ORIGINAL REPORT
Predictors of Local Invasion in Infiltrative Basal Cell Carcinoma: Tumour Budding Outperforms the WHO Subtyping
Maria-Teresa FERNÁNDEZ-FIGUERAS1,2, Noelia PÉREZ-MUÑOZ1,2, Luis PUIG3,4, Rodolfo POSADA-CAEZ1, Rosa BALLESTER VICTORIA1, Martha HENRIQUEZ2 and Eva MUSULEN1,5
1Department of Pathology, Hospital Universitari General de Catalunya, Grupo Quironsalud, Sant Cugat del Vallés, Barcelona, 2Affiliation Universitat Internacional de Catalunya, Sant Cugat del Vallés, Barcelona, 3Affiliation Department of Dermatology, IIB Sant Pau, Hospital de la Santa Creu i Sant Pau, Barcelona, 4Affiliation Universitat Autònoma de Barcelona, Barcelona, Spain, and 5Affiliation Institut de Recerca contra la Leucèmia Josep Carreras, Barcelona, Spain
Tumour budding (TB) correlates with increased local invasion in various neoplasms. Certain basal cell carcinomas (BCCs) exhibit local aggressiveness. Detecting adverse prognostic factors in partial biopsies could aid in identifying cases with heightened local risk. The absolute number of TB (≤ 3 tumour cells) in excision specimens of 271 infiltrative BCCs (0: absent; 1: 1–2 foci; 2: ≥ 3 foci; 3: ≥ 10 foci), the histopathological subtype and depth of infiltration, perineural invasion, and other histological features were evaluated. A significant correlation was found between TB and both depth of infiltration (rho 0.445, p < 0.001) and perineural invasion (p = 0.009). In the multivariate analysis of depth and perineural invasion (multiple regression, stepwise), TB was identified as a significant covariate together with diameter, inflammation, and perineural invasion for the former, and depth for the latter. Conversely, no correlation existed between the WHO histological subtypes (infiltrating, sclerosing, and micronodular), and depth of infiltration or perineural invasion. This study demonstrates the value of TB as a biomarker for local invasiveness in BCC. In routine practice, a count of ≥ 3 TB foci in lesions incompletely excised or with narrow tumour-free surgical margins would be a straightforward and reproducible method to guide BCC treatment.
Key words: basal cell carcinoma; classification; risk assessment; treatment; tumour budding; World Health Organization.
SIGNIFICANCE
Basal cell carcinomas are typically low-aggressiveness tumours; however, infiltrative variants such as sclerosing, infiltrating, and micronodular subtypes can cause significant local destruction, necessitating repeated surgeries and impacting patient well-being. Our study highlights the utility of assessing tumour budding in partial basal cell carcinoma specimens to identify those at higher local risk, aiding treatment decisions and reducing the need for additional procedures. Conversely, we demonstrated that the WHO subclassification of infiltrative basal cell carcinoma variants lacks correlation with histological features indicative of local aggressiveness.
Citation: Acta Derm Venereol 2024; 104: adv40172. DOI https://doi.org/10.2340/actadv.v104.40172.
Copyright: © 2024 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Feb 25, 2024; Accepted after revision: Jun 10, 2024; Published: Jul 2, 2024
Corr: Maria-Teresa Fernández-Figueras, Hospital Universitari General de Catalunya, Carrer Pedro i Pons, Sant Cugat del Vallés (Barcelona), ES-08195, Spain. E-mail: maiteffig@gmail.com
Competing interests and funding: The authors have no conflicts of interest to declare.
Martha Henriquez was a recipient of the Horacio Oliva grant for medical students from the Sociedad Española de Anatomía Patológica (SEAP), which supported her 2-month tenure at the Department of Anatomical Pathology, Hospital Universitari General de Catalunya, grupo Quironsalud.
INTRODUCTION
Basal cell carcinoma (BCC) stands as the most prevalent human cancer type, exhibiting a steadily increasing incidence. Correspondingly, there has been a rise in the occurrence of locally aggressive cases, usually characterized by infiltrative growth. A significant number of BCCs are histologically diagnosed in incisional biopsies, or their excisions reveal involved margins. Discovering a histological biomarker capable of stratifying the risk of deep local invasion in these cases could greatly aid in guiding therapeutic decisions, at the same time averting aggressive procedures and tumour relapses.
The treatment of BCC has been traditionally influenced by the histological subtype, especially in “difficult to treat” cases and those located in “high risk areas” (1). Nonetheless, interobserver agreement in subtyping tends to be low, mostly due to the frequent overlap of different subtypes in the same tumour. Recently, a group of experienced dermatopathologists elaborated a position statement declaring that unifying the infiltrating, sclerosing, and micronodular subtypes into a single infiltrative category improves the consensus among observers, without significant therapeutic implications (2). However, the identification of a histopathological biomarker to categorize the risk of local invasiveness in this group of infiltrative BCCs remains an unmet need to adjust the best treatment for each tumour.
Recent studies on infiltrative BCCs (3) have revealed that the amplification of a subpopulation of tumour cells promoting epithelial-mesenchymal transition (EMT) is associated with increased invasiveness and a greater likelihood of recurrence. Tumour budding (TB) is a manifestation of partial epithelial-to-mesenchymal transition (p-EMT), an intermediate stage that provides invasiveness, stemness, and chemoresistance (4). TB is a widely recognized prognostic factor in several human neoplasms, displaying a direct correlation with local invasion, metastases, and overall survival (5), and can be a surrogate marker of local aggressiveness in BCC.
Histologically, TB is characterized by the presence of single cells or small clusters (3–5 cells) at the invasive front of tumours, although intratumoral budding has also relevance (5). Poorly differentiated clusters (5–15 cells) have also been studied in some tumours and are considered to represent an earlier stage of p-EMT (6). Since its original description in 1954 (7), the TB parameter has been incorporated into the study of gastrointestinal and head and neck tumours (6, 8), and more recently in urothelial carcinomas (9), gynaecological neoplasms (10), and other malignancies (11, 12).
The importance of TB is recognized in skin tumours, but only a limited number of studies have been published on this subject. Most of them are focused on cutaneous squamous cell carcinoma, demonstrating a high specificity and sensitivity in predicting recurrences (13), and a significant correlation with metastases and mortality risk (14–16). In addition, 1 study of a series of 742 melanomas revealed that a high degree of TB (> 5 buds in 0.785 mm2) is an independent factor that correlates with recurrence and 5-year overall survival (17).
The main objective of this study was to investigate the relation between TB and features of local aggressivity in BCCs exhibiting infiltrative growth. The primary implication would be the identification, among incisional biopsies or excisions of BCCs with narrow or positive margins, of cases at a higher risk of deep local involvement and perineural invasion, based on the presence of TB. Assessing this parameter could significantly contribute to selecting the most appropriate treatment for individual cases.
MATERIALS AND METHODS
Case selection
We reviewed 675 cases of cutaneous BCC from the files of the Pathology Department at Hospital Universitari General de Catalunya, Grupo Quironsalud, received between 1 February 2022, and 31 December 2022. Among them, 271 cases met the criteria established for inclusion in the study.
The inclusion criteria were: (i) BCCs with infiltrative growth-infiltrating, micronodular and sclerosing, and (ii) complete resection specimens. The exclusion criteria were (i) the presence of fibrous scars from previous biopsies occupying more than 30% of the tumour, (ii) nodular or superficial BCCs with infiltrative areas smaller than 0.5 mm in width, and (iii) basosquamous cell carcinomas. No clinical data were investigated for these patients; however, to the best of our knowledge, metastases did not develop in any of these cases.
Histopathologic evaluation
Digital images of haematoxylin-eosin-stained slides scanned using the microscope slide scanner IntelliSite (Philips Healthcare, Amsterdam, the Netherlands) were retrieved from the institutional repository. In every case, tumour tissue had been completely included in paraffin blocks and all sections were examined. The digitalized images were evaluated separately by at least 2 dermatopathologists. When agreement was not achieved, the case was discussed to reach a consensus, or it was shown to a third researcher. After examination of all available sections, the following items were evaluated: tumour budding, infiltrative subtype (most abundant pattern), diameter of the infiltrative area, depth, ulceration, inflammatory response, degree of squamatisation, and perineural invasion. Perineural invasion was defined by the presence of tumour cells within the nerve sheath. For nerves < 0.1 mm, a minimum of 33% involvement of the circumference was required, while indisputable presence of tumour cells within the nerve sheath was necessary for larger nerves.
The absolute number of TBs was evaluated, focusing on the periphery of the tumour. TB was considered to be present when isolated cells or small clusters of 2 or 3 tumour cells were found, separated from the main tumour mass. The intensity of TB was scored as follows: 0 (absent), 1 (1 or 2 tumour buds), 2 (3 to 9 tumour buds), and 3 (≥ 10 tumour buds) (Fig. 1). These values were determined following the initial evaluation of a smaller series of cases, aiming to identify the optimal thresholds for stratifying positive cases into 3 groups. Subsequently, categories 0/1 and 2/3 were combined for further analysis, treating budding as a dichotomous variable (present/absent).
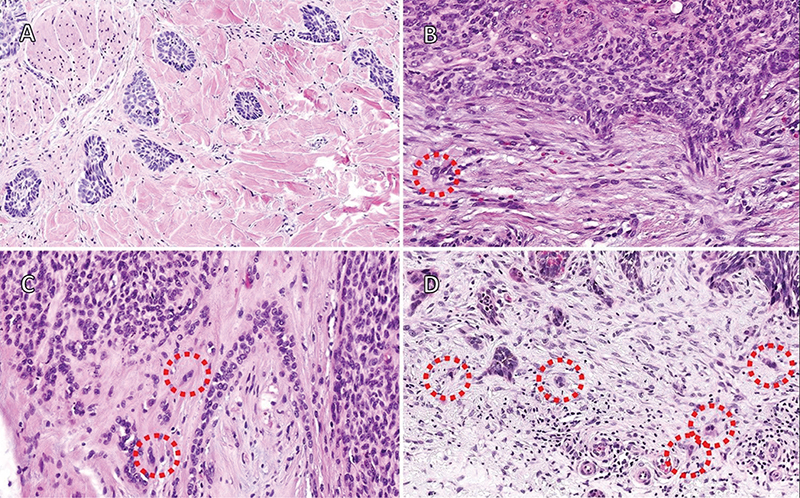
Fig. 1. Tumor budding (TB) quantification. Quantification of TB was performed after examining all sections, with the criterion that only groups of 3 or fewer tumour cells were considered for scoring. (A) Score 0 corresponds to cases with a total absence of tumour budding, as seen in this example of infiltrating BCC with a micronodular growth pattern. (B) Cases with score 1 presented only 1 or 2 tumour buds (red circle), sometimes with an equivocal appearance, making it challenging to distinguish them from reactive endothelial cells or fibroblasts. (C) Score 2 was assigned when the total number of tumour buds ranged between 3 and 10. This field contains many groups of more than 3 cells and 2 clear tumour buds (red circles) close to an area of perineural invasion. (D) Score 3 was assigned when the total number of tumour buds (red circles) exceeded 10.
The infiltrative pattern (infiltrating, sclerosing, or micronodular) was recorded, and in cases of tumour heterogeneity, the predominant pattern was selected. Tumour diameter in millimetres (mm) was measured in multiple sections, and the macroscopic tumour diameter was also consulted and compared to ensure accuracy. The greatest tumour depth in mm was evaluated either from the level of the adjacent epithelial surface when feasible or from the nearest point to it, not considering the exophytic areas, which have little implication on the depth of invasion. Ulceration was recorded as “present” when complete epidermal disappearance or tumour erosion reaching the tumour stroma occurred; otherwise, it was noted as “absent”. The inflammatory infiltrate (both intratumoral and peritumoral) and squamatisation (presence of squamous cells at the centre of tumour lobules with or without keratin whorls) were evaluated using a scale: 0 (absent), 1 (mild), 2 (moderate), and 3 (intense). However, the inflammatory response to ulceration was considered a secondary phenomenon and was not included in the evaluation. Additionally, the presence or absence of perineural invasion was documented.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows, Version 26.0 (IBM Corp, Armonk, NY, USA). The χ2 test was used to compare proportions of categoric variables and the Mann–Whitney test or Kruskal–Wallis ANOVA test to compare means of parametric or ordinal variables; correlation between continuous or ordinal variables was assessed with Pearson and Spearman methods. Multiple regression or logistic regression were used for multivariable analysis. Statistical significance was set at p < 0.05 (two-tailed).
RESULTS
Some degree of TB was observed in 186 of the 271 BCCs examined. However, in 64 cases, only 1 or 2 TB foci were identified upon meticulous section examination (score 1). The small number of tumour buds in this subgroup led to interobserver disagreement in some cases, due to the similarity between certain tumour buds and activated fibroblasts or endothelial cells (Fig. 1B). These instances were considered equivocal but were recorded to enable differentiation from cases with zero budding, which had been considered relevant in previous studies (18). In contrast, in 122 cases there was complete agreement among observers regarding the presence of TB. Among these cases, 81 exhibited between 3 and 9 TB foci (score 2), while 41 tumours displayed more than 10 TB foci (score 3).
According to WHO (19) subtype classification, 179 cases were infiltrative (66.1%), 16 sclerosing (5.9%), and 76 micronodular (28.0%); ulceration was observed in 114 cases (42.1%), and perineural invasion in 26 (9.6%).
The mean diameter of lesions was 7.11 mm (standard deviation [SD] 4.17; median 6.30; range 0.8 to 27.0 mm); the mean depth 2.26mm (SD 1.73; median 1.80; range 0.4 to 20.0 mm); the mean intensity of inflammation was 1.29 (median 1.00, SD 1.07). The mean score of squamatisation was 0.73 (median 1.00, SD 0.82).
The main results of our statistical analysis are displayed in Tables I and II. The intensity of TB was greater in tumours with perineural invasion and in ulcerated tumours (Table I).
The intensity of TB was correlated with depth and diameter of lesions (Table I). The median depths were 1.60 mm for grade 0, 1.55 mm for grade 1, 2.10 mm for grade 2, and 2.50 mm for grade 3; Kruskal–Wallis p < 0.001; groups 0,1 and 2,3 were homogeneous according to Dunn’s test. The median diameters by grades of TB were 5.30 mm for grade 0, 6.1 mm for grade 1, 7.3 mm for grade 2, and 7.5 mm for grade 3; Kruskal–Wallis p = 0.006; groups 2 and 3 were homogeneous according to Dunn’s test. Grades 0/1 and 2/3 of TB were combined for further analysis of TB as a dichotomous variable (present/absent). TB was more frequently present in infiltrative BCC (52.5%) and sclerosing BCC (81.3%) than in micronodular BCC (19.7%) (p < 0.001). The presence of TB was also significantly associated with perineural invasion and ulceration (Table I).
There was a correlation between diameter and depth of lesions, but not with grade of inflammation. There was no association between diameter of lesions and presence of perineural invasion, but ulcerated lesions were larger than non-ulcerated lesions (Table I). As regards depth, both ulceration and perineural invasion were associated with greater mean thickness of lesions (Table I). The degree of inflammation decreased with increasing depth and varied across histologic subtypes, being greater for infiltrative BCC (Table II); there were no differences according to the presence/absence of ulceration or perineural invasion.
No association was found between histologic subtypes and diameter, depth, and perineural invasion, but ulceration was more frequently present in infiltrative BCC (Table II).
There was a correlation between the degree of squamatisation and depth of lesions as well as grade and presence of TB, but not with diameter of lesions and degree of inflammation; no differences were found regarding ulceration or perineural invasion (Table I).
There was no significant association between ulceration and perineural invasion.
Multivariable analysis with depth as dependent variable (multiple regression, stepwise) yielded diameter (standardized coefficient Beta 0.228, p < 0.001), perineural invasion (Beta 0.153, p = 0.007), inflammation (Beta –0.176, p = 0.002), and presence of TB (Beta 0.151, p = 0.009) as independent predictive variables (R 0.431, p < 0.001).
Multivariable analysis with perineural invasion as dependent variable (logistic regression, forward step) yielded a model with depth (OR 1.287, 95% confidence interval [CI] 1.054–1.573, p = 0.014) and presence of TB (OR 2.686, 95% CI 1.095–6.589, p = 0.031) as independent predictive variables (goodness of fit χ2 7.478, p = 0.486; AUCROC = 0.718, p < 0.001).
DISCUSSION
The increasing life expectancy and the cumulative effects of sun damage are associated with a rising incidence of BCCs and, in parallel, an increase in the number of aggressive and difficult-to-treat tumours, a trend likely to persist. BCCs sharing similar clinicopathological characteristics may have a completely different outcome, highlighting an urgent need to explore novel methods for effectively stratifying the risk of individual cases. The vast majority of aggressive BCCs exhibit histologically an infiltrative growth pattern, which has been subtyped as infiltrating, sclerosing, or micronodular, according to the WHO (19). However, distinguishing between these 3 subtypes has a very limited reproducibility and offers minimal clinical relevance. In contrast, TB emerges as a promising histopathological biomarker, potentially aiding in the identification of tumours with higher invasive potential.
In our series, TB was found to correlate with an increased risk of perineural invasion and greater tumour depth, the most relevant microscopical features of local aggressiveness. These results were confirmed by a multivariate analysis where TB emerged as an independent factor associated with perineural invasion and increased tumour depth, alongside depth for the former and diameter and perineural invasion for the latter.
The presence of TB, regarded as a manifestation of p-EMT, has previously been linked to local and distant aggressiveness in both cutaneous and extracutaneous neoplasms (4–6, 8–17). Nevertheless, its investigation within the context of BCCs has remained unexplored until the present study. The significance of EMT in BCC oncogenesis is underscored by recent findings from a comprehensive study utilizing single-cell RNA analysis in a large series of infiltrating BCCs, revealing the existence of 2 distinct malignant basal subtypes. Notably, subtype 2, which favours EMT, is linked to increased invasion and recurrence, in contrast to subtype 1, which promotes differentiation (3). Therefore, TB, which can be considered as a surrogate marker of p-EMT, would be an indirect indicator of higher recurrence risk.
Additionally, detection and classification of BCC using computer vision and neural networks represent a promising field of research (20). Previous investigations have shown that TB evaluation can be substantially improved with the implementation of deep learning algorithms (21). This holds great potential for facilitating routine evaluation and significantly enhancing the accuracy and consistency of TB detection and quantification.
Interestingly, histopathological subtyping failed to show any significance for these variables, thus providing further justification to avoid including this subclassification in the diagnostic process. It is noteworthy that certain cases exhibited mixed infiltrative patterns. The presence of small foci corresponding to patterns other than the predominant one may have impacted some of the results. Nevertheless, if any of these patterns possessed a higher potential for local invasion, it would have been evident in cases where that particular pattern was predominant.
Of note, the increased presence of inflammatory infiltrate in the infiltrative subtype can play a role in the progression of this type of tumours (3). Besides, micronodular BCCs exhibited very low levels of tumour budding, potentially aligning with the concept of a “poorly differentiated cluster” (6). Other of our results could be guessed intuitively, such as the relation between tumour depth and tumour diameter, ulceration, perineural invasion, and inflammation. While ulceration may not inherently indicate high-risk behaviour, it is commonly linked to non-superficial tumours, prompting us to explore its significance (22). Regarding squamatisation, it did not correlate significantly with the main parameters of aggressiveness in the multivariate analysis, suggesting it may primarily represent a histopathological peculiarity.
Based on our results, TB emerges as a valuable histological biomarker for infiltrative BCCs, surpassing the differentiation of subtypes in its effectiveness. Assessing TB using a threshold of ≥ 3 foci and ≤ 3 cells across all tumour sections proves to be a straightforward and potentially reproducible method that could offer valuable insights for therapeutic decision-making, particularly in cases with partial resections or surgical margins very close to the tumour. Future studies are anticipated to validate our findings, potentially establish alternative cutoff values, and incorporate poorly differentiated clusters into the analysis.
ACKNOWLEDGEMENTS
Ignasi Gich, from the CIBER Epidemiología y Salud Pública (CIBERESP), Department of Clinical Epidemiology and Public Health, HSCSP, Sant Pau Biomedical Research Institute (IIB Sant Pau), Barcelona, Spain, contributed to the statistical analysis.
IRB approval status: With approval from the Institutional Ethics Committee (Approval ID: 2023/97-APA-HUGC).
REFERENCES
- Peris K, Fargnoli MC, Garbe C, Kaufmann R, Bastholt L, Seguin NB, et al. Diagnosis and treatment of basal cell carcinoma: European consensus-based interdisciplinary guidelines. Eur J Cancer, 2019; 118: 10–34.
- Fernández-Figueras MT, Malvehi J, Tschandl P, Rutten A, Rongioletti F, Requena L, et al. Position paper on a simplified histopathological classification of basal cell carcinoma: results of the European Consensus Project. J Eur Acad Dermatol Venereol 2022; 36: 351–359.
- Huang L, Wang X, Pei S, Li X, Dong L, Bian X, et al. Single-cell profiling reveals sustained immune infiltration, surveillance, and tumor heterogeneity in infiltrative basal cell carcinoma. J Invest Dermatol 2023; 143: 2283–2294.
- Pal A, Barrett TF, Paolini R, Parikh A, Puram SV. Partial EMT in head and neck cancer biology: a spectrum instead of a switch. Oncogene 2021; 40: 5049–5065.
- Zlobec I, Berger MD, Lugli A. Tumour budding and its clinical implications in gastrointestinal cancers. Br J Cancer 2020; 123: 700–708.
- Feakins RM, Loughrey MB, Silver A. Buds, clusters, and transitions in 21st century colorectal carcinoma: revolution or reinvention? J Pathol 2023; 261: 121–124.
- Imai T. The growth of human carcinoma: a morphological analysis. Fukuoka Igaku Zasshi 1954; 45: 72–102.
- Stögbauer F, Beck S, Ourailidis I, Hess J, Poremba C, Lauterbach M, et al. Tumour budding-based grading as independent prognostic biomarker in HPV-positive and HPV-negative head and neck cancer. Br J Cancer 2023; 128: 2295–2306.
- Eckstein M, Matek C, Wagner P, Erber R, Büttner-Herold M, Wild PJ, et al. Proposal for a novel histological scoring system as a potential grading approach for muscle-invasive urothelial bladder cancer correlating with disease aggressiveness and patient outcomes. Eur Urol Oncol 2024; 7: 128–138.
- Ailia MJ, Thakur N, Chong Y, Yim K. Tumor budding in gynecologic cancer as a marker for poor survival: a systematic review and meta-analysis of the perspectives of epithelial-mesenchymal transition. Cancers (Basel) 2022; 14: 1431.
- Zombori-Tóth N, Hegedűs F, Almási S, Sejben A, Tiszlavicz L, Furák J, et al. Proposal of a grading system for squamous cell carcinoma of the lung: the prognostic importance of tumour budding, single cell invasion, and nuclear diameter. Virchows Arch 2023; 483: 393–404.
- Jiang D, Xu S, Zhang C, Hu C, Li L, Zhang M, et al. Association between the expression levels of ADAMTS16 and BMP2 and tumor budding in hepatocellular carcinoma. Oncol Lett 2023; 25: 256.
- Karayannopoulou G, Panteris E, Kanitakis J. Tumour budding is an independent predictive factor of cutaneous squamous-cell carcinoma aggressiveness. Anticancer Res 2020; 40: 2695–2699.
- Fujimoto M, Yamamoto Y, Takai T, Fujimoto N, Ogawa K, Yoshikawa T, et al. Tumor budding is an objective high-risk factor associated with metastasis and poor clinical prognosis in cutaneous squamous cell carcinoma sized <4 cm. Am J Surg Pathol 2019; 43: 975–983.
- Gonzalez-Guerrero M, Martínez-Camblor P, Vivanco B, Fernández-Vega I, Munguía-Calzada P, Gonzalez-Gutierrez MP, et al. The adverse prognostic effect of tumor budding on the evolution of cutaneous head and neck squamous cell carcinoma. J Am Acad Dermatol 2017; 76: 1139–1145.
- Farah M, Milton DR, Gross ND, Nagarajan P, Gu J, Curry JL, et al. Histopathologic features predictive of metastasis and survival in 230 patients with cutaneous squamous cell carcinoma of the head and neck and non-head and neck locations: a single-center retrospective study. J Eur Acad Dermatol Venereol 2022; 36: 1246–1255.
- Lino-Silva LS, Zepeda-Najar C, Caro-Sánchez CH, Herrera-Gómez Á, Salcedo-Hernández RA. Prognostic significance of tumor budding in melanoma. Melanoma Res 2022; 32: 318–323.
- Zlobec I, Bächli M, Galuppini F, Berger MD, Dawson HE, Nagtegaal ID, et al. Refining the ITBCC tumor budding scoring system with a “zero-budding” category in colorectal cancer. Virchows Arch 2021; 479: 1085–1090
- Messina J, Epstein E, Kossard S, McKenzie C, Patel R, Patterson J, et al. Basal cell carcinoma. In: Elder DE, Massi D, Scolyer RA, Willemze R, editors. WHO classification of skin tumours. Lyon: International Agency for Research on Cancer; 2018: p. 26–34.
- Pai RK, Banerjee I, Shivji S, Jain S, Hartman D, Buchanan DD, et al. Quantitative pathologic analysis of digitized images of colorectal carcinoma improves prediction of recurrence-free survival. Gastroenterology 2022; 163: 1531–1546.e8.
- Urech M, Kyrgidis A, Argenziano G, Reggiani C, Moscarella E, Longo C, et al. Dermoscopic ulceration is a predictor of basal cell carcinoma response to imiquimod: a retrospective study. Acta Derm Venereol 2017; 97: 117–119.