ORIGINAL REPORT
Effects of Magnoliae Flos on Atopic Dermatitis-Like Inflammation Evaluated via Extracellular Signal-regulated Kinase or Signal Transducers and Activators of Transcription 1/3 Signalling Pathways
Tae-Young GIL1, Bo-Ram JIN1, Yea-Jin PARK2, Hye-Min KIM2 and Hyo-Jin AN1,3
1Department of Oriental Pharmaceutical Science, College of Pharmacy, Kyung Hee University, Dongdaemun-gu, Seoul, 2Department of Rehabilitative Medicine of Korean Medicine and Neuropsychiatry, College of Korean Medicine, Sangji University, Wonju, Gangwon state and 3Department of Integrated Drug Development and Natural Products, Graduate School, Kyung Hee University, Seoul,Republic of Korea
Atopic dermatitis is a chronic inflammatory skin disease. Skin is the largest organ and plays a pivotal role in protecting the body. Not only does the skin act as a physical barrier against the external environment, but it also has its own immune system. Atopic dermatitis is caused by prolonged excessive inflammatory responses that worsen under imbalanced cutaneous immune system skin conditions. Although the prevalence and burden of atopic dermatitis is increasing, the standard therapeutic agents remain unclear due to the complicated pathophysiology of the condition. The objective of this study is to examine the use of Magnoliae flos, the dried flower bud of Magnolia biondii or related plants. The effects and underlying mechanism of action of aqueous extract of the buds of Magnoliae flos (MF) were evaluated. Immortalized human keratinocytes (HaCaT) stimulated with tumour necrosis factor-α and interferon-γ mixture and NC/Nga mice stimulated with 2,4-dinitrochlorobenzene were used as atopic dermatitis models, in vitro and in vivo, respectively. The effects of MF were determined by measuring the suppression of pro-inflammatory signalling pathways, such as extracellular signal-regulated kinase or signal transducers and activators of transcription 1/3 and restoring skin barrier molecules. In conclusion, MF is a potential therapeutic alternative for the treatment of atopic dermatitis through repressing inflammatory pathways.
SIGNIFICANCE
The chronic inflammatory skin disease atopic dermatitis reduces patients’ quality of life due to severe effects of itch and frequent relapse. There is a need for therapeutic agents that will relieve the symptoms of atopic dermatitis in the long-term without side-effects. Magnoliae flos has been used for a long time to treat inflammatory diseases with safety and low toxicity for; therefore this study examined its potential in treating atopic dermatitis. MF showed repressive effects on cutaneous inflammatory responses and recovery on skin barrier molecules which were done regulating through ERK or STAT1/3 signalling pathways.
Key words: atopic dermatitis; inflammation; Magnoliae flos; MAPK; STATs.
Citation: Acta Derm Venereol 2023; 103: adv11593. DOI: https://doi.org/10.2340/actadv.v103.11593.
Copyright: © Published by Medical Journals Sweden, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: Sep 12, 2023; Published: Nov 13, 2023
Corr: Hyo-Jin An, Department of Oriental Pharmaceutical Science, College of Pharmacy, Kyung Hee University, 26 Kyungheedae-ro, Dongdaemun-gu, Seoul 02447, Republic of Korea. E-mails: hjan@khu.ac.kr; sangjipharm@gmail.com
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
A topic dermatitis (AD) is a chronic relapsing inflammatory skin disorder characterized by recurrent eczematous lesions and severe pruritus (1). AD is associated with complex interactions of innate and adaptive responses, resulting in immunological abnormalities following immunoglobulin (Ig)E sensitization or epithelial barrier dysfunction (2, 3). The incidence and prevalence of AD has increased over recent decades (4). The Global Burden of Disease study reported that skin and related diseases are the world’s fourth major cause of non-fatal disease burden (5). In addition to the intense itch and skin lesions, the socioeconomic burden of AD reduces an individual’s quality of life (6). There is currently no cure for AD; however, there are treatments and resolution strategies that aim to alleviate symptoms, avoid trigger factors, or repair barriers in combination with anti-inflammation (7).
One of the first-line therapeutic treatments for AD is corticosteroids, such as dexamethasone (Dexa.) (8). Despite effectiveness in treating flare-ups of AD, the prolonged use of corticosteroids may cause side-effects, such as acne, delayed wound healing, or skin atrophy (9). Other therapeutic agents for AD target the Janus kinase and signal transducer and activator of transcription protein (JAK-STAT) pathways, which are implicated in inflammatory or neoplastic skin diseases (10). Upadacitinib, baricitinib, and abrocitinib are small molecules that inhibit the JAK-STAT pathway by specifically blocking JAK1 or JAK2 (11). Phase III clinical trials are ongoing for these therapies.
Magnoliae flos aqueous extract (MF) is proposed in the current study as a potential alternative for the treatment of AD owing to its targeting of the JAK-STAT signalling pathways. MF is extracted from the dried flower buds of Magnolia biondii or related plants and has been used to treat allergic rhinitis, sinusitis, and headaches (12). MF contains magnolin, which inhibits allergy (13) or has anti-inflammatory activity (14). However, the effect of MF on AD has not been elucidated.
Since AD is known to be a chronic inflammatory disease, therapeutic treatments have focused on regulating immunological responses (7). A skewed balance between T helper type 2 (Th2) and T helper type 1 (Th1) cells occurs in the presence of a specific immunoglobulin E (IgE) response in inflammatory diseases, and many pro-inflammatory cytokines and chemokines are produced in AD (15). The secretion of interleukin (IL)-4, 5, and 13 by B-cells, eosinophils and mast cells indicates acute AD (16). In addition, a predominance of Th1 cells or interferon (IFN)-γ leads to chronic AD skin lesions that undergo tissue remodelling related to chronic inflammation (17).
The management of AD requires the control of inflammation and maintenance of the homeostasis of the skin barrier. The skin is the largest organ of the body and forms a protective outermost layer, acting as physical barrier and possessing a healthy balance of various immune cells (18). Molecular defects occur in the dermal layers, including dysregulation of cornified envelope proteins, stratum corneum lipids, or tight junctions. In the epidermis, excessive activation of serine proteases, including kallikreins (KLKs), disrupts the skin barrier by conserving homeostasis through desquamation, anti-microbial defence, or innate immune responses (19). The current study used a model based on HaCaT immortalized cells as an in vitro representation of human skin with fewer variabilities.
AD is a multifactorial disease with a complicated aetiology; hence a range of different approaches to disease management may be needed. The objective of this study was to determine the effects on AD of treatment with MF, including the effects on regulation of inflammation and restoration of the skin barrier. The effects of MF were studied in vitro in HaCaT keratinocytes stimulated with a mixture of tumour necrosis factor (TNF)-α and IFN-γ (TNF-α+IFN-γ), and in vivo in 2,4-dinitrochlorobenzen (DNCB)-induced NC/Nga mice.
MATERIALS AND METHODS
Preparation of Magnoliae flos aqueous extract
The dried flower buds of M. biondii (Nanumherb, Cheongju-si, Chungcheongbuk-do, Republic of Korea) (HA1801980101, country of origin: China) were processed and extracted with water at 100°C for 4 h using a reflux condenser. The extract was filtered with Whatman filter paper (Cytiva Whatman, USA and lyophilized. The percentage yield was 13.73% w/w. The freeze-dried MF was dissolved with saline prior to treating the cells (125, 250, 500 μg/mL) or mice (100, 200 mg/kg).
Cell culture and MF treatment
HaCaT keratinocytes were pre-treated with MF for 1 h. Thereafter, the mRNA and total protein expression levels were determined using qRT-PCR and western blot, respectively.
2,4-dinitrochlorobenzen-induced atopic dermatitis animal model and treatment
Female 6-week-old NC/Nga mice, weight 18–23 g (Daehan Biolink Co., Daejeon, Korea) were kept under standard conditions according to the guidelines adopted and promulgated by Sangji University, Wonju-si, Gangwon-do, Korea. Prior to the study, all the experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of Sangji University (approval protocol number: 2020-14; 17 September 2020). The mice were housed and maintained at a temperature of 20–25°C, humidity of 40–60 %, and a 12-h light/dark cycle. The mice were randomly assigned to 5 groups (6-7 mice per group): (i) control; (ii) 2,4- dinitrochlorobenzene (DNCB)-induced AD model; (iii) DNCB-induced AD model with dexamethasone (5 mg/kg) as positive control (DNCB+ Dexa.); (iv) DNCB-induced AD model with MF (100 mg/kg) (DNCB+MF100); and (v) DNCB-induced AD model with MF (200 mg/kg) (DNCB+MF200). To induce AD-like skin inflammation, the dorsal skin was topically sensitized with 100 μL 1% DNCB, twice in the first week (on days 1 and 4). For the process of challenging, 100 μL 0.4% DNCB was applied topically 3 times a week for an additional 3 weeks. Normal saline was administered to the control group during the experimental period. The mice in the 5 groups were euthanized by cervical dislocation 4 weeks after first sensitization with DNCB. On the day of sacrificing the animal after 4 weeks, blood and dorsal skin tissues were collected for further assays.
Statistical analysis
Data were expressed as the mean ± standard error of the mean (SEM) or standard deviation (SD). Comparisons among groups were carried out using 1-way analysis of variance (ANOVA) followed by Dunnett’s post-hoc test served in GraphPad Prism5 (GraphPad Software, San Diego, CA, USA). p-values < 0.05 were considered statically significant.
The materials and methods have been published previously (20).
RESULTS
Effects of Magnoliae flos on pro-inflammatory cytokines and related proteins in human keratinocytes
As a representative chronic inflammatory disease, AD is associated with the production of pro-inflammatory cytokines and proteins. The effects of MF on pro- inflammatory cytokines in human keratinocytes were thus determined. The mRNA levels of Th1-related cytokines, such as TNF-α and IL-6, increased on stimulation with TNF-α+IFN-γ and decreased with pretreatment with 250 μg/mL and 500 μg/mL MF (Fig. 1A). Based on the results of pro-inflammatory cytokines at the transcriptional level, the pro-inflammatory cytokines and their related proteins were determined at the translational level (Fig. 1B). Using enzyme-linked immunoassay (ELISA) kits, increased production of TNF-α and IL-6 was found in the supernatant from TNF-α+IFN-γ-stimulated HaCaT keratinocytes. However, production of TNF-α and IL-6 was significantly downregulated by treatment with MF at 250 μg/mL and 500 μg/mL concentrations (Fig. 1B). The relative mRNA expression levels of IL-4 and IL-13, which are Th-2 specific cytokines (Fig. 1C), as well as IL-17 and IL-22, which are Th-17-associated cytokines (Fig. 1D), were increased by TNF-α+IFN-γ. Furthermore, 250 μg/mL or 500 μg/mL of MF inhibited the relative mRNA expression of the induced cytokine production. The effects of MF on mRNA expression of AD-specific chemokines, such as regulation upon activation, normal T cell expression and secretion (RANTES/CCL5) or macrophage-derived chemokine (MDC/CCL22) were determined. The relative levels of the mRNAs were upregulated by TNF-α+IFN-γ and downregulated by MF treatment in HaCaT keratinocytes in a dose-dependent manner (Fig. 1E). In addition to Th1-specific cytokines, the effects of MF on Th2 skewing inflammatory responses and related proteins were determined. One of them is thymic stromal lymphopoietin (TSLP), a critical initial molecule in allergic skin inflammation in keratinocytes (21). Increased TSLP production and mRNA expression were observed in TNF-α+IFN-γ-stimulated HaCaT keratinocytes. However, these changes were suppressed by treatment with MF (Fig. 1F and 1G). As markers involved in keratinocyte differentiation and inflammation, upregulated periostin and Akt phosphorylation levels were induced by TNF-α+ IFN-γ stimulation. Furthermore, MF repressed the activation of Akt and periostin in human immortalized keratinocytes (Fig. 1H).
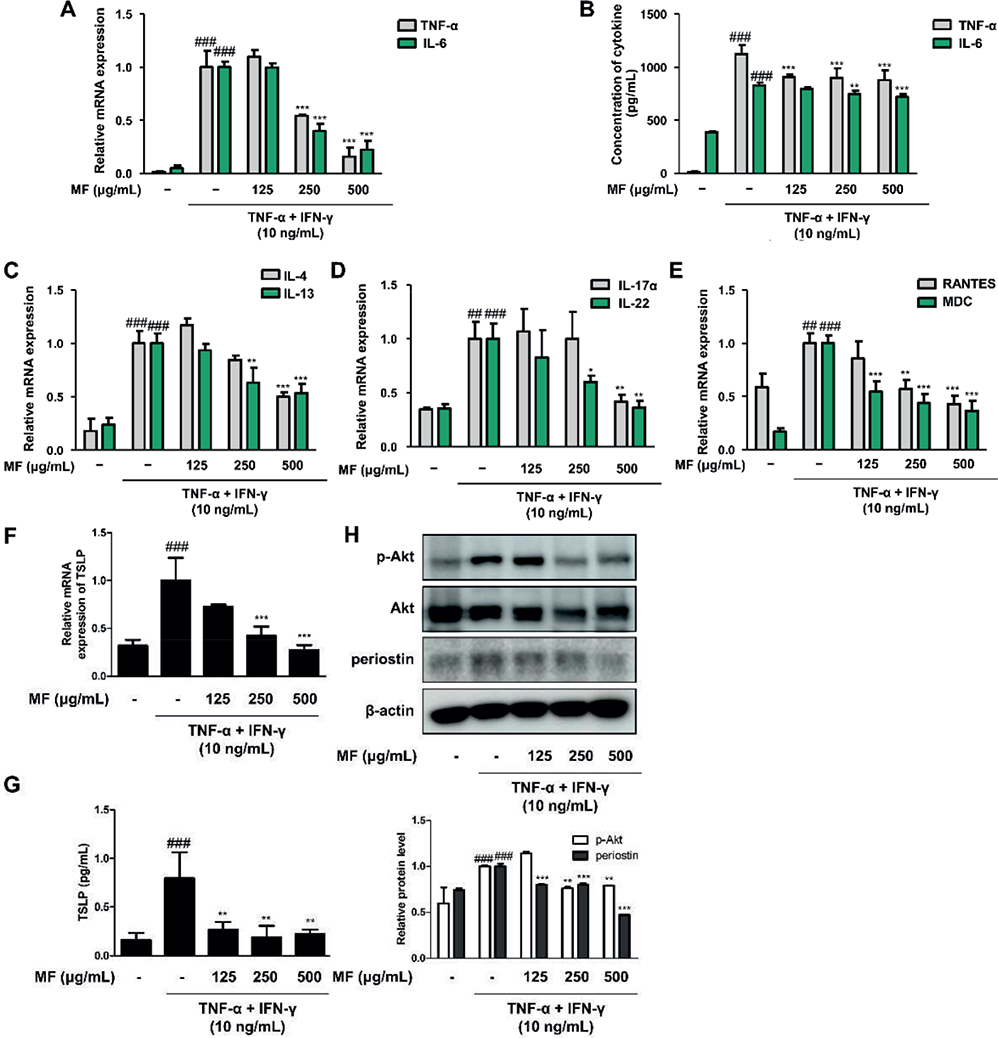
Fig. 1. Effects of Magnoliae flos (MF) on pro-inflammatory cytokines and proteins in tumour necrosis factor (TNF)-α+ interferon (IFN)-γ-stimulated immortalized human keratinocytes (HaCaT). The mRNA levels of: (A) T helper type 1 (Th1)-related cytokines, TNF-α and interleukin (IL)-6; (C) Th2-related cytokines, interleukin (IL)-4 and IL-13; (D) Th17-related cytokines, IL-17a and IL-22; and (E) AD-related chemokines, regulation upon activation, normal T cell expression and secretion (RANTES) and macrophage-derived chemokine (MDC); and (F) thymic stromal lymphopoietin (TSLP) were analysed by quantitative reverse transcription-PCR (qRT-PCR). Three repeats of an experiment were conducted. Cells were pre-treated with 125, 250, or 500 μg/mL MF for 1 h prior to the addition of TNF-α+IFN-γ and incubated for 18–24 h (B, G) and 10–30 min (H). Production of pro-inflammatory cytokines, such as (B) TNF-α, IL-6, and (G) TSLP, were examined using EIA kits. (H) Total proteins were prepared and western blotted for p-Akt, Akt, and periostin. Akt and β-actin were used as internal controls. Densitometric analysis was performed using ImageJ software (1.49, Java program, NIH Image). Data are presented as mean ± standard error of the mean (SEM). ##p < 0.01, ###p < 0.001 vs the control group; *p < 0.05, **p < 0.01, ***p < 0.001 vs the TNF-α+IFN-γ-stimulated group.
Effects of Magnoliae flos on skin barrier molecules and serine proteases in human keratinocytes
As AD is caused by a disrupted skin barrier and is elucidated as a cornified cell envelope composed of filaggrin (FLG), loricrin (LOR), involucrin (INV), and other proteins, these proteins were studied. Stimulation by the TNF-α+IFN-γ mixture downregulated the expression of skin barrier molecules; however, mRNA (Fig. 2A–C) and protein (Fig. 2D) expression levels were restored by treatment with MF. In addition, mRNA expression levels of KLK5 and KLK7 were determined during TNF-α+IFN-γ-induced inflammation. KLKs play a role in regulating skin desquamation and inflammation during cutaneous repair. Their increased expression by pro-inflammatory stimulation was downregulated by treatment with MF (Fig. 2E and F).
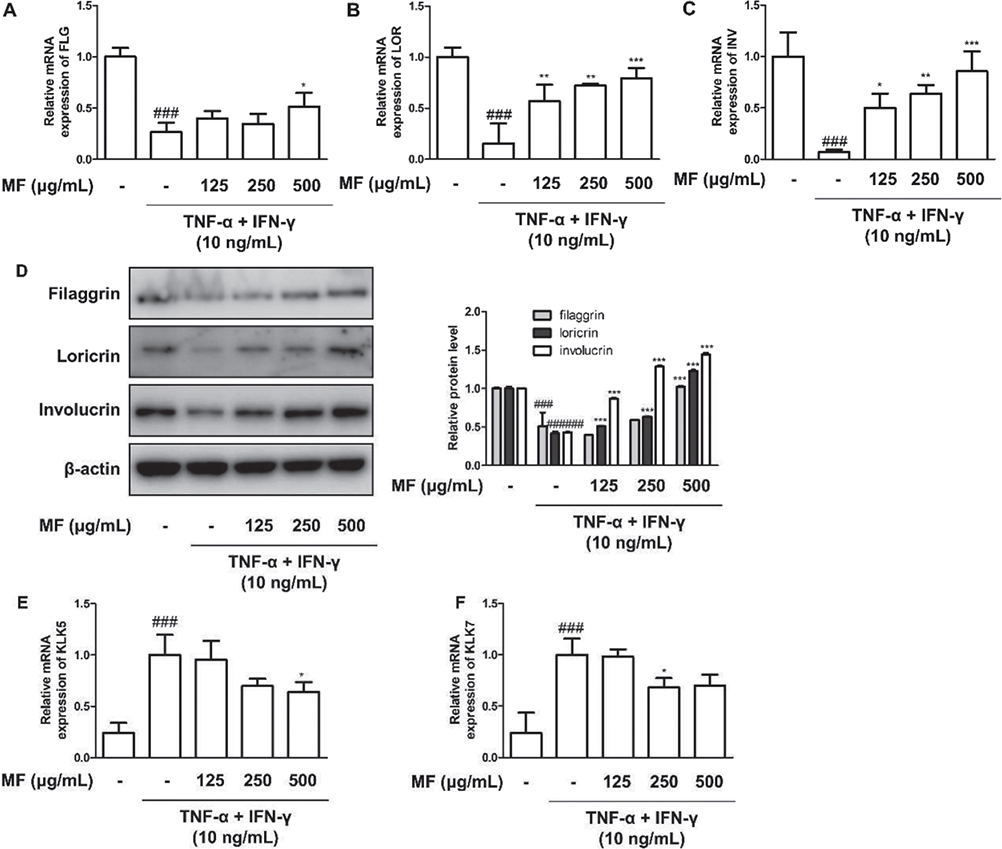
Fig. 2. Effects of Magnoliae flos (MF) on skin barrier molecules in tumour necrosis factor (TNF)-α+ interferon (IFN)-γ-stimulated immortalized human keratinocytes (HaCaT). Cells were pre-treated with 125, 250, or 500 μg/mL MF for 1 h prior to the addition of TNF-α+IFN-γ and incubated for 18–24 h. The mRNA level of: (A) filaggrin (FLG), (B) loricrin (LOR), (C) involucrin (INV), (E) kallikrein-related peptidase 5 (KLK5), and (F) KLK7 were quantified using quantitative reverse transcription-PCR (qRT-PCR). (D) Total protein of skin barrier molecules including filaggrin, loricrin, and involucrin were evaluated with western blot assay. β-actin was used as an internal control. Densitometric analysis was performed using ImageJ software. The data shown represent mean ± standard error of the mean (SEM) of triplicate independent experiments (n = 3). ###p < 0.001 vs the control group; *p < 0.05, **p < 0.01, ***p < 0.001 vs the TNF-α+IFN-γ-stimulated group.
Effects of Magnoliae flos on phosphorylation of NF-κB, MEK1/2-ERK1/2 MAPK, and JAK2-STAT1/3 signalling pathways in human keratinocytes
Focusing on the inflammatory disease (22), the effects MF was evaluated the suppressing the phosphorylated NF-κB, MAPK, and STAT1/3 in protein expressions in HaCaT cells (22).
NF-κB p65 translocation from the nucleus to the cytosol was found at the protein level in TNF-α+IFN-γ-stimulated HaCaT keratinocytes. In the mammalian system, which has another pro-inflammatory signalling pathway, MAPKs have 3 well-known subfamilies: extracellular signal-regulated kinases (ERKs), c-Jun N-terminal kinases (JNKs), and p38 MAPKs. The 3 subunits were thus assessed according to their influence on MF. Only ERK1/2 phosphorylation was found to be downregulated by MF during TNF-α+IFN-γ-induced inflammation, and was observed when MF concentrations of 125 μg/mL and 500 μg/mL were administered. As MAPKs are activated by MAPK kinases (MAPKKs) through serial phosphorylation, the current study evaluated the effect of MF on the phosphorylation of MEK1/2, a MAPKK. MF was found to inhibit the activation of MEK1/2 at the highest concentration of MF alone (Fig. 3B). As an immune-mediated signalling pathway, the JAK-STAT signalling pathway is involved in AD (23). In the current study, the phosphorylation of JAK2, STAT1, and STAT3 was assessed at the protein level. JAK2 activation by TNF-α+ IFN-γ stimulation was suppressed in a dose-dependent manner by pre-treatment with MF (Fig. 3C). Phosphorylated STAT1 and STAT3 on both tyrosine and serine by TNF-α+IFN-γ had increased protein levels; however, MF downregulated their expression (Fig. 3C).
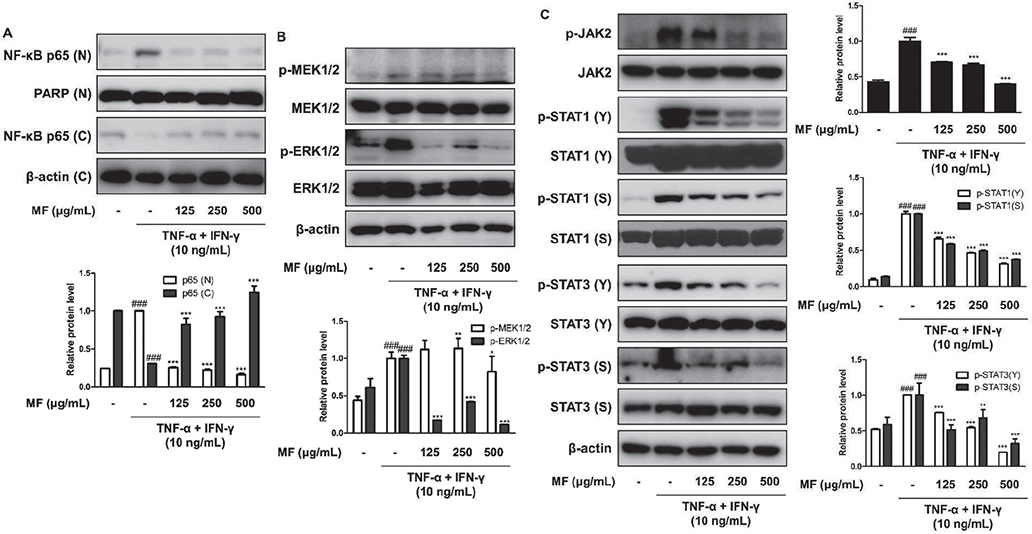
Fig. 3. Effects of Magnoliae flos (MF) on activation of NF-κB, MEK-ERK, and Janus kinase-signal transducers and activators of transcription (STAT) signalling pathways in tumour necrosis factor (TNF)-α+ interferon (IFN)-γ-stimulated immortalized human keratinocytes (HaCaT). Cells were pre-treated with 125, 250, or 500 μg/mL MF for 1 h prior to the addition of TNF-α+IFN-γ, and incubated for the indicated time. Activation of (A) NF-κB, (B) mitogen-activated and extracellular signal-regulated kinase (MEK)1/2-extracellular signal-regulated kinase (ERK) 1/2, and (C) JAK2-STAT1/3 were analysed by western blot assay. (A) Proteins were separated from nucleus and cytosol to evaluate the activation of NF-κB. (B) Total proteins were extracted and western blotted for p-MEK1/2, MEK1/2, p-ERK1/2, and ERK1/2. PARP, MEK1/2, ERK1/2, and β-actin were used as internal controls. (C) Total protein was extracted for western blotting assay for analysing phosphorylation of JAK2, STAT1, and STAT3. JAK2, STAT1, STAT3, and β-actin were used as internal controls. Densitometric analysis was performed using ImageJ software (1.49, Java software, NIH Image). The data shown represent mean ± standard error of the mean (SEM) of triplicate independent experiments (n = 3). ###p < 0.001 vs the control group; *p < 0.05, **p < 0.01, ***p < 0.001 vs the TNF-α+IFN-γ-stimulated group.
Effects of MF on atopic dermatitis like skin inflammation in AD mouse model
DNCB (0.4% or 1.0%) was applied topically on the dorsal side of NC/Nga mice, a suitable animal model for spontaneously occurring AD (24) (Fig. 4A). The changes in dorsal skin lesions were observed at the end of each week with photographs (Fig. 4B) and scoring (Fig. 4C). On the final day of application of DNCB, Dexa. and MF, AD-like skin lesions appeared severe with excoriation or erosion compared with those in the control group. The application of a high concentration of MF (MF 200 mg/kg) alone had no effects on DNCB-induced AD-like skin lesions. One of the typical characteristics of AD is disrupted skin barrier, which leads to dry skin and augmented TEWL. Increased TEWL by DNCB application was inhibited by the application of the positive control, Dexa.(5mg/kg), or MF (100mg/kg, 200mg/kg) (Fig. 4D). Other features of AD-like skin lesions include epidermal hyperplasia, oedema, and accumulation of inflammatory cells (15). Histopathological changes were examined by haematoxylin and eosin (H&E) staining (Fig. 4E). MF suppressed epidermal hyperplasia and edema, whereas DNCB induced thickening of epidermis.
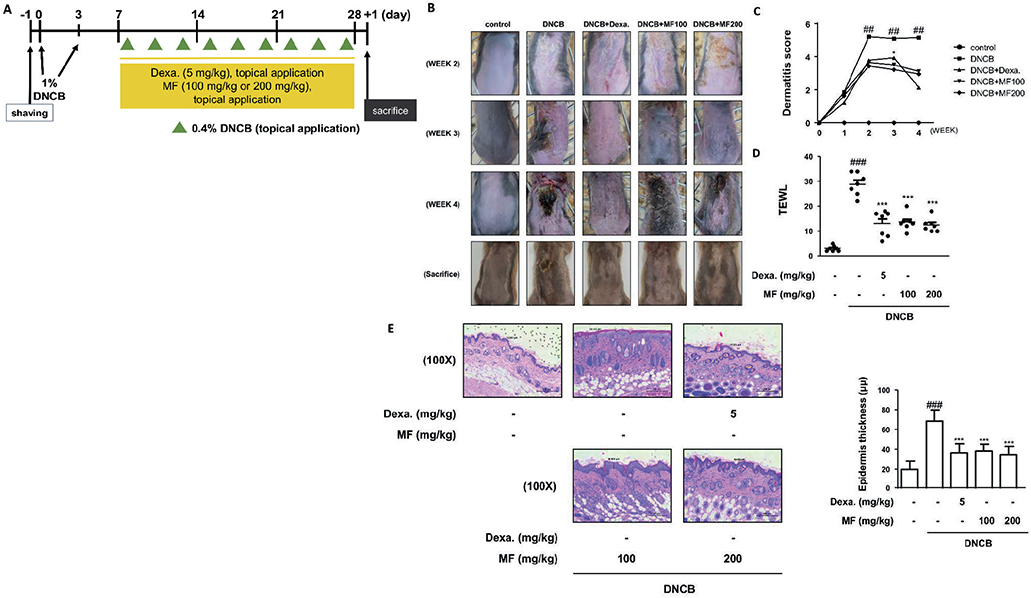
Fig. 4. Effects of Magnoliae flos (MF) on 2,4-dinitrochlorobenzen (DNCB)-induced atopic dermatitis (AD)-like skin lesions in NC/Nga mice. (A) Experimental scheme for the induction of AD and treatment with MF in NC/Nga mice. (B) Weekly comparison of DNCB-induced dermatitis in NC/Nga mice after topical application of MF. Representative photographs of dorsal regions of the mice from each group after induction of AD. (C) Dermatitis scores over 4 weeks. The dermatitis scores were determined as the sum of scores graded as 0 (none), 1 (mild), 2 (moderate), or 3 (severe) for each of the 4 symptoms; erythema/haemorrhage, scarring/dryness, oedema, and excoriation/erosion. (D) Transepidermal water loss (TEWL) was measured with a gpskin barrier light (gpower, Repulic of Korea) on day 28 after induction of AD (n = 6 or 7). (E) Dorsal skin was excised, fixed in 10% formaldehyde, embedded in paraffin, and sectioned. The tissue sections were assayed by haematoxylin and eosin (H&E) staining. The stained sections were visualized with a microscope at 100× magnification. Data shown represent mean ± standard deviation (SD) (n = 6 or 7). ###p < 0.001 vs the control group; ***p < 0.001 vs the DNCB-induced AD group.
Effects of MF on pruritus symptoms in AD mouse model
Pruritus is one of the major features of AD, and itching is the primary target of managing AD (25). DNCB application increased scratching behaviour compared with the control. However, the application of Dexa.(5mg/kg) or MF (200 mg/kg) reduced the frequency of this behaviour (Fig. 5A). As mast cells interact with sensory nerves and cutaneous cells, provoking itch and inflammation, the infiltration of mast cells was evaluated via toluidine blue staining. Mast cells are indicated by red arrows in Fig. 5B. Typical itch studies have revealed the interplay between histamine release and IgE-mediated mast cell activation. In terms of itch, serum IgE level was determined in the DNCB-induced AD mouse model. The group treated with MF had downregulated serum IgE levels compared with the DNCB-stimulated group (Fig. 5C).
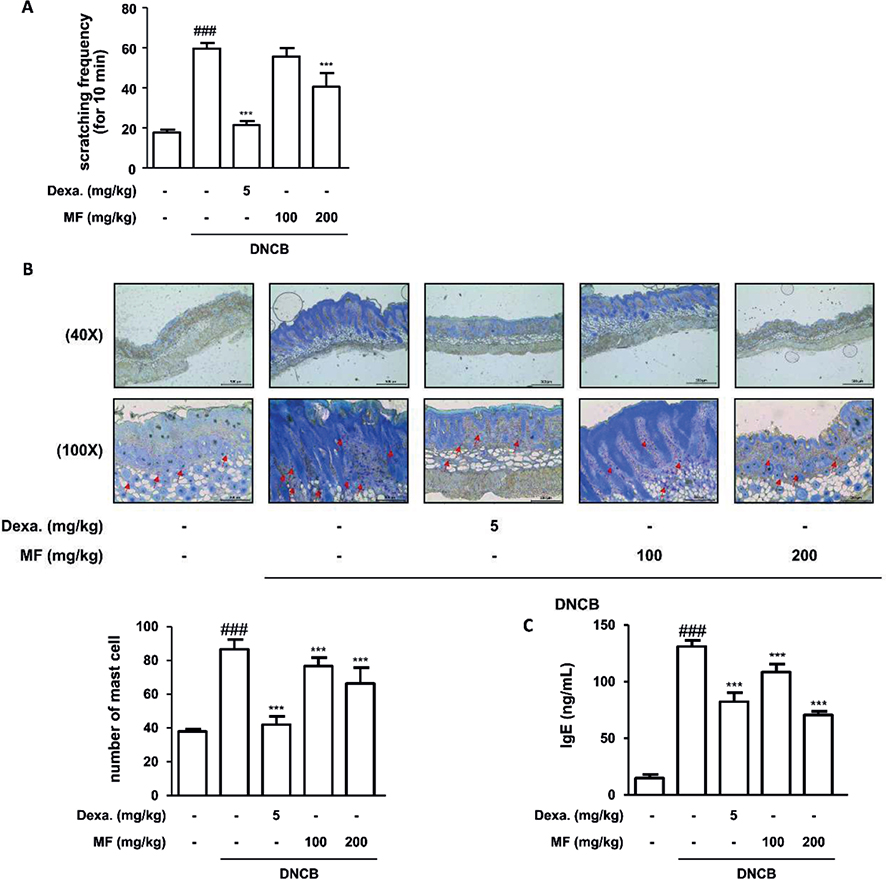
Fig. 5. Effects of Magnoliae flos (MF) on pruritus characteristics in 2,4-dinitrochlorobenzen (DNCB)-induced atopic dermatitis (AD). (A) Frequency of scratching behaviour in mice with DNCB-induced AD. (B) The dorsal skin was excised, fixed in 10% formaldehyde, embedded in paraffin, and sliced into sections. The tissue sections were stained with toluidine blue. The mean number of infiltrated mast cells was determined from 5 randomly selected areas per mouse. The stained sections were visualized with microscope at 40× (upper line) and 100× (lower line) magnifications. (C) Serum levels of immunoglobulin (Ig)E were measured by enzyme-linked immunoassay (ELISA). Data are expressed as mean ± SD (n = 6 or 7). ###p < 0.001 vs the control group; ***p < 0.001 vs the DNCB-induced AD group.
Effects of MF on the skin barrier molecules in AD mouse model
Skin barrier molecules were evaluated in mRNA and protein expression levels. In particular, FLG, LOR, and INV in the dorsal skin were assessed herein. DNCB application suppressed the skin barrier molecules compared with control treatment. However, the positive control, Dexa., caused significant recovery of the molecules (***p < 0.001). Furthermore, a high concentration of MF (200 mg/kg) restored the expression of skin barrier molecules at the mRNA (Fig. 6A–C) and protein levels (Fig. 6D).
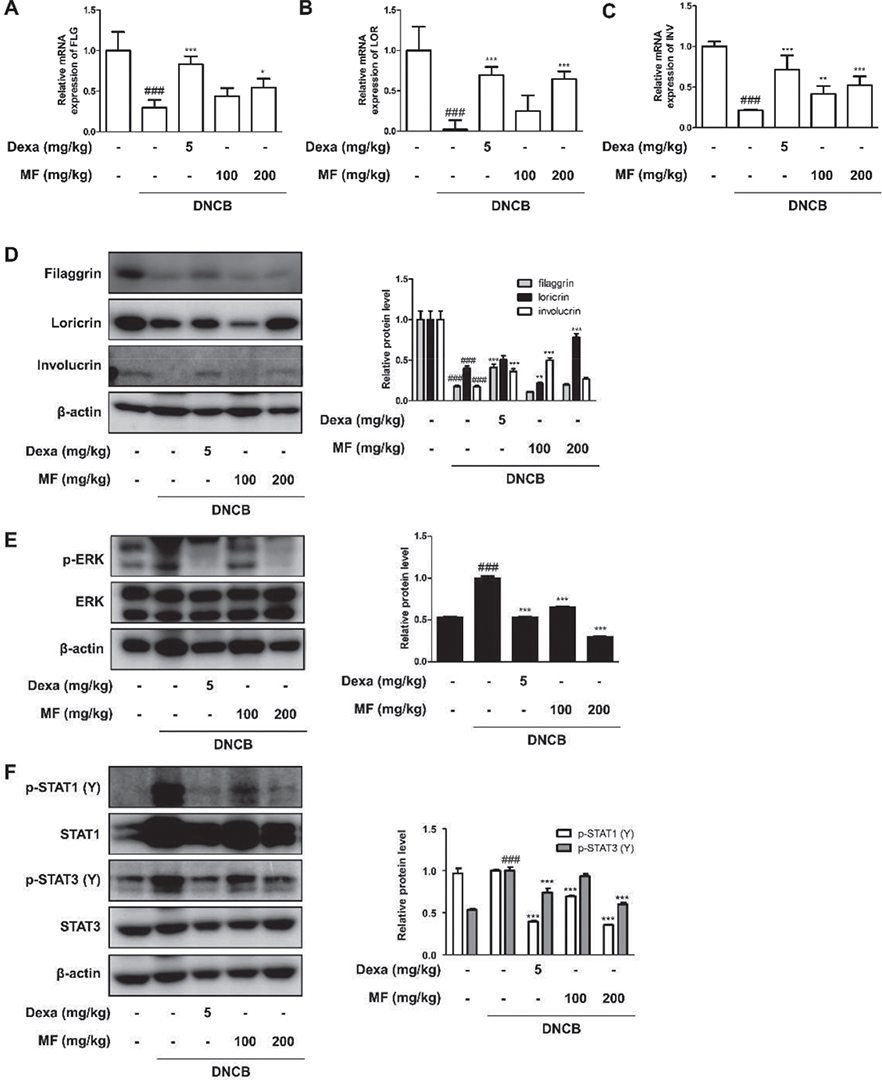
Fig. 6. Effects of Magnoliae flos (MF) on skin barrier molecules and extracellular signal-regulated kinase (ERK)1/2 or STAT signalling pathways in 2,4-dinitrochlorobenzene (DNCB)-induced atopic dermatitis (AD). Total RNA prepared from the dorsal tissue and the mRNA expression of (A) filaggrin (FLG), (B) loricrin (LOR) and (C) involucrin (INV) were determined by quantitative reverse transcription-PCR. Dorsal skin tissue lysates were immunoblotted with (D) FLG, LOR and INV to investigate effects of MF on the skin barrier molecules in DNCB-induced AD-like NC/Nga mouse model. The protein levels of (E) ERK1/2 MAPK and (F) STAT1/3 family were determined via western blot with specific antibodies in dorsal skin tissue lysates. ERK1/2, STAT1, STAT3, and β-actin were used as internal controls. Data are expressed as mean ± SD (n = 6 or 7). ###p < 0.001 vs the control group; *p < 0.05, **p < 0.01, ***p < 0.001 vs the DNCB-induced AD group.
Effects of MF on the ERK and STAT1/3 signalling pathways in AD mouse model
Based on the results obtained for MF from the in vitro experiments (Fig. 3), the effects of MF on the DNCB-induced in vivo model are shown in Fig. 6. The suppressive effects of MF on the translocation of p65, which were found in human keratinocytes, were not observed in DNCB-induced NC/Nga mice (data not shown). Phosphorylation of ERK/MAPK was inhibited by the application of MF (Fig. 6E). The effects of MF on the phosphorylation of STATs were also determined. Fig. 6F shows that the increased phosphorylation of STAT1/3 on tyrosine stimulated by DNCB was repressed by the application of Dexa. or MF.
DISCUSSION
MF is an aqueous extract of M. biondii or dried flower buds of plants that contain bioactive compounds, such as magnolin (26). Using hot water is one of generally used methods of decoction, we applied it for making MF from the buds (27). The flower bud of M. biondii, called shin-I, has been used as treatment for nasal congestion and headache, and has been found to inhibit prostate cancer cell growth and lung cancer cell migration (28). The effects of the compounds from flower bud have been identified to be antioxidative and anti-inflammatory (29). As AD is an chronic inflammatory disease and is included in atopic march, the anti-rhinitis effects of MF may have potential in treatment of AD. Atopic march is a pathological condition that progresses from AD to allergic rhinitis and asthma (30). This condition is based on the underlying concept of atopy, which is considered to be crucial in linking AD, allergic rhinitis and asthma (31). In the current study, MF suppressed the effects of pro-inflammatory cytokines and related signalling pathways, such as ERK/MAPK and JAK-STAT pathways, in TNF-α+IFN-γ-stimulated human keratinocytes. The involvements of inhibitors against ERK or STAT and the effects of MF on them were shown in Fig. S1. Furthermore, MF alleviated AD-like skin lesions and itch behaviour in DNCB-induced NC/Nga mice.
AD is also known as atopic eczema owing to its unusual and complicated aetiology. Therefore, various management strategies for AD are necessary. First, the control of pro-inflammatory cytokines and chemokines is required. Because different immune cells are involved in AD, Th1/2/17/22-related cytokines and chemokines are increased under the pro-inflammatory environment, mimicking AD-like skin lesions (20). The typical cytokines were found to be Th1/2/17 (Fig. 1A–D) and chemokines (Fig. 1E–G). MF inhibits pro-inflammatory cytokines, which are produced by activated inflammatory signalling pathways, such as NF-κB, MAPKs, or JAK-STATs (32). Although the suppressive effect of MF on NF-κB p65 translocation was found only in human keratinocytes, and not in the AD mouse model (Fig. 3), another inflammatory transcription factor family, the STAT family, was revealed to have a downregulation effect on MF (Fig. 3C and Fig. 6F). However, species difference might be responsible for the differing results, and further research is necessary.
In this study, the allergic chemical, DNCB, was repeatedly applied to the dorsal skin of an AD-like mouse model. DNCB causes hyperkeratosis, thickened epidermis, and infiltrated lymphocytes and mast cells; similar phenotypes to human AD symptoms (33). Besides erythema/haemorrhage, scarring/dryness, and excoriation/erosion, which were observed in DNCB-induced mice (Fig. 4), one of the major characteristics of AD is pruritus. Itching is mediated by mast cells on nerve fibres in peripheral tissues and the central nervous system (34). DNCB caused increased infiltration of mast cells. Furthermore, the increased serum IgE levels were downregulated by treatment with MF, which was accompanied by scratching behaviour (Fig. 5). TSLP stimulates the sensory neurones, provoking itch, scratching behaviour, and inflammation, and was demonstrated as a functional pruritogen (35) that correlates with mast cells (36). The effects of MF on periostin protein expression (Fig. 1H) were determined in the current study. As periostin is an intrinsic inducer of TSLP from keratinocytes, it induces the activation of the Akt pathways, resulting in keratinocyte survival and differentiation (37).
In addition to the pathogenesis of AD, skin barrier molecules, including filaggrin, loricrin, involucrin, and serine proteases, were identified in the current study. Figs 2 and 6 depict the restorative effects of MF on disrupted molecules owing to cornification in the epidermis (38). According to recent studies, FLG is related to proteases and protease inhibitors in AD, inducing a loss in skin homeostasis (39). As a serine protease, KLK5 regulates the KLK cascade in the stratum corneum, leading to skin permeability and inflammation. KLK7 is an important chymotryptic enzyme in AD-like skin lesions that destroys the homeostasis of the epidermal barrier (40). In the current study, MF was found to restore the disrupted skin barrier molecules and downregulate serine proteases (Figs 2 and 6).
In conclusion, the objective of this study was to determine the effects of MF on AD-like inflammatory conditions, using an in vitro model of TNF-α+IFN-γ-stimulated human keratinocytes, and an in vivo model of DNCB-induced AD-like skin lesions in NC/Nga mice. MF was found to suppress pro-inflammatory cytokines and chemokines, and the signalling pathways involved in inflammation. MF also showed regulatory effects on cornified envelope-composing molecules and epidermal serine proteases. Taken together, these results suggest that MF or its compounds can alleviate AD-like responses, and thus are potential treatments for AD.
ACKNOWLEDGEMENTS
This work was supported by the Basic Science Research Program of the National Research Foundation (NFR) of Korea (grant number NRF-2021R1F1A1058077).
REFERENCES
- Puar N, Chovatiya R, Paller AS. New treatments in atopic dermatitis. Ann Allergy Asthma Immunol 2021; 126: 21–31.
- Leung DY, Geha RS. Immunoregulatory abnormalities in atopic dermatitis. Clin Rev Allergy 1986; 4: 67–86.
- Czarnowicki T, Krueger JG, Guttman-Yassky E. Novel concepts of prevention and treatment of atopic dermatitis through barrier and immune manipulations with implications for the atopic march. J Allergy Clin Immunol 2017; 139: 1723–1734.
- Ständer S. Atopic dermatitis. N Engl J Med 2021; 384: 1136–1143.
- Seth D, Cheldize K, Brown D, Freeman EF. Global burden of skin disease: inequities and innovations. Curr Dermatol Rep 2017; 6: 204–210.
- Kader HA, Azeem M, Jwayed SA, Al-Shehhi A, Tabassum A, Ayoub MA, et al. Current insights into immunology and novel therapeutics of atopic dermatitis. Cells 2021; 10: 1392.
- Langan SM, Irvine AD, Weidinger S. Atopic dermatitis. Lancet 2020; 396: 345–360.
- Frazier W, Bhardwaj N. Atopic dermatitis: diagnosis and treatment. Am Fam Physician 2020; 101: 590–598.
- Coondoo A, Phiske M, Verma S, Lahiri K. Side-effects of topical steroids: a long overdue revisit. Indian Dermatol Online J 2014; 5: 416–425.
- Gomez-Garcia F, Gomez-Arias PJ, Hernandez J, Montilla AM, Gay-Mimbrera J, Aguilar-Luque M, et al. Drugs targeting the JAK/STAT pathway for the treatment of immune-mediated inflammatory skin diseases: protocol for a scoping review. BMJ Open 2019; 9: e028303.
- Ferreira S, Guttman-Yassky E, Torres T. Selective JAK1 inhibitors for the treatment of atopic dermatitis: focus on upadacitinib and abrocitinib. Am J Clin Dermatol 2020; 21: 783–798.
- Kim HJ, Nam YR, Nam JH. Flos Magnoliae inhibits chloride secretion via ANO1 inhibition in Calu-3 cells. Am J Chin Med 2018; 46: 1079–1092.
- Ma P, Che D, Zhao T, Zhang Y, Li C, An H, et al. Magnolin inhibits IgE/Ag-induced allergy in vivo and in vitro. Int Immunopharmacol 2019; 76: 105867.
- Kim JY, Lim HJ, Lee da Y, Kim JS, Kim DH, Lee HJ, et al. In vitro anti-inflammatory activity of lignans isolated from Magnolia fargesii. Bioorg Med Chem Lett 2009; 19: 937–940.
- An HJ, Kim JY, Kim WH, Gwon MG, Gu HM, Jeon MJ, et al. Therapeutic effects of bee venom and its major component, melittin, on atopic dermatitis in vivo and in vitro. Br J Pharmacol 2018; 175: 4310–4324.
- Gittler JK, Shemer A, Suarez-Farinas M, Fuentes-Duculan J, Gulewicz KJ, Wang CQ, et al. Progressive activation of T(H)2/T(H)22 cytokines and selective epidermal proteins characterizes acute and chronic atopic dermatitis. J Allergy Clin Immunol 2012; 130: 1344–1354.
- Coondoo A. The role of cytokines in the pathomechanism of cutaneous disorders. Indian J Dermatol 2012; 57: 90–96.
- Boguniewicz M, Leung DY. Atopic dermatitis: a disease of altered skin barrier and immune dysregulation. Immunol Rev 2011; 242: 233–246.
- Di Paolo CT, Diamandis EP, Prassas I. The role of kallikreins in inflammatory skin disorders and their potential as therapeutic targets. Crit Rev Clin Lab Sci 2021; 58: 1–16.
- Gil TY, Hong CH, An HJ. Anti-inflammatory effects of ellagic acid on keratinocytes via MAPK and STAT pathways. Int J Mol Sci 2021; 22: 1277.
- Liu YJ, Soumelis V, Watanabe N, Ito T, Wang YH, Malefyt Rde W, et al. TSLP: an epithelial cell cytokine that regulates T cell differentiation by conditioning dendritic cell maturation. Annu Rev Immunol 2007; 25: 193–219.
- Ye J, Piao H, Jiang J, Jin G, Zheng M, Yang J, et al. Polydatin inhibits mast cell-mediated allergic inflammation by targeting PI3K/Akt, MAPK, NF-kappaB and Nrf2/HO-1 pathways. Sci Rep 2017; 7: 11895.
- Montilla AM, Gomez-Garcia F, Gomez-Arias PJ, Gay-Mimbrera J, Hernandez-Parada J, Isla-Tejera B, et al. Scoping review on the use of drugs targeting JAK/STAT pathway in atopic dermatitis, vitiligo, and alopecia areata. Dermatol Ther 2019; 9: 655–683.
- Jin H, He R, Oyoshi M, Geha RS. Animal models of atopic dermatitis. J Invest Dermatol 2009; 129: 31–40.
- Pavlis J, Yosipovitch G. Management of itch in atopic dermatitis. Am J Clin Dermatol 2018; 19: 319–332.
- Huang Y, Zou X, Zhang X, Wang F, Zhu W, Zhang G, et al. Magnolin inhibits prostate cancer cell growth in vitro and in vivo. Biomed Pharmacother 2017; 87: 714–720.
- Wang L, Zhao Z, Zhao H, Liu M, Lin C, Li L, et al. Pectin polysaccharide from flos magnoliae (Xin Yi, magnolia biondii pamp. flower buds): hot-compressed water extraction, purification and partial structural characterization. Food Hydrocolloids 2022; 122: 107061.
- Lee CJ, Lee MH, Yoo SM, Choi KI, Song JH, Jang JH, et al. Magnolin inhibits cell migration and invasion by targeting the ERKs/RSK2 signaling pathway. BMC Cancer 2015; 15: 576.
- Kim JS, Kim JY, Lee HJ, Lim HJ, Lee DY, Kim DH, et al. Suppression of inducible nitric oxide synthase expression by furfuran lignans from flower buds of Magnolia fargesii in BV-2 microglial cells. Phytother Res 2010; 24: 748–753.
- Bantz SK, Zhu Z, Zheng T. The Atopic march: progression from atopic dermatitis to allergic rhinitis and asthma. J Clin Cell Immunol 2014; 5: 202.
- Spergel JM. Atopic march: link to upper airways. Curr Opin Allergy Clin Immunol 2005; 5: 17–21.
- Abe S, Yuasa K. Sudachitin, a polymethoxyflavone from Citrus sudachi, induces apoptosis via the regulation of MAPK pathways in human keratinocyte HaCaT cells. Biochem Biophys Res Commun 2019; 519: 344–350.
- Lee K-S, Jeong E-S, Heo S-H, Seo J-H, Jeong D-G, Choi Y-K. A novel model for human atopic dermatitis: application of repeated DNCB patch in BALB/c mice, in comparison with NC/Nga mice. Lab Anim Res 2010; 26: 95–102.
- Voisin T, Chiu IM. Mast cells get on your nerves in itch. Immunity 2019; 50: 1117–1119.
- Wang SH, Zuo YG. Thymic stromal lymphopoietin in cutaneous immune-mediated diseases. Front Immunol 2021; 12: 698522.
- Xia Q, Liu T, Wang J, Sun L, Chen X, Liu Y, et al. Mast cells and thymic stromal lymphopoietin (TSLP) expression positively correlates with pruritus intensity in dermatitis herpetiformis. Eur J Dermatol 2020; 30: 499–504.
- Masuoka M, Shiraishi H, Ohta S, Suzuki S, Arima K, Aoki S, et al. Periostin promotes chronic allergic inflammation in response to Th2 cytokines. J Clin Invest 2012; 122: 2590–2600.
- Gutowska-Owsiak D, de La Serna JB, Fritzsche M, Naeem A, Podobas EI, Leeming M, et al. Orchestrated control of filaggrin-actin scaffolds underpins cornification. Cell Death Dis 2018; 9: 412.
- Lee HJ, Lee SH. Epidermal permeability barrier defects and barrier repair therapy in atopic dermatitis. Allergy Asthma Immunol Res 2014; 6: 276–287.
- Morizane S. The role of kallikrein-related peptidases in atopic dermatitis. Acta Med Okayama 2019; 73: 1–6.