The Management of Scabies in the 21st Century: Past, Advances and Potentials
Charlotte Bernigaud1–5, Katja Fischer2,4 and Olivier Chosidow1,4,5
1Dermatology Department, AP-HP, Henri Mondor hospital, Paris-Est University, Créteil, France, 2QIMR Berghofer Medical Research Institute, Infectious Diseases Program, Herston, Brisbane, Australia, 33Research group Dynamyc, EA7380, Faculté de Santé, EnvA, USC ANSES, Paris-Est University, Créteil, France, 4IACS (International Alliance for the Control of Scabies, and 5GrIDIST, Groupe Infectiologie Dermatologique – Infections Sexuellement Transmissibles, French Society of Dermatology
Scabies is one of the most common skin diseases worldwide, affecting 150–200 million people yearly. Scabies affects young children in particular, and has the greatest impact in poor overcrowded living conditions. The burden of the disease is now well characterized, including group A Streptococcus and Staphylococcus aureus bacterial superinfections, with reports of nephritis, acute rheumatic fever, or fatal invasive sepsis secondary to scabies. Management of scabies remains largely suboptimal from diagnosis to treatment, and progress in the development of new therapeutic measures leading to cure is urgently needed. This review gives an overview of the current limitations in the management of scabies, an update on recent advances, and outlines prospects for potential improvements.
Key words: scabies; Sarcoptes scabiei; neglected tropical disease; ivermectin; permethrin; moxidectin; acaricide discovery and development; ovicidal.
Accepted Mar 19, 2020; Epub ahead of print Mar 24, 2020
Acta Derm Venereol 2020; 100: adv00112
Corr: Charlotte Bernigaud, Department of Dermatology, Hôpital Henri Mondor, 51 Avenue du Maréchal de Lattre de Tassigny, FR-94010 Créteil, France. E-mail: charlotte.bernigaud@aphp.fr
Scabies is more than just a disease that provokes a horrendous itch. For more than a century, researchers, clinicians and public health physicians, together with policymakers, have worked to improve the management of scabies. Finally, in 2017, scabies was added to the WHO list of neglected tropical diseases after a long and still ongoing process of documenting the morbidities and burden caused by the disease. This additional and increased research activity, resulting in high-impact publications, has increased our knowledge of the biology, pathology and management of scabies, and has opened doors to new strategies.
Scabies (Latin scabere “ to scratch”) is a common parasitic disease caused by the microscopic mite Sarcoptes scabiei var. hominis (1). The burrowing mite causes intense itching, associated with typical skin lesions. The disease has been known for over 2,500 years; the Greeks and Romans were the first to write about its contagious nature. The mite was first identified and illustrated in the 17th century (2). Despite marked advances in parasitology in the 19th and 20th centuries, research into scabies has been hampered largely by limited access to the parasite and by low interest in an ectoparasite that mainly affects the poor. The earliest understanding of the mite biology and transmission was provided by Kenneth Mellanby, a British entomologist, in the 1940s during World War II (3, 4). Further advances in the physiopathology and host-parasite interactions have been made in the last 30 years (5, 6), mainly through the development of experimental animal models (7, 8). The therapeutic options for the management of scabies increased considerably in the 1970–80s with the discovery of ivermectin, one of the most important drugs currently used to treat scabies; the researchers were recently awarded, 35 years after its discovery, the 2015 Nobel Prize for Physiology and Medicine (9).
Currently, the recent expansion of multi-omics techniques will enable scientists to design large-scale mite and host molecular and biochemical analyses to develop new diagnostic tools or treatments in the near future (10–13). Scabies was, for a long time, not appropriately considered to be a true health target. In the past 10 years, stupendous efforts, made by a group of experts brought together in the International Alliance for the Control of Scabies (IACS), have given scabies the recognition it deserves (14). Thus, in 2017, the WHO decided to add scabies to the list of neglected tropical diseases and has called for large-scale action to achieve control and eradication (15).
Scabies is a prevalent disease, which is present in all parts of the world, with greatest prominence in disadvantaged populations living in tropical and subtropical regions, and has a documented significant burden. The latest estimates suggest that 150–200 million people have scabies in the world every year, and that the scabies burden is particularly high in Asia, Oceania, and Latin America (16). Young children in underprivileged populations living in crowded conditions are more often at risk (17). Transmission of scabies occurs mainly via skin-to-skin contact and, less frequently, via fomites within a patient mite-contaminated environment (generally in the context of severe forms of scabies; see below). As scabies is contagious, persons sharing the same household with patients may frequently be affected. This is especially the case in severe scabies, i.e. profuse or crusted scabies, in which the mite burden per person is dramatically increased, small epidemics around a single case can easily develop, and are fuelled by overcrowded households and transient lifestyles. The risk of transmission is known to depend on the patient’s mite load, household size and population concentration, and how individuals interact with each other. Indeed, people living in clustered communities or in crowded housing conditions are at higher risk of scabies and outbreaks. In high-income nations, high endemicity of scabies is often reported in closed communities and institutional settings, such as hospitals, child-care and elderly-care residential facilities (18, 19), prisons, schools, homeless populations, and refugee camps (20–22).
For a long time, the scabies mite has been erroneously perceived as an ectoparasite that just causes itching. However, recent epidemiological studies indicate increas-ingly substantial morbidity, and even mortality (23), due to scabies infection, mostly caused by bacterial infections appearing with the parasitic infestation (14, 24). It has been hypothesized that scratching of lesions in response to the immense itch is present more often in scabies than in any other pruritic skin affections (25). The discomfort caused by the intense itch can have direct consequences, i.e. depriving patients of sleep (26), interfering with concentration at work or school, leading to a negative impact on attendance, performance (27) and quality of life (28). Scratching scabies lesions themselves leads to breaches in skin barrier that creates an entry point for opportunistic commensal or pathogenic bacteria that can become invasive, such as group A Streptococcus (GAS) and S. aureus (29). These bacteria lead to secondary infection of the epidermis, also known as pyoderma or impetigo, which can become more severe and cause skin and soft-tissue infections (including necrotizing fasciitis), septicaemia, or more invasive bacterial infections (24). In some cases, immune-mediated diseases can occur follow-ing infection, such as glomerulonephritis (30) or acute rheumatic fever (31), both of which can become chronic. This association between scabies parasites and bacterial pathogens is observed mainly in tropical or subtropical areas of the globe and in remote locations (17); with some data suggesting that up to 40% of impetigo lesions can be linked with scabies, especially among young children (32, 33). This particular link was established early in the 1970s, with epidemiological studies showing epidemics of acute glomerulonephritis in Trinidad (34) or Southern Africa (35) contemporaneously with scabies outbreaks, or in interventional studies in the field showing reduction in childhood haematuria following scabies treatment (30), or reduction in impetigo or skin sores prevalence paralleling a reduction in scabies numbers during mass drug administration (MDA) campaigns (36–38). More recent fundamental experimental work has supported and, in part, explained these observations with evidence of direct effects of mite gut proteins (serpins and serine proteases) in downregulating the innate host immunity including complement defence and neutrophil function, thereby modulating the microenvironment around the mite, allowing associated bacteria to flourish (39–43). Beyond the itch, scabies still causes a significant social impact, affecting quality of life and school or job absenteeism amongst infested patients. Its marked social, economic and psychological ramifications are underscored, but are sufficient to justify global improvement in its management.
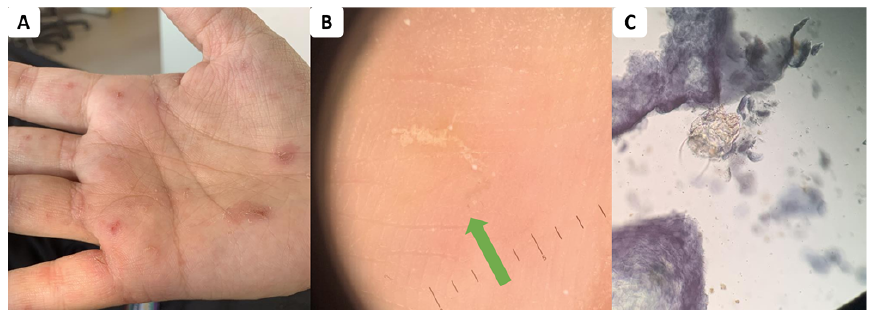
Typically, the first symptoms of scabies are severe itch that worsens at night (44) and typical skin lesions caused by the penetration and progress of the mite through the epidermis. Most scabies lesions are found in classical sites, such as finger webs, hands, wrists, periumbilical skin, buttocks, genitals, periareolar region in females, or feet (1). In adult patients, the head is usually not affected, although it may be involved in infants, and babies (45). Burrows, vesicles, pustules, nodules, or excoriated pruritic papules are the most common lesions observed, all of which indicate the presence of a mite within the epidermis (Fig. 1A). The severe forms of scabies include profuse and crusted scabies. Both presentations are characterized by a very high parasite burden, with hundreds, thousands or even millions of mites per patient, and the development of extensive lesions. In such forms of scabies, hyperkeratotic skin (rather than real crusts) may be restricted to a finger, toe, or the scalp, for example, or diffusely affect multiple skin sites, including the face and palms and soles (1). This condition is seen in the elderly or in patients with underlying immunodeficiency from any cause (transplant recipients, corticosteroid use including topically-applied medication, HIV-, or HTLV-1-infested patients) (46–48).
The diagnosis of scabies is easy if a burrow, the specific lesion of scabies, is observed at a typical site of predilection. However, burrows may not be visible and diagnosis of scabies can be challenging, leading to misdiagnosis and mismanagement. Clinical diagnostic algorithms have been created to assist health-workers in recognizing scabies using a combination of parameters of patient history and clinical arguments, such as, for example, a history of diffuse itch, presence of lesions in typical skin areas, and itching in household members (49, 50). These diagnostic aids, which have proved reliable in endemic regions, have to be extrapolated and optimized across a larger range of settings, from the dermatologist’s daily practice offices to resource-poor field settings, regardless of local prevalence. With this in mind, consensus criteria for the diagnosis of scabies were developed recently using a 4-round Delphi process including 34 international experts under the aegis of IACS (51). The IACS 2019 Criteria includes 3 levels of definition (confirmed scabies, clinical scabies, and suspected scabies case) and 8 subcategories. The accuracy and reproducibility of scabies diagnosis using these criteria have yet to be validated, as well as the ease of using it for GPs, experts in dermatology and other specialties, and non-expert health-workers (52).
As yet, a simple test for scabies based on molecular markers is still not available. Non-invasive methods have been developed for directly identifying the mite in diagnostic scrapings (53). The gold standard remains the visualization of the parasite (adults or immature forms), eggs, eggshell fragments or mite faecal pellets by light microscopic examination of a skin scraping. New technologies have been customized. Their sensitivity and specificity are summarized in Table I. Light microscopy examination has an excellent specificity (Fig. 1B), but is highly operator-dependent and is time-consuming, as repeated scrapings may be necessary. Dermoscopy or epiluminescence microscopy are tools used in daily clinical practice by dermatologists for a variety of cutaneous disorders, including parasitic infestations (54). The diagnosis of scabies using dermoscopy is confirmed by the observation of the “jet-with-contrail” pattern in the skin, representing a mite and its burrow, or an image of a black or brown triangle, the “delta-wing jet” sign representing the head of the mite (Fig. 1C) (55). Videodermatoscopy utilizes a dermoscope with a video camera connected to a computer that allows very high magnification and can be used to assess the viability of living mites. These are expensive techniques and therefore some authors have adapted low-cost equipment, used in botanical or entomology investigation, for use in the medical assessment of scabies (56). Reflectance confocal microscopy has been developed more recently for pigmented skin lesions, to differentiate malignant melanoma from benign naevi. The system uses an 830-nm wavelength diode laser and provides high optical resolution to penetrate to a depth of 200–300 µm into the skin. Imaging of the scabies mites and eggs using this device has been described (57). Some authors have tried to develop diagnostic techniques using molecular tools, such as matrix-assisted laser desorption ionization – time of flight (MALDI-TOF), antigen detection system or PCR specifically targeting scabies DNA. With PCR, while most studies have found a very high specificity, often close to 100%, sensitivity was continually low, ranging from 30% to 60% (58–60), poorer compared with parasite observation either by microscopic or dermoscopic examination (61). No biomarker-based diagnostic kits have been developed for use as a simple and rapid method to identify mite infection without dermatological skills.
Improvement in scabies management is essential and will come from better identification of which patients need to be treated. Thus, the accurate and definitive diagnosis of scabies is crucial. Non-invasive diagnostic tools have been developed, but will have to be improved further.
Treatment of small clusters (individual and family level)
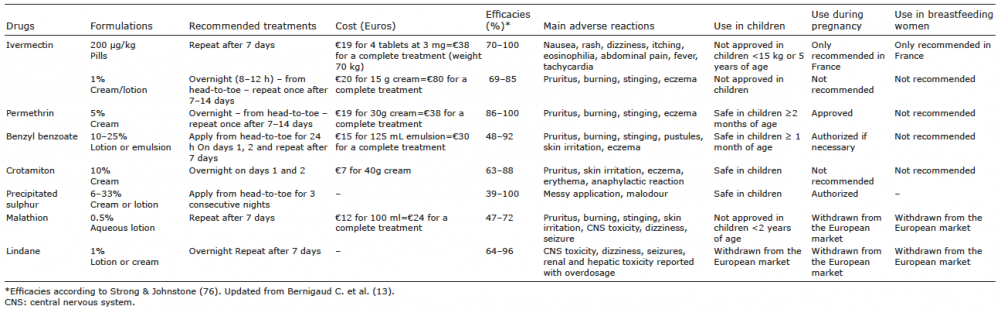
Treatment must be prescribed for all confirmed cases of scabies, and should be given to all household and family contacts. The options available for treatment of scabies are summarized in Table II. Topical medicines were considered first-line treatment until the arrival of oral ivermectin in 1981, which was, at first, reserved for recurrent, difficult-to-treat cases, those with superinfected or eczematous skin, or for patients with crusted scabies (67). Topical agents should be applied to the entire skin surface, from “head-to-toe”, avoiding the eyes, nose, and mouth. The application period depends on the specific instructions from the manufacturer. Adverse events are reported with all topical medicines for scabies, but they appear to be limited. Oral ivermectin is given at a standard dose of 0.2 mg/kg and may be associated with lower rates of complete cure if given only once (68–70). This could be explained by the limited ovicidal activity of the drug (71) and the short half-life of ivermectin in the skin, which was shown in 2 experimental trials in a porcine scabies model (72, 73). Giving the drug with a high-fat content meal has been proposed in order to increase its absorption and, accordingly, this might increase its efficacy (74, 75). Most scabicides act by affecting the nerve and muscle function of the parasite, and they are active only against mobile stages (larva, nymph and adults) and not eggs (13). The optimal interval between dosing in the 2-dose regimen still needs to be optimized and should be a short window between larval hatching (occurring at day 2–4 of the mite life-cycle) and the development of the adult stage that can be fertilized (at day 5–8, maximum day 15). Two recent Cochrane systematic reviews of data from respectively 22 (76) and 15 (77) randomized controlled trials (RCTs) placed topical 5% permethrin and ivermectin at the same level of efficacy and safety, and are consequently considered as the reference treatments. Between these 2 systematic reviews, performed in 2010 and 2017, no newer trials were included in the evaluation, but the conclusions were different based on the regimen of the drugs used. Strong & Johnstone (76) concluded, in 2010, that topical permethrin 5% was the most efficacious agent for the treatment of scabies. Rosumeck et al. in 2017 (77), reviewing the same trials as in 2010, concluded that topical permethrin was equal to ivermectin when 2 doses were given. Overall, among all the therapeutic trials analysed, a significant heterogeneity in the methods and outcome measurements was found, making the conclusions difficult to evaluate. A French randomized clinical trial, cluster-designed with a robust protocol, is currently recruiting patients with common scabies to establish finally which treatment, 5% topical permethrin or 0.2 mg/kg oral ivermectin, both given twice at 10 days’ interval is the most efficacious (SCRATCH, NCT02407782) (78). Treatment also depends on the availability of the drug in the different countries. For example, since it was first approved in France for the treatment of scabies in 2001, oral ivermectin has been licensed only for this indication in 10 nations as first-line treatment, and is mostly used off-label and may not even be accessible in other countries. In those countries, available and cheaper medications are preferred, such as sulphur preparations and benzyl benzoate. To widen access to this key effective medication, in June 2019 the WHO added ivermectin to the 21st WHO Essential Medicines List (79).
Follow-up is necessary after treatment, to evaluate the cure of the patient and to prevent re-infestation. Treat-ment success should be expected in approximately two weeks. Itching can persist for up to one month after successful treatment. Causes of apparent treatment failure with an effective treatment include incorrect diagnosis, dermatitis secondary to the mite or topical agent, incorrect application of the topical agent, poor penetration of the agent into hyperkeratotic skin or nails, and re-infestation from scabies-infested close contacts (80). Parasite resistance has been reported for both permethrin (81, 82) and ivermectin (83, 84), but its clinical importance remains a matter of debate. Studies are lacking and surveillance for better documentation is warranted. Treatment failure has been observed, mainly because drugs are not 100% effective (74– 93% clearance is observed with permethrin and 68–86% with ivermectin (77)). Some authors recently tried to determine which factors were associated with treatment failure (85, 86). These 2 studies found that incorrect decontamination of furniture or fomites was a key factor in treatment failure. While living mites can be found in samples of environmental dust from floors and furniture of patients with scabies (87), the indirect transmission of mites by fomites is thought to be rare (3), at least in common scabies. Studies are needed to evaluate the impact of environment-decontamination procedures on the success of the treatment (88) in non-profuse cases of scabies and in cases of severe scabies with high mite-load, in order to optimize cure rates. Simplified and generalized algorithms, based on high-throughput experimental data that can be used in a large range of settings, including resource-poor population, were suggested recently (89).
Appropriate treatment should also be given for severe secondary bacterial infection. Topical antibiotic creams (e.g. mupirocin or fusidic acid) are not recommended in cases with profuse lesions. Systemic antibiotics have to target GAS and S. aureus (including MRSA in specific areas). Oral trimethoprim-sulphamethoxazole (cotrimoxazole) or intramuscular benzathine penicillin G are used in tropical endemic regions (90), whereas pristinamycin, amoxicillin/clavulanic acid or cephalexin may be used in non-tropical regions. For the itch, there is no specific treatment. Antihistamines can assist, but it is their sedative properties that are effective, rather than an anti-pruritic mechanism (91).
For the treatment of crusted scabies, there is no consensus as, to date, no randomized controlled trials have been performed. Most records come from small studies, and experience in northern Australia, where highly infested patients are seen (47). They suggest a regimen of multiple doses of oral ivermectin with repeated topical permethrin and keratolytic therapy.
For the treatment of children, only topical treatments have been approved. The application time in children can differ from that in adults. Oral ivermectin cannot be used if the patient’s body weight is less than 15 kg. Recent reports using ivermectin off-label in infants and young children (aged 1–64 months, body weight 4–14.5 kg)
are highly reassuring on the safety and efficacy of this treatment in this age group (92). Recommendations for use of ivermectin in infants may change in the near future. For the treatment of pregnant women, only one country allows the use of ivermectin, as a second-line treatment after 5% topical permethrin, at any trimester of the pregnancy supported by an expert recommendation (93). Most exclusions of women from ivermectin treatment are for basic precaution rather than any anticipated foetal toxicity. In practice, thousands of women have been treated inadvertently before their pregnancy becomes known in onchocerciasis eradication programmes in Africa. Occurrence of miscarriage, stillbirth, or birth defects in the reference population did not differ significantly (94, 95). Continued surveillance is necessary, as well as more fundamental work to enable this target population to be treated adequately.
Treatment of large clusters (collectivity level)
As scabies is a contagious disease spread by skin-to-skin contact, people living in crowded communities are at greater risk. In these collectivity settings, because patients and their close contacts have to be treated all at the same time and because prevalence can be very high in some communities, the opportunity of MDA has emerged to control scabies in endemic spaces (96). The first MDA was performed in a scabies-endemic region of Panama in the 1970s by Taplin et al. (97). Successively, multiple programmes have evolved in all parts of the world to control scabies by MDA, firstly with lindane, topical permethrin, followed by the use of oral ivermectin (summarized in Table SI). Only one trial, the Skin Health Intervention Fiji Trial (SHIFT) has randomized 3 islands in Fiji to 1 of 2 intervention strategies, i.e. oral ivermectin and topical permethrin, compared with standard care as control (36). At 12 months, mass treatment with ivermectin was found to be the most effective, with a relative reduction of 94% from baseline in the prevalence of scabies and 67% in impetigo. During all these years, many MDA programmes have resulted in successful and significant reduction in the prevalence of scabies in highly endemic or endemic settings. Controversial results have been reported only from Australia (see Table SI), presumably because the adherence of the target population with the treatment regimen was poor. Fewer data exist on the sustainability of success of MDA in the longer term, but this approach seems to be efficient (98, 99), especially in communities with a scabies prevalence higher than 5% (100). Interestingly, the treatment of scabies alone also seems to result in a significant reduction in GAS impetigo (36) and kidney complications, signified by the reduction in haematuria (30). A recent study has indicated that it is not necessary to add antibiotics during MDA for scabies to reduce the prevalence of impetigo (37). Although, as most MDAs target only one disease, some programmes have looked at the potential to integrate scabies MDA in other schemes for neglected tropical diseases eradication programmes (101), such as onchocerciasis, lymphatic filariasis, trachoma, schistosomiasis, yaws, or infection with soil-transmitted helminths. On a smaller scale, combining ivermectin and albendazole administration to treat both lymphatic filariasis and onchocerciasis, or scabies and lymphatic filariasis (102), or scabies and strongyloidiasis (103) were found to be effective and safe. On a larger scale, the co-administration of azithromycin and ivermectin for targeting trachoma and scabies in the Choiseul Province, Solomon Islands, was also found to be effective, feasible and secure in 26,188 enrolled participants (104). Remarkably, MDAs for scabies control have had additional unintended downstream effects, as they have been found to be efficient in controlling head lice (105) and Anopheles farauti, the vector of malaria in the Solomon Islands (106). All these interesting programmes provide positive results and robust evidence to encourage MDA with ivermectin to control scabies in highly endemic populations on a larger scale. Optimization of these programmes will be required in order to understand factors associated with success, defining the appropriate regimen to use, and determining the numbers of rounds of MDA (24).
Similarly, controlling scabies at a larger scale by MDAs has been extrapolated for smaller outbreaks in closed institutions, such as schools (107), prisons (108), age-care facilities (18), and, more recently, in asylum seekers (109).
As mentioned above, treatment either with a topical acaricide or oral treatment with ivermectin are the current standards of care for common scabies. Most patients with scabies infection will recover with a suitable medical intervention, but patients often require multiple treatments and/or a combination of topical and systemic drugs. The major limitations of current therapies are poor compliance with repeated treatments, limited activity against eggs, and half-lives too short to cover the whole 14-day life cycle of the mite (13). The recent development of an experimental porcine scabies model provides real potential to conduct translational preclinical and pharmacokinetic studies with new drug candidates.
In order to optimize and improve the therapeutic options for scabies treatment, the concept of translating existing drugs used in the veterinary clinic to humans was investigated (72, 73). Moxidectin is a molecule compound suitable for oral administration. It is a member of the same family as ivermectin, and was recently developed and approved by the US Food and Drug Administration (FDA) for treatment of onchocerciasis. Moxidectin has a very interesting pharmacological profile: rapid absorption, large distribution, and a much longer half-life in plasma and, importantly, in the skin than ivermectin (72), potentially covering the entire life-cycle of the scabies mite. In a pilot trial in the experimental pig model for scabies performed in France, moxidectin used orally, at a single dose of 0.3 mg/kg, was found to be more effective than the conventional 2 doses of ivermectin at 1-week interval (0.2 mg/kg) (72). A multicentre clinical phase II trial in humans is in progress in Australia and France, with the aim of developing moxidectin as a new single-dose treatment for scabies (NCT03905265) (117).
The use of higher doses of ivermectin for treatment of scabies is also an interesting option and currently under investigation (118). The clinical development of ivermectin for scabies and other parasites might have been rushed, and the dose of 0.2 mg/kg was not derived after high-level dose-ranging studies; it was based on a reasoned, but arbitrary, decision. An emerging hypothesis is that the parasite infection may need a higher dose of ivermectin to achieve a cure. This concept was first raised for head lice infestation, as the standard dose of oral ivermectin (0.2 mg per kg body weight) was found to be poorly effective. Further studies found that treatment with 0.4 mg/kg ivermectin (a double dose) was approximately 95–100% effective (119). Similar results were reported for other parasitic infections (118). Dose-ranging experimental studies in the pig model are ongoing to determine whether higher doses of ivermectin are more effective at controlling scabies infestation. In France, a French Ministry of Health-approved randomized controlled clinical trial is in process, comparing the efficacy of ivermectin given orally as the higher double dose of 0.4 mg/kg with the conventional treatment dose of 0.2 mg/kg, given 3 times 7 days apart (on D0, D7 and D14), supplemented in both arms with daily application of emollient therapy and topical 5% permethrin on D0 and D7 (GALECRUSTED, NCT02841215) (120).
Other novel treatments are also in development, using herbal compounds (121, 122) and even entomopathogenic fungus (123). The use of advanced molecular and biochemical technologies will help to design new therapeutic tools. These next-generation drugs are needed immediately and should be tailored to scabies mites.
The worldwide prevalence of scabies remains high, and currently available treatments may not be sufficiently effective to control the disease. During the past 20 years, at the beginning of the 21st century, a lot of important work has been completed concerning the management of scabies, mainly driven by IACS, a global advocacy body formed in 2012. We hope that the next 10 years will provide a significant improvement for patients infested with scabies, and that new drugs and diagnostics will enhance the therapeutic options for the benefit of patients and their families.
Conflicts of interest: CB received research support from Bioderma Laboratoire Dermatologique and Codexial Dermatologie. OC received drugs, donated free of charge, for research from Codexial Dermatologie and lecture fees from Zambon Laboratoire, Codexial Dermatologie and MSD France. CB and OC act as unpaid scientific advisors for Medicines Development for Global Health. The authors have no other potential conflicts of interest to declare.
- Chosidow O. Clinical practices. Scabies. N Engl J Med 2006; 354: 1718–1727.
View article Google Scholar - Currier RW, Walton SF, Currie BJ. Scabies in animals and humans: history, evolutionary perspectives, and modern clinical management. Ann N Y Acad Sci 2011; 1230: E50–60.
View article Google Scholar - Mellanby K. Transmission of scabies. Br Med J 1941; 2: 405–406.
View article Google Scholar - Mellanby K, Johnson CG, Bartley WC, Brown P. Experiments on the survival and behavior of the itch mite, Sarcoptes scabiel DeG. var hominis. Bull Entomol Res 1942; 33: 267–271.
View article Google Scholar - Arlian LG, Morgan MS. A review of Sarcoptes scabiei: past, present and future. Parasit Vectors 2017; 10: 297.
View article Google Scholar - Holt DC, Fischer K. Novel insights into an old disease: recent developments in scabies mite biology. Curr Opin Infect Dis 2013; 26: 110–115.
View article Google Scholar - Mounsey K, Ho M-F, Kelly A, Willis C, Pasay C, Kemp DJ, et al. A tractable experimental model for study of human and animal scabies. PLoS Negl Trop Dis 2010 Jul 20; 4: e756.
View article Google Scholar - Arlian LG, Runyan RA, Achar S, Estes SA. Survival and infectivity of Sarcoptes scabiei var. canis and var. hominis. J Am Acad Dermatol 1984; 11: 210–215.
View article Google Scholar - Laing R, Gillan V, Devaney E. Ivermectin – old drug, new tricks? Trends Parasitol 2017; 33: 463–472.
View article Google Scholar - Fernando DD, Marr EJ, Zakrzewski M, Reynolds SL, Burgess STG, Fischer K. Gene silencing by RNA interference in Sarcoptes scabiei: a molecular tool to identify novel therapeutic targets. Parasit Vectors 2017; 10: 289.
View article Google Scholar - Mofiz E, Holt DC, Seemann T, Currie BJ, Fischer K, Papenfuss AT. Genomic resources and draft assemblies of the human and porcine varieties of scabies mites, Sarcoptes scabiei var. hominis and var. suis. GigaScience 2016; 5: 23.
View article Google Scholar - Swe PM, Zakrzewski M, Waddell R, Sriprakash KS, Fischer K. High-throughput metagenome analysis of the Sarcoptes scabiei internal microbiota and in-situ identification of intestinal Streptomyces sp. Sci Rep 2019; 9: 11744.
View article Google Scholar - Bernigaud C, Samarawickrama GR, Jones MK, Gasser RB, Fischer K. The challenge of developing a single-dose treatment for scabies. Trends Parasitol 2019; 35: 931–943.
View article Google Scholar - Engelman D, Kiang K, Chosidow O, McCarthy J, Fuller C, Lammie P, et al. Toward the global control of human scabies: introducing the International Alliance for the Control of Scabies. PLoS Negl Trop Dis 2013; 7: e2167.
View article Google Scholar - NTD_STAG_report_2017.pdf. [cited 2019 Sep 4]. Available from: https://www.who.int/neglected_diseases/NTD_STAG_report_2017.pdf?ua=1.
View article Google Scholar - Karimkhani C, Colombara DV, Drucker AM, Norton SA, Hay R, Engelman D, et al. The global burden of scabies: a cross-sectional analysis from the Global Burden of Disease Study 2015. Lancet Infect Dis 2017; 17: 1247–1254.
View article Google Scholar - Romani L, Steer AC, Whitfeld MJ, Kaldor JM. Prevalence of scabies and impetigo worldwide: a systematic review. Lancet Infect Dis 2015; 15: 960–967.
View article Google Scholar - Cassell JA, Middleton J, Nalabanda A, Lanza S, Head MG, Bostock J, et al. Scabies outbreaks in ten care homes for elderly people: a prospective study of clinical features, epidemiology, and treatment outcomes. Lancet Infect Dis 2018; 18: 894–902.
View article Google Scholar - Bouvresse S, Chosidow O. Scabies in healthcare settings. Curr Opin Infect Dis 2010; 23: 111–118.
View article Google Scholar - Di Meco E, Di Napoli A, Amato LM, Fortino A, Costanzo G, Rossi A, et al. Infectious and dermatological diseases among arriving migrants on the Italian coasts. Eur J Public Health 2018; 28: 910–916.
View article Google Scholar - Lugovi?-Mihi? L. The increase in Croatia’s scabies incidence: how did refugees and traveling contribute? Travel Med Infect Dis 2019; 29: 74.
View article Google Scholar - Arnaud A, Chosidow O, Détrez M-A, Bitar D, Huber F, Foulet F, et al. Prevalences of scabies and pediculosis corporis among homeless people in the Paris region: results from two randomized cross-sectional surveys (HYTPEAC study). Br J Dermatol 2016; 174: 104–112.
View article Google Scholar - Lynar S, Currie BJ, Baird R. Scabies and mortality. Lancet Infect Dis 2017; 17: 1234.
View article Google Scholar - Engelman D, Cantey PT, Marks M, Solomon AW, Chang AY, Chosidow O, et al. The public health control of scabies: priorities for research and action. Lancet Lond Engl 2019; 394: 81–92.
View article Google Scholar - Brenaut E, Garlantezec R, Talour K, Misery L. Itch characteristics in five dermatoses: non-atopic eczema, atopic dermatitis, urticaria, psoriasis and scabies. Acta Derm Venereol 2013; 93: 573–574.
View article Google Scholar - Worth C, Heukelbach J, Fengler G, Walter B, Liesenfeld O, Feldmeier H. Impaired quality of life in adults and children with scabies from an impoverished community in Brazil. Int J Dermatol 2012; 51: 275–282.
View article Google Scholar - Worth C, Heukelbach J, Fengler G, Walter B, Liesenfeld O, Hengge U, et al. Acute morbidity associated with scabies and other ectoparasitoses rapidly improves after treatment with ivermectin. Pediatr Dermatol 2012; 29: 430–436.
View article Google Scholar - Jin-Gang A, Sheng-Xiang X, Sheng-Bin X, Jun-Min W, Song-Mei G, Ying-Ying D, et al. Quality of life of patients with scabies. J Eur Acad Dermatol Venereol 2010; 24: 1187–1191.
View article Google Scholar - Chosidow O, Fuller LC. Scratching the itch: is scabies a truly neglected disease? Lancet Infect Dis 2017; 17: 1220–1221.
View article Google Scholar - Lawrence G, Leafasia J, Sheridan J, Hills S, Wate J, Wate C, et al. Control of scabies, skin sores and haematuria in children in the Solomon Islands: another role for ivermectin. Bull World Health Organ 2005; 83: 34–42.
View article Google Scholar - Thornley S, Marshall R, Jarrett P, Sundborn G, Reynolds E, Schofield G. Scabies is strongly associated with acute rheumatic fever in a cohort study of Auckland children. J Paediatr Child Health 2018; 54: 625–632.
View article Google Scholar - Mason DS, Marks M, Sokana O, Solomon AW, Mabey DC, Romani L, et al. The prevalence of scabies and impetigo in the Solomon Islands: a population-based survey. PLoS Negl Trop Dis 2016; 10: e0004803.
View article Google Scholar - Romani L, Koroivueta J, Steer AC, Kama M, Kaldor JM, Wand H, et al. Scabies and impetigo prevalence and risk factors in Fiji: a national survey. PLoS Negl Trop Dis 2015; 9: e0003452.
View article Google Scholar - Svartman M, Finklea JF, Earle DP, Potter EV, Poon-King T. Epidemic scabies and acute glomerulonephritis in Trinidad. Lancet Lond Engl 1972; 1: 249–251.
View article Google Scholar - Turnbull PR. Aetiology of acute glomerulonephritis. Br Med J 1973; 2: 666.
View article Google Scholar - Romani L, Whitfeld MJ, Koroivueta J, Kama M, Wand H, Tikoduadua L, et al. Mass drug administration for scabies control in a population with endemic disease. N Engl J Med 2015; 373: 2305–2313.
View article Google Scholar - Marks M, Toloka H, Baker C, Kositz C, Asugeni J, Puiahi E, et al. Randomized trial of community treatment with azithromycin and ivermectin mass drug administration for control of scabies and impetigo. Clin Infect Dis 2019; 68: 927–933.
View article Google Scholar - Romani L, Marks M, Sokana O, Nasi T, Kamoriki B, Cordell B, et al. Efficacy of mass drug administration with ivermectin for control of scabies and impetigo, with coadministration of azithromycin: a single-arm community intervention trial. Lancet Infect Dis 2019; 19: 510–518.
View article Google Scholar - Mika A, Reynolds SL, Mohlin FC, Willis C, Swe PM, Pickering DA, et al. Novel scabies mite serpins inhibit the three pathways of the human complement system. PloS One 2012; 7: e40489.
View article Google Scholar - Reynolds SL, Pike RN, Mika A, Blom AM, Hofmann A, Wijeyewickrema LC, et al. Scabies mite inactive serine proteases are potent inhibitors of the human complement lectin pathway. PLoS Negl Trop Dis 2014; 8: e2872.
View article Google Scholar - Swe PM, Fischer K. A scabies mite serpin interferes with complement-mediated neutrophil functions and promotes staphylococcal growth. PLoS Negl Trop Dis 2014; 8: e2928.
View article Google Scholar - Swe PM, Reynolds SL, Fischer K. Parasitic scabies mites and associated bacteria joining forces against host complement defence. Parasite Immunol 2014; 36: 585–593.
View article Google Scholar - Swe PM, Christian LD, Lu HC, Sriprakash KS, Fischer K. Complement inhibition by Sarcoptes scabiei protects Streptococcus pyogenes – an in vitro study to unravel the molecular mechanisms behind the poorly understood predilection of S. pyogenes to infect mite-induced skin lesions. PLoS Negl Trop Dis 2017; 11: e0005437.
View article Google Scholar - Jannic A, Bernigaud C, Brenaut E, Chosidow O. Scabies itch. Dermatol Clin 2018; 36: 301–308.
View article Google Scholar - Boralevi F, Diallo A, Miquel J, Guerin-Moreau M, Bessis D, Chiavérini C, et al. Clinical phenotype of scabies by age. Pediatrics 2014; 133: e910–916.
View article Google Scholar - Elosua-González M, García-Zamora E. Crusted Scabies. N Engl J Med 2017; 377: 476.
View article Google Scholar - Roberts LJ, Huffam SE, Walton SF, Currie BJ. Crusted scabies: clinical and immunological findings in seventy-eight patients and a review of the literature. J Infect 2005; 50: 375–381.
View article Google Scholar - del Giudice P, Sainte Marie D, Gérard Y, Couppié P, Pradinaud R. Is crusted (Norwegian) scabies a marker of adult T cell leukemia/lymphoma in human T lymphotropic virus type I-seropositive patients? J Infect Dis 1997; 176: 1090–1092.
View article Google Scholar - Mahé A, Faye O, N’Diaye HT, Ly F, Konaré H, Kéita S, et al. Definition of an algorithm for the management of common skin diseases at primary health care level in sub-Saharan Africa. Trans R Soc Trop Med Hyg 2005; 99: 39–47.
View article Google Scholar - Steer AC, Tikoduadua LV, Manalac EM, Colquhoun S, Carapetis JR, Maclennan C. Validation of an integrated management of childhood illness algorithm for managing common skin conditions in Fiji. Bull World Health Organ 2009; 87: 173–179.
View article Google Scholar - Engelman D, Fuller LC, Steer AC, International Alliance for the Control of Scabies Delphi panel. Consensus criteria for the diagnosis of scabies: a Delphi study of international experts. PLoS Negl Trop Dis 2018; 12: e0006549.
View article Google Scholar - Osti MH, Sokana O, Gorae C, Whitfeld MJ, Steer AC, Engelman D. The diagnosis of scabies by non-expert examiners: a study of diagnostic accuracy. PLoS Negl Trop Dis 2019; 13: e0007635.
View article Google Scholar - Micali G, Lacarrubba F, Verzì AE, Chosidow O, Schwartz RA. Scabies: advances in noninvasive diagnosis. PLoS Negl Trop Dis 2016; 10: e0004691.
View article Google Scholar - Micali G, Lacarrubba F, Massimino D, Schwartz RA. Dermatoscopy: alternative uses in daily clinical practice. J Am Acad Dermatol 2011; 64: 1135–1146.
View article Google Scholar - Dupuy A, Dehen L, Bourrat E, Lacroix C, Benderdouche M, Dubertret L, et al. Accuracy of standard dermoscopy for diagnosing scabies. J Am Acad Dermatol 2007; 56: 53–62.
View article Google Scholar - Micali G, Lacarrubba F, Verzì AE, Nasca MR. Low-cost equipment for diagnosis and management of endemic scabies outbreaks in underserved populations. Clin Infect Dis 2015; 60: 327–329.
View article Google Scholar - Lacarrubba F, Verzì AE, Micali G. Detailed analysis of in vivo reflectance confocal microscopy for Sarcoptes scabiei hominis. Am J Med Sci 2015; 350: 414.
View article Google Scholar - Wong SSY, Poon RWS, Chau S, Wong SCY, To KKW, Cheng VCC, et al. Development of conventional and real-time quantitative PCR assays for diagnosis and monitoring of scabies. J Clin Microbiol 2015; 53: 2095–2102.
View article Google Scholar - Hahm JE, Kim CW, Kim SS. The efficacy of a nested polymerase chain reaction in detecting the cytochrome c oxidase subunit 1 gene of Sarcoptes scabiei var. hominis for diagnosing scabies. Br J Dermatol 2018; 179: 889–895.
View article Google Scholar - Angelone-Alasaad S, Molinar Min A, Pasquetti M, Alagaili AN, D’Amelio S, Berrilli F, et al. Universal conventional and real-time PCR diagnosis tools for Sarcoptes scabiei. Parasit Vectors 2015; 8: 587.
View article Google Scholar - Fukuyama S, Nishimura T, Yotsumoto H, Gushi A, Tsuji M, Kanekura T, et al. Diagnostic usefulness of a nested polymerase chain reaction assay for detecting Sarcoptes scabiei DNA in skin scrapings from clinically suspected scabies. Br J Dermatol 2010; 163: 892–894.
View article Google Scholar - Chosidow O, Sbidian E. La gale: une reconnaissance méritée! Ann Dermatol Venereol 2012; 139: 425–427.
View article Google Scholar - Walter B, Heukelbach J, Fengler G, Worth C, Hengge U, Feldmeier H. Comparison of dermoscopy, skin scraping, and the adhesive tape test for the diagnosis of scabies in a resource-poor setting. Arch Dermatol 2011; 147: 468–473.
View article Google Scholar - Woodley D, Saurat JH. The Burrow Ink Test and the scabies mite. J Am Acad Dermatol 1981; 4: 715–722.
View article Google Scholar - Cinotti E, Labeille B, Cambazard F, Biron AC, Chol C, Leclerq A, et al. Videodermoscopy compared to reflectance confocal microscopy for the diagnosis of scabies. J Eur Acad Dermatol Venereol 2016; 30: 1573–1577.
View article Google Scholar - Delaunay P, Hérissé AL, Hasseine L, Chiaverini C, Tran A, Mary C, et al. Scabies polymerase chain reaction with standardized dry swab sampling: an easy tool for cluster diagnosis of human scabies. Br J Dermatol 2020; 182: 197–201.
View article Google Scholar - Currie BJ, McCarthy JS. Permethrin and ivermectin for scabies. N Engl J Med 2010; 362: 717–725.
View article Google Scholar - Usha V, Gopalakrishnan Nair TV. A comparative study of oral ivermectin and topical permethrin cream in the treatment of scabies. J Am Acad Dermatol 2000; 42: 236–240.
View article Google Scholar - Bachewar NP, Thawani VR, Mali SN, Gharpure KJ, Shingade VP, Dakhale GN. Comparison of safety, efficacy, and cost effectiveness of benzyl benzoate, permethrin, and ivermectin in patients of scabies. Indian J Pharmacol 2009; 41: 9–14.
View article Google Scholar - Sharma R, Singal A. Topical permethrin and oral ivermectin in the management of scabies: a prospective, randomized, double blind, controlled study. Indian J Dermatol Venereol Leprol 2011; 77: 581–586.
View article Google Scholar - Bernigaud C, Fernando DD, Lu H, Taylor S, Hartel G, Guillot J, et al. In vitro ovicidal activity of current and under-development scabicides – which treatments kill scabies eggs? Br J Dermatol 2020; 182: 511–513.
View article Google Scholar - Bernigaud C, Fang F, Fischer K, Lespine A, Aho LS, Dreau D, et al. Preclinical study of single-dose moxidectin, a new oral treatment for scabies: efficacy, safety, and pharmacokinetics compared to two-dose ivermectin in a porcine model. PLoS Negl Trop Dis 2016; 10: e0005030.
View article Google Scholar - Bernigaud C, Fang F, Fischer K, Lespine A, Aho LS, Mullins AJ, et al. Efficacy and pharmacokinetics evaluation of a single oral dose of afoxolaner against Sarcoptes scabiei in the porcine scabies model for human infestation. Antimicrob Agents Chemother 2018; 62. pii: e02334.
View article Google Scholar - Guzzo CA, Furtek CI, Porras AG, Chen C, Tipping R, Clineschmidt CM, et al. Safety, tolerability, and pharmacokinetics of escalating high doses of ivermectin in healthy adult subjects. J Clin Pharmacol 2002; 42: 1122–1133.
View article Google Scholar - Miyajima A, Hirota T, Sugioka A, Fukuzawa M, Sekine M, Yamamoto Y, et al. Effect of high-fat meal intake on the pharmacokinetic profile of ivermectin in Japanese patients with scabies. J Dermatol 2016; 43: 1030–1036.
View article Google Scholar - Strong M, Johnstone P. Interventions for treating scabies. Cochrane Database Syst Rev 2007; 3: CD000320.
View article Google Scholar - Rosumeck S, Nast A, Dressler C. Ivermectin and permethrin for treating scabies. Cochrane Database Syst Rev 2018; 4: CD012994.
View article Google Scholar - F. Boralevi, University Hospital, Bordeaux. Oral ivermectin versus topical permethrin to treat scabies in children – ClinicalTrials.gov. [cited 2019 Sep 01]. Available from: https://clinicaltrials.gov/ct2/show/NCT02407782.
View article Google Scholar - WHO-MVP-EMP-IAU-2019.06-eng.pdf. [cited 2019 Sep 01]. Available from: https://apps.who.int/iris/bitstream/handle/10665/325771/WHO-MVP-EMP-IAU-2019.06-eng.pdf?ua=1.
View article Google Scholar - Chosidow O. Scabies and pediculosis. Lancet Lond Engl 2000; 355: 819–826.
View article Google Scholar - Pasay C, Walton S, Fischer K, Holt D, McCarthy J. PCR-based assay to survey for knockdown resistance to pyrethroid acaricides in human scabies mites (Sarcoptes scabiei var hominis). Am J Trop Med Hyg 2006; 74: 649–657.
View article Google Scholar - Andriantsoanirina V, Izri A, Botterel F, Foulet F, Chosidow O, Durand R. Molecular survey of knockdown resistance to pyrethroids in human scabies mites. Clin Microbiol Infect 2014; 20: O139–141.
View article Google Scholar - Currie BJ, Harumal P, McKinnon M, Walton SF. First documentation of in vivo and in vitro ivermectin resistance in Sarcoptes scabiei. Clin Infect Dis Off Publ Infect Dis Soc Am 2004; 39: e8–12.
View article Google Scholar - Mounsey KE, Holt DC, McCarthy JS, Currie BJ, Walton SF. Longitudinal evidence of increasing in vitro tolerance of scabies mites to ivermectin in scabies-endemic communities. Arch Dermatol 2009; 145: 840–841.
View article Google Scholar - De Sainte Marie B, Mallet S, Gaudy-Marqueste C, Baumstarck K, Bentaleb N, Loundou A, et al. Gales en échec de traitement: étude observationnelle. Ann Dermatol Venereol 2016; 143: 9–15.
View article Google Scholar - Aussy A, Houivet E, Hébert V, Colas-Cailleux H, Laaengh N, Richard C, et al. Risk factors for treatment failure in scabies: a cohort study. Br J Dermatol 2019; 180: 888–893.
View article Google Scholar - Arlian LG, Estes SA, Vyszenski-Moher DL. Prevalence of Sarcoptes scabiei in the homes and nursing homes of scabietic patients. J Am Acad Dermatol 1988; 19: 806–811.
View article Google Scholar - Williams C, Fuller LC. Why does scabies treatment fail? Br J Dermatol 2019; 180: 710–711.
View article Google Scholar - Bernigaud C, Fernando DD, Lu HC, Chosidow O, Fischer K. Revisiting guidelines for the environmental control of scabies 2018 ASP Annual Conference of the Australian Society of Parasitology. St Kilda, Melbourne, Australia; September 2018.
View article Google Scholar - Bowen AC, Tong SYC, Andrews RM, O’Meara IM, McDonald MI, Chatfield MD, et al. Short-course oral co-trimoxazole versus intramuscular benzathine benzylpenicillin for impetigo in a highly endemic region: an open-label, randomised, controlled, non-inferiority trial. Lancet Lond Engl 2014; 384: 2132–2140.
View article Google Scholar - Sanders KM, Nattkemper LA, Rosen JD, Andersen HH, Hsiang J, Romanelli P, et al. Non-histaminergic itch mediators elevated in the skin of a porcine model of scabies and of human scabies patients. J Invest Dermatol 2019; 139: 971–973.
View article Google Scholar - Levy M, Martin L, Bursztejn A-C, Chiaverini C, Miquel J, Mahé E, et al. Ivermectin safety in infants and children under 15 kg treated for scabies: a multicentric observational study. Br J Dermatol 2019 Jul 25. [Epub ahead of print].
View article Google Scholar - Scabicides et grossesse [cited 2019 Sep 01]. Available from: https://lecrat.fr/spip.php?page=article&id_article=585.
View article Google Scholar - Pacqué M, Muñoz B, Poetschke G, Foose J, Greene BM, Taylor HR. Pregnancy outcome after inadvertent ivermectin treatment during community-based distribution. Lancet Lond Engl 1990; 336: 1486–1489.
View article Google Scholar - Kamgno J, Pion SD, Chesnais CB, Bakalar MH, D’Ambrosio MV, Mackenzie CD, et al. A test-and-not-treat strategy for onchocerciasis in Loa loa-endemic areas. N Engl J Med 2017; 377: 2044–2052.
View article Google Scholar - Engelman D, Steer AC. Control strategies for scabies. Trop Med Infect Dis 2018; 3. pii: E98.
View article Google Scholar - Taplin D, Arrue C, Walker JG, Roth WI, Rivera A. Eradication of scabies with a single treatment schedule. J Am Acad Dermatol 1983; 9: 546–550.
View article Google Scholar - Marks M, Romani L, Sokana O, Neko L, Harrington R, Nasi T, et al. Prevalence of scabies and impetigo three years after mass drug administration with ivermectin and azithromycin. Clin Infect Dis Off Publ Infect Dis Soc Am 2019 May 25. pii: ciz444.
View article Google Scholar - Romani L, Whitfeld MJ, Koroivueta J, Kama M, Wand H, Tikoduadua L, et al. Mass drug administration for scabies – 2 years of follow-up. N Engl J Med 2019; 381: 186–187.
View article Google Scholar - Martin D, Wiegand R, Goodhew B, Lammie P, Mkocha H, Kasubi M. Impact of ivermectin mass drug administration for lymphatic filariasis on scabies in eight villages in Kongwa District, Tanzania. Am J Trop Med Hyg 2018; 99: 937–939.
View article Google Scholar - Engelman D, Fuller LC, Solomon AW, McCarthy JS, Hay RJ, Lammie PJ, et al. Opportunities for integrated control of neglected tropical diseases that affect the skin. Trends Parasitol 2016; 32: 843–854.
View article Google Scholar - Mohammed KA, Deb RM, Stanton MC, Molyneux DH. Soil transmitted helminths and scabies in Zanzibar, Tanzania following mass drug administration for lymphatic filariasis – a rapid assessment methodology to assess impact. Parasit Vectors 2012; 5: 299.
View article Google Scholar - Kearns TM, Speare R, Cheng AC, McCarthy J, Carapetis JR, Holt DC, et al. Impact of an ivermectin mass drug administration on scabies prevalence in a remote Australian Aboriginal community. PLoS Negl Trop Dis 2015; 9: e0004151.
View article Google Scholar - Romani L, Marks M, Sokana O, Nasi T, Kamoriki B, Wand H, et al. Feasibility and safety of mass drug coadministration with azithromycin and ivermectin for the control of neglected tropical diseases: a single-arm intervention trial. Lancet Glob Health 2018; 6: e1132–1138.
View article Google Scholar - Coscione S, Esau T, Kekeubata E, Diau J, Asugeni R, MacLaren D, et al. Impact of ivermectin administered for scabies treatment on the prevalence of head lice in Atoifi, Solomon Islands. PLoS Negl Trop Dis 2018; 12: e0006825.
View article Google Scholar - Kositz C, Talina J, Diau J, Asugeni R, Whitehorn C, Mabey D, et al. Incidental mosquitocidal effect of an ivermectin mass drug administration on Anopheles farauti conducted for scabies control in the Solomon Islands. Trans R Soc Trop Med Hyg 2017; 111: 97–101.
View article Google Scholar - Kaburi BB, Ameme DK, Adu-Asumah G, Dadzie D, Tender EK, Addeh SV, et al. Outbreak of scabies among preschool children, Accra, Ghana, 2017. BMC Public Health 2019; 19: 746.
View article Google Scholar - Leppard B, Naburi AE. The use of ivermectin in controlling an outbreak of scabies in a prison. Br J Dermatol 2000; 143: 520–523.
View article Google Scholar - Beeres DT, Ravensbergen SJ, Heidema A, Cornish D, Vonk M, Wijnholds LD, et al. Efficacy of ivermectin mass-drug administration to control scabies in asylum seekers in the Netherlands: a retrospective cohort study between January 2014 – March 2016. PLoS Negl Trop Dis 2018; 12: e0006401.
View article Google Scholar - Taplin D, Porcelain SL, Meinking TL, Athey RL, Chen JA, Castillero PM, et al. Community control of scabies: a model based on use of permethrin cream. Lancet Lond Engl 1991; 337: 1016–1018.
View article Google Scholar - Carapetis JR, Connors C, Yarmirr D, Krause V, Currie BJ. Success of a scabies control program in an Australian aboriginal community. Pediatr Infect Dis J 1997; 16: 494–499.
View article Google Scholar - Wong LC, Amega B, Connors C, Barker R, Dulla ME, Ninnal A, et al. Outcome of an interventional program for scabies in an Indigenous community. Med J Aust 2001; 175: 367–370.
View article Google Scholar - Wong L-C, Amega B, Barker R, Connors C, Dulla ME, Ninnal A, et al. Factors supporting sustainability of a community-based scabies control program. Australas J Dermatol 2002; 43: 274–277.
View article Google Scholar - Marks M, Taotao-Wini B, Satorara L, Engelman D, Nasi T, Mabey DC, et al. Long term control of scabies fifteen years after an intensive treatment programme. PLoS Negl Trop Dis 2015; 9: e0004246.
View article Google Scholar - Andrews RM, Kearns T, Connors C, Parker C, Carville K, Currie BJ, et al. A regional initiative to reduce skin infections amongst aboriginal children living in remote communities of the Northern Territory, Australia. PLoS Negl Trop Dis 2009; 3: e554.
View article Google Scholar - La Vincente S, Kearns T, Connors C, Cameron S, Carapetis J, Andrews R. Community management of endemic scabies in remote aboriginal communities of northern Australia: low treatment uptake and high ongoing acquisition. PLoS Negl Trop Dis 2009; 3: e444.
View article Google Scholar - Dose-finding study of moxidectin for treatment of scabies – ClinicalTrials.gov. [cited 2019 Sep 01]. Available from: https://clinicaltrials.gov/ct2/show/NCT03905265.
View article Google Scholar - Chosidow O, Bernigaud C, Do-Pham G. High-dose ivermectin in malaria and other parasitic diseases: a new step in the development of a neglected drug. Parasite Paris Fr 2018; 25: 33.
View article Google Scholar - Chosidow O, Giraudeau B, Cottrell J, Izri A, Hofmann R, Mann SG, et al. Oral ivermectin versus malathion lotion for difficult-to-treat head lice. N Engl J Med 2010; 362: 896–905.
View article Google Scholar - Efficacy study between two different dosages of an antiparasitic in patients with crusted scabies – ClinicalTrials.gov. [cited 2019 Sep 01]. Available from: https://clinicaltrials.gov/ct2/show/NCT02841215.
View article Google Scholar - Fang F, Candy K, Melloul E, Bernigaud C, Chai L, Darmon C, et al. In vitro activity of ten essential oils against Sarcoptes scabiei. Parasit Vectors 2016; 9: 594.
View article Google Scholar - Thomas J, Carson CF, Peterson GM, Walton SF, Hammer KA, Naunton M, et al. Therapeutic potential of tea tree oil for scabies. Am J Trop Med Hyg 2016; 94: 258–266.
View article Google Scholar - Al Khoury C, Guillot J, Nemer N. Lethal activity of beauvericin, a Beauveria bassiana mycotoxin, against the two-spotted spider mites, Tetranychus urticae Koch. J Appl Entomol 2019; 143: 974–983.
View article Google Scholar