There are concerns about the psychological effects of the COVID-19 pandemic and quarantines on patients with psoriasis, given this population’s increased pre-pandemic depression risk and putatively high suicidality (1, 2). Furthermore, many tertiary patients are considered at increased risk of COVID-19 due to immunomodulatory treatments, and were advised to follow stringent social distancing. A previous survey reported an early pandemic depression rate of 27.5% in psoriasis (2); however, the pre-pandemic rate in this sample was unknown; depression prevalence in psoriasis generally ranges from 22% to 34% (3). To our knowledge, there are no longitudinal data on mental health in psoriasis before and during the pandemic. The aim of this study was to investigate whether patients’ depressive symptom levels and dermatology-related quality of life changed compared with pre-pandemic rates and during different pandemic waves. The study also captured 12-month suicidality among these patients during COVID-19.
MATERIALS AND METHODS and ReSults
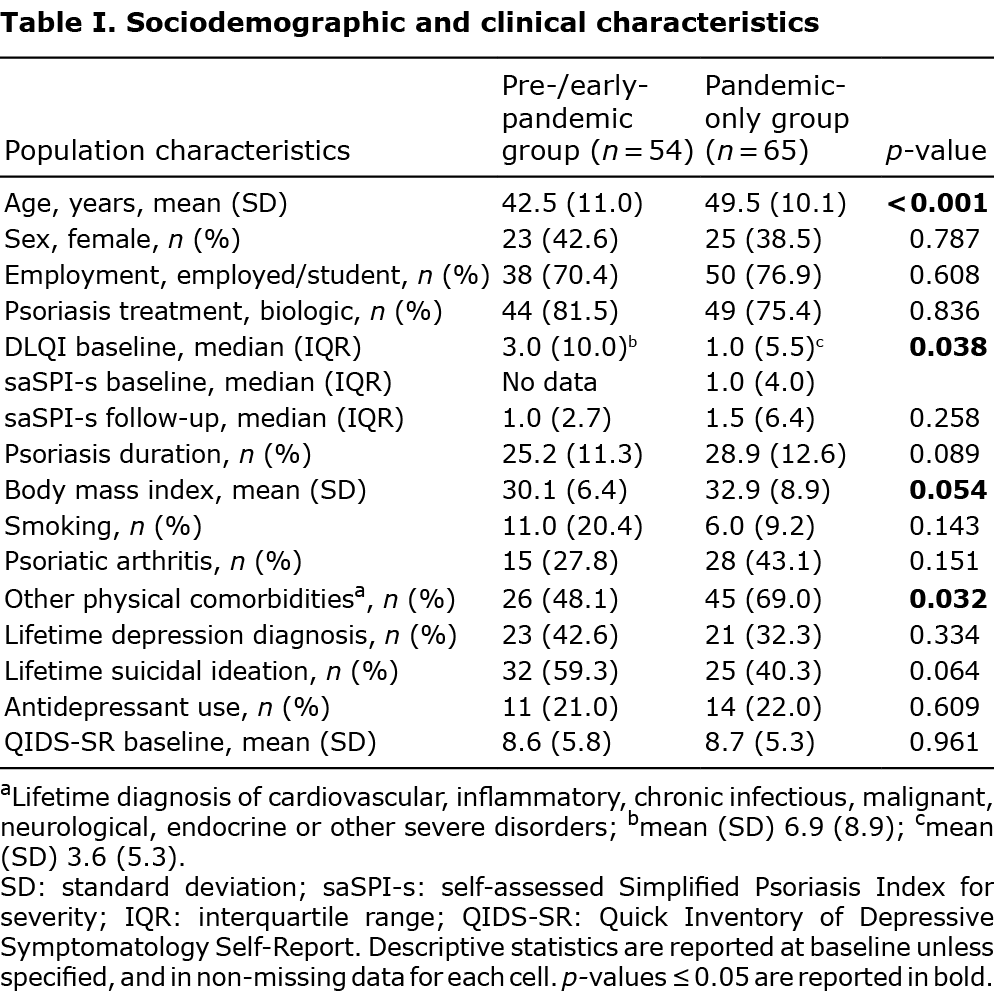
Patients with dermatologist-confirmed psoriasis attending tertiary psoriasis and psoriatic arthritis clinics completed questionnaires at baseline and at a 1-year follow-up. Two cohorts were developed: a group recruited before COVID-19 was used to assess differences before vs during the pandemic (“pre-/early-pandemic” group); and a group recruited at the end of the first pandemic wave was used to assess change during the course of the pandemic (“pandemic-only” group) (Fig. S1). Baseline data were collected as part of another study, whose methods have been described previously (1). The Quick Inventory of Depressive Symptomatology-Self-Report (QIDS-SR) (4) and the Dermatology Life Quality Index (DLQI) (5) were used at both time-points. Lifetime suicidality (ideation and behaviour) at baseline and 12-month suicidality at follow-up were assessed with the Sheehan-Suicidality Tracking Scale (S-STS) (6). The pandemic-only group completed the Impact of Events Scale-Revised (IES-R; pandemic-adapted) (7).
Baseline vs follow-up questionnaire scores within each group were compared, using paired t-tests for total QIDS-SR; and Wilcoxon matched-pairs signed-rank tests for DLQI and QIDS-SR item scores with range 0–3. To assess baseline vs follow-up between-group differences in total scores, linear mixed effects models were fitted (predictor: time-group interaction; other fixed effects: age, sex and physical comorbidity). In a secondary analysis, cumulative link models were used to assess between-group differences for individual QIDS-SR items where Wilcoxon tests were significant after multiple comparisons correction, including covariates as for total scores. In the pandemic-only group, Spearman’s correlation between mood change and psoriasis severity change was measured. Between-group difference in 12-month suicidality prevalence was investigated with age- and lifetime suicidality-adjusted logistic regression. No covariate data were missing; outcome missingness is reported in Table SI. p-values were corrected separately for within- and between-group comparisons, controlling for the false-discovery rate (FDR).
Results
Out of 119 respondents, 54 were enrolled pre-pandemic (September 2019–February 2020) and were followed-up during the second pandemic wave (October 2020–February 2021); 65 patients enrolled post-first wave (May–September 2020) and were followed-up pre-third wave, after lockdowns in England were eased (August–October 2021). Patient characteristics are shown in Table I. Baseline QIDS-SR was similar in the groups. Psoriasis severity did not differ between baseline and follow-up in the pandemic-only group, which had available data (p = 0.17).
No significant difference was found in follow-up vs baseline total QIDS-SR scores in either group (mean ± standard deviation (SD) pre-/early-pandemic group: 8.9 ± 5.2 vs 7.9 ± 5.6, p = 0.32; pandemic-only group: 9.1 ± 5.5 vs 8.5 ± 5.2, p = 0.42). There was no between-group difference in QIDS-SR longitudinal trends (β (95% confidence intervals; CI)) –0.31 (–1.75, 1.14), Table SI). When individual depressive symptoms were investigated (Table SII), concentration was poorer during than before COVID-19 (p = 0.054). Mood had worsened significantly by August–October 2021 in the pandemic-only group (item mean ± SD 0.95 ± 0.94 vs 0.68 ± 0.83; p = 0.03); the between-group difference in mood trajectories was significant (time*group interaction: cumulative odds ratio (95% CI) 1.53 (1.09, 2.14); p = 0.045). Mood deterioration did not correlate with the numerical change in psoriasis severity in the pandemic-only group (r=0.10, p > 0.9).
The DLQI trajectories showed an opposing trend between groups (p = 0.098; puncorrected=0.059), with numerical improvement of DLQI during compared with pre-pandemic (median ± interquartile range (IQR)) 1.0 ± 6.0 vs 2.0 ± 7.0, p = 0.36), but numerical worsening in the pandemic-only group (median ± IQR 2.0 ± 5.0 follow-up vs 1.0 ± 5.5 baseline, p = 0.36). The 12-month suicidality rate was higher during COVID-19 compared with the 12-month interval including pre-pandemic months, even after controlling for lifetime suicidality and FDR (27.9% in the pandemic-only group, 3 of whom had suicidal behaviour vs 15.2% in the pre-/early-pandemic group, none of whom had suicidal behaviour; odds ratio (95% CI) 5.72 (1.48, 27.40), p = 0.045). In 23.5% of patients with 12-month suicidality, this was a new occurrence. By August 2021, 20% of the pandemic-only group had probable post-traumatic stress disorder (IES-R ≥33).
DISCUSSION
To our knowledge, this is the first study examining depression and suicidality trajectories in patients with psoriasis during COVID-19. Whilst patients’ overall depressive symptom levels did not differ before vs during COVID-19 or between the examined pandemic waves, mood and 12-month suicidality deteriorated as the pandemic progressed. Our findings for the pre-/early-pandemic group align with recovery trends of the English general population affective burden by the end of the first wave (8), and the early psychological stability in other patient groups (9). Less is known about later pandemic stages. In a UK survey, whilst mental health remained stable in the general population by October 2020, it tended to worsen among the physically vulnerable (10). In the current study, depression levels during October 2020–February 2021, although numerically higher, did not significantly differ from pre-pandemic levels, and skin-related quality of life numerically improved. As most patients had chronic moderate-to-severe psoriasis (mean baseline disease duration 27.2 years, 78.1% on biologics), pre-developed coping strategies in response to long-standing illness (9) may have contributed to resilience. Notably, DLQI values were overall relatively low (1), aligning with similar tertiary cohorts before COVID-19 in England (11) and Spain (12). The groups’ DLQI trends may further reflect a reduction in distressing social stimuli during lockdown or a lower attribution of patients’ distress and impairment to psoriasis in the light of acute, pandemic-driven concerns and lifestyle restrictions (13).
The subsequent mood deterioration may suggest symptom-specific pandemic effects. Identifying worsening mental health dimensions is important for early intervention. A study in Wuhan, China, identified depressed mood and somatic symptoms as principal pandemic depression drivers (14). Neither psoriasis severity nor somatic depression dimensions worsened significantly in the current pandemic-only group. Also given the reversal trend of the DLQI trajectory later in the pandemic, stress-driven mood changes may have subsequently affected the perceived skin-related life quality during COVID-19 rather than the other way round. Twelve-month suicidality was higher in the pandemic-only group, despite lower baseline lifetime suicidality and depression compared with the pre-/early-pandemic group. In the general population, although suicides did not increase early into the pandemic, reports of other suicidal phenomena are inconclusive, with some studies showing an increase in suicidal thoughts (15).
These data provide important insights about psychological pandemic effects in psoriasis. The main strengths of this study are the longitudinal design and validated questionnaires. Study limitations include the possibility of false-negative results for small effects owing to the sample size, potentially limited generalizability in mild psoriasis and the lack of controls with other skin disease. It is currently critical for clinicians to monitor mood in psoriasis patients, offer timely treatments and remain vigilant about suicidality in the post-pandemic era. Increasing suicidality rates over time are concerning. Further research into long-term psychological outcomes and suicidality incidence is required.
ACKNOWLEDGEMENTS
GL, CEK, RBW and HC are supported by the National Institute for Health and Care Research (NIHR) Manchester Biomedical Research Centre Funding Scheme. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Conflicts of interest: CEK has received honoraria, consultant and/or research funding from Janssen, Eli Lilly, LEO, Novartis, Abbvie, UCB, Almirall, Pfizer, and L’Oréal. HC has received personal compensation for activities with Novartis, UCB, Lilly, Biogen, Orphazyme as a speaker, advisory board member or consultancy, grants via The University of Manchester from Novartis, UCB and MedImmune, and has received travel support from Abbvie and Janssen. RBW has received research grants from AbbVie, Almirall, Amgen, Celgene, Janssen, Lilly, Leo, Medac, Novartis, Pfizer, and UCB and consulting fees from AbbVie, Almirall, Amgen, Arena, Astellas, Avillion, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, DiCE, GSK, Janssen, Lilly, Leo, Medac, Novartis, Pfizer, Sanofi, Sun Pharma, UCB, and UNION. GL has received speaker honoraria from Janssen, Lilly, Leo, and Novartis. PST has no conflicts of interest.
REFERENCES
- Lada G, Chinoy H, Heal C, Warren RB, Talbot PS, Kleyn CE. Depression and suicidality in patients with psoriasis and the role of psoriatic arthritis; a cross-sectional study in a tertiary setting. J Acad Consult Liaison Psychiatry 2022; 63: 372–383.
- Mahil SK, Yates M, Yiu Z, Langan SM, Tsakok T, Dand N, et al. Describing the burden of the COVID-19 pandemic in people with psoriasis: findings from a global cross-sectional study. J Eur Acad Dermatol Venereol 2021; 35: e636–e640.
- Dowlatshahi EA, Wakkee M, Arends LR, Nijsten T. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol 2014; 134: 1542–1551.
- Rush AJ, Trivedi MH, Ibrahim HM, Carmody TJ, Arnow B, Klein DN, et al. The 16-item quick inventory of depressive symptomatology (QIDS), clinician rating (QIDS-C), and self-report (QIDS-SR): a psychometric evaluation in patients with chronic major depression. Biol Psychiatry 2003; 54: 573–583.
- Finlay AY, Khan G. Dermatology Life Quality Index (DLQI) – a simple practical measure for routine clinical use. Clin Exp Dermatol 1994; 19: 210–216.
- Sheehan DV, Giddens JM, Sheehan IS. Status Update on the Sheehan-Suicidality Tracking Scale (S-STS) 2014. Innov Clin Neurosci 2014; 11: 93.
- Weiss DS. The impact of event scale: revised. Cross-cultural assessment of psychological trauma and PTSD: Springer; 2007: p. 219–238.
- Fancourt D, Steptoe A, Bu F. Trajectories of anxiety and depressive symptoms during enforced isolation due to COVID-19 in England: a longitudinal observational study. Lancet Psychiatry 2021; 8: 141–149.
- Sloan M, Gordon C, Lever E, Harwood R, Bosley MA, Pilling M, et al. COVID-19 and shielding: experiences of UK patients with lupus and related diseases. Rheumatol Adv Pract 2021; 5: rkab003.
- Pierce M, McManus S, Hope H, Hotopf M, Ford T, Hatch SL, et al. Mental health responses to the COVID-19 pandemic: a latent class trajectory analysis using longitudinal UK data. Lancet Psychiatry 2021; 8: 610–619.
- Lamb R, Matcham F, Turner M, Rayner L, Simpson A, Hotopf M, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol 2017; 176: 1028–1034.
- Tribó MJ, Turroja M, Castaño-Vinyals G, Bulbena A, Ros Cucurull E, García-Martínez P. Patients with moderate to severe psoriasis associate with higher risk of depression and anxiety symptoms: results of a multivariate study of 300 Spanish individuals with psoriasis. Acta Derm Venereol 2019; 99: 417–422.
- Lada G, Chinoy H, Talbot PS, Warren RB, Kleyn CE. The effect of the Covid-19 pandemic on illness perceptions of psoriasis and the role of depression: findings from a cross-sectional study. Skin Health Dis 2022; 2: e145.
- Zhao N, Li W, Zhang S-F, Yang BX, Sha S, Cheung T, et al. Network analysis of depressive symptoms among residents of Wuhan in the later stage of the COVID-19 pandemic. Front Psychiatr 2021; 12: 735973.
- John A, Eyles E, Webb RT, Okolie C, Schmidt L, Arensman E, et al. The impact of the COVID-19 pandemic on self-harm and suicidal behaviour: update of living systematic review. F1000Research 2021; 9: 1097.