SHORT COMMUNICATION
Bullous Pemphigoid that Developed During Nemolizumab Treatment for Atopic Dermatitis: Two Case Reports
Michie KATSUTA1*, Ryoichi KAMIDE2, Yozo ISHIUJI1, Yoshimasa NOBEYAMA1 and Akihiko ASAHINA1
1Department of Dermatology, The Jikei University School of Medicine, 25-8 Nishi-Shimbashi 3-chome, Minato-ku, Tokyo 105-8461, Japan, and 2Hihunoclinic Ningyocho, Tokyo, Japan.
*E-mail: mkatta1219@gmail.com
Citation: Acta Derm Venereol 2024; 104: adv40634. DOI https://doi.org/10.2340/actadv.v104.40634.
Copyright: © 2024 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Apr 23, 2024; Accepted after revision: Aug 6, 2024; Published: Sep 2, 2024
Competing interests and funding: NK and YN received research funding and honorarium as a speaker from Maruho. YI received an honorarium as a speaker from Maruho, Sanofi, AbbVie, Pfizer, Eli Lilly, and Otsuka. The other authors declare they have no conflicts of interest.
INTRODUCTION
Nemolizumab is a monoclonal antibody targeting interleukin (IL)-31 receptor α. The drug is useful for the treatment of moderate to severe atopic dermatitis (AD). In particular, the agent is known to be highly effective for itch in AD (1, 2). On the other hand, there are many cases of cutaneous adverse events (3). Here, we report 2 cases of bullous pemphigoid (BP) diagnosed during nemolizumab treatment for AD and discuss the pathogenesis of the development of BP.
CASE REPORTS
Case 1. A 70-year-old man was referred to us with a 2-year history of persistent itchy eczema associated with AD (Fig. 1a). The patient had had diabetes and dry skin for at least 30 years, which had been treated with an oral dipeptidyl peptidase 4 (DPP-4) inhibitor for 2 years and a topical corticosteroid, respectively. We prescribed nemolizumab (60 mg/4 weeks). Four weeks after the initiation of the administration, both eruption and itch were significantly improved. In the day after the second administration, the patient suffered from non-itchy erythema accompanied by bulla on the feet. At that time, we suspected the onset of BP, but blood test showed a normal range of serum antibody against BP180-NC16a domain (< 9 U/mL). Despite both treatment of topical corticosteroid and cessation of the initiate of the DPP-4 inhibitor, bullous lesions were increasing. One week later, the patient showed an elevated serum level of antibody against BP180-NC16a domain (489 U/mL) and BP disease area index (BPDAI) 43/26/0 (Fig. 1b–d). Blood examination showed higher levels of eosinophil count, thymus and activation chemokine (TARC), and IgE compared with the baseline before nemolizumab initiation (Fig. 1). Histopathological examination and direct immunofluorescence (DIF) were compatible with BP (Fig. 1e, f). Prior to the onset of BP, he had neither experienced COVID-19 nor received a COVID-19 vaccine before the onset. Therefore, we made the diagnosis of BP due to nemolizumab, and then discontinued the administration of nemolizumab. To treat BP, we administered oral prednisolone 1.0 mg/kg/day and intravenous immunoglobulin (600 mg/day for 3 days). Three months after the initiation of treatment, the eruption was significantly improved, and blood test showed normal range of anti-BP 180 antibody.
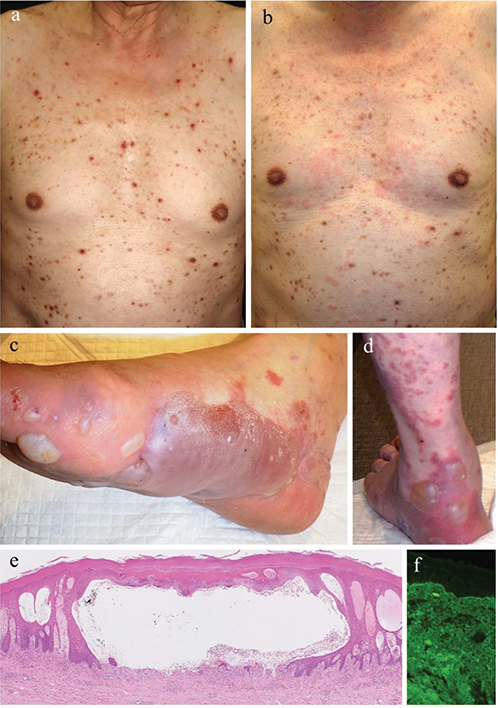
Fig. 1. (a) Clinical findings before nemolizumab treatment (Eczema Area and Severity Index 36.8, Investigator’s Global Assessment 3, and itch Visual Analog Scale 90, eosinophil count 998/μL, TARC 1,658 pg/mL, IgE 356 IU/mL). (b–d) Clinical findings at the time of blister appearance (itch Visual Analog Scale 10, eosinophil count 1,558/μL, TARC 6338 pg/mL, IgE 794 IU/mL). (e) Histopathological findings (loupe image, haematoxylin, and eosin staining). A subepidermal blister and eosinophilic infiltration in the upper dermis is evident. (f) Direct immunofluorescence (×100). Linear IgG deposition is shown at the dermal–epidermal junction.
Case 2. A-49-year-old man was referred to us with a 2-year history of eczema with severe itch associated with AD. Physical examination showed oedematous erythema with scratch marks on the trunk and extremities (Fig. 2a). We treated the patient with nemolizumab (60 mg/4 weeks). Four weeks after initiation, both itch and erythema on the trunk and extremities tended to improve, but a small bulla appeared on the left thigh. Seven days after the second course, many small bullae without itch appeared on the extremities (Fig. 2b–e). Blood examination showed an elevated level of anti-BP 180 antibody of 67.6 U/mL, and higher levels of eosinophil count, TARC, and IgE compared with the baseline before nemolizumab initiation (Fig. 2). Histopathological and DIF findings were compatible with BP (Fig. 2 f, g). Therefore, we diagnosed the patient as having BP due to nemolizumab (BPDAI 14/15/0). Despite treatment with a topical corticosteroid, erythema accompanied by bullae was exacerbated, and we then discontinued the administration of nemolizumab. To treat the BP, we administered prednisolone at 0.5 mg/kg/day. After the initiation of treatment, the eruption improved, and blood test showed a normal range of anti-BP180 antibody.
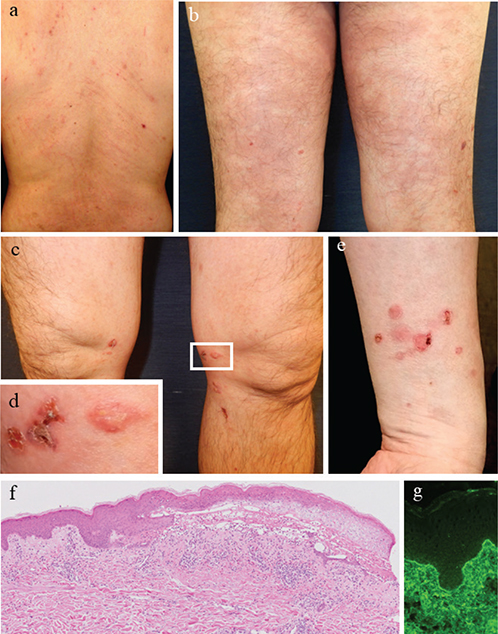
Fig. 2. (a) Clinical findings before nemolizumab treatment (Eczema Area and Severity Index 11, Investigator’s Global Assessment 3, and itch Visual Analog Scale 85, eosinophil 835/μL, TARC 1,134 pg/mL, IgE 3977 IU/mL). (b–e) Clinical findings at the time of blister appearance (eosinophil 1938/μL, TARC 4,463 pg/mL, IgE 5726 IU/mL. (f) Histopathological findings (loupe image, haematoxylin, and eosin staining). A subepidermal blister and eosinophilic infiltration in the upper dermis were evident. (g) Direct immunofluorescence (×100). Linear IgG deposition is evident at the dermal–epidermal junction.
DISCUSSION
This case report describes the onset of BP during the course of AD treated with nemolizumab. Previously, Masuyuki et al. (4) reported a case of BP after the initiation of nemolizumab and hypothesized that clinical BP became apparent from occult BP due to nemolizumab. How-ever, we observed that anti-BP 180 antibody changed from a negative to a positive result during nemolizumab treatment in Case 1. Also, BP appeared at the early age of 49 years in Case 2. Given that BP commonly occurs at in older age, the specificity in our case is distinct. Indeed, Marazza et al. (5) reported that individuals in their forties rarely suffer from BP with 1/50-fold compared with those in their seventies. These facts demonstrate that de novo BP occurs following nemolizumab administration rather than being a manifestation of occult BP.
We did not confirm IgG deposition at the dermal–epidermal junction before nemolizumab administration. Therefore, the causative factors of BP should be carefully discussed in both cases. In Case 1, DPP-4 inhibitor was being administered at the onset of bullous lesions. DPP-4 inhibitor-associated BP is sometimes caused by an autoantibody other than antibodies against BP180-NC16a domain (6). In Case 2, the onset age of BP is distinctively lower. Based on these instances, we might perform DIF for both cases to detect latent BP before the initiation of nemolizumab.
Both AD and BP share a common clinical symptom of itch associated with IL-31 (7). Nemolizumab activates IL-13 function in neurogenic inflammation through inhibiting IL-31 function (8). Consequently, nemolizumab can lead to a Th2-predominant condition and exacerbation of eczema in AD, although it remarkably inhibits itch. TARC has been reported to increase in BP (9) as well as AD treated with nemolizumab (1). In our cases, the levels of TARC were significantly elevated along with eosinophil counts. Based on this, we regard an obligatory Th2-predominant condition due to nemolizumab as part of the mechanism of BP onset.
The present report described 2 cases where BP developed during treatment with nemolizumab, along with elevated eosinophils and TARC. Dermatologists should be aware of the possibility of the development of BP in AD patients treated with nemolizumab.
ACKNOWLEDGEMENTS
Yukiko Tateno, MD and Hizuru Tomita, MD contributed to data collection at the Jikei University School of Medicine. The authors would like to thank David Price (www.english-for-services.com) for English language editing.
IRB approval status: The Ethics Committee of the Jikei University School of Medicine approved the study protocol (approval number, #25-210).
REFERENCES
- Kabashima K, Matsumura T, Komazaki H, Kawashima M, Nemolizumab JPSG. Trial of nemolizumab and topical agents for atopic dermatitis with pruritus. N Engl J Med 2020; 383: 141–150. https://doi.org/10.1056/NEJMoa1917006
- Watanabe Y, Ishiuji Y, Ogawa-Tominaga M, Katsuta M, Asahina A. Real-world clinical efficacy of nemolizumab in Japanese patients with atopic dermatitis. Itch 2023; 33: 691–692. https://doi.org/10.1097/itx.0000000000000071
- Kabashima K, Matsumura T, Komazaki H, Kawashima M, Nemolizumab JPaSG. Nemolizumab plus topical agents in patients with atopic dermatitis (AD) and moderate-to-severe pruritus provide improvement in pruritus and signs of AD for up to 68 weeks: results from two phase III, long-term studies. Br J Dermatol 2022; 186: 642–651. https://doi.org/10.1111/bjd.20873
- Masuyuki R, Sato E, Imafuku S. A case of bullous pemphigoid following administration of anti-IL-31 receptor A antibody. J Dermatol 2024 Mar 20 [Online ahead of print]. https://doi.org/10.1111/1346-8138.17171
- Marazza G, Pham HC, Schärer L, Pedrazzetti PP, Hunziker T, Trüeb RM, et al. Incidence of bullous pemphigoid and pemphigus in Switzerland: a 2-year prospective study. Br J Dermatol 2009; 161: 861–868. https://doi.org/10.1111/j.1365-2133.2009.09300.x
- Tasanen K, Varpuluoma O, Nishie W. Dipeptidyl peptidase-4 inhibitor-associated bullous pemphigoid. Front Immunol 2019; 10: 1238. https://doi.org/10.3389/fimmu.2019.01238
- Rüdrich U, Gehring M, Papakonstantinou E, Illerhaus A, Engmann J, Kapp A, et al. Eosinophils are a major source of interleukin-31 in bullous pemphigoid. Acta Derm Venereol 2018; 98: 766–771. https://doi.org/10.2340/00015555-2951
- Fassett MS, Braz JM, Castellanos CA, Salvatierra JJ, Sadeghi M, Yu X, et al. IL-31-dependent neurogenic inflammation restrains cutaneous type 2 immune response in allergic dermatitis. Sci Immunol 2023; 8: eabi6887. https://doi.org/10.1126/sciimmunol.abi6887
- Kakinuma T, Wakugawa M, Nakamura K, Hino H, Matsushima K, Tamaki K. High level of thymus and activation-regulated chemokine in blister fluid and sera of patients with bullous pemphigoid. Br J Dermatol 2003; 148: 203–210. https://doi.org/10.1046/j.1365-2133.2003.05066.x