ORIGINAL REPORT
Clinical Characteristics and Therapeutic Management of Atopic Dermatitis in Elderly Patients Compared with Young Adult Patients: A Prospective Multicentre Study
Clément POLLYN-MILLOT1, François MACCARI2,3, Jean-Luc PERROT4, Ziad REGUIAI5, Claire BOULARD6, Pierre-André BECHEREL7, Claire POREAUX8, Laure MERY-BOSSARD9, Domitille BEAULIEU10, Diane POURCHOT11, Anne-Claire FOUGEROUSSE2, Edouard BEGON12, Anne-Laure LIEGEON13, Charlotte FITE14, Ines ZAARA14, Dominique LONS-DANIC14, Beatrice WALLS14, Caroline JACOBZONE15, Charlotte LEPELLEY16, Daphné DENIS16 and Guillaume CHABY1; for the OMCCI Group
1Department of Dermatology, University Hospital of Amiens, Amiens, 2Department of Dermatology, Military Teaching Hospital Bégin, Saint Mandé, 3Dermatology, Private Practice, Saint-Maur-des-Fossés, 4Department of Dermatology, University Hospital of Saint-Etienne, Saint-Etienne, 5Department of Dermatology, Polyclinique Courlancy-Bezannes, Reims, 6Department of Dermatology, Jacques Monod Hospital, Le Havre, 7Department of Dermatology, Antony Private Hospital, Antony, 8Department of Dermatology, Stanislas-Nancy Medical Centre, Nancy, 9Department of Dermatology, François Quesnay Hospital, Mantes La Jolie, 10Department of Dermatology, Poissy/Saint-Germain-en-Laye Hospital, Saint-Germain-en-Laye, 11Department of Dermatology, Victor Dupouy Hospital, Argenteuil, 12Department of Dermatology, Pontoise Hospital, Pontoise, 13Department of Dermatology, Metz-Thionville Regional Hospital Centre, Thionville, 14Department of Dermatology, Paris Saint Joseph, Hospital Paris, Paris, 15Department of Dermatology, Groupe Hospitalier Bretagne Sud-Hôpital du Scorff, Lorient, and 16Dermatology, Private Practice, Vannes, France
Atopic dermatitis (AD) is the most common chronic inflammatory dermatitis in developed countries, and has a major impact on those affected. Little is known about AD in elderly patients. This prospective multicentre observational study described the clinical characteristics and burden of AD in elderly subjects ≥ 65 years, as well as the therapeutic options chosen for this population in routine care, and compared findings with those in young adults with AD < 30 years. Cohort data from adult patients with moderate-to-severe AD enrolled in a French national prospective registry (December 2020 to May 2023) were analysed. Patients ≥ 65 years made up 12.5% of the total adult cohort and presented less head-and-neck and extremity involvement, and were less affected by generalized forms than young adult patients. Elderly patients predominantly had late-onset AD and had similar disease severity to younger adults. Although the overall impact of AD appeared to be lower in elderly patients and treatment was initially less used in this age group, the substantial impact on sleep and psychiatric comorbidities was similar in older and younger adult patients. Better understanding of AD in elderly patients and the establishment of age-specific treatment guidelines may help dermatologists manage the disease in older people.
SIGNIFICANCE
Although atopic dermatitis is common and has a major impact on those affected, little is known about atopic dermatitis in the elderly. Our study sought to address this using information from a French national health registry. We found that, although the overall impact of atopic dermatitis seems to be lower in elderly patients, the impact of atopic dermatitis on sleep, and mood and depression is similar in elderly and young adult patients, with many being severely affected. Better understanding of atopic dermatitis in people aged ≥ 65 years and the establishment of age-specific treatment guidelines may help dermatologists manage atopic dermatitis in older people.
Key words: aged; dermatitis; atopic; observational study; prevalence; registries; therapeutics.
Citation: Acta Derm Venereol 2024; 104: adv40420. DOI: https://doi.org/10.2340/actadv.v104.40420.
Copyright: © 2024 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Mar 4, 2024; Accepted after revision: Aug 23, 2024; Published: Sep 9, 2024
Corr: Guillaume Chaby, Department of Dermatology, Centre Hospitalier Universitaire Amiens-Picardie, FR-80054 Amiens Cedex 1, France. E-mail: chaby.guillaume@chu-amiens.fr
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
Atopic dermatitis (AD) is the most common form of chronic inflammatory dermatitis in developed countries, and its prevalence continues to grow across most continents in the 21st century (1). AD has major repercussions on the quality of life (QoL) of patients and their families, with negative effects in terms of social, academic, and professional life (2–4).
Although AD has always been a particularly significant concern in the paediatric population, it can also persist or appear in adulthood, affecting individuals of all ages, without exception. Its clinical manifestations tend to change with age. Three distinct age-associated phases – infantile, childhood, and adult – define the usual distribution of AD lesions (5). It is likely that the prevalence of AD in elderly patients is underestimated (6, 7), and AD characteristics and impact in this specific population are still insufficiently described. Indeed, while there is comprehensive information available on adult AD, there remains an under-representation of elderly subjects ≥ 65 years in clinical studies, both observational and interventional (8). We do not know whether AD in the elderly can be considered to be a fourth distinct form of this inflammatory dermatitis. In particular, the question is raised as to whether AD in the elderly presents any clinical similarities or distinctions in comparison with the classic adult form, and more specifically with the form observed in young adults < 30 years, which is inextricably linked to AD in adolescents. Additionally, the burden of AD on elderly patients has not been accurately described to date. Lastly, treatment guidelines do not specifically address the issue of AD management for elderly patients, and the consequences for treatment options chosen in routine care have not yet been established (9).
The aim of our prospective multicentre observational study was to describe the clinical characteristics and the burden of AD in elderly subjects ≥ 65 years, as well as the therapeutic options chosen for this population in routine care, and to look for distinctive features relative to young adult patients with AD.
MATERIALS AND METHODS
This was a prospective multicentre observational study based on cohort data from the Observatoire des Maladies Cutanées Chroniques Inflammatoires (French Observatory of Chronic Inflammatory Skin Diseases) (OMCCI). The OMCCI is a French national prospective registry, launched in December 2020, with contributions from 22 dermatology investigation centres, comprising hospitals (n = 16) and private centres (n = 6). Adult patients with moderate-to-severe AD, who were seen in consultation and gave informed consent to participate in the study, were enrolled following the initiation or modification of systemic conventional, biologic, or Janus kinase inhibitor (JAKi) treatment. Patients taking part in interventional therapeutic trials were excluded. The study was approved by the French National Data Protection Authority and the national Institutional Review Board (CNRIPH 20.05.27.35855 / ID 8375).
At the inclusion, for all subjects ≥ 65 years and < 30 years satisfying entry criteria in the registry, the baseline severity of AD was assessed by a dermatologist, using the Eczema Area and Severity Index (EASI) (an EASI score < 7 indicates mild AD, an EASI score ≥ 7 and < 21 indicates moderate AD, and an EASI score ≥ 21 indicates severe AD). The anatomical regions affected were classified into 1 of 2 categories: segmental (1 or more areas affected from the limbs, torso, head and neck, extremities, and external genitalia) or generalized (involvement of the torso, all 4 limbs, head, and neck). In addition, all treatments prescribed for AD over the previous 6 months before the enrolment were recorded, as well as any changes that occurred at the time of the enrolment visit. The number of hospitalizations related to a severe worsening of atopic dermatitis in the previous 6 months was also recorded.
Patients reported the impact of AD on several aspects of their lives at the inclusion visit. QoL was assessed using the Dermatology Life Quality Index (DLQI) (range: 0–30; ≤ 5 indicates no or little impact, 6–10 indicates a moderate impact, and ≥ 11 indicates significant impact on QoL). The impact of AD on sleep and on low mood or depression over the previous 7 days was stratified into 4 levels of severity according to patients’ responses: none, mild (“rarely”), moderate (“sometimes”), or severe (“very often or all the time”). The overall discomfort caused by AD was stratified into 4 levels of severity: none, mild (“a little uncomfortable”), moderate (“quite uncomfortable”), and severe (“very uncomfortable”).
These physician- and patient-reported results were included in a conventional electronic case report form (eCRF) and were subject to standard control procedures.
Statistical analysis
Data management and statistical analyses were performed using SAS® software (version 9.4; SAS Institute, Cary, NC, USA). Qualitative variables were expressed as numbers (n) and percentages (%). Quantitative variables were expressed as means with standard deviations (SD) or medians with interquartile ranges (IQR). The results obtained in patients aged ≥ 65 years were compared with those obtained in young adults aged 18 to 29 years. The definition of young adults was determined by the age range distribution of the French population, according to the National Institute of Statistics and Economic Studies (INSEE) (10). The test χ2 or Fisher’s exact test (when the number of participants was less than 5) was used for qualitative variables, and the Mann–Whitney test was used for quantitative variables. The type I error (α) was set at 0.05 for all tests.
RESULTS
Between December 2020 and May 2023, 587 adult patients with AD were seen in consultation and provided data for analysis. The overall mean (SD) age of the population was 39.9 (17.5) years, with a median (IQR) age of 36 (25–50) years.
A total of 73 patients were aged ≥ 65 years (12.5%), with a mean (SD) age of 74.1 (7.3) years (extremes 65–91 years). The group aged < 30 years consisted of 207 patients (35.5%), with a mean (SD) age of 23.4 (3.2) years. The age distribution of the patients with AD is shown in Fig. 1. The sex ratio was 0.83 males to 1 female in those aged ≥ 65 years, and 0.97 males to 1 female in those aged < 30 years. This difference in gender distribution was not significant.
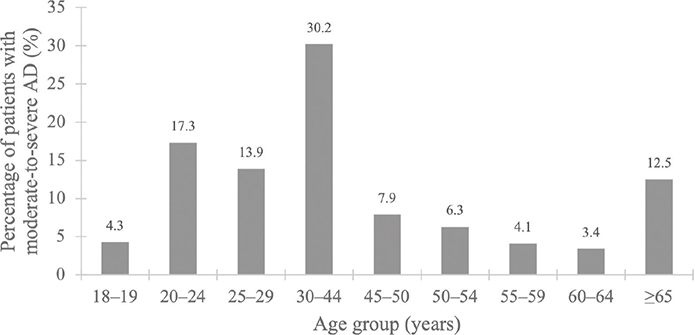
Fig. 1. Distribution of patients with moderate-to-severe AD, by age range. AD: atopic dermatitis in the OMCCI register.
Clinical characteristics
The mean (SD) age at AD diagnosis was 40.8 (31.6) years in patients aged ≥ 65 years and 6.2 (7.6) years in those aged < 30 years. In patients aged ≥ 65 years, 63.6% were diagnosed with AD in adulthood (≥ 18 years), vs 11.7% of those aged < 30 years. In elderly patients, over half were diagnosed with AD after the age of 51 years and 34.8% received this diagnosis after the age of 65 years. For patients aged < 30 years, the disease was diagnosed in childhood (< 12 years) in 76.0% of cases, vs 31.8% for those aged ≥ 65 years (p < 0.001, Table I).
The mean (SD) EASI score was 17.8 (10.3) among patients aged ≥ 65 years and 20.1 (13.4) among patients aged < 30 years (p = 0.343). The distribution of severity scores was also broadly similar in the 2 groups (p = 0.130). A severe EASI score was observed in 31.5% and 42.6% of patients aged ≥ 65 years and < 30 years, respectively. A significantly higher proportion of patients aged ≥ 65 years had been hospitalized at least once over the past 6 months (12.3%) compared with patients aged < 30 years (4.9%) (p = 0.032).
The anatomical sites affected differed significantly between age groups (p = 0.043). Patients aged ≥ 65 years had less involvement of the extremities (55.6% vs 68.9%, p = 0.059) and significantly less head and neck involvement (62.3% vs 85.6%, p < 0.001) than patients aged < 30 years. The limbs and torso were found to be frequently involved in both age groups. Generalized involvement was reported in a smaller proportion of elderly patients than young adults (9.6% vs 20.5%), while segmental involvement was more frequent (87.7% vs 78.6%) (p = 0.043).
Burden of disease
The impact of AD on QoL, as assessed by the DLQI, was lower in patients aged ≥ 65 years than in patients aged < 30 years (mean [SD] scores of 8.2 [4.8] vs 12.3 [6.7]; p < 0.001). More patients aged ≥ 65 years (35.6%) reported a mild impact on QoL (DLQI ≤ 5) than patients aged < 30 years (18.0%). A severe score (DLQI ≥ 11) was reported by 27.4% of patients aged ≥ 65 years vs 57.8% of patients aged < 30 years. The extent of impact on QoL was significantly affected by age group (p < 0.001). To go into further detail, patients aged ≥ 65 years reported significantly fewer complexes (p = 0.003), and less impact on their choice of clothing (p < 0.031), on their leisure activities (p = 0.005), and on their relationships with friends and family (p = 0.019) than patients aged < 30 years (Table II). Regarding the functional signs of AD (itching, pain, burning sensations), there were no differences in the frequency of symptom intensity, with 71.2% and 76.0% of patients aged ≥ 65 years and < 30 years, respectively, reporting that they experience these symptoms “a great deal” or “a lot” (p = 0.576) (Table II).
The impact of AD on sleep was comparable between the 2 age groups, reported as being severe in 53.5% of patients aged ≥ 65 years and in 65.9% of patients aged < 30 years, and non-existent in only 12.7% and 9.3% of patients, respectively.
Reports of low mood and depression were not significantly different between the 2 age groups. Mood was severely impacted by AD in approximately 30% of both patients aged ≥ 65 years and < 30 years, and fewer than 20% of patients in both age groups reported their mood never being impacted.
The overall impact of AD reported by patients aged ≥ 65 years was less severe than that reported by patients aged < 30 years. Specifically, 17.1% of patients aged ≥ 65 years reported a mild general impact and 32.9% of this group reported a severe general impact, as compared with 13.3% and 46.8% of patients aged < 30 years, respectively. Only 2.9% of patients aged ≥ 65 years and 1.5% of patients aged < 30 years reported no general repercussions, with no significant effect of age group on overall disease impact.
Treatment
Over the course of the 6 months prior to the enrolment visit, the overall distribution of the different therapeutic classes administered to patients did not reveal any significant differences between the 2 age groups (p = 0.063) (Fig. 2). Among patients aged ≥ 65 years, 57.5% were treated with topical treatments only, 6.8% with systemic immunomodulators (3 patients with methotrexate and 2 with cyclosporine), and 6.8% with biologics (5 patients with dupilumab). Only 1 patient aged ≥ 65 years was treated with JAKi (baricitinib). The number of patients aged < 30 years who were treated with topical treatments only was significantly lower (43.5%, p = 0.039).
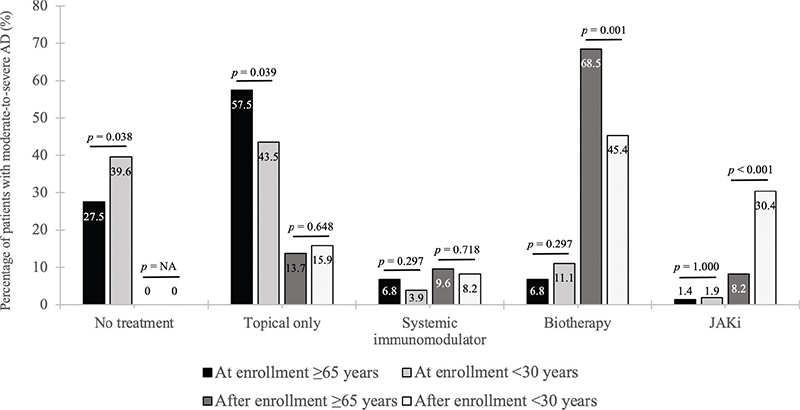
Fig. 2. Treatment options at enrolment and after the enrolment visit for patients with moderate-to-severe AD, by age group. AD: atopic dermatitis; JAKi: Janus kinase inhibitor; NA: not applicable.
After the enrolment visit, 86.4% of patients aged ≥ 65 years and 84.1% of those aged < 30 years received systemic treatment. Among patients aged ≥ 65 years, 9.6% were treated with a systemic immunomodulator (5 with methotrexate and 2 with alitretinoin) vs 8.2% of patients aged < 30 years (14 with ciclosporin, 1 with methotrexate, and 1 with alitretinoin) (p = 0.718), and 68.5% were treated with a biologic (36 with dupilumab and 14 with tralokinumab) vs 45.4% (75 with dupilumab and 19 with tralokinumab), respectively (p = 0.001). The increase in the rate of biologic prescriptions after enrolment among both those aged ≥ 65 and those < 30 years was significant (p < 0.001 for each age group). The proportion of patients who were prescribed JAKi treatment was lowest in those aged ≥ 65 years (8.2% [5 with baricitinib and 1 with upadacitinib] vs 30.4% in those aged < 30 years [41 with baricitinib, 16 with upadacitinib, and 6 with abrocitinib]; p < 0.001) (Fig. 2).
DISCUSSION
In our cohort of adult patients with moderate-to-severe AD, 12.5% were elderly. In France, the “objectifs peau” (Skin Objectives) study found a relatively similar estimate, based on self-assessment questionnaires (9.1% of the ≥ 15-year-old population diagnosed with AD) (11). In studies from other European countries, this proportion varied according to the methodology used and the defined age range for the study population; however, it generally remained similar to that of our series (7, 12–14). For example, in Finland, national registries show that patients aged ≥ 65 years represent 15.3% of all patients aged over 14 years with AD (7). Meanwhile, in Italy, they account for almost 11.4% of the adult population with severe AD (14).
We observed 2 main modes of AD onset in elderly patients, as defined by the age at diagnosis: paediatric-onset AD, which tended to persist or recur more or less in late adulthood, and late-onset AD, which appeared in adulthood. Our study revealed that AD in the elderly is predominantly a late-onset condition, with approximately two-thirds of cases occurring in adulthood and one-third occurring from the age of 65 years or later. However, age at diagnosis is not necessarily indicative of the age at onset of the disease. In some cases, diagnosis may be delayed after onset, especially when the diagnosis is not easy. This may explain the relatively high proportion of AD diagnoses in the elderly.
In our series, there was no difference in the overall clinical severity of AD between the elderly patients and the young adults, although the intensity of AD in elderly patients resulted in more hospital admissions. However, several phenotypic differences between the 2 age groups were identified in our study. It is well known that patients with AD display different clinical presentations depending on age. The clinical course of infantile, childhood, and adult atopic dermatitis can be speculated to be influenced by the maturation of the adaptative immune system, changes in the sex hormone milieu, age-related barrier dysfunctions in the skin and gut, and environmental stimuli (15). In elderly AD, immunosenescence in combination with a defective aged epidermal barrier dysregulation of innate immune cells, disturbance of sweat function, and skewing of adaptive immunity to a Th2 response, might play an important role in the pathogenesis and the clinical presentation (16). Some studies have reported a tendency for involvement of the cervico-cephalic extremity and the hands in adults AD, as compared with paediatric AD (17, 18). This description does not match our findings in those aged ≥ 65 years. However, this difference may be related to the fact that these previous studies included a very small number of patients ≥ 65 years with atopic dermatitis.
Our data also pointed to a notable frequency of genital involvement in adults, although this appeared to diminish with age. To date, there have been very few reports that provide information on the prevalence of genital eczema in DA. The relatively low interest shown by dermatologists in genital eczema in AD may contribute to underestimating this condition in real-life practice studies (19). A US study reported that the involvement of genital lesions in AD was 10.3% in adults, close to the percentage we observed (20). This topography should not be overlooked when examining patients of any age, as it certainly contributes to the burden of adulthood AD.
A third of the elderly patients with AD in our study reported a severe impact on their QoL. Nevertheless, the overall DLQI score was lower among patients aged ≥ 65 years compared with young adults, although this result was probably underestimated as the item on occupational consequences did not concern much of our older population. It can nevertheless be observed that the aspects of quality of life most affected during the course of atopic dermatitis in both young and older adults are symptoms and emotional impact, with less impact on social functioning. However, social functioning, such as the complex induced by AD, is more affected in young adults. The results of the International Study of Life with Atopic Eczema (ISOLATE) demonstrated several years ago that more than one-third of patients report that AD has eroded their self-confidence and could lead to social isolation among the youngest (21). Symptoms such as itching, burning sensations, and pain, which are major factors in the burden and activity of AD, are experienced on an equally severe level by both elderly patients and young adults. These symptoms are frequently correlated with substantial sleep disturbances, which affected more than half of our patients, both young and elderly, supporting the notion that AD is a risk factor for sleep disturbances for patients of any age (22). Similarly, the strong association between AD and psychiatric comorbidities was found in the same proportions of young and older adults alike, nearly a third of whom found AD to have a major impact on mood.
At the time of enrolment, 85.0% of patients aged ≥ 65 years were not receiving any treatment or were receiving only topical treatments, whereas almost 9 out of 10 patients started systemic treatment after the enrolment visit. This observation raises the question of the probable inadequacy of therapeutic management for adults aged ≥ 65 years with AD, given the severity of their condition. A recent French publication showed that a quarter of dermatologists demonstrated therapeutic inertia with their patients with moderate-to-severe AD (23). This therapeutic inertia affects all adults with atopic dermatitis, regardless of their age, as our study seems to confirm. Besides, some patients were prescribed topical treatment only after the enrolment visit because they were waiting for a pre-therapeutic assessment or because they were no longer eligible for systemic treatment. At the end of the enrolment visit, age did not appear to be an issue for the initiation of any systemic treatment, with just 2 exceptions: ciclosporin, which was not used by any elderly patient, undoubtedly due to the higher risk of adverse effects in these patients; and JAKi, for which the number of prescriptions remained relatively marginal in comparison with those for methotrexate or biologics in patients aged ≥ 65 years. However, it should be pointed out that part of the enrolment period coincided with the publication of a warning concerning the prescription of JAKi, particularly in patients aged ≥ 65 years (24). Dupilumab was by far the preferred treatment out of the innovative therapies used by elderly patients in our study. This preference can be correlated with the reassuring data from a real-life study showing good efficacy and no serious adverse events in elderly patients treated with dupilumab (14).
Interestingly, our cohort includes patients from both hospital and private care centres across France, providing real-life data that are representative of the population with AD treated in routine dermatological care in France. Moreover, the diagnosis of AD was made by a dermatologist, guaranteeing optimal diagnostic confidence, unlike in very large-scale studies, which are generally based on data from self-assessment questionnaires or national registries, typically completed by non-dermatologists. The diagnosis of late-onset or elderly AD can be difficult for a non-dermatologist, as physicians have to rely on ruling out several differential diagnoses, without any specific clinical or paraclinical diagnostic criteria (25, 26).
Limitations
However, our study results may be limited by possible memory bias, which may interfere with the progressive history of AD. This issue has been raised in several studies, which have found a higher age of AD onset with self-reporting methods than with methods using a physician assessment at follow-up (27). This is likely to be due to the changing nature of AD, as there may be no disease activity at the time of the clinical examination, but it may be present during interval periods. The second limitation concerns generalizability, as the findings do not necessarily apply to patients with a mild form of the disease who are treated by general practitioners.
Conclusion
Our results highlight that clinical characteristics and therapeutic management of AD in elderly patients are different from those of younger patients. Although the overall impact of the disease seems to be less severe, the impact of AD on sleep and psychiatric comorbidities is similar in older and younger adult patients, with severe impact occurring in the same proportions of each group. Our study shows that dermatologists are adapting and transposing current general recommendations for AD treatment into real life, preferring biologics to ciclosporin or JAKi. Age should not be an obstacle for the prescription of effective systemic treatments if required. More studies in this specific field may help to better describe AD in elderly patients and potentially adapt guidelines in this particular population
ACKNOWLEDGEMENTS
IRB approval status: The study was approved by the Commission nationale de l’informatique et des libertés (National Commission on Data Processing and Liberty; French National Data Protection Authority) and the national Institutional Review Board (Comité de Protection des Personnes; ref CNRIPH 20.05.27.35855 / ID 8375).
The authors thank all the members of the recruiting network who contributed to including patients in the registry.
REFERENCES
- Bylund S, Von Kobyletzki LB, Svalstedt M, Svensson A. Prevalence and incidence of atopic dermatitis: a systematic review. Acta Derm Venereol 2020; 100: 320–329. https://doi.org/10.2340/00015555-3510
- de Bruin-Weller M, Gadkari A, Auziere S, Simpson EL, Puig L, Barbarot S, et al. The patient-reported disease burden in adults with atopic dermatitis: a cross-sectional study in Europe and Canada. J Eur Acad Dermatol Venereol 2020; 34: 1026–1036. https://doi.org/10.1111/jdv.16003
- Drucker AM, Wang AR, Li WQ, Sevetson E, Block JK, Qureshi AA. The burden of atopic dermatitis: summary of a report for the National Eczema Association. J Invest Dermatol 2017; 137: 26–30. https://doi.org/10.1016/j.jid.2016.07.012
- Laughter MR, Maymone MBC, Mashayekhi S, Arents BWM, Karimkhani C, Langan SM, et al. The global burden of atopic dermatitis: lessons from the Global Burden of Disease Study 1990–2017. Br J Dermatol 2021; 184: 304–309. https://doi.org/10.1111/bjd.19580
- Bieber T. Atopic dermatitis. N Engl J Med 2008; 358: 1483–1494. https://doi.org/10.1056/NEJMra074081
- Bieber T, D’Erme AM, Akdis CA, Traidl-Hoffmann C, Lauener R, Schäppi G, et al. Clinical phenotypes and endophenotypes of atopic dermatitis: where are we, and where should we go? J Allergy Clin Immunol 2017; 139: S58–64. https://doi.org/10.1016/j.jaci.2017.01.008
- Salava A, Rieppo R, Lauerma A, Salo V. Age-dependent distribution of atopic dermatitis in primary care: a nationwide population-based study from Finland. Acta Derm Venereol 2022; 102: adv00738. https://doi.org/10.2340/actadv.v102.2287
- Lam M, Zhu JW, Maqbool T, Adam G, Tadrous M, Rochon P, et al. Inclusion of older adults in randomized clinical trials for systemic medications for atopic dermatitis: a systematic review. JAMA Dermatol 2020; 156: 1240–1245. https://doi.org/10.1001/jamadermatol.2020.2940
- Wollenber A, Christen-Zäch S, Taieb A, Paul C, Thyssen JP, de Bruin-Weller M, et al. ETFAD/EADV Eczema task force 2020 position paper on diagnosis and treatment of atopic dermatitis in adults and children. J Eur Acad Dermatol Venereol 2020; 34: 2717–2744. https://doi.org/10.1111/jdv.16892
- Institut National de la Statistique et des Etudes économiques. Jeunes de 18 à 29 ans – France, portrait social. Edition 2019 [in French] [cited 2023 Dec 27]. Available from: https://www.insee.fr/fr/statistiques/4238379?sommaire=4238781
- Richard MA, Corgibet F, Beylot-Barry M, Barbaud A, Bodemer C, Chaussade V, et al. Sex- and age-adjusted prevalence estimates of five chronic inflammatory skin diseases in France: results of the « OBJECTIFS PEAU » study. J Eur Acad Dermatol Venereol 2018; 32: 1967–1971. https://doi.org/10.1111/jdv.14959
- Theodosiou G, Montgomery S, Metsini A, Dalgard FJ, Svensson Å, Kobyletzki LB. Burden of atopic dermatitis in Swedish adults: a population-based study. Acta Derm Venereol 2019; 99: 964–970. https://doi.org/10.2340/00015555-3257
- Zink A, Arents B, Fink-Wagner A, Seitz I, Mensing U, Wettemann N, et al. Out-of-pocket costs for individuals with atopic eczema: a cross-sectional study in nine European countries. Acta Derm Venerol 2019; 99: 263–267. https://doi.org/10.2340/00015555-3102
- Patruno C, Napolitano M, Argenziano G, Peris K, Ortoncelli M, Girolomoni G, et al. Dupilumab therapy of atopic dermatitis of the elderly: a multicentre, real-life study. J Eur Acad Dermatol Venereol 2021; 35: 958–964. https://doi.org/10.1111/jdv.17094
- Weidinger S, Natalija N. Atopic dermatitis. Lancet 2016; 387: 1109–1122. https://doi.org/10.1016/S0140-6736(15)00149-X
- Williamson S, Merritt J, De Benedetto A. Atopic dermatitis in the elderly: a review of clinical and pathophysiology hallmarks. Br J Dermatol 2020; 182: 47–54. https://doi.org/10.1111/bjd.18652
- Chan AR, Sandhu VK, Drucker AM, Fleming P, Lynde CW. Adult-onset atopic dermatitis: presentations and progress. J Cutan Med Surg 2020; 24: 267–272. https://doi.org/10.1177/1203475420911896
- Silverberg JI, Vakharia PP, Chopra R, Sacotte R, Patel N, Immaneni S, et al. Phenotypical differences of child- and adult-onset atopic dermatitis. J Allergy Clin Immunol Pract 2018; 6: 1306–1312. https://doi.org/10.1016/j.jaip.2017.10.005
- Woo YR, Han Y, Lee JH, Lee YB, Kim JE, Kim M, et al. Real-world prevalence and burden of genital eczema in atopic dermatitis: a multicenter questionnaire-based study. J Dermatol 2021; 48: 625–632. https://doi.org/10.1111/1346-8138.15817
- Silverberg JI, Margolis D, Boguniewicz M, Fonacier L, Grayson M, Ong P, et al. Distribution of atopic dermatitis lesions in United States adults. J Eur Acad Dermatol Venereol 2019; 33: 1341–1348. https://doi.org/10.1111/jdv.15574
- Zuberbier T, Orlow SJ, Paller AS, Taïeb A, Allen G, Hernanz-Hermosa JM, et al. Patient perspectives on the management of atopic dermatitis. J Allergy Clin Immunol 2006;118: 226–232. https://doi.org/10.1016/j.jaci.2006.02.031
- Silverberg JI, Garg NK, Paller AS, Fishbein AB, Zee PC. Sleep disturbances in adults with eczema are associated with impaired overall health: a US population-based study. J Invest Dermatol 2015; 135: 56–66. https://doi.org/10.1038/jid.2014.325
- Halioua B, Corgibet F, Taieb C, Thenie C, Veniard C, Noel M, et al. Therapeutic inertia in the management of patients with inadequately controlled atopic dermatitis. J Eur Acad Dermatol Venereol 2022; 36: e817–e818. https://doi.org/10.1111/jdv.18303
- Food and Drug Administration. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. 09.01.2021 FDA Drug Safety Communication [cited 2023 Dec 27]. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/fda-requires-warnings-about-increased-risk-serious-heart-related-events-cancer-blood-clots-and-death
- Fishbein AB, Silverberg JI, Wilson EJ, Ong PY. Update on atopic dermatitis: diagnosis, severity assessment, and treatment selection. J Allergy Clin Immunol Pract 2020; 8: 91–101. https://doi.org/10.1016/j.jaip.2019.06.044
- Williams HC, Burney PG, Hay RJ, Archer CB, Shipley MJ, Hunter JJ, et al. The U.K. working party’s diagnostic criteria for atopic dermatitis: derivation of a minimum set of discriminators for atopic dermatitis. Br J Dermatol 1994;131: 383–396. https://doi.org/10.1111/j.1365-2133.1994.tb08530.x
- Kim JP, Chao LX, Simpson EL, Silverberg JI. Persistence of atopic dermatitis (AD): a systematic review and meta-analysis J Am Acad Dermatol 2016; 75: 681–687. https://doi.org/10.1016/j.jaad.2016.05.028