ORIGINAL REPORT
Real-world Experience of Abrocitinib Treatment in Patients with Atopic Dermatitis and Hand Eczema: Up to 28-week Results from the BioDay Registry
Esmé KAMPHUIS1#, Celeste M. BOESJES2#, Laura LOMAN1, Marijke KAMSTEEG3, Inge HAECK4, Anneke M. T. VAN LYNDEN-VAN NES5, Klaziena POLITIEK6, Liana F. VAN DER GANG2, Marlies DE GRAAF2, Marjolein S. DE BRUIN-WELLER2* and L. A. SCHUTTELAAR1*
Departments of Dermatology, 1University Medical Center Groningen, Groningen, 2University Medical Center Utrecht, Utrecht, 3Radboud University Medical Center, Nijmegen, 4Reinier de Graaf Gasthuis, Delft, 5Meander Medical Center, Amersfoort, and 6Medical Center Leeuwarden, Leeuwarden, The Netherlands
#These authors share first authorship. *These authors share last authorship.
Limited daily practice data on the effect of abrocitinib in patients with atopic dermatitis are available. The aim of this multicentre prospective study is to evaluate the effectiveness and safety of abrocitinib in patients with atopic dermatitis treated in daily practice. In a subgroup, the effectiveness of abrocitinib on hand eczema was evaluated. A total of 103 patients from the BioDay registry were included in the study: week 4 (n = 95), week 16 (n = 61) and week 28 (n = 39). At week 28, the Eczema Area and Severity Index (EASI)-50/75/90 was achieved by 81.8%, 57.6%, and 18.2%, respectively, and the weekly average pruritus numerical rating scale ≤ 4 by 62.9%. The effectiveness of abrocitinib was not significantly different between dupilumab non-responders and dupilumab-naïve patients/responders, and between upadacitinib non-responders and upadacitinib-naïve patients/responders. Mean ± standard deviation Hand Eczema Severity Index decreased from 27.4 ± 27.7 at baseline to 7.7 ± 12.1 at week 28 (n = 31). Thirty-two patients (31.1%) discontinued treatment due to ineffectiveness (n = 17), adverse events (n = 9) or both (n = 3). The most frequently reported adverse event was nausea (n = 28). In conclusion, abrocitinib is an effective treatment for atopic dermatitis and can be effective for patients with previous inadequate response to dupilumab or upadacitinib. Furthermore, hand eczema can improve in patients treated with abrocitinib for atopic dermatitis.
Key words: atopic dermatitis; hand eczema; abrocitinib; JAKinhibitor; daily practice.
SIGNIFICANCE
Daily practice studies on the effectiveness and safety of abrocitinib for atopic dermatitis and hand eczema in patients with atopic dermatitis are limited. This study showed that abrocitinib can be an effective treatment for patients with atopic dermatitis, including those with previous inadequate response to dupilumab or upadacitinib. In addition, abrocitinib can show an improvement on hand eczema in patients with atopic dermatitis. The majority of patients experienced at least 1 adverse event. Almost one-third (31.1%) of patients discontinued abrocitinib treatment (16.5% due to ineffectiveness and 8.7% due to adverse events).
Citation: Acta Derm Venereol 2024; 104: adv19454. DOI https://doi.org/10.2340/actadv.v104.19454.
Copyright: © Published by Medical Journals Sweden, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Submitted: Sep 15, 2023; Accepted: Dec 13, 2023; Published: Feb 7, 2024
Corr: Marie L. A. Schuttelaar, Department of Dermatology, University Medical Center Groningen, NL-9700 RB Groningen, The Netherlands
Competing interests and funding: CMB Boesjes is a speaker for AbbVie and Eli Lilly. IH is a consultant, advisory board member, and/or speaker for AbbVie, Eli Lilly, Janssen, LEO Pharma, Regeneron Pharmaceuticals, and Sanofi. Klaziena Politiek received consultancy fees for AbbVie, LEO Pharma and Sanofi Genzyme. LFvdG is a speaker for AbbVie, Regeneron Pharmaceuticals, and Sanofi. MdG is a consultant, advisory board member, and/or speaker for AbbVie Almirall, Eli Lilly, LEO Pharma, Novartis, Pfizer, Regeneron Pharmaceuticals, and Sanofi. MSdB-W is a consultant, advisory board member, and/or speaker for AbbVie, Almirall, Aslan, Arena, Eli Lilly, Galderma, Janssen, Leo Pharma, Pfizer, Regeneron, and Sanofi. MLAS is a consultant, advisory board member and/or speaker for AbbVie, Eli Lilly, Galderma, LEO Pharma, Pfizer, and Sanofi. The other authors have nothing to disclose.
INTRODUCTION
Atopic dermatitis (AD) is one of the most common chronic inflammatory skin conditions worldwide (1). The complex pathogenesis of AD is associated with immune dysregulation, epidermal barrier dysfunction, and genetic and environmental factors (2). Hand eczema (HE) is common in patients with AD; it is present in more than 50% of patients with AD in the clinical population (3). HE is often multifactorial with more than 1 underlying aetiology. In addition to atopic HE, other aetiological HE subtypes are irritant contact dermatitis (ICD), allergic contact dermatitis (ACD), and protein contact dermatitis (PCD) (4). Similar to AD, atopic HE is characterized by Th2 and Th22 activation. ICD has a Th1/Th17 immune profile and ACD shows a variable immune profile depending on the allergen.
After years without targeted systemic treatments for moderate-to-severe AD, new advanced systemic treatments are transforming AD therapy. Recently, 3 oral Janus kinase (JAK)-inhibitors have been approved for the treatment of AD (baricitinib, upadacitinib and abrocitinib) (5). Several pro-inflammatory cytokines that signal via the JAK-signal transducers and activators of transcription (JAK-STAT) pathways contribute to AD pathogenesis. Because of their involvement in multiple pathways, JAK-inhibitors provide a promising treatment option for patients with AD, as well as for patients who have failed on other systemic treatments. In addition, JAK-inhibitors might therefore effectively treat HE, which often has a multifactorial aetiology. Abrocitinib, a selective JAK1-inhibitor, has proven to be an effective treatment for patients with AD in clinical trials and is approved for patients aged ≥ 18 years with moderate-to-severe AD (6–10). Currently, only 2 small single-centre studies are available regarding the effect of abrocitinib on AD in daily practice (11, 12). In addition, 1 case report on the effectiveness of abrocitinib on HE has been published (13).
The aim of this study is to evaluate the effectiveness and safety of abrocitinib in patients with AD, including those with previous inadequate response to dupilumab or upadacitinib, in daily practice. Furthermore, for a subgroup of patients with AD, the effectiveness of abrocitinib on HE was evaluated.
MATERIALS AND METHODS
Study population and treatment
All patients with AD receiving abrocitinib, between March 2022 and June 2023, from the dermatology departments of 3 academic and 3 non-academic hospitals in the Netherlands were included. All patients with HE who received abrocitinib for their AD at the University Medical Center Groningen (UMCG) were included in the HE analyses. The patients were included in the Dutch BioDay registry, and the study was considered to be non-interventional by the local medical ethics committee (METC 18/239). The study was conducted in accordance with the principles of the Declaration of Helsinki.
Patients were administered 200 mg abrocitinib once daily (QD). In cases where patients were over 65 years of age, had comorbidities, or were taking medications that interacted with abrocitinib, patients were administered 100 mg or 50 mg QD. During treatment the dosage could be adjusted. Previous systemic therapy was discontinued prior to abrocitinib initiation. Patients were recorded as being in the washout of previous systemic therapy at baseline when prednisolone, cyclosporine A, or JAK-inhibitors had been used within 1 week, methotrexate within 4 weeks and biologics within 10 weeks prior to baseline. The wash-out period for ultraviolet (UV) therapy was 4 weeks. Concomitant usage of topical corticosteroids and topical calcineurin inhibitors was permitted. Patients visited the outpatient clinic at baseline, after 4, 8, and 16 weeks, and thereafter every 3 months.
Outcome measures
Outcome measures were assessed at every visit. AD disease severity was assessed using the Eczema Area and Severity Index (EASI) (14) and the 6-point Investigator Global Assessment (IGA) (15). Endpoints were an EASI improvement of ≥ 50%, ≥ 75%, or ≥ 90% (EASI-50, EASI-75, EASI-90, respectively) compared with baseline. Absolute cut-off scores were EASI ≤ 7 (16, 17), EASI ≤ 4 (18), and IGA ≤ 1 (19). In addition, patient-reported outcomes measures (PROMs) included the average pruritus numerical rating scale (NRS) (20) for the past week, the Patient-Oriented Eczema Measure (POEM) (21), the Dermatology Life Quality Index (DLQI) (22), the Atopic Dermatitis Control Tool (ADCT) (23) (only assessed at baseline and week 16), and the Patient Global Assessment of Disease Status (PGADS) (24). Absolute cut-off scores were NRS-pruritus ≤ 4 (16), NRS-pruritus ≤ 1 (16, 17), POEM ≤ 7 (16), DLQI ≤ 5 (16), ADCT < 7 (23), or PGADS ≥ 3 (24). HE disease severity was assessed with the Hand Eczema Severity Index (HECSI) (25) and the Photographic guide (26). Endpoints were a HECSI score improvement of ≥ 50%, ≥ 75%, or ≥ 90% (HECSI-50, HECSI-75, HECSI-90, respectively) compared with baseline and (almost) clear on the Photographic guide.
Dupilumab and upadacitinib (non-)responders
Outcomes of the EASI, NRS-pruritus, DLQI, and POEM were stratified by dupilumab or upadacitinib non-responders (dup-NR, upa-NR) vs dupilumab- or upadacitinib-naïve patients/responders (dup-naïve/R, upa-naïve/R). Non-responders were defined as patients who discontinued treatment due to ineffectiveness or a combination of ineffectiveness/adverse events (AEs). Naïve patients were defined as patients with no previous dupilumab or upadacitinib treatment. Responders were defined as patients who discontinued treatment due to AEs or other reasons.
Safety
AEs and laboratory parameters were monitored during each visit. AEs were quantified as the rate of AEs per 100 patient-years (PY) and the proportion of patients who reported experiencing ≥ 1 AE. AEs that led to treatment discontinuation were considered to be severe. AEs that required treatment were considered to be moderate. All other AEs were considered to be mild.
Statistical analysis
Statistical analyses were performed with IBM SPSS Statistics for Windows (version 27.0) (Armonk, NY, USA.), SAS v9.4, and Rstudio (Boston, MA, USA). Figures were made using GraphPad Prism (version 8; Boston, MA, USA). For a subgroup of patients, analyses were performed to correct for dropouts over time as this provides more accurate estimates of the effect of abrocitinib and to analyse the differences between dup/upa-naïve/R and dup/upa-NR. The subgroup included patients who started abrocitinib treatment at least 16 weeks before data lock. The analyses were based on 16 weeks instead of 28 weeks data, due to the substantially lower number of patients who reached the 28-week visit. Firstly, multiple imputation (MI) was applied on outcomes (EASI, NRS-pruritus, DLQI, POEM) (16) to reduce risk of bias and loss of statistical power. The variables age, sex, wash-out of oral immunosuppressive therapy at baseline, number/reason of dropouts, and dupilumab/upadacitinib non-responders were used as predictors. The data were imputed 30 times, based on the percentage of missing data of these variables (29.8%) (27). After MI, all imputed outcomes of patients after discontinuation of abrocitinib treatment were excluded from the analyses to avoid bias. A linear regression model was used for continuous outcomes (mean EASI, mean NRS-pruritus, mean DLQI, mean POEM), in which a residual covariance (i.e. GEE-type) matrix was included to correct for multiple measurements per patient over time. The effect of follow-up time and interaction of the follow-up time with dupilumab and upadacitinib (non-)responders was tested with likelihood ratio tests. A logistic regression with a random intercept was used for dichotomous outcomes (EASI ≤ 7, NRS-pruritus ≤ 4, DLQI ≤ 5, POEM ≤ 7) (16). Thereafter, the interaction of time with dupilumab or upadacitinib (non-)responders was included. Outcomes were used to estimate means for continuous outcomes and probabilities for dichotomous outcomes with 95% confidence intervals (95% CI). The model did not converge for POEM ≤ 7 in the upa-NR group, due to the small sample size reaching this endpoint, therefore POEM ≤ 7 was excluded from the upadacitinib (non-)responder analysis. To calculate p-values, miceadds package in R was used to pool the likelihood ratio tests (28). A p-value < 0.05 was considered statistically significant. For the total population, raw data up to 28 weeks were summarized as mean ± standard deviation (SD) or as absolute number and percentage.
RESULTS
Patient and baseline characteristics
A total of 103 patients were included: week 4 (n = 95), week 8 (n = 84), week 16 (n = 61), and week 28 (n = 39). The mean ± SD treatment period was 19.7 ± 15.3 weeks. Ninety patients were treated in an academic hospital and 13 in a non-academic hospital. Baseline and patient characteristics are described in Table I.
Abrocitinib dosage
At baseline, 96 patients received abrocitinib 200 mg QD, 6 patients 100 mg QD, and 1 patient 50 mg QD. Four patients switched from 200 mg QD to 100 mg QD (due to AEs n = 3 or patient’s wish n = 1) and 2 patients switched from 100 mg QD to 200 mg QD due to inadequate response.
Effectiveness at week 16 based on analyses
Seventy-eight patients were included for analyses. Baseline and patient characteristics, and a flowchart of this subgroup of patients, are shown in Table SI and Fig. S1. Of these patients, EASI, NRS-pruritus, DLQI, and POEM showed a significant improvement (p < 0.001) during 16 weeks of abrocitinib treatment, with the largest change from baseline to week 4 (Table II). At week 16, the probability of achieving EASI ≤ 7, NRS-pruritus ≤ 4, DLQI ≤ 5, and POEM ≤ 7 was 83.8% (95% CI 67.9–92.7), 60.9% (95% CI 44.7–74.9), 54.9% (95% CI 36.2–72.3), and 35.0% (95% CI 15.7–60.8), respectively. In total, 62 (79.5%) of the subgroup of patients had been treated with dupilumab. Thirty-seven (47.4%) patients were defined as dupilumab non-responders. Sixteen (20.5%) patients were dupilumab-naïve patients and 25 (32.1%) were defined as dupilumab responders. In addition, 27 (34.6%) of the subgroup of patients had been treated with upadacitinib. Twenty-one (26.9%) patients were defined as upadacitinib non-responders. In the upa-NR group, 28.6% (6/21) patients previously received 15 mg upadacitinib. Fifty-one (65.4%) patients were upadacitinib-naïve patients and 6 (7.7%) were defined as upadacitinib responders. No significant differences in EASI, NRS-pruritus, DLQI, and POEM over time were found between the dup-NR vs dup-naïve/R and upa-NR vs upa-naïve/R groups (Fig. 1A/B and Table II).
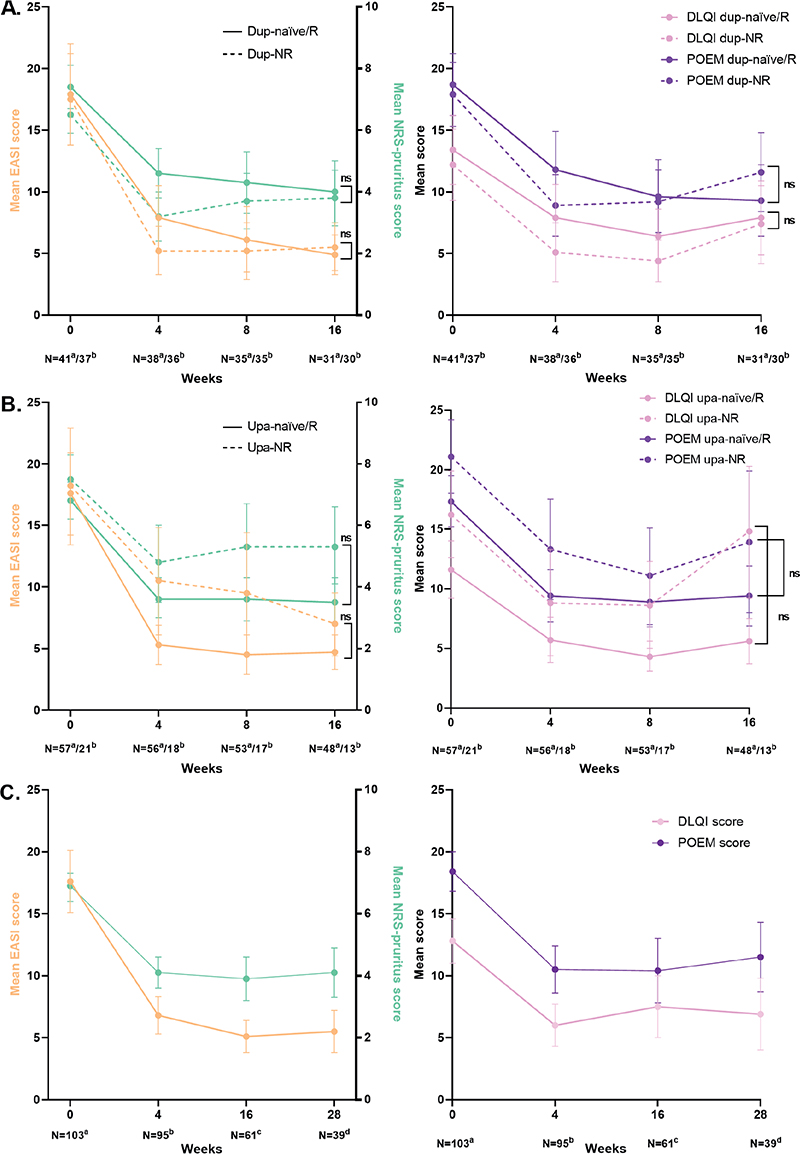
Fig. 1. Effectiveness outcomes of abrocitinib treatment. (A) Effectiveness at week 16 based on sub-analyses stratified by adupilumab-naïve patients/responders and bdupilumab non-responders (analysed with linear regression model). Data after multiple imputation. (B) Effectiveness at week 16 based on sub-analyses stratified by aupadacitinib-naïve patients/responders and bupadacitinib non-responders (analysed with linear regression model). Data after multiple imputation. (C) Effectiveness up to 28 weeks of the total cohort (raw data). Eczema Area and Severity Index (EASI): aMissing = 6 bMissing = 4 cMissing = 2 dMissing = 6. Numerical rating scale (NRS)-pruritus: aMissing = 5 bMissing = 0 cMissing = 0 dMissing = 4. Dermatology Life Quality Index (DLQI): aMissing = 32 bMissing = 39 cMissing = 27 dMissing = 16. Patient-Oriented Eczema Measure (POEM): aMissing = 33 bMissing = 40 cMissing = 27 dMissing = 16. dup-naïve/R: dupilumab-naïve patients/responders; dup-NR: dupilumab non-responders; upa-naïve/R: upadacitinib-naïve patients/responders; upa-NR: upadacitinib non-responders; N: number; ns: non-significant.
| Baseline n = 78 |
Week 4 n = 74 |
Week 8 n = 70 |
Week 16 n = 61 |
p-valuea | |
| Patients who discontinued treatment, n (%) | 4 (5.1) | 4 (5.1) | 9 (11.5) | 6 (7.7) | – |
| Concomitant immunosuppressive therapy, n (%) | 41 (52.6) | 9 (11.5) | 3 (3.8) | 1 (1.3) | – |
| EASI score, mean (95% CI) | 17.7 (14.9–20.5) | 6.6 (4.9–8.3) | 5.7 (3.9–7.5) | 5.2 (4.0–6.5) | < 0.001 |
| EASI ≤ 7, probability % (95% CI) | 7.6 (3.0–18.0) | 74.6 (58.2–86.1) | 82.2 (66.6–91.5) | 83.8 (67.9–92.7) | – |
| Weekly average NRS-pruritus, mean (95% CI) | 7.0 (6.5–7.5) | 3.9 (3.3–4.5) | 4.0 (3.3–4.7) | 3.9 (3.3–4.6) | < 0.001 |
| Weekly average NRS-pruritus ≤ 4, probability % (95% CI) | 11.5 (5.9–21.5) | 67.0 (52.7–78.8) | 63.4 (47.9–76.6) | 60.9 (44.7–74.9) | – |
| DLQI score, mean (95% CI) | 12.8 (10.7–14.9) | 6.6 (4.7–8.5) | 5.4 (4.0–6.9) | 7.6 (5.6–9.7) | < 0.001 |
| DLQI ≤ 5, probability % (95% CI) | 21.1 (10.8–37.2) | 61.4 (42.0–77.8) | 63.6 (43.6–79.9) | 54.9 (36.2–72.3) | – |
| POEM score, mean (95% CI) | 18.3 (16.4–20.2) | 10.4 (8.4–12.4) | 9.5 (7.5–11.4) | 10.4 (8.1–12.7) | < 0.001 |
| POEM ≤ 7, probability % (95% CI) | 2.4 (0.5–11.2) | 43.3 (24.1–64.8) | 46.3 (25.9–68.1) | 35.0 (15.7–60.8) | – |
| Responder subgroups, n (%) Dup-NR Dup-naïve/R Dup-R Dup-naïve Upa-NR 15 mgb 30 mgb Unknown Upa-naïve/R Upa-R 15 mgb 30 mgb Upa-naïve |
n = 37 (47.4) n = 41 (52.6) n = 25 (32.1) n = 16 (20.5) n = 21 (26.9) n = 6 (7.7) n = 14 (17.9) n = 1 (1.3) n = 57 (73.1) n = 6 (7.7) n = 5 (6.4) n = 1 (1.3) n = 51 (65.4) |
n = 36 (48.6) n = 38 (51.4) n = 23 (31.1) n = 15 (20.3) n = 18 (24.3) n = 6 (8.1) n = 12 (16.2) n = 0 (0.0) n = 56 (75.7) n = 6 (8.1) n = 5 (6.8) n = 1 (1.4) n = 50 (67.6) |
n = 35 (50.0) n = 35 (50.0) n = 21 (30.0) n = 14 (20.0) n = 17 (24.3) n = 6 (8.6) n = 11 (15.7) n = 0 (0.0) n = 53 (75.7) n = 6 (8.6) n = 5 (7.1) n = 1 (1.4) n = 47 (67.1) |
n = 30 (49.2) n = 31 (50.8) n = 18 (29.5) n = 13 (21.3) n = 13 (21.3) n = 4 (6.6) n = 9 (14.8) n = 0 (0.0) n = 48 (78.7) n = 6 (9.8) n = 5 (8.2) n = 1 (1.6) n = 42 (68.9) |
– – – – – – – – – – – – – |
| Patients who discontinued treatment, n (%) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
1 (2.7) 3 (7.3) 3 (14.3) 1 (1.8) |
1 (2.7) 3 (7.3) 1 (4.8) 3 (5.3) |
5 (13.5) 4 (9.8) 4 (19.0) 5 (8.8) |
3 (8.1) 3 (7.3) 2 (9.5) 4 (7.0) |
– – – – |
| EASI score, mean (95% CI) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
17.5 (13.8–21.2) 17.9 (13.8–22.0) 18.2 (13.4–22.9) 17.6 (14.2–20.9) |
5.2 (3.3–7.2) 7.9 (5.2–10.5) 10.5 (6.1–14.8) 5.3 (3.7–6.9) |
5.2 (2.9–7.5) 6.1 (3.5–8.8) 9.5 (4.5–14.4) 4.5 (2.9–6.1) |
5.5 (3.6–7.5) 4.9 (3.3–6.5) 7.0 (4.4–9.5) 4.7 (3.3–6.1) |
0.535c 0.254d |
| EASI ≤ 7, probability % (95% CI) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
7.0 (2.0–21.6) 16.9 (7.6–33.7) 8.4 (1.8–31.4) 13.7 (6.5–26.5) |
76.8 (57.1–89.2) 63.3 (42.9–79.8) 42.9 (19.3–70.3) 78.2 (62.9–88.3) |
77.1 (56.6–89.7) 77.1 (56.5–89.7) 60.9 (29.8–85.1) 81.7 (66.6–91.0) |
76.4 (53.9–89.9) 81.3 (60.1–92.6) 68.1 (32.9–90.2) 81.7 (65.9–91.1) |
– – – – |
| Weekly average NRS-pruritus, mean (95% CI) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
6.5 (5.9–7.2) 7.4 (6.7–8.1) 7.5 (6.8–8.3) 6.8 (6.2–7.4) |
3.2 (2.4–4.0) 4.6 (3.8–5.4) 4.8 (3.5–6.0) 3.6 (3.0–4.3) |
3.7 (2.8–4.6) 4.3 (3.3–5.3) 5.3 (3.8–6.7) 3.6 (2.9–4.3) |
3.8 (2.9–4.7) 4.0 (3.0–5.0) 5.3 (4.1–6.6) 3.5 (2.8–4.3) |
0.372c 0.640d |
| Weekly average NRS-pruritus ≤ 4, probability % (95% CI) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
13.7 (5.4–30.7) 9.5 (3.4–23.7) 3.7 (0.4–25.0) 15.0 (7.3–28.1) |
78.1 (58.3–90.1) 56.1 (36.0–74.4) 56.5 (28.8–80.7) 70.2 (53.9–82.5) |
69.5 (48.0–84.9) 57.5 (35.5–76.8) 42.4 (17.2–72.3) 69.9 (52.7–82.9) |
62.9 (40.1–81.1) 58.9 (36.2–78.4) 27.2 (8.2–61.0) 70.6 (52.7–83.8) |
– – – – |
| DLQI score, mean (95% CI) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
12.2 (9.3–15.1) 13.4 (10.6–16.2) 16.2 (12.6–19.9) 11.6 (9.2–14.0) |
5.1 (2.7–7.5) 7.9 (5.3–10.6) 8.8 (4.4–13.3) 5.7 (3.8–7.6) |
4.4 (2.7–6.1) 6.4 (4.3–8.6) 8.6 (5.0–12.3) 4.3 (3.1–5.6) |
7.4 (4.2–10.5) 7.9 (4.9–10.9) 14.8 (9.2–20.3) 5.6 (3.7–7.5) |
0.639c 0.372d |
| DLQI ≤ 5, probability % (95% CI) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
24.9 (10.5–48.3) 17.5 (6.3–40.0) 9.4 (1.7–39.0) 25.2 (12.9–43.6) |
70.1 (42.3–88.2) 53.2 (29.2–75.8) 46.9 (17.3–78.9) 68.8 (49.3–83.4) |
73.5 (46.6–89.8) 53.7 (28.1–77.5) 49.3 (18.0–81.2) 71.3 (49.4–86.3) |
59.0 (31.3–82.0) 51.0 (24.6–76.8) 31.6 (5.5–78.6) 62.6 (38.3–81.9) |
– – – – |
| POEM score, mean (95% CI) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
17.9 (15.3–20.5) 18.7 (16.2–21.2) 21.1 (18.0–24.2) 17.3 (15.2–19.5) |
8.9 (6.4–11.4) 11.8 (8.8–14.9) 13.3 (9.1–17.5) 9.4 (7.2–11.6) |
9.2 (6.7–11.8) 9.6 (6.7–12.6) 11.1 (7.0–15.1) 8.9 (6.8–11.1) |
11.6 (8.4–14.8) 9.3 (6.4–12.2) 13.9 (8.0–19.9) 9.4 (6.9–11.9) |
0.291c 0.682d |
| POEM ≤ 7, probability % (95% CI) Dup-NR Dup-naïve/R Upa-NR Upa-naïve/R |
1.3 (0.1–16.1) 3.2 (0.5–17.6) – – |
54.6 (24.4–81.7) 32.7 (10.9–66.0) – – |
51.5 (23.0–79.1) 41.1 (14.0–75.0) – – |
34.6 (10.8–69.7) 34.9 (10.7–70.4) – – |
– – – – |
| Data after multiple imputation. n: number; 95% CI: 95% confidence interval; dup: dupilumab; upa: upadacitinib; NR: non-responders; naïve/R: naïve patients/responders; EASI: Eczema Area and Severity Index; NRS: numerical rating scale; DLQI: Dermatology Life Quality Index; POEM: Patient-Oriented Eczema Measure. | |||||
| ap-values based on overall likelihood ratio tests for follow-up time. bHighest previously received dose. cp-value for the difference between dup-NR and dup-naïve/R. dp-value for the difference between upa-NR and upa-naïve-R. | |||||
Effectiveness up to 28 weeks of the total cohort
AD effectiveness outcomes based on raw data are shown in Fig. 1C and Fig. 2. In the total cohort, the mean ± SD EASI changed from 17.6 ± 12.7 to 5.5 ± 5.1 after 28 weeks of treatment. EASI-50/75/90 was achieved by 68.2%, 44.7%, and 21.2% at week 4, respectively, 82.5%, 52.6%, and 22.8% at week 16, respectively, and 81.8%, 57.6%, and 18.2% at week 28, respectively. After 28 weeks, 66.7% achieved EASI ≤ 7, 51.5% achieved EASI ≤ 4, and 26.5% achieved IGA ≤ 1. At week 28, NRS-pruritus ≤ 4 was achieved by 62.9%, NRS-pruritus ≤ 1 by 11.4%, POEM ≤ 7 by 34.8%, DLQI ≤ 5 by 65.2%, and PGADS rating of at least “good” (≥ 3) by 47.8%. ADCT < 7 was achieved by 56.3% at week 16.
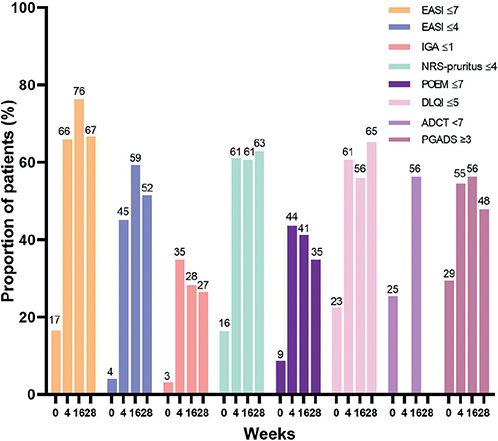
Fig. 2. Effectiveness up to 28 weeks of the total cohort. Proportion of patients who achieved an Eczema Area and Severity Index (EASI) ≤ 7, EASI ≤ 4, Investigator Global Assessment (IGA) ≤ 1, numerical rating scale (NRS)-pruritus ≤ 4, Patient-Oriented Eczema Measure (POEM) ≤ 7, Dermatology Life Quality Index (DLQI) ≤ 5, Atopic Dermatitis Control Tool (ADCT) < 7 or Patient Global Assessment of Disease (PGAD) ≥ 3.
HE outcome measures are shown in Fig. S2. The mean ± SD HECSI changed from 27.4 ± 27.7 to 15.1 ± 23.9 after 4 weeks of treatment, and to 7.7 ± 12.1 after 28 weeks of treatment. HECSI-50/75/90 was achieved by 65.2%, 52.2%, and 43.5% at week 4, respectively, 86.7%, 66.7%, and 60.0% at week 16, respectively, and 80.0%, 80.0%, and 70.0% at week 28, respectively. A score of (almost) clear on the Photographic guide was achieved by 76.5% (13/17) of the patients at week 16 and 63.6% (7/11) at week 28.
Safety
All AEs are listed in Table III. A total of 206 AEs were reported. Seventy-six patients (73.8%) experienced at least 1 AE. The most frequently reported AEs were nausea (n = 28 (71.8/100PY)) and acneiform eruption (n = 26 (66.7/100PY). In total, 42 laboratory abnormalities were documented, with increased triglyceride levels and increased creatinine phosphokinase (CPK) levels most frequently reported (both n = 14 (35.9/100PY)). The majority of AEs (81.6%) were evaluated as mild. In 4 patients abrocitinib dosage was reduced due to an AE.
| Events, n (/100PY) | Patients with ≥ 1 AEs, n (%) | |
| Total Severity of AEs Mild Moderate Severe |
206 (528.2) 168 (430.8) 26 (66.7) 12 (30.8) |
76 (73.8) – – – |
| Gastrointestinal-related conditions Nausea Abdominal pain Intestinal complaints Gastro enteritis Diarrhoea Pyrosis Dysphagia |
28 (71.8) 7 (17.9) 3 (7.7) 2 (5.1) 2 (5.1) 2 (5.1) 1 (2.7) |
28 (27.2) 7 (6.8) 3 (2.9) 2 (1.9) 2 (1.9) 2 (1.9) 1 (1.0) |
| Skin-related conditions Acneiform eruption Verrucae Mollusca contagiosa Mouth ulcers Abscess Alopecia Furunculosis Inflamed sebaceous gland cyst |
26 (66.7) 2 (5.1) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) |
26 (25.2) 2 (1.9) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) |
| Infections Herpes simplex infection Upper airway infection Folliculitis Herpes zoster infection Periodontitis Bursitis Urinary tract infection Tonsillitis Unspecified eye infection Unspecified infection finger |
15 (38.5) 5 (12.8) 4 (10.3) 3 (7.7) 2 (5.1) 2 (5.1) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) |
13 (12.6) 5 (4.9) 4 (3.9) 2 (1.9) 2 (1.9) 2 (1.9) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) |
| General conditions Headache Fatigue Weight gain Common cold Flu |
15 (38.5) 7 (17.9) 5 (12.8) 2 (5.1) 1 (2.7) |
15 (14.6) 7 (6.8) 5 (4.9) 2 (1.9) 1 (1.0) |
| Others Myalgia Dizziness Dyspnoea Oedema hands Increased allergy outbreaks Epistaxis Joint pain Fever attacks Unspecified pain eyes Ocular migraine Cognitive problems Hospitalization COVID-19 infection |
5 (12.8) 4 (10.3) 2 (5.1) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) 1 (2.7) |
5 (4.9) 4 (3.9) 2 (1.9) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) |
| Laboratory abnormalities Hypertriglyceridaemiaa Increase in CPKb Anaemiac Hypercholesterolaemiad Increase in ALATb |
14 (35.9) 14 (35.9) 9 (23.1) 3 (7.7) 2 (5.1) |
14 (13.6) 14 (13.6) 9 (8.7) 3 (2.9) 2 (1.9) |
| aTriglycerides > 2.0 mmol/L. bIncrease > 3 times upper limit of normal (ULN). cAnaemia: haemoglobin < 8.5 mmol/L (men) or < 7.5 mmol/L (women). dHypercholesterolaemia > 8.0 mmol/L. Other reference categories; leukocytopaenia < 2.0×109/l, thrombocytosis > 600×109/l, neutropaenia < 1.0×109/L, lymphocytopaenia < 0.5×109/L, creatinine increase in > 130%. AE: adverse event; CPK: creatinine phosphokinase; ALAT: alanine aminotransferase. | ||
Drop-out
A flowchart of patients is shown in Fig. 3. In total, 32 (31.1%) patients discontinued abrocitinib treatment. Seventeen patients (16.5%) discontinued treatment due to ineffectiveness (including 2 patients who received abrocitinib 100 mg QD because of interaction with comedication). Nine patients (8.7%) discontinued treatment due to AEs (acneiform eruption, fever attacks, abscesses, nausea, elevated liver enzymes, folliculitis/intestinal complaints, herpes zoster infection, nausea/dizziness, acneiform eruption/weight gain). Of those 32 patients, 6 patients were further treated with tralokinumab, 9 with dupilumab, 1 with baricitinib, and 9 with upadacitinib.

Fig. 3. Flowchart of patients during abrocitinib treatment (n = 103).
DISCUSSION
This daily practice study evaluated the effectiveness and safety of abrocitinib on AD and HE in patients with AD. Abrocitinib treatment improved clinical outcomes and PROMs of patients with AD, with a rapid change at week 4, even in patients with previous inadequate response to dupilumab or upadacitinib treatment. In addition, HE disease severity decreased in patients treat-ed with abrocitinib for AD. Nonetheless, the majority of patients (73.8%) experienced at least 1 AE. Almost one-third of patients (31.1%) discontinued abrocitinib treatment, with ineffectiveness as the most frequently reported reason.
So far, only 2 studies have been published on the effectiveness of abrocitinib in daily practice (11, 12). A prospective, observational study by Tong et al. included 16 patients with moderate-to-severe AD, all of whom had a history of inadequate response to dupilumab (11). All patients received abrocitinib 100 mg QD for 12 weeks. EASI-75 was reached by 29.4% at week 12. The current study reported a higher proportion of patients reaching EASI-75 at week 16 (52.6%). A reason for this is the higher dosage of abrocitinib (200 mg QD) in the majority of patients in the current study. Another prospective, observational, single-centre study by Olydam et al. (12) included 41 patients with AD treated with abrocitinib, of whom 51.2% started with abrocitinib 100 mg QD. The median treatment duration was 28 weeks. The study show-ed outcomes that were based on the last review of the patients. This has left the longitudinal course of disease activity unclear and made it impossible to compare this study with the current study.
Several clinical trials have been conducted with good results for abrocitinib treatment in patients with AD (6–10). A comparison between a daily practice study and clinical trials is difficult. However, the ability of abrocitinib to quickly reduce disease severity and itch, a hallmark of abrocitinib in clinical trials, is also seen in the current study, with a rapid improvement in clinical outcomes and PROMs after 4 weeks of treatment. In the current study, the drop-out rate is higher compared with clinical trials (31.1% vs approximately 10%, respectively) (6, 7, 10). This high drop-out rate was also seen in other daily practice studies with JAK-inhibitors (12, 29, 30). A reason for this may be that, during clinical trials, there were not as many alternative treatment options in daily practice as are currently available.
In this study, no significant differences were found in effectiveness of abrocitinib between dup-NR and dup-naïve/R patients. Dup-NR showed an improvement in effectiveness after 16 weeks of treatment. This supports the hypothesis that abrocitinib can be a good treatment option for patients with previous inadequate response to dupilumab, since JAK-inhibitors are involved in multiple pathways of AD pathogenesis and not only cover the targets of dupilumab. Interestingly, there were also no significant differences in effectiveness between upa-NR and upa-naïve/R. Since abrocitinib and upadacitinib are both JAK1-inhibitors, a hypothesis of similar efficacy could be argued. However, abrocitinib reduced disease severity and increased the probability of achieving endpoints in both upa-NR and upa-naïve/R, although the upa-NR tended to have a less favourable response. The non-significance might be related to the small sample size. Another reason for this could be that abrocitinib and upadacitinib both have slightly different pharmacodynamics (e.g. selectivity for the different JAKs (JAK1, JAK2, JAK3, TYK2)) (31, 32). Important to note is that a subgroup of upa-NR (n = 6) previously received 15 mg upadacitinib, and that these patients were all treated with abrocitinib 200 mg. Due to the small numbers it was not possible to perform subgroup analyses. Results of a systematic literature review and network meta-analyses of clinical trials suggested that the EASI response was highest for upadacitinib 30 mg and abrocitinib 200 mg compared with all other treatments (33). Future studies should include a larger sample size of patients who previously failed on upadacitinib 30 mg, in order to confirm the findings of the current study.
To date, only 1 case report has been published on the effectiveness of abrocitinib on HE, demonstrating positive results in atopic HE (13). The current study showed that abrocitinib improved HE in patients with AD. HECSI-75 was achieved by 80.0% of the patients at week 28. However, the subgroup of patients with HE was rather small and more than 40% had “almost clear” HE based on the Photographic guide at baseline. Therefore, no firm conclusions can be drawn. Interestingly, approximately one-third of the patients with (atopic) HE had concomitant ICD of the hands. ICD has a Th1/Th17 immune profile, while atopic HE is, like AD, characterized by Th2 and Th22 activation (34). Since JAK-inhibitors target multiple cytokine pathways beyond the Th2 pathway, it is hypothesized that JAK-inhibitors could effectively treat HE irrespective of the aetiological subtype. It would be interesting for future studies to further investigate the efficacy of both topical and oral JAK-inhibitors in different aetiological subtypes of HE.
A considerable proportion of patients (73.8%) reported at least 1 AE during abrocitinib treatment. However, not all AEs were related or possibly related to abrocitinib. Most frequently reported AEs in the current study were similar, as observed in clinical trials and the other daily practice studies (6–12). In the current study, AEs were the reason for discontinuation of abrocitinib treatment in 8.7% of the total cohort. In the daily practice study by Tong et al. (11) no AE led to discontinuation of abrocitinib. However, patients in that study were treated with a lower dosage of abrocitinib (100 mg QD) for a treatment period of only 12 weeks. In the study by Olydam et al. (12), 17.1% of the patients discontinued abrocitinib (100–200 mg QD) treatment due to AEs, which is higher than in the current study.
A strength of this study is the sub-analysis of dupilumab and upadacitinib non-responders. Further strengths are the multicentre and prospective design of the study, together with the use of many validated clinical outcomes and PROMs. In addition, the included patients were treated in both academic and non-academic hospitals. A limitation of this study is that the majority of patients might still have experienced therapeutic effects of their previous AD treatments at baseline, which could have influenced the current results. However, this limitation reflects real-world daily practice. Lastly, the high drop-out rate may have introduced a positive selection bias, therefore MI and statistical models were used to provide more accurate outcomes.
In conclusion, abrocitinib is an effective treatment for patients with AD in daily practice, and it can be effective in those with previous inadequate response to dupilumab or upadacitinib treatment. Furthermore, HE can improve in patients treated with abrocitinib for AD. However, the majority of patients reported at least 1 AE. Almost one-third of patients discontinued abrocitinib treatment. Future studies should focus on patients’ characteristics as possible predictors for effectiveness and AEs.
ACKNOWLEDGEMENTS
Funding sources: Patients included in this study participated in the BioDay registry sponsored by Eli Lilly, Sanofi Genzyme, Leo Pharma, Abbvie and Pfizer.
The study was approved by the local medical research ethics committee as a non-interventional study (METC 18-239) and was performed according to the principles of the Declaration of Helsinki.
REFERENCES
- Eichenfield LF, Tom WL, Chamlin SL, Feldman SR, Hanifin JM, Simpson EL, et al. Guidelines of care for the management of atopic dermatitis: section 1. Diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol 2014; 70: 338–351.
- Werfel T, Allam JP, Biedermann T, Eyerich K, Gilles S, Guttman-Yassky E, et al. Cellular and molecular immunologic mechanisms in patients with atopic dermatitis. J Allergy Clin Immunol 2016; 138: 336–349.
- Simpson EL, Thompson MM, Hanifin JM. Prevalence and morphology of hand eczema in patients with atopic dermatitis. Dermatitis 2006; 17: 123–127.
- Thyssen JP, Schuttelaar MLA, Alfonso JH, Andersen KE, Angelova-Fischer I, Arents BWM, et al. Guidelines for diagnosis, prevention, and treatment of hand eczema. Contact Dermatitis 2022; 86: 357–378.
- Mikhaylov D, Ungar B, Renert-Yuval Y, Guttman-Yassky E. Oral Janus kinase inhibitors for atopic dermatitis. Ann Allergy Asthma Immunol 2023; 130: 577–592.
- Simpson EL, Sinclair R, Forman S, Wollenberg A, Aschoff R, Cork M, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis: results from JADE MONO-1, a randomized, phase 3 clinical trial. Lancet 2020; 396: 255–266.
- Silverberg JI, Simpson EL, Thyssen JP, Gooderham M, Chan G, Feeney C, et al. Efficacy and safety of abrocitinib in patients with moderate-to-severe atopic dermatitis: a randomized phase 3 clinical trial. JAMA Dermatol 2020; 156: 863–873.
- Blauvelt A, Silverberg JI, Lynde CW, Bieber T, Eisman S, Zdybski J, et al. Abrocitinib induction, randomized withdrawal, and retreatment in patients with moderate-to-severe atopic dermatitis: Results from the JAK1 Atopic Dermatitis Efficacy and Safety (JADE) REGIMEN phase 3 trial. J Am Acad Dermatol 2022; 86: 104–112.
- Bieber T, Simpson EL, Silverberg JI, Thaçi D, Paul C, Pink AE, et al. Abrocitinib versus Placebo or Dupilumab for Atopic Dermatitis. N Engl J Med 2021; 384: 1101–1112.
- Reich K, Thyssen JP, Blauvelt A, Eyerich K, Soong W, Rice ZP, et al. Efficacy and safety of abrocitinib versus dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized, double-blind, multicentre phase 3 trial. Lancet 2022; 400: 273–282.
- Tong Z, Zhang Y, Zhou K, Zou Y, Wu Z, Chen J, et al. An observational study of abrocitinib in adults with moderate-to-severe atopic dermatitis after switching from dupilumab. J Am Acad Dermatol 2023; 89: 826–828.
- Olydam JI, Schlösser AR, Custurone P, Nijsten TEC, Hijnen D. Real-world effectiveness of abrocitinib treatment in patients with difficult-to-treat atopic dermatitis. J Eur Acad Dermatol Venereol 2023; 37: 2537–2542.
- Sitaru S, Preis S, Eberlein B. Successful treatment of atopic hand and foot eczema with oral janus kinase 1 inhibition. Dermatitis 2023; 34: 560.
- Hanifin JM, Thurston M, Omoto M, Cherill R, Tofte SJ, Graeber M. The eczema area and severity index (EASI): assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp Dermatol 2001; 10: 11–18.
- Futamara M, Leshem YA, Thomas KS, Nankervis H, Williams HC, Simpson EL. A systematic review of Investigator Global Assessment (IGA) in atopic dermatitis (AD) trials: many options, no standards. JAAD 2016; 74: 288–294.
- De Bruin-Weller M, Biedermann T, Bissonnette R, Deleuran M, Foley P, Girolomoni G, et al. Treat-to-target in atopic dermatitis: an international consensus on a set of core decision points for systemic therapies. Acta Derm Venereol 2021; 101: adv00402.
- Silverberg JI, Gooderham M, Katoh N, Aoki V, Pink AE, Binamer Y, et al. Optimizing the management of atopic dermatitis with a new minimal disease activity concept and criteria and consensus-based recommendations for systemic therapy. Br J Dermatol 2023; 188: ljac140.022.
- Spekhorst LS, Boesjes CM, Loman L, Zuithoff NPA, Bakker DS, Kamphuis E, et al. Successful tapering of dupilumab in atopic dermatitis patients with low disease activity: a large pragmatic daily practice study from the BioDay registry. Br J Dermatol 2023; 189: 327–335.
- Beck LA, Deleuran M, Bisonette R, de Bruin-Weller M, Galus R, Nakahara T, et al. Dupilumab provides acceptable safety and sustained efficacy for up to 4 years in an open-label study of adults with moderate-to-severe atopic dermatitis. Am J Clin Dermatol 2022; 23: 393–408.
- Phan NQ, Blome C, Fritz F, Gerss J, Reich A, Ebata T, et al. Assessment of pruritus intensity: prospective study on validity and reliability of the visual analogue scale, numerical rating scale and verbal rating scale in 471 patients with chronic pruritus. Acta Derm Venereol 2012; 92: 502–507.
- Charman CR, Venn AJ, Williams HC. The patient-oriented eczema measure: development and initial validation of a new tool for measuring atopic eczema severity from the patients’ perspective. Arch Dermatol 2004; 140: 1513–1519.
- Finlay AY, Khan GK. Dermatology Life Quality Index (DLQI) – a simple practical measure for routine clinical use. Clin Exp Dermatol 1994; 19: 210–206.
- Pariser DM, Simpson EL, Gadkari A, Bieber T, Margolis DJ, Brown M, et al. Evaluating patient-perceived control of atopic dermatitis: design, validation, and scoring of the Atopic Dermatitis Control Tool (ADCT). Curr Med Res Opin 2020; 36: 367–376.
- Griffiths C, de Bruin-Weller M, Deleuran M, Fargnoli MC, Staumont-Sallé D, Hong CH, et al. Dupilumab in adults with moderate-to-severe atopic dermatitis and prior use of systemic non-steroidal immunosuppressants: analysis of four phase 3 trials. Dermatol Ther (Heidelb) 2021; 11: 1357–1372.
- Held E, Skoet R, Johansen JD, Agner T. The hand eczema severity index (HECSI): a scoring system for clinical assessment of hand eczema. A study of inter- and intraobserver reliability. Br J Dermatol 2005; 152: 302–307.
- Coenraads PJ, Van Der Walle H, Thestrup-Pedersen K, Ruzicka T, Dreno B, De La Loge C, et al. Construction and validation of a photographic guide for assessing severity of chronic hand dermatitis. Br J Dermatol 2005; 152: 296–301.
- Donders AR, van der Heijden GJ, Stijnen T, Moons KG. Review: a gentle introduction to imputation of missing values. J Clin Epidemiol 2006; 59: 1087–1091.
- Robitzsch A, Grund S. Miceadds: Some additional multiple imputation functions, especially for ‘mice’. R package version 3.13-1. 2022. [accessed 2023 June 20]. Available from: https://cran.r-project.org/web/packages/miceadds/index.html
- Boesjes CM, Van der Gang LF, Zuithoff NPA, Bakker DS, Spekhorst LS, Haeck I, et al. Effectiveness of upadacitinib in patients with atopic dermatitis including those with inadequate response to dupilumab and/or baricitinib: results from the BioDay Registry. Acta Derm Venereol 2023; 103: adv00872.
- Boesjes CM, Kamphuis E, Zuithoff NPA, Bakker DS, Loman L, Spekhorst LS, et al. Daily practice experience of baricitinib treatment for patients with difficult-to-treat atopic dermatitis: results from the BioDay Registry. Acta Derm Venereol 2022; 102: adv00820.
- Schmieder GJ, Draelos ZD, Pariser DM, Banfield C, Cox L, Hodge M, et al. Efficacy and safety of the Janus kinase 1 inhibitor PF-04965842 in patients with moderate-to-severe psoriasis: phase II, randomized, double-blind, placebo-controlled study. Br J Dermatol 2018; 179: 54–62.
- Parmentier JM, Voss J, Graff C, Schwartz A, Argiriadi M, Friedman M, et al. In vitro and in vivo characterization of the JAK1 selectivity of upadacitinib (ABT-494). BMC Rheumatol 2018; 2: 23.
- Silverberg JI, Hong HC, Calimlim BM, Lee WJ, Teixeira HD, Collins EB, et al. Comparative efficacy of targeted systemic therapies for moderate-to-severe atopic dermatitis without topical corticosteroids: an updated network meta-analysis. Dermatol Ther (Heidelb) 2023; 13: 2247–2264.
- Cheng J, Facheris P, Ungar B, Guttman-Yassky E. Current emerging and investigational drugs for the treatment of chronic hand eczema. Expert Opin Investig Drugs 2022; 31: 843–853.