ORIGINAL ARTICLE
Chronic Use of Hydrochlorothiazide and Risk of Skin Cancer in Caucasian Adults: A PharmLines Initiative Inception Cohort Study
Marjolijn S. HAISMA1#, Nathalie GREVEN1#, Mathanhy LOGENDRAN1, Jens BOS2, Bert V.D. VEGT3, Barbara HORVÁTH1, Stijn DE VOS2, Geertruida H. DE BOCK4, Eelko HAK2 and Emőke RÁCZ1
1Department of Dermatology, University of Groningen, University Medical Center Groningen, 2Groningen Research Institute of Pharmacy, Unit PharmacoTherapy, -Epidemiology & -Economics, University of Groningen, 3Department of Pathology, and 4Department of Epidemiology, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands
#These authors contributed equally to this study.
Photosensitizing properties of hydrochlorothiazide may increase skin cancer risk. To date, study findings on the association between hydrochlorothiazide use and skin cancer risk are inconsistent, notably regarding confounding and dose-response. The aim of this study was to investigate the association between hydrochlorothiazide use and incidence of skin cancer in a cohort of unselected Caucasian adults, taking dosing into account. As part of the PharmLines Initiative, which links data from the Lifelines Cohort Study and prescription database IADB.nl, patients aged ≥ 40 years were included from Lifelines, a prospective population-based cohort study in the north of the Netherlands. Skin cancer incidence was compared between subjects starting hydrochlorothiazide treatment (n = 608), subjects starting treatment with other antihypertensives (n = 508), and non-antihypertensive long-term medication users (n = 1,710). Cox regression analyses were performed to obtain hazard ratios, adjusted for potential confounders. The risk of any skin cancer, keratinocyte carcinoma, basal cell carcinoma and squamous cell carcinoma was not significantly increased in general hydrochlorothiazide users. A clear association was observed between high cumulative hydrochlorothiazide use (≥ 5,000 defined daily dose; ≥ 125,000 mg) and the risk of any skin cancer (adjusted hazard ratio 5.32, 95% confidence interval (95% CI) 2.40–11.81), keratinocyte carcinoma (adjusted hazard ratio 7.31, 95% CI 3.12–17.13), basal cell carcinoma (adjusted hazard ratio 7.72, 95% CI 3.11–19.16) and squamous cell carcinoma (adjusted hazard ratio 19.63, 95% CI 3.12–123.56). These findings should lead to awareness with high use of hydrochlorothiazide in Caucasian adults.
Key words: basal cell carcinoma; hydrochlorothiazide; skin cancer; squamous cell carcinoma.
SIGNIFICANCE
Hydrochlorothiazide is a widely used antihypertensive drug with photosensitizing properties, which may therefore increase skin cancer risk. However, studies are inconsistent regarding this possible association. The current study investigated skin cancer risk in hydrochlorothiazide users. This risk was compared with the skin cancer risk in subjects starting treatment with other antihypertensives and non-antihypertensive long-term medication, in order to avoid detection bias, and corrected for multiple possible confounders. A significant association with elevated adjusted hazard ratio’s in Cox regression analysis was found for patients with high cumulative hydrochlorothiazide use and risk of any skin cancer, keratinocyte carcinoma, basal cell carcinoma and squamous cell carcinoma, suggesting that providing extra information on skin cancer and its prevention is important for this group.
Citation: Acta Derm Venereol 2023; 103: adv3933. DOI https://doi.org/10.2340/actadv.v103.3933.
Copyright: © Published by Medical Journals Sweden, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: Jan 11, 2023; Published: Apr 4, 2023
Corr: Emőke Rácz, Department of Dermatology, University Medical Center Groningen, Hanzeplein 1, NL-9700 RB Groningen, The Netherlands. E-mail: e.racz@umcg.nl
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
Hydrochlorothiazide (HCTZ) is a thiazide diuretic that is regarded as a mainstay of antihypertensive treatment (1). As a result, HCTZ and other thiazide diuretics belong to the most prescribed antihypertensive drugs in the USA and Western European countries (2). HCTZ has photosensitizing properties (3). As HCTZ promotes increased absorption of ultraviolet (UV) radiation, which is a major risk factor for all types of skin cancer, it is possible that prolonged use of HCTZ might be linked to skin cancer (4).
In 2019 a meta-analysis study found that the use of thiazide diuretics is associated with an increased risk of squamous cell carcinoma (SCC, adjusted odds ratio (OR) 1.86), basal cell carcinoma (BCC, adjusted OR 1.19) and melanoma (adjusted OR 1.14) (5). However, as many of the included studies had a case-control design with the use of healthy controls, an important limitation is the possible introduction of detection bias, as HCTZ users usually have more frequent physician contact than do healthy controls, with a higher possibility of detecting skin cancer. This could have led to an overestimation of the actual association. Also, often no information was collected on other important risk factors, such as race, smoking, UV exposure and co-medication. Most of the studies included in this meta-analysis found a positive association between thiazide use and skin cancer risk, although other studies contradicted these findings (6, 7). These differences in association are probably due to different definitions of drug exposure and cancer outcomes, as several studies investigated HCTZ use separately, while others studied thiazide diuretics as a single group. The same applies to skin cancer outcomes, which are grouped differently by studies.
In order to elucidate these inconsistent results, the aim of this study was investigate the association between general use of HCTZ (for at least 1 year), high cumulative use of HCTZ, and the incidence of skin cancer, which, in order to avoid detection bias, was compared with the incidence of skin cancer in users of other antihypertensives and users of non-antihypertensive long-term medication.
MATERIALS AND METHODS
Study design and data sources
A retrospective inception cohort study was performed combining information from 4 national and regional data sources: the Lifelines cohort study, the Pathological Anatomical National Automated Archive (PALGA): nationwide network and registry of histo- and cytopathology in the Netherlands, prescription database IADB.nl (formerly known as InterAction Database) and Statistics Netherlands. The study population and baseline data were selected from the Lifelines cohort between 1 January 2007 and the end of follow-up.
In 2017, the PharmLines Initiative was established to link the data of the Lifelines Cohort Study to the IADB.nl to provide more accurate information about drug exposure data (8). The University of Groningen IADB.nl pharmacy prescription database is an expanding database that contains prescription data for more than 20 years from 1996 until 2019 from approximately 100 community pharmacies and covers an estimated population of 930,000 patients (9). Registration in the database is irrespective of healthcare insurance. Age, sex and prescription rates among the database population have been found to be representative of the Netherlands as a whole. The database has been widely used for research. Each person is individually tracked throughout the database period, and prescription records contain information on the date of dispensing a prescription, the quantity of medication dispensed, the dose regimen, the number of days the prescription is valid, the prescribing physician and the Anatomical Therapeutic Chemical code (ATC code). Each patient has a unique anonymous identifier; date of birth and sex are known. Due to the high patient-pharmacy commitment in the Netherlands, the medication records for each patient are virtually complete, except for over-the-counter (OTC) drugs and medication dispensed during hospitalization.
Lifelines is a multi-disciplinary prospective population-based cohort study with a 3-generation design, examining the health and health-related behaviours of 167,729 persons living in the north of the Netherlands. Lifelines employs a broad range of investigative procedures in assessing the biomedical, socio-demographic, behavioural, physical and psychological factors that contribute to the health and disease of the general population, with a special focus on multi-morbidity and complex genetics (10). Data collection in Lifelines was conducted according to the guidelines of the Declaration of Helsinki, and all procedures were approved by the medical ethics committee of the University Medical Center Groningen (2007/152).
To obtain the most accurate baseline data, the date of the baseline measurements of the Lifelines cohort was matched as accurately as possible to the index date (date of start of drug prescription, which defined the index date; see below).
The primary outcomes of interest were: development of any skin cancer, BCC, SCC (combined as keratinocyte carcinoma (KC)), malignant melanoma and melanoma in situ (combined as melanoma), Merkel cell carcinoma and malignant adnexal skin tumours. Skin cancer diagnoses were retrieved from the PALGA Foundation during the study period (11). This allowed us to gain reliable and complete information regarding skin cancer diagnoses, since all histopathology data of the Netherlands are archived in PALGA, which is unique to have one foundation storing all national pathology data.
Information about mortality was obtained from Statistics Netherlands, which also acted as a trusted third party, in which linkage of Lifelines, IADB.nl, PALGA and Statistics Netherlands was performed. After the linkage, all patient identifiers were removed and each participant was given a unique identifier code, to ensure data were completely anonymized. Analyses were conducted in a password secured electronic environment, which was provided by Statistics Netherlands (12).
Study population: inclusion and exclusion
Patients were selected based on the index date, which is the date of the first prescription of HCTZ (index group), other antihypertensives or non-antihypertensive long-term medication (2 reference groups). The first reference group “subjects starting treatment with other non-HCTZ antihypertensives” was defined as starting with a minimum of 2 prescriptions of non-thiazide containing antihypertensive drugs. The second reference group “subjects starting treatment with non-antihypertensive long-term medication” was defined as starting with a minimum of 2 prescriptions within the first year of a non-antihypertensive long-term drug for a chronic disease that requires regular prescriptions. Further details and definitions of index and reference groups are shown in Appendix S1.
Follow-up started on the index date and ended on the date of skin cancer diagnosis, date of death, if applicable, or end of follow-up, which was 31 December 2020.
Only patients who started these drugs at age 40 years or older were eligible for inclusion. To ensure no antihypertensive or other long-term medication treatment was initiated before the index date, only patients who were not prescribed these medications before the index date and who were present > 365 days before and 185 days after index date in the IADB.nl database were included.
Patients who were not Caucasian were further excluded, as it has been shown that skin cancer is not as prevalent in other racial groups and most Lifelines participants were Caucasian (13). Patients with high risk of skin cancer, including those with a history of organ transplantation (14), history of human immunodeficiency virus (HIV) (15), chronic lymphocytic leukaemia (16), and a previous diagnosis of melanoma or KC were also excluded. Patients who had a skin cancer diagnosis within 6 months after the index date were also excluded, to ensure a reasonable time from exposure to cancer outcome (17). Furthermore, patients who ever used HCTZ, HCTZ combination preparations, or other thiazide(-like) diuretics (at least 2 prescriptions) were excluded from the reference groups. Selection of the study population is shown in Fig. 1.
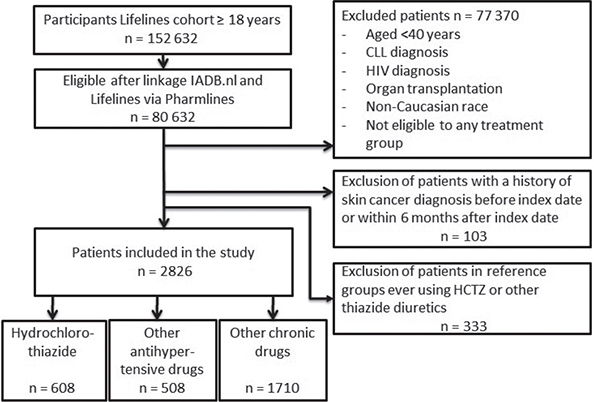
Fig. 1. Flowchart of patient selection. CLL: chronic lymphocytic leukaemia; HIV: human immunodeficiency virus.
Statistical analysis
To compare baseline characteristics across HCTZ users and the 2 reference groups, the χ2 test was used for categorical data and the Kruskal–Wallis test for continuous data.
Skin cancer incidence was estimated per 100 months at risk (incidence density) for the different groups. To analyse the association between HCTZ use and skin cancer risk, hazard ratios (HRs) were estimated using a Cox regression model. In case of multiple skin cancer, time to the first skin cancer diagnosis was used to define the time at risk. Both univariable and multivariable Cox regression analysis was used to estimate skin cancer risk in the different study groups, adjusted for covariables, which were significantly different between the index and reference groups (possible confounders), to estimate adjusted HRs (aHRs). The following potential confounders were included (see Appendix S1): age at index date, sex, smoking status, use of immunosuppressant drugs, use of oral glucocorticoids, use of (other) diuretics, statins, comorbidities, such as diabetes, education level, UV exposure, ever outdoor profession and coffee consumption.
To investigate a potential dose-response relationship of HCTZ use and skin cancer, HRs were computed for cumulative defined daily dose (DDD; < 5000 DDD and ≥ 5000 DDD, equal to < 125,000 mg and ≥ 125,000 mg, respectively, in which 1 DDD is equal to 25 mg HCTZ) in univariable Cox regression analysis, and adjusted for the most prominent and frequent confounders in multivariable analysis (groups were too small to adjust for all possible confounders). A p-value < 0.05 was considered statistically significant. Statistical analyses were performed using SPSS, version 25 (SPSS, Inc., Chicago, IL, USA).
RESULTS
Baseline characteristics
After selection of inclusion and exclusion criteria, a total of 2,826 participants were included (Fig. 1). Of this group, 608 patients started with HCTZ, 508 patients with another antihypertensive drug, and 1,710 with other long-term medication. Median age at index was 55 years for HCTZ users, 54 years for antihypertensive drug users, and 50 years for other long-term medication users (p < 0.001, Table I).
| HCTZ users n = 608 | Other antihyper-tensive users n = 508 | Other long-term medication users n = 1,710 | p-value | |
| Age at index, years, median (IQR) | 55 (49–63) | 54 (47–61) | 50 (45–58) | < 0.001 |
| Female sex, n (%) | 344 (56.6) | 249 (49.0) | 976 (57.1) | 0.005 |
| Education level, n (%) | ||||
| Low | 239 (39.3) | 164 (32.3) | 541 (31.6) | |
| Middle | 177 (29.11) | 140 (27.6) | 550 (32.2) | |
| High | 187 (30.8) | 196 (38.6) | 604 (35.3) | |
| Unknowna | < 10 | < 10 | 15 (0.9) | 0.002 |
| Smoking status, n (%) | ||||
| Never | 199 (32.7) | 191 (37.6) | 668 (39.1) | |
| Ever | 409 (67.3) | 317 (62.5) | 1,042 (60.9) | 0.021 |
| UV exposureb, median (IQR) | 7 (5–10) | 7 (5–10) | 7 (5–10) | 0.121 |
| Ever outdoor profession, n (%) | 20 (3.3) | 22 (4.3) | 73 (4.3) | 0.546 |
| Coffee consumptionc, median (IQR) | 28 (21–35) | 28 (21–35) | 28 (21–35) | 0.174 |
| Self-reported comorbidities | ||||
| Diabetes | 44 (7.2) | 26 (5.1) | 58 (3.4) | < 0.001 |
| Chronic obstructive pulmonary disease | 37 (6.1) | 10 (2.0) | 59 (3.5) | 0.002 |
| Heart attack | < 10 | 13 (2.6) | 31 (1.8) | 0.0985 |
| Stroke | 11 (1.8) | < 10 | 14 (0.8) | 0.158 |
| Heart failure | 17 (2.8) | 19 (3.7) | 37 (2.2) | 0.165 |
| Use of other drugs, n (%) | ||||
| Use of immuno-suppressive drugs, n (%) | 24 (3.9) | 13 (2.6) | 38 (2.2) | 0.075 |
| Oral glucocorticoids | 137 (22.5) | 35 (6.9) | 172 (10.1) | < 0.001 |
| Other diuretics | 68 (11.2) | 34 (6.7) | 40 (2.3) | < 0.001 |
| Statins | 279 (45.9) | 181 (35.6) | 237 (13.9) | < 0.001 |
| aExcluded from analysis. bNumber of days (> 30 min/day)/2 weeks, median. cDefined as mean cups of coffee per week. | ||||
| HCTZ: hydrochlorothiazide; IQR: interquartile range; UV: ultraviolet. | ||||
The HCTZ group and other long-term medication group comprised a slightly higher proportion of female users (56.6% and 57.1%, respectively) compared with the other antihypertensive drug users (49.0%, p = 0.005, Table I). The other antihypertensive drug users contained the largest group of patients with high education level (38.6%, p = 0.002, Table I). Smokers were most frequent in the HCTZ group (67.3%, p = 0.021, Table I). Coffee consumption, UV exposure and ever outdoor profession did not differ significantly between the groups. The occurrence of comorbidities and the use of other medication in the index and reference groups are also shown in Table I. Median follow-up time was 105 months for HCTZ users, 97 months for other antihypertensive drug users, and 110 months for other long-term medication users (Table II).
| HCTZ users n = 608 | Other antihypertensive users n = 508 | Other long-term medication users n = 1,710 | |
| Age at first diagnosis, years, median (IQR) | 67 (60–74) | 64 (54–72) | 63 (55–71) |
| Follow-up time, months, median (IQR) | 105 (66–161) | 97 (56–138) | 110 (68–155) |
| Time to first skin cancers, months, median (IQR) | 93 (59–152) | 89 (57–122) | 90 (45–145) |
| Skin cancer incidence density per 100 months in patientsa | |||
| Any skin cancer | 0.068 | 0.063 | 0.047 |
| Keratinocyte cancer | 0.056 | 0.055 | 0.042 |
| Basal cell carcinoma | 0.045 | 0.049 | 0.037 |
| Squamous cell carcinoma | 0.015 | 0.0076 | 0.0070 |
| Melanomab | 0.011 | 0.0076 | 0.0065 |
| Merkel cell carcinoma | – | – | – |
| Malignant adnexal skin cancer | – | – | – |
| Skin cancer incidence density per 100 months in patients with multiple skin cancersc | |||
| No | 0.046 | 0.049 | 0.035 |
| Yes | 0.021 | 0.013 | 0.012 |
| Total amount of skin cancer, n (%) | |||
| Any skin cancer | 72 (100) | 46 (100) | 133 (100) |
| Keratinocyte cancer | 64 (88.9) | 41 (89.1) | 120 (90.2) |
| Basal cell carcinoma | 53 (73.6) | 36 (78.3) | 105 (78.9) |
| Squamous cell carcinoma | 11 (15.3) | 5 (10.9) | 15 (11.3) |
| Melanomab | 8 (11.1) | 5 (10.9) | 13 (9.8) |
| aIncluding patients with multiple diagnoses of BCC, SCC and melanoma combined. bMelanoma = melanoma in situ + invasive melanoma. c≥ 2 diagnosis of any skin cancer. | |||
| HCTZ: hydrochlorothiazide; N/A: not applicable. | |||
Skin cancer
Table II summarizes the skin cancer incidence density and total skin cancer numbers among the different groups. Among all types of skin cancer, BCC was mostly identified in all study groups. No cases of Merkel cell carcinoma or malignant adnexal skin cancer were identified. Skin cancer incidence density per 100 months was slightly higher in HCTZ users for any skin cancer, KC, SCC, and melanoma, compared with the reference groups. Furthermore, the HCTZ group included the highest incidence of multiple skin tumours.
Cox regression analyses
After adjusting for the following possible confounders: age, sex, smoking status and other diuretics (first-mentioned aHR) and additional oral glucocorticoids, statins, diabetes, chronic obstructive pulmonary disease (COPD) and education level (second-mentioned aHR), in general HCTZ users, no significant aHRs for any skin cancer (aHR 1.12, 95% confidence interval (95% CI) 0.78–1.60 and aHR 1.29, 95% CI 0.61–2.71), KC (aHR 1.02, 95% CI 0.69–1.51 and aHR 1.19, 95% CI 0.54–2.64), BCC (aHR 0.93, 95% CI 0.60–1.43 and aHR 0.83, 95% CI 0.33–2.07) and SCC (aHR 1.35, 95% CI 0.59–3.08 and aHR 6.25, 95% CI 0.58–66.89) were found in multivariable analyses (Table III).
| Treatment group | Crude HR (95% CI) | aHRa (95% CI) | aHRb (95% CI) |
| Any skin cancer | |||
| HCTZ | 1.45 (1.02–2.05) | 1.12 (0.78–1.60) | 1.29 (0.61–2.71) |
| Other antihypertensives | 1.40 (0.94–2.09) | 1.14 (0.76–1.71) | 1.01 (0.42–2.43) |
| Other long-term medication | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) |
| Keratinocyte carcinoma | |||
| HCTZ | 1.35 (0.93–1.97) | 1.02 (0.69–1.51) | 1.19 (0.54–2.64) |
| Other antihypertensives | 1.38 (0.91–2.22) | 1.10 (0.72–1.70) | 1.09 (0.44–2.69) |
| Other long-term medication | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) |
| Basal cell carcinoma | |||
| HCTZ | 1.21 (0.80–1.83) | 0.93 (0.60–1.43) | 0.83 (0.33–2.07) |
| Other antihypertensives | 1.39 (0.89–2.18) | 1.14 (0.73–1.80) | 0.90 (0.34–2.36) |
| Other long-term medication | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) |
| Squamous cell carcinoma | |||
| HCTZ | 2.28 (1.07–4.87) | 1.35 (CI 0.59–3.08) | 6.25 (0.58–66.89) |
| Other antihypertensives | 1.11 (0.37–3.35) | 0.75 (0.24–2.30) | 2.71 (0.14–53.94) |
| Other long-term medication | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) |
| aAdjusted for age, sex, smoking status and other diuretics. bAdjusted for age, sex, smoking status, other diuretics, oral glucocorticoids, statins, diabetes, chronic obstructive pulmonary disease and education level. | |||
| aHR: adjusted hazard ratio; 95% CI: 95% confidence interval; HCTZ: hydrochlorothiazide. Bold values are statistically significant. | |||
Due to low numbers, risk of melanoma was not analysed using Cox regression analysis.
Cumulative defined daily dose
Table IV shows 2 subgroups of cumulative DDDs and the risk of any skin cancer, KC, BCC and SCC.
| DDD, mean (SD) 1,907 (5,132) DDD, median (IQR) 952.5 (465–2,032) |
||
| Cumulative DDD per skin cancer type | Crude HR (95% CI) | aHRa (95% CI) |
| Any skin cancer | ||
| < 5,000b (n = 441) | 1.00 (reference) | 1.00 (reference) |
| ≥ 5,000 (n = 28c) | 4.20 (1.96–8.97) | 5.32 (2.40–11.81) |
| Keratinocyte carcinoma | ||
| < 5,000b (n = 441) | 1.00 (reference) | 1.00 (reference) |
| ≥ 5,000 (n = 28) | 5.39 (2.42–12.02) | 7.31 (3.12–17.13) |
| Basal cell carcinoma | ||
| < 5,000b (n = 441) | 1.00 (reference) | 1.00 (reference) |
| ≥ 5,000 (n = 28) | 5.44 (2.32–12.75) | 7.72 (3.11–19.16) |
| Squamous cell carcinoma | ||
| < 5,000b (n = 441) | 1.00 (reference) | 1.00 (reference) |
| ≥ 5,000 (n = 28) | 14.53 (2.55–82.83) | 19.63 (3.12–123.56) |
| aAdjusted for age, sex, smoking status and other diuretics. b146–4,999 DDD. cIn the hydrochlorothiazide (HCTZ) group, only patients using HCTZ for a minimal of 292 days were included. It was assumed that patients with a documented cumulative DDD of less than 292/2 = 146 DDD (a mean of 12.5 mg HCTZ/day for 292 days), were incorrectly entered into the prescription database IADB. These patients were excluded from analysis. Therefore, the total amount of patients is < 608. | ||
| aHR: adjusted hazard ratio; IQR: interquartile range; 95% CI: 95% confidence interval. 1 DDD is equal to 25 mg HCTZ. | ||
| Bold values are statistically significant. | ||
The median DDD of HCTZ use was 952.5 DDD. Overall, 6% of the patients were high-dose users of HCTZ (≥ 5,000 DDD, equal to ≥ 125,000 mg). Of patients with any skin cancer, 34% were high-dose users, yielding a significant aHR of 5.32 (95% CI 2.40–11.81). Forty-one percent of patients with KC were high cumulative dose users of HCTZ, with a significant aHR of 7.31 (95% CI 3.12–17.13).
Of patients with BCC and SCC, 42% and 67% were high cumulative dose users, respectively. Significant aHRs of 7.72 (95% CI 3.11–19.16) for BCC and 19.63 (95% CI 3.12–123.56) for SCC were found.
Survival analysis: Kaplan–Meier curves
The Kaplan–Meier curves of skin cancer risk in the 3 study groups showed that the survival curve belonging to the hypertensive drug users crossed the other curves 1 or multiple times. The survival curves of the different cumulative DDD HCTZ groups did not cross (Appendix S1; Fig. S1).
DISCUSSION
This retrospective cohort study found that general use of HCTZ (for at least 1 year) was not an independent risk factor for the development of skin cancer. However, a clear association was observed between high cumulative use of HCTZ (≥ 5,000 DDD; ≥ 125,000 mg) and any skin cancer, KC, BCC, and SCC. This finding shows the importance of increased exposure by prolonged and/or high use of HCTZ and skin cancer risk.
Other studies are inconsistent regarding skin cancer risk and the use of HCTZ. Multiple studies have found an increased risk of skin cancer, especially of SCC, in HCTZ or other thiazide diuretic users (5, 18–22). However, most of these studies did not include UV exposure as a possible important confounder (18–22). Other studies have found no association of thiazide or HCTZ use and skin cancer risk (6, 7, 23–25). Part of these studies did not specifically analyse high cumulative use of (hydrochloro)thiazide (6, 23) and 1 study only analysed BCC risk (7). Two of these studies were conducted in predominantly Asian populations, which found no significant associations or even decreased risks of KC in high cumulative users of HCTZ compared with non-HCTZ users and concluded that HCTZ use is safe in the Asian population (24, 25).
Although this study included only patients who had been using HCTZ for at least 1 year, an explanation for the insignificant risk of skin cancer in the entire group of HCTZ users in the current study is probably that most patients (94%) were low cumulative HCTZ users, leading to a lower susceptibility of developing skin cancer. This is confirmed by the results from the current dose-response analyses, which showed that, in patients who were prescribed more than 5,000 cumulative DDD of HCTZ (≥ 125,000 mg), a significant and a much higher risk of skin cancer was observed, especially for SCC with an aHR of 19.63. This is in line with the finding that cumulative UV damage plays the largest role in the development of SCC (26).
The association of high exposure to HCTZ and the risk of skin cancer, especially of SCC, has been confirmed by multiple studies. The study of Pedersen et al. (19) found a significant adjusted OR of 1.54 for BCC and 7.38 for SCC with high cumulative use (≥ 200,000 mg) of HCTZ. In a study by Jensen et al. (18) an incidence rate ratio (IRR) of 1.67 was found for SCC in patients using HCTZ for at least 1 year and 1.92 in patients using HCTZ for more than 5 years. The IRR of BCC was not significantly increased. Another study found an OR of 2.1 for SCC of the lip in ever-HCTZ users, which increased to 3.9 for higher cumulative use of HCTZ (≥ 25,000 mg) (20). A recent study also found increased HRs for SCC in patients with high duration (HR of 3.70 for HCTZ use > 10 years) and high cumulative use (HR of 4.96 for cumulative use ≥ 100,000 mg) of HCTZ. They did not find an increased risk of BCC or melanoma. However, their reference group consisted of users of other thiazide diuretics, who may also have an increased risk of developing skin cancer (27). Another recent study found comparable results, in which an IRR of 1.95 for SCC was found in long-term users of HCTZ (≥ 20 prescriptions), but no associations with BCC and melanoma were found (22).
In consultation with the European Medicines Agency and the Dutch Medicines Evaluation Board, marketing authorization holders of HCTZ-containing drugs have sent a letter on 30 October 2018 to healthcare professionals in the Netherlands, warning about the risk of KC in patients who are long-term users of HCTZ (28). Since the time between the warning and the included skin cancers (until 2020) was relatively short, it was not possible to investigate the influence of this warning on skin cancer incidence. The effect of the warning should be evaluated in future studies, which could have led to different sun exposure and protection behaviour patterns in patients using HCTZ in the last few years.
Although not significant, this study also found increased aHRs for the development of any skin cancer and KC in the other antihypertensive group. Su et al. (23) demonstrated a weak risk of SCC with the use of angiotensin converting enzyme (ACE)-inhibitors (aHR 1.10, 95% CI 1.01–1.19), which were included in the other antihypertensive group in the current study. Schmidt et al. (29) found an increased risk of malignant melanoma with long-term low intensity use of angiotensin II receptor blockers (OR, 1.53; 95% CI 1.05–2.23), which were also included in our other antihypertensive group. These associations might provide an explanation for the somewhat increased aHRs in the other antihypertensive group of our study. More studies investigating skin cancer risk in non-thiazide antihypertensive drugs are needed before these drugs are considered as possible safe alternatives to HCTZ.
The main strengths of the current study include the population-based design, the long follow-up period, and the use of very reliable, complete and large cancer and prescription registries, which minimizes selection bias and maximizes validity of the results. This study also considered a variety of confounders and an induction period of at least 6 months for a skin cancer diagnosis to be included, in order to consider a reasonable time from exposure to cancer outcome, which is of particular importance for skin cancer outcomes. Detection bias was prevented by using other long-term medication users and users of other hypertensive drugs as reference groups, since long-term medication users usually have more frequent physician contact than healthy controls.
Study limitations
This study has some limitations. Due to the use of the Lifelines cohort, some risk factors did not apply to the study population. Only Caucasian adults were included, since most of the Lifelines participants are Caucasian, and significance could not be achieved with other skin types. This study was unable to collect information regarding the use of sunscreen, tanning behaviour, or the tendency to sunburn. As the current study used mean time spent outdoors as a substitute measure of UV exposure (of which the timeframe for UV exposure vs HCTZ exposure is unclear), this could have introduced misclassification. In addition, comorbidities were self-reported in the Lifelines cohort, making these outcomes less reliable. Our groups were relatively small and therefore this study could not correct for all possible confounders in Cox regression analysis regarding cumulative DDD; also, the study did not stratify the analysis according to all possible confounders, such as age, sex, smoking status or educational level, although this might have led to identifying more specific groups at risk as they were significantly different between the HCTZ group and the control group. Some HR estimators have large CIs and may have been significant in a larger study population. Also, the risk of melanoma could not be analysed due to low incidence numbers. Furthermore, as the IADB.nl database contained only prescription data from public pharmacies, data regarding prescription records from hospital pharmacies were missing, which could lead to an incomplete picture of the co-medication history of some study participants. Finally, using the Kaplan–Meier curve for the risk of skin cancer, the lines of the different study groups were crossing, making the results of the univariable Cox regression analysis less reliable to interpret.
Conclusion
This study showed no evidence for an association between general HCTZ use (for at least 1 year) and the risk of skin cancer in a Caucasian population. A clear association was observed for any skin cancer, KC, BCC, and SCC, with high cumulative HCTZ use (≥ 5000 DDD; ≥ 125 000 mg), suggesting that the insignificant risk association between skin cancer and general HCTZ use was probably caused by the large proportion of low cumulative users of HCTZ.
Consequently, these results show that caution is advised with prolonged and high use of HCTZ in Caucasian adults. In these cases, alternative antihypertensive treatments should be considered. Further analyses in larger European ancestry populations are needed to validate the current findings. In addition, further analysis of other potential risk factors, such as smoking, sun exposure, educational level and comorbidity in larger cohorts of HCTZ users might lead to the identification of even more specific groups at risk of skin cancer in association with HCTZ use. In the current cohort, this study corrected only for potential confounders, but did not stratify the current results for all other potential risk factors. In addition, more studies investigating skin cancer risk in non-thiazide antihypertensive drugs are needed before these drugs are considered as safe alternatives to HCTZ, since it has been shown previously that other antihypertensive drugs may also increase skin cancer risk to a lesser extent.
ACKNOWLEDGEMENTS
The authors wish to acknowledge the services of the Lifelines Cohort Study, the contributing research centres delivering data to Lifelines, all the study participants, PALGA and the participating IADB.nl pharmacies for kindly providing their data for research.
The Lifelines initiative has been made possible by subsidy from the Dutch Ministry of Health, Welfare and Sport, the Dutch Ministry of Economic Affairs, the University Medical Center Groningen (UMCG), Groningen University and the Provinces in the North of the Netherlands (Drenthe, Friesland, Groningen). The IADB.nl is funded by the University of Groningen. Since this particular study was not funded, these sources had no role in the preparation of data or the manuscript.
The data collection in Lifelines was conducted according to the guidelines of the Declaration of Helsinki, and all procedures were approved by the Medical Ethics Committee of the University Medical Center Groningen (2007/152). All Lifelines participants have each signed an informed consent stating that he/she approves use of his/her (anonymized) data and material for scientific purposes.
Data of the IADB.nl is collected according to the national and European guidelines on privacy with human data valid at the time of collection.
The data that support the findings of this study are available from PharmLines, PALGA, CBS and IADB.nl. Restrictions apply to the availability of these data, which were used under license for this study. Data are available with the permission of PharmLines, PALGA, CBS and IADB.nl.
REFERENCES
- Akbari P, Khorasani-Zadeh A. Thiazide diuretics. 2021 Jul 13. In: StatPearls. Treasure Island (FL): 2022 Jan. [Assessed on 30 June 2022] Available at: https://www.statpearls.com/point-of-care/30055.
- Wang YR, Alexander, G Caleb, Stafford RS. Outpatient hypertension treatment, treatment intensification, and control in Western Europe and the United States. Available at https://jamanetwork.com/
- George EA, Baranwal N, Kang JH, Qureshi AA, Drucker AM, Cho E. Photosensitizing medications and skin cancer: a comprehensive review. Cancers (Basel). MDPI; 2021.
- Armstrong BK, Kricker A. The epidemiology of UV induced skin cancer. J Photochem Photobiol B. 2001; 63: 8–18.
- Shin D, Lee ES, Kim J, Guerra L, Naik D, Prida X. Association between the use of thiazide diuretics and the risk of skin cancers: a meta-analysis of observational studies. J Clin Med Res 2019; 11: 247–255.
- Schmidt SAJ, Schmidt M, Mehnert F, Lemeshow S, Sørensen HT. Use of antihypertensive drugs and risk of skin cancer. J Eur Acad Dermatol Venereol 2015; 29: 1545–1554.
- Ruiter R, Visser LE, Eijgelsheim M, Rodenburg EM, Hofman A, Coebergh JWW, et al. High-ceiling diuretics are associated with an increased risk of basal cell carcinoma in a population-based follow-up study. Eur J Cancer 2010; 46: 2467–2472.
- Sediq R, van der Schans J, Dotinga A, Alingh RA, Wilffert B, Bos JHJ, et al. Concordance assessment of self-reported medication use in the netherlands three-generation lifelines cohort study with the pharmacy database iadb.Nl: the pharmlines initiative. Clin Epidemiol Dove 2018; 10: 981–989.
- IADB. Background. Visited on 15 November 2021. Available via: https://www.iadb.nl/
- Scholtens S, Smidt N, Swertz MA, Bakker SJL, Dotinga A, Vonk JM, et al. Cohort Profile: LifeLines, a three-generation cohort study and biobank. Int J Epidemiol 2015; 44: 1172–1180.
- Casparie M, Tiebosch ATMG, Burger G, Blauwgeers H, van de Pol A, van Krieken JHJM, et al. Pathology databanking and biobanking in The Netherlands, a central role for PALGA, the nationwide histopathology and cytopathology data network and archive. Cell Oncol 2007; 29: 19–24.
- Centraal Bureau voor de Statistiek (CBS). CBS home. [Assessed on 06 June 2021] Available at: https://www.cbs.nl/nl-nl
- Gloster HM, Jr, Neal K. Skin cancer in skin of color. J Am Acad Dermatol 2006; 55: 741–764.
- Adami J, Gäbel H, Lindelöf B, Ekström K, Rydh B, Glimelius B, et al. Cancer risk following organ transplantation: a nationwide cohort study in Sweden. Br J Cancer 2003; 89: 1221–1227.
- Wilkins K, Turner R, Dolev JC, LeBoit PE, Berger TG, Maurer TA. Cutaneous malignancy and human immunodeficiency virus disease. J Am Acad Dermatol 2006; 54: 189–206.
- Royle JA, Baade PD, Joske D, Girschik J, Fritschi L. Second cancer incidence and cancer mortality among chronic lymphocytic leukaemia patients: a population-based study. Br J Cancer 2011; 105: 1076–1081.
- Pottegård A, Hallas J. New use of prescription drugs prior to a cancer diagnosis. Pharmacoepidemiol Drug Saf 2017; 26: 223–227.
- Jensen A, Thomsen HF, Engebjerg MC, Olesen AB, Sørensen HT, Karagas MR. Use of photosensitising diuretics and risk of skin cancer: a population-based case-control study. Br J Cancer 2008; 99: 1522–1528.
- Pedersen SA, Gaist D, Schmidt SAJ, Hölmich LR, Friis S, Pottegård A. Hydrochlorothiazide use and risk of nonmelanoma skin cancer: a nationwide case-control study from Denmark. J Am Acad Dermatol 2018; 78: 673–681.
- Pottegård A, Hallas J, Olesen M, Svendsen MT, Habel LA, Friedman GD, et al. Hydrochlorothiazide use is strongly associated with risk of lip cancer 2017; 282: 322–331.
- Rouette J, Yin H, Pottegård A, Nirantharakumar K, Azoulay L. Use of hydrochlorothiazide and risk of melanoma and nonmelanoma skin cancer. Drug Saf 2021; 44: 245–254.
- Schneider R, Reinau D, Stoffel S, Jick SS, Meier CR, Spoendlin J. Risk of skin cancer in new users of thiazides and thiazide-like diuretics: a cohort study using an active comparator group*. Br J Dermatol 2021; 185: 343–352.
- Su KA, Habel LA, Achacoso NS, Friedman GD, Asgari MM. Photosensitizing antihypertensive drug use and risk of cutaneous squamous cell carcinoma. Br J Dermatol 2018; 179: 1088–1094.
- Pottegård A, Pedersen SA, Schmidt SAJ, Lee CN, Hsu CK, Liao TC, et al. Use of hydrochlorothiazide and risk of skin cancer: a nationwide Taiwanese case-control study. Br J Cancer 2019; 121: 973–978.
- Park E, Lee Y, Jue MS. Hydrochlorothiazide use and the risk of skin cancer in patients with hypertensive disorder: a nationwide retrospective cohort study from Korea. Korean J Intern Med 2020; 35: 917–928.
- Rosso S, Zanetti R, Martinez C, Tormo MJ, Schraub S, Sancho-Garnier H, et al. The multicentre south European study “Helios”. II: Different sun exposure patterns in the aetiology of basal cell and squamous cell carcinomas of the skin. Br J Cancer 1996; 73: 1447–1454.
- Rouette J, Yin H, Pottegård A, Nirantharakumar K, Azoulay L. Use of hydrochlorothiazide and risk of melanoma and nonmelanoma skin cancer. Drug Saf 2021; 44: 245–254.
- Medicines Evaluation Board. Hydrochloorthiazide. [Assessed on 15 Nov 2021] Available from: https://www.cbg-meb.nl/documenten/videos/2018/10/30/hct-ton-de-boer.
- Schmidt SAJ, Schmidt M, Mehnert F, Lemeshow S, Sørensen HT. Use of antihypertensive drugs and risk of skin cancer. J Eur Acad Dermatol Venereol 2015; 29: 1545–1554.