016/j.euros.2021.09.005