ORIGINAL REPORT
Drug Survival and Long-term Outcome of Tofacitinib in Patients with Alopecia Areata: A Retrospective Study
Jundong HUANG1#, Sihan DENG1#, Ji LI1–3, Yan TANG1,3, Fangfen LIU1,3, Yu LIU1, Shijia RAO4 and Wei SHI1,3
1Department of Dermatology, 2Hunan Key Laboratory of Aging Biology, 3National Clinical Research Center for Geriatric Disorders, Xiangya Hospital, Central South University and 4Department of Dermatology, The Second Xiangya Hospital, Central South University, Hunan Key Laboratory of Medical Epigenomics, Changsha, Hunan, PR China
Several non-randomized clinical trials and retrospective studies have demonstrated encouraging efficacy and well-tolerated safety of tofacitinib in the treatment of alopecia areata. However, there are scarce data on a large cohort of patients with alopecia areata in long-term real-world practice. This single-centre, retrospective, observational cohort study included 126 patients with alopecia areata treated with tofacitinib between February 2021 and December 2022. The aims of this study are to evaluate drug survival, effectiveness and safety of tofacitinib for treatment of alopecia areata, and to identify potential factors influencing long-term outcomes. Median duration of treat-ment was 23.00 (interquartile range (IQR) 15.00, 47.25) weeks. Median all-cause survival time of 126 patients treated with tofacitinib was 44 weeks (95% confidence interval (95% CI) 36.3, 51.7), and the all-cause drug retention rate at 12 weeks, 24 weeks and 48 weeks were 90.0%, 66.4% and 42.3%, respectively. The most common reason for discontinuation was complete remission/satisfaction. A total of 80 patients treated with tofacitinib for over 6 months were included in the efficacy analysis, the overall complete response rate at 24 weeks was 33.8% (27/80). No life-threatening serious adverse events occurred. Sex is an independent risk factor in predicting patient outcomes. This real-world study confirmed the high effectiveness and acceptable safety profile of tofacitinib in alopecia areata, with a satisfactory drug survival rate, and provides supporting data for the clinical application of tofacitinib in Chinese patients with alopecia areata.
Key words: alopecia areata; tofacitinib; JAK inhibitor; drug survival; effectiveness.
SIGNIFICANCE
Several studies have demonstrated the efficacy of tofacitinib in the treatment of alopecia areata. However, there are scarce data on a large cohort of patients with alopecia areata in long-term real-world practice. This study included 126 patients with alopecia areata treated with tofacitinib. The study assessed the drug survival, effectiveness, and safety of treatment with tofacitinib and identified potential factors influencing long-term outcomes. The results confirm the efficacy of tofacitinib and that it is well-tolerated in a real-life setting for alopecia areata, with a satisfactory drug survival rate. This provides important data for the long-term use of tofacitinib in patients with alopecia areata.
Citation: Acta Derm Venereol 2023; 103: adv13475. DOI https://doi.org/10.2340/actadv.v103.13475.
Copyright: © Published by Medical Journals Sweden, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: Oct 10, 2023; Published: Nov 13, 2023
Corr: Shijia Rao, Department of Dermatology, The Second Xiangya Hospital, Central South University, Hunan Key Laboratory of Medical Epigenomics, Changsha, Hunan, PR China and Wei Shi, MD, PhD, Department of Dermatology, Xiangya Hospital, Central South University, Changsha, Hunan, PR China. E-mails: scarlett0512@csu.edu.cn; shiwei@csu.edu.cn
#These authors contributed equally to this work and share first authorship.
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
Alopecia areata (AA) is a chronic autoimmune dermatosis characterized by non-scarring hair-loss patches on the scalp and/or any other hair-bearing skin (1). Disease prevalence is increasing, reaching an estimation of 0.22% in the USA, approximately 5% of whom will progress to alopecia universalis (AU) or alopecia totalis (AT) (2, 3), which are the most refractory to current treatment regimens and have the worst prognosis. The management of AA is notoriously challenging, with high rates of therapeutic failures or relapses, imposing a heavy psychological and economic burden on patients and their families.
The Janus kinase (JAK) and signal transducer and activator of transcription (STAT) pathways mediate the signalling of many cytokines from cell membrane receptors to the nucleus and play a crucial role in the immunopathogenesis of AA (4). Tofacitinib primarily targets JAK 1/3 and reduces the immune response and resultant inflammation, emerging as an alternative for the treatment of refractory cases of AA (5). Several non-randomized clinical trials have demonstrated encouraging efficacy and well-tolerated safety of tofacitinib, especially for patients who have previously failed to respond to other systemic treatments (6–8). One prospective cohort study assessed the efficacy of tofacitinib at 6 months in 48 patients with AA, and 62.2% patients had achieved at least 50% regrowth of hair after treatment (9). However, the drug response and safety of treatment with tofacitinib treatment for AA observed in clinical trials may not accurately reflect real-world clinical practice.
Despite the rapid development of novel JAK inhibitors, including baricitinib and ritlecitinib, tofacitinib remains an effective and the longest-used JAK inhibitor for AA (10–12). Nevertheless, there are limited retrospective studies to support the long-term efficacy and sustainability of tofacitinib for the treatment of AA (6). Drug survival/ retention refers to the duration between the initiation and discontinuation of a drug, while drug retention rate is the proportion of patients who keep using a drug for a period of time. Drug survival provides a proxy measure for long-term effectiveness, safety, and tolerability of a drug (13). To our knowledge, there is currently a lack of research on the drug survival of tofacitinib in patients with AA.
The objectives of this study were to examine the drug survival, effectiveness and safety of tofacitinib for the treatment of AA and to assess the correlation between tofacitinib survival and patients’ characteristics in a real-world setting.
METHODS
Study design, patients and data collection
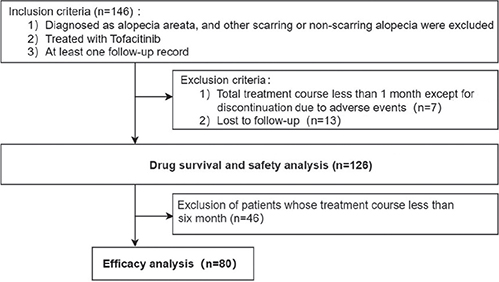
This single-centre, retrospective, observational study was conducted at the Department of Dermatology, Xiangya Hospital, Central South University, Changsha, China. Data were retrieved for all patients with dermatologically confirmed AA who received at least 1 month of treatment with tofacitinib at our institution between February 2021 and December 2022. A total of 126 people were included in the safety and drug survival analysis, and 80 of them were included in the efficacy analysis (Fig. 1). All patients were administered tofacitinib according to their weight, with an initial dose of 5 mg twice a day (weight > 40 kg) or 5 mg per day (weight < 40 kg) (14, 15). Patients with missing clinical data, or whose prognosis could not be tracked were excluded. The study protocol was approved by the hospital’s research ethics committee and written informed consent was obtained from all patients.
For each patient, demographic and clinical data, including age at the time of initiation of JAK inhibitors, sex, body mass index (BMI), age of onset, disease duration, AA subtypes, Severity of Alopecia Tool (SALT) grade, external scalp involvement, duration of the current episode, family history, concomitant therapy, prior treatment history, and comorbidity were recorded. Regular follow-up laboratory testing (including blood routine, liver and kidney function, and blood lipids) was conducted every month within the first 3 months and every 2–3 months thereafter. During therapy, data were collected for each patient on the regimen, regarding treatment duration, discontinuation time, efficacy, and adverse events (AEs).
Outcomes
The primary outcome was the drug survival of tofacitinib, which was defined as the time from initiation to discontinuation of tofacitinib. Temporary treatment interruptions of less than 2 weeks for any reason were allowed and this interruption time was not included in the analysis. Secondary outcomes were the number of patients achieving complete remission (CR) at 24 weeks and AEs. SALT was used to quantify scalp hair loss degree, according to the percentage of hair loss area divided into 6 subclasses, which was independently verified by 2 authors (16). CR was defined as complete hair regrowth achieved by treatment (S0). Partial remission (PR) was defined as significant hair regrowth after treatment (SALT grade decreased by at least 1 grade). Ineffectiveness was defined as no significant hair regrowth after treatment (SALT grade unchanged). Deterioration was defined as an increase in hair-loss area (SALT grade increase at least 1 grade). Effective rate = CR+PR; remission rate = CR. Discontinuation associated with ineffectiveness or AEs was evaluated, and the reasons for discontinuation were recorded.
Refractory AA was defined as those in whom the existing routine treatment was ineffective or unsatisfactory and who had been treated for more than 1 year (18). The remaining patients were classified as the general group.
Statistical analysis
Data were analysed using SPSS version 25.0 (IBM® Inc., Chicago, IL, USA). Categorical variables were expressed as numbers or proportions and were evaluated with the χ2 test or Fisher’s precision probability test. Measurement data were reported as mean ± standard deviation (SD) or median (interquartile range; IQR), and comparisons between groups were evaluated with the Student’s t-test or Mann–Whitney U test. The Marginal Homogeneity test was used to compare the changes in SALT grading before and after treatment. Multivariate logistic regression was performed to analyse the factors affecting the curative effect.
Drug survival time was defined as the duration of tofacitinib treatment and was calculated in weeks. Kaplan–Meier method was used to plot the survival curves of all-cause drug withdrawal and drug discontinuation due to treatment inefficacy/AEs. According to different drug withdrawal reasons, Kaplan–Meier curves were built to graphically analyse drug survival. A log-rank test was used to compare the differences in the distribution of survival time. Cox proportional risk model was used to evaluate the factors influencing drug discontinuation. Results were expressed as hazard ratio and 95% CI. All graphs were produced using the GraphPad Prism 8 program (8.0.1 for Windows, GraphPad Software, San Diego, California USA).
RESULTS
A total of 126 patients with AA receiving tofacitinib were involved in the drug retention analysis, including the general group (n = 71) and refractory group (n = 55), their baseline demographics and clinical characteristics are shown in Table I. Of these, 78 (61.9%) were female, with a median age of 25.5 (18.0, 35.25) years. A total of 31 patients were treated for more than 48 weeks, 6 patients were treated for more than 72 weeks, and the median duration of treatment was 23.00 (IQR 15.00, 47.25) weeks. Out of the study population, 34 patients (27.0%) had total or universal alopecia. The most common extra-scalp-involved areas were eyebrows (50.8%), followed by eyelashes (26.2%), body hair (18.3%), and nails (14.3%). Atopy (11.1%) and thyroid disease (7.9%) were the most common comorbidities.
| Variables | Patient (n = 126) |
| Age, year, mean (IQR) | 25.5 (18.0, 35.25) |
| < 25 years, n (%) | 60 (47.6) |
| Sex, n (%) | |
| Male | 48 (38.1) |
| Female | 78 (61.9) |
| Body mass index, kg/m2, mean ± SD | 21.4 ± 3.5 |
| Age of onset, age, mean (IQR) | 20.5 (13.0, 32.25) |
| <18 years, n (%) | 56 (44.4) |
| Duration of disease, months, n (%) | |
| <12 | 36 (28.6) |
| 12~60 | 51 (40.5) |
| >60 | 39 (30.9) |
| Current episode duration, months, n (%) | |
| <12 | 59 (46.8) |
| 12~60 | 52 (41.3) |
| >60 | 15 (11.9) |
| SALT grade, n (%) | |
| S1 | 0 (0) |
| S2 | 33 (26.2) |
| S3 | 43 (34.1) |
| S4 | 16 (12.7) |
| S5 | 34 (27.0) |
| Subtype, n (%) | |
| Alopecia totalis | 5 (3.9) |
| Alopecia universalis | 29 (23.1) |
| Patch alopecia areata | 92 (73.0) |
| Body hair involvement, n (%) | |
| Eyebrows | 64 (50.8) |
| Eyelashes | 33 (26.2) |
| Body hair | 23 (18.3) |
| Nail involvement, n (%) | 18 (14.3) |
| Prior treatment, n (%) | |
| Topical medication* | 94 (74.6) |
| Oral glucocorticoids | 68 (54.0) |
| Immunosuppressant** | 9 (7.1) |
| Family history, n (%) | 11 (8.7) |
| Comorbidity, n (%) | |
| Thyroid disease | 10 (7.9) |
| Systemic lupus erythematosus | 1 (0.8) |
| Vitiligo | 1 (0.8) |
| Atopy | 14 (11.1) |
| Concomitant therapy, n (%) | |
| Topical corticosteroids or minoxidil | 78 (61.9) |
| SALT: severity of alopecia tool; IQR: interquartile range; SD: standard deviation. | |
| *Including topical or intracutaneous injection of glucocorticoid and topical minoxidil. | |
| **Including cyclosporine and methotrexate. | |
The median duration of all-cause drug survival was 44 weeks (95% CI 36.3–51.7), and the all-cause drug retention rate at 12, 24 and 48 weeks were 90.0%, 66.4% and 42.3%, respectively. The ineffective and adverse drug retention rates combined were 96.7%, 86.4% and 82.8% after 12, 24 and 48 weeks, respectively (Fig. 2a). Specifically, all-cause drug retention rates were compared between the general (median retention 42.0 weeks, 95% CI 34.1–49.8) and refractory (median survival 49.0 weeks, 95% CI 35.9–62.1) patients, but no statistical difference was observed (log rank p = 0.445) (Fig. 2b). A total of 64 patients (50.8%) discontinued the treatment during the follow-up. The most common reason for discontinuation was the attainment of CR or satisfactory efficacy (n = 33), followed by ineffective treatment (n = 13). The impact of the COVID-19 (SARS-CoV-2) epidemic, including challenges related to return visits, drug procurement, and the risk of infection, also contributed to treatment discontinuation (n = 8). Only 2 patients discontinued due to adverse reactions. The remaining 8 patients discontinued for personal preference reasons, such as pregnancy preparation and concerns regarding long-term adverse drug reactions.
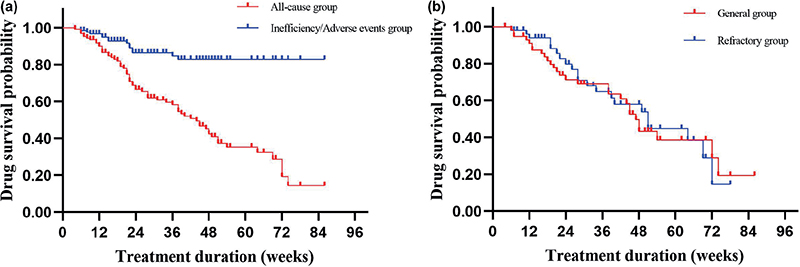
Fig. 2. Kaplan–Meier plot of drug survival for tofacitinib in patients with alopecia areata (n = 126). (a) Curves split for different reasons of discontinuation. (b) All-cause drug survival rate curves of the general group and the refractory group.
Eighty patients were included in the efficacy evaluation (age 26 (18.0, 34.0) years; 63.8% female), including 38 individuals in the general group and 42 in the refractory group (Table SI). Patients in the refractory group had a younger age of onset, a longer duration of the disease, and more common eyebrows and eyelash involvement. After 6 months of treatment, the general group achieved a CR rate of 47.4% (18/38), while the refractory group achieved a CR rate of 21.4% (9/42). Overall, the proportion of severe alopecia decreased from 72.5% to 7.5%. In only 3 patients was treatment ineffective and 1 patient worsened; they are all in the refractory group (Fig. S1, Tables SII–SIV). Multivariate logistic regression showed that women ((odds ratio (OR) 5.166, 95% CI 1.337–19.959, p < 0.05)) and patients without nail changes (OR 0.077, 95% CI 0.008–0.741, p < 0.05) were more likely to achieve clinical remission (Table SV).
Of the 126 patients, 70 patients reported adverse reactions and no life-threatening serious AE occurred during the follow-up. Scalp seborrhoeic dermatitis/folliculitis (22.2%) and facial acne/acne-like rash (12.7%) were the most common AE. Most of the patients with acne reported were mild or moderate. Twenty patients (15.9%) reported infection-related AEs, with upper respiratory tract infections (10.3%) being the most common. Dyslipidaemia (7.9%) is the most common laboratory-related adverse reaction. Other adverse reactions include menstrual disorders (3.2%), Bell’s palsy (0.8%), gastrointestinal symptoms (0.8%) and headache (0.8%) (Table SVI). Among patients who experienced AEs, only 2 patients discontinued as a result. One developed herpes zoster in the second month of treatment. The other discontinued in the fifth month due to repeated infection, fever, and a mild increase in creatinine, and the creatinine returned to normal after a month of discontinuation.
Univariate Cox proportional hazard analyses showed that duration less than 12 months was a risk factor for discontinuation (HR = 0.488, 95% CI 0.290–0.820, p = 0.007). In particular, when incorporating risk factors with a p-value < 0.1 into multivariate Cox regression, only AEs (HR = 0.602, 95% CI 0.364–0.996, p = 0.048) were observed as a statistically positive predictor for drug discontinuation. Age, sex, BMI, age of onset, course of the disease, baseline SALT grade, external scalp involvement, comorbidities, previous treatment, and refractory AA did not significantly influence drug survival (Table II).
| Variable | Univariate analyses | Multivariable analyses | ||||
| HR | 95% CI | p-value | HR | 95% CI | p-value | |
| Agea | 0.747 | 0.454–0.1229 | 0.250 | – | – | – |
| Sex (M/F) | 0.868 | 0.518–1.452 | 0.589 | – | – | – |
| BMI, kg/m2) | 1.033 | 0.960–1.111 | 0.388 | – | – | – |
| Age of onset | 1.644 | 0.980–2.759 | 0.060 | 1.400 | 0.827–2.370 | 0.211 |
| Duration of diseaseb | 0.488 | 0.290–0.820 | 0.007 | 0.522 | 0.266–1.026 | 0.059 |
| Current episode durationb | 0.620 | 0.377–1.020 | 0.060 | 0.906 | 0.473–1.736 | 0.766 |
| Baseline SALT grade | ||||||
| S3 | 0.649 | 0.325–1.298 | 0.222 | – | – | – |
| S4 | 0.752 | 0.400–1.414 | 0.376 | – | – | – |
| S5 | 0.696 | 0.298–1.623 | 0.402 | – | – | – |
| AU or AT | 1.418 | 0.809–2.485 | 0.223 | – | – | – |
| Eyebrows involvement | 1.066 | 0.650–1.748 | 0.800 | – | – | – |
| Eyelashes involvement | 1.057 | 0.598–1.870 | 0.848 | – | – | – |
| Body hair involvement | 1.214 | 0.632–2.335 | 0.560 | – | – | – |
| Nails involvement | 0.711 | 0.337–1.498 | 0.370 | – | – | – |
| Family history | 1.455 | 0.661–3.201 | 0.351 | – | – | – |
| Atopy disease | 0.611 | 0.263–1.419 | 0.252 | – | – | – |
| Thyroid disease | 0.850 | 0.307–2.347 | 0.753 | – | – | – |
| Prior systemic therapy | 0.894 | 0.545–1.468 | 0.659 | – | – | – |
| Refractory AA | 0.824 | 0.498–1.364 | 0.451 | – | – | – |
| Concomitant therapy | 1.001 | 0.600–1.670 | 0.997 | – | – | – |
| AEs reported | 0.617 | 0.376–1.015 | 0.057 | 0.602 | 0.364–0.996 | 0.048 |
| a<18/ ≥ 18 years. b< 12 months/ ≥ 12 months. | ||||||
| Values in bold indicate hazard ratios with a p<0.10 in univariate analyses and p<0.05 in multivariable analyses. HR: hazard ratio; BMI: body mass index; SALT: Severity of Alopecia Tool; AA: alopecia areata; AU: alopecia universalis; AT: alopecia totalis; AE: adverse events. | ||||||
DISCUSSION
This retrospective study analysed the long-term efficacy, safety and drug retention of tofacitinib in patients with AA, and is the first research to describe drug survival of JAK inhibitors in AA and shows that tofacitinib has high treatment durability in real-world clinical practice. The median all-cause drug retention time for tofacitinib in patients with AA was 44 weeks. The ineffective/AEs drug retention rate and all-cause drug retention rate at 48 weeks were 82.8% and 42.3%, respectively. Among 80 patients enrolled in the efficacy study, 27 patients (33.75%) achieved a CR and 36 patients (45%) had a decrease in SALT score to S1 after 6 months of treatment, indicating positive responses to tofacitinib in patients with AA. These results indicate the overall effectiveness of the treatment and that it was well-tolerated.
Approximately 60% of patients with AA experience disease onset before the age of 30 years, and the peak incidence age is 25–29 years old, with no obvious tendency with regard to sex (1). In the current study, the demography features were basically consistent with previous research (9), except for the higher proportion of female patients included. This difference is not surprising, given that female patients often exhibit a stronger willingness to treatment and concerns about obesity-related AEs caused by glucocorticoids.
Drug survival serves as long-term evidence of a drug and is influenced by several real-life factors, including treatment efficacy, safety, patient satisfaction, and physicians’ preference (18). To date, no study has described the drug survival of JAK inhibitors in AA. The current data shows that tofacitinib has high treatment persistence in patients with AA in real-life conditions. In addition, tofacitinib does not cause corticosteroid-induced obesity and offers convenience of use, which also contributes to its high retention rate. The majority of patients discontinued treatment due to symptom relief after treatment. Other reasons for discontinuation included treatment ineffectiveness (10.4%), epidemic impact (6.3%), AEs (1.6%), and personal choices (6.3%). Interestingly, this study showed that patients who reported adverse reactions had a lower risk of discontinuation. This may because the adverse reaction record in the current study comes mainly from the self-report of patients and regular blood sampling examination, and this type of patient has relatively good compliance. Recently, a study showed that the drug retention of tofacitinib did not differ between biological disease-modifying anti-rheumatic drugs-failure and drugs-naive patients (19). Similarly, there is no statistical difference observed in the all-cause drug retention rates between the general and refractory group of patients with AA, it is not clear if this phenomenon is present in other diseases.
Consistent with previous literature, these data indicate that tofacitinib is effective for AA treatment, with an all-cause response rate of 95% at 6 months, with more than one-third of patients achieving a CR. A recent meta-analysis showed the effectiveness of tofacitinib in the treatment of AA (SALT score decrease > 5%) was up to 80%, and the significant effectiveness (SALT score decrease > 50%) was 54% (14). The differences from the above results may be attributed to the different evaluation indicators and baseline population characteristics. SALT grading was used instead of SALT score in this study; this may underestimate the efficacy of tofacitinib, especially in patients with less severe hair loss at baseline (16). In addition, a delayed response to tofacitinib was observed in some patients, whose hair growth occurred only 3 months after treatment. This suggests the heterogeneity of response time between individuals, so the efficacy would be better after sufficient observation time (usually > 3 months). However, despite the continuous use of tofacitinib for 6 months, there are patients who still do not respond, options including oral corticosteroids, increased dose, or other immunosuppressant agents were considered for subsequent treatment in such patients (20, 21).
In addition, this study evaluated the determinants of poor prognosis, which may help physicians with individualized treatment of AA. Some factors have been reported to influence the treatment efficacy of AA, including disease severity, onset in childhood, longer disease duration, diffuse pattern of hair loss, family history, and involvement of the nails (3, 22). The current results show that sex and nail involvement are the key factors influencing treatment efficacy, with sex being an independent predictor of patient response. Some researchers suggest that sex is a biological variable between males and females in congenital and acquired immune responses (23), which may contribute to variations in the occurrence and treatment response of individuals with autoimmune diseases. Although several clinical trial results support the safety of tofacitinib (24–26), long-term real-world safety data are currently lacking. According to previous literature reports (15, 27), the most common AEs are infection-related diseases, such as upper respiratory tract infection, herpes zoster, etc., while other serious side-effects, such as cardiovascular adverse events, the spread of tuberculosis, and malignant tumours, have also been reported. It is worth noting that the risk of adverse reactions is significantly positively correlated with the dose, especially the risk of venous thrombosis (28). Data from a phase IIIb–IV postmarketing safety study of tofacitinib showed a significantly increased risk of thromboembolism and death in patients over 50 years of age with at least one cardiovascular risk factor (28). In 2021, data from another study comparing tofacitinib with tumour necrosis factor (TNF) inhibitors in rheumatoid arthritis showed a higher risk of major cardiovascular events and cancer in the tofacitinib group (29). However, most of these results are based on long-term observations of drug use in patients with rheumatoid arthritis, compared with the AA population, which is younger and generally without underlying disease.
This study showed that tofacitinib (5 mg, twice daily) has good safety in patients with AA, the side-effects are usually mild, and no severe AEs occurred. Facial acne/acne-like rash and scalp seborrhoeic dermatitis/folliculitis were the most commonly reported AEs, with a higher incidence than other reports (6, 30). The incidence of upper respiratory tract infections was lower than that reported, which may be because of the increasing use of masks by Chinese residents to prevent COVID-19 infection. In the current study, only 1 patient had a mild haemoglobin decline. No other adverse haematological reactions were reported. JAK2 is involved in the erythropoietin hematopoietic pathway, and its inhibition can lead to a variety of haematological adverse reactions (31, 32). Tofacitinib is mainly against JAK1 and JAK3 pathways, which may explain its relatively safe haematological system characteristics. Slightly lower than previous studies, the current study also showed that 7.9% (10/126) of patients developed dyslipidaemia during treatment (30). Although it has been shown that JAK inhibitors may reduce foam cell accumulation and ultimately improve vascular stiffness by inhibiting γ-IFN signalling (33), the mechanism of blood lipid changes caused by tofacitinib and whether it will increase the risk of related cardiovascular events are not clear. In addition, 1 patient developed idiopathic facial neuritis (Bell palsy) during medication, which has not been reported previously.
This study has some limitations. Firstly, the sample size was small and only single-centre clinical data were included. Secondly, 13 patients used in the early stage were lost to follow-up. Nevertheless, the study results suggest that JAK inhibitors could be an ideal systemic treatment for some patients if long-term security in larger cohorts is confirmed in further investigations.
In conclusion, this study confirmed the efficacy and safety of tofacitinib in a real-life setting for AA, with a satisfactory drug survival rate, providing supporting data for the clinical application of tofacitinib in patients with AA. Potential prognostic characteristics associated with long-term outcomes include nail involvement and the sex of the patient. Further studies on the drug retention rate of other JAK inhibitors in the treatment of AA are needed to investigate the long-term outcomes of different types of JAK inhibitors and provide evidence for treatment options in real-world settings.
ACKNOWLEDGEMENTS
Reviewed and approved by the institutional research ethics boards of Xiangya Hospital, Central South University, (Changsha, China); approval number: 202303049. This study was also conducted in adherence to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
REFERENCES
- Gilhar A, Etzioni A, Paus R. Alopecia areata. N Engl J Med 2012; 366: 1515–1525.
- Mostaghimi A, Gao W, Ray M, Bartolome L, Wang T, Carley C, et al. Trends in prevalence and incidence of alopecia areata, alopecia totalis, and alopecia universalis among adults and children in a US employer-sponsored insured population. JAMA Dermatol 2023; 159: 411–418.
- Strazzulla LC, Wang EHC, Avila L, Lo Sicco K, Brinster N, Christiano AM, et al. Alopecia areata: disease characteristics, clinical evaluation, and new perspectives on pathogenesis. J Am Acad Dermatol 2018; 78: 1–12.
- Lensing M, Jabbari A. An overview of JAK/STAT pathways and JAK inhibition in alopecia areata. Front Immunol 2022; 13: 955035.
- Damsky W, King BA. JAK inhibitors in dermatology: the promise of a new drug class. J Am Acad Dermatol 2017; 76: 736–744.
- Liu LY, Craiglow BG, Dai F, King BA. Tofacitinib for the treatment of severe alopecia areata and variants: a study of 90 patients. J Am Acad Dermatol 2017; 76: 22–28.
- Sanchez-Diaz M, Diaz-Calvillo P, Rodriguez-Pozo JA, Tercedor-Sánchez J, Cantudo-Cuenca MR, Molina-Leyva A, et al. Tofacitinib for treatment of alopecia areata: real-world evidence and factors associated with therapeutic response. Acta Derm Venereol 2022; 102: adv00736.
- Park HS, Kim MW, Lee JS, Yoon HS, Huh CH, Kwon O, et al. Oral tofacitinib monotherapy in Korean patients with refractory moderate-to-severe alopecia areata: a case series. J Am Acad Dermatol 2017; 77: 978–980.
- AlMarzoug A, AlOrainy M, AlTawil L, AlHayaza G, AlAnazi R, AlIssa A, et al. Alopecia areata and tofacitinib: a prospective multicenter study from a Saudi population. Int J Dermatol 2022; 61: 886–894.
- King B, Zhang X, Harcha WG, Szepietowski JC, Shapiro J, Lynde C, et al. Efficacy and safety of ritlecitinib in adults and adolescents with alopecia areata: a randomised, double-blind, multicentre, phase 2b-3 trial. Lancet 2023; 401: 1518–1529.
- King B, Ohyama M, Kwon O, Zlotogorski A, Ko J, Mesinkovska NA, et al. Two phase 3 trials of baricitinib for alopecia areata. N Engl J Med 2022; 386: 1687–1699.
- King BA, Craiglow BG. Janus kinase inhibitors for alopecia areata. J Am Acad Dermatol 2023; 89: S29–s32.
- Egeberg A, Ottosen MB, Gniadecki R, Broesby-Olsen S, Dam TN, Bryld LE, et al. Safety, efficacy and drug survival of biologics and biosimilars for moderate-to-severe plaque psoriasis. Br J Dermatol 2018; 178: 509–519.
- Guo L, Feng S, Sun B, Jiang X, Liu Y. Benefit and risk profile of tofacitinib for the treatment of alopecia areata: a systemic review and meta-analysis. J Eur Acad Dermatol Venereol 2020; 34: 192–201.
- Nash P, Kerschbaumer A, Dörner T, Dougados M, Fleischmann RM, Geissler K, et al. Points to consider for the treatment of immune-mediated inflammatory diseases with Janus kinase inhibitors: a consensus statement. Ann Rheum Dis 2021; 80: 71–87.
- Olsen EA, Hordinsky MK, Price VH, Roberts JL, Shapiro J, Canfield D, et al. Alopecia areata investigational assessment guidelines – Part II. National Alopecia Areata Foundation. J Am Acad Dermatol 2004; 51: 440–447.
- Taneja N, Gupta S. Apremilast is efficacious in refractory alopecia areata. J Dermatolog Treat 2020; 31: 727–729.
- van den Reek J, Kievit W, Gniadecki R, Goeman JJ, Zweegers J, van de Kerkhof PCM, et al. Drug survival studies in dermatology: principles, purposes, and pitfalls. J Invest Dermatol 2015; 135: 1–5.
- Jung JY, Lee E, Kim JW, Suh CH, Kim HA. Efficacy and drug retention of tofacitinib in rheumatoid arthritis: from the nationwide Korean College of Rheumatology Biologics registry. Clin Exp Rheumatol 2023; 41: 1034–1041.
- Zhang W, Li X, Chen B, Zhang J, Torres-Culala KMT, Zhou C. Oral tofacitinib and systemic corticosteroids, alone or in combination, in patients with moderate-to-severe alopecia areata: a retrospective study. Front Med (Lausanne) 2022; 9: 891434.
- Jabbari A, Sansaricq F, Cerise J, Chen JC, Bitterman A, Ulerio G, et al. An open-label pilot study to evaluate the efficacy of tofacitinib in moderate to severe patch-type alopecia areata, totalis, and universalis. J Invest Dermatol 2018; 138: 1539–1545.
- Zhou C, Li X, Wang C, Zhang J. Alopecia areata: an update on etiopathogenesis, diagnosis, and management. Clin Rev Allergy Immunol 2021; 61: 403–423.
- Klein SL, Flanagan KL. Sex differences in immune responses. Nat Rev Immunol 2016; 16: 626–638.
- Sandborn WJ, Su C, Panes J. Tofacitinib as induction and maintenance therapy for ulcerative colitis. N Engl J Med 2017; 377: 496–497.
- Ruperto N, Brunner HI, Synoverska O, Ting TV, Mendoza CA, Spindler A, et al. Tofacitinib in juvenile idiopathic arthritis: a double-blind, placebo-controlled, withdrawal phase 3 randomised trial. Lancet 2021; 398: 1984–1996.
- Deodhar A, Sliwinska-Stanczyk P, Xu H, Baraliakos X, Gensler LS, Fleishaker D, et al. Tofacitinib for the treatment of ankylosing spondylitis: a phase III, randomised, double-blind, placebo-controlled study. Ann Rheum Dis 2021; 80: 1004–1013.
- Taneja V, El-Dallal M, Haq Z, Tripathi K, Systrom HK, Wang LF, et al. Effectiveness and safety of tofacitinib for ulcerative colitis: systematic review and meta-analysis. J Clin Gastroenterol 2022; 56: e323–e333.
- Winthrop KL, Cohen SB. Oral surveillance and JAK inhibitor safety: the theory of relativity. Nat Rev Rheumatol 2022; 18: 301–304.
- Ozdede A, Yazıcı H. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med 2022; 386: 1766.
- Yan D, Fan H, Chen M, Xia L, Wang S, Dong W, et al. The efficacy and safety of JAK inhibitors for alopecia areata: a systematic review and meta-analysis of prospective studies. Front Pharmacol 2022; 13: 950450.
- Akada H, Akada S, Hutchison RE, Sakamoto K, Wagner KU, Mohi G. Critical role of Jak2 in the maintenance and function of adult hematopoietic stem cells. Stem Cells 2014; 32: 1878–1889.
- Miyake M, Goodison S, Lawton A, Zhang G, Gomes-Giacoia E, Rosser CJ. Erythropoietin is a JAK2 and ERK1/2 effector that can promote renal tumor cell proliferation under hypoxic conditions. J Hematol Oncol 2013; 6: 65.
- Hao XR, Cao DL, Hu YW, Li XX, Liu XH, Xiao J, et al. IFN-gamma down-regulates ABCA1 expression by inhibiting LXRalpha in a JAK/STAT signaling pathway-dependent manner. Atherosclerosis 2009; 203: 417–428.