ORIGINAL REPORT
10 Years of Mohs Micrographic Surgery in Denmark: Results from a Nationwide Cohort
Yuki M. F. ANDERSEN1, Katrine KARMISHOLT1, Trine HØGSBERG2, Mia-Louise NIELSEN1, Amanda BROSBØL1, Sascha STAVE1, Louise SCHØSLER2, Silje OMLAND1, Christine Aarenstrup DAUGAARD2 and Martin GLUD1
1Department of Dermatology and Venereology, Bispebjerg Hospital, Copenhagen, and 2Department of Dermatology and Venereology, Aarhus University Hospital, Aarhus, Denmark
Basal cell carcinoma is the most common skin malignancy and constitutes a burden for patients and society. Mohs micrographic surgery is a recommended treatment for high-risk basal cell carcinoma, but long-term outcomes of Mohs micrographic surgery in Denmark are unknown. This study aimed to estimate the 5-year recurrence rate of basal cell carcinoma following Mohs micrographic surgery, and to investigate patient and procedure characteristics since the introduction of the procedure in Denmark. The Danish Registry for Mohs Surgery was established and all Mohs micrographic surgery cases nationwide from January 2012 to December 2022 were included. A total of 1,774 patients were included in the cohort, and 2,203 high-risk basal cell carcinomas were treated using Mohs micrographic surgery techniques. The overall 5-year recurrence of basal cell carcinoma following Mohs micrographic surgery was 3.8% (95% CI 2.8–5.0), 3.1% (95% CI 2.1–4.7) for primary basal cell carcinomas, and 5.3% (95% CI 3.6–7.8) for recurrent basal cell carcinomas. The primary basal cell carcinomas showed a tendency towards lower recurrence rates and better surgical outcomes than recurrent basal cell carcinomas, although not significantly. The recurrence rate estimates correspond to international levels, supporting Mohs micrographic surgery as a treatment option for high-risk basal cell carcinomas in Danish dermatological practice. The newly established patient registry serves as a cohort for future research in this field.
SIGNIFICANCE
Since Mohs micrographic surgery was introduced to treat basal cell carcinomas (BCCs) in Denmark a decade ago, almost no data regarding Mohs micrographic surgery outcomes have been available. By investigating data from a newly established patient registry, we found that the 5-year recurrence rates were approximately 3% and 5% for primary and recurrent basal cell carcinoma, respectively, following Mohs micrographic surgery. We also found that primary basal cell carcinomas projected better surgical outcomes than recurrent basal cell carcinomas. These novel real-world data underline the benefits of Mohs micrographic surgery and can contribute to clinical guidelines for basal cell carcinoma management. The newly established registry may also serve as a source for future studies.
Key words: basal cell carcinoma; epidemiology; Mohs micrographic surgery; recurrence rate; registry.
Citation: Acta Derm Venereol 2025; 105: adv41118. DOI: https://doi.org/10.2340/actadv.v105.41118.
Copyright: © 2025 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Jul 3, 2024. Accepted after revision: Dec 12, 2024. Published: Jan 10, 2025.
Corr: Yuki M. F. Andersen, Department of Dermatology and Venereology, Bispebjerg Hospital, Nielsine Nielsens Vej 4, DK-2400 Copenhagen, Denmark. E-mail: yuki.egeberg@gmail.com
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
Basal cell carcinoma (BCC) is the most common skin malignancy, with an estimated lifetime prevalence up to 30% in fair-skinned populations (1). Although rarely fatal, BCCs constitute a large socioeconomic burden and may affect patients’ quality of life significantly (2, 3). Well-established risk factors for BCC include sun exposure, fair skin colour, genetic predisposition, and immunosuppression (4, 5).
A range of therapeutic options are available for BCCs including curettage, surgery, cryo-surgery, photodynamic therapy, topical imiquimod, radiation therapy, and systemic hedgehog inhibitors. Treatment strategies usually depend on prognostic stratification of recurrence risk among other factors such as patient characteristics and preference (6, 7). A BCC is typically classified as being at high risk of recurrence if it is localized in the H-zone in the face, is larger than 2 cm, if it is a recurrent tumour, has ill-defined borders, perineural invasion, or has an aggressive histological subtype (8). Mohs micrographic surgery (MMS) is considered the gold standard for treatment of high-risk BCCs in the facial area, due to its high cure rate and tissue-sparing benefits (9–12).
A decade ago, MMS was introduced as a treatment option in Danish hospital settings, and it is currently performed in 2 university hospitals in Denmark. The knowledge of MMS outcomes in Denmark is limited, and long-term efficacy and safety of MMS in Danish hospitals are unknown (13). The aim of this study was to provide novel nationwide descriptions of patients undergoing MMS, and to estimate the 5-year recurrence rate following MMS in Denmark.
MATERIALS AND METHODS
The study was registered and approved by the Capital Region’s Research Inventory prior to study initiation (Videncenter for Dataanmeldelser, ref. P-2021-856). All legal and ethical requirements for the study were met.
Danish Registry for Mohs Surgery
The Danish Registry for Mohs Surgery is a nationwide patient cohort established in December 2022. The objective of the registry is to collect information regarding BCCs treated by MMS in Danish public hospitals, and to generate data for scientific research and quality assurance. The registry includes information on patient characteristics, BCC characteristics, MMS procedures, and postoperative follow-up visits. Two centres are currently performing MMS in Denmark, Bispebjerg University Hospital in Copenhagen and Aarhus University Hospital in Aarhus. At the time of establishment of the registry, no systematic data were available regarding MMS operations in Denmark. We therefore entered data based on retrospective chart reviews of virtually all MMS operations carried out in Denmark since the method was introduced in 2012 and until December 2022.
Patient inclusion and data entry
For the retrospective chart reviews, research assistants entered relevant information using search functions in the electronic patient charts. If any of the information was unavailable or unclear, fields were classified as “unknown” or “missing”. All data were collected using the web-based application Research Electronic Data Capture (REDCap, version 13.7.14; https://project-redcap.org/) which was available at both MMS centres.
Baseline data
Information regarding participants’ age, gender, risk factors for BCC, smoking status, and anticoagulant therapy was collected.
Basal cell carcinoma characteristics
Each histologically verified BCC that was treated with MMS was divided into primary BCC (pBCC) or recurrent BCC (rBCC). If information regarding previous treatment attempts was unclear, cases were classified as unknown. BCCs that were incompletely excised during the first attempt at treatment were also categorized as rBCC. The anatomical location of each BCC as well as histological type and previous treatment modalities for rBCC were also identified. Several patients had undergone more than 1 MMS procedure, leading to a larger number of BCC cases than participants. Each BCC was entered as a separate case, even if, for example, 2 tumours were treated during a single operative session.
Mohs micrographic surgeries
Information regarding each operation was entered into the registry, including hospital department, date of procedure, measurements of the tumour and defect, number of Mohs stages, histological findings, and closure technique. Furthermore, the use of preoperative and postoperative medication was registered.
Follow-ups
For each visit following every MMS operation, information on postoperative complications, patient satisfaction, and signs of BCC recurrence was collected. The Danish Dermatological Association recommends follow-up visits at 6 and 12 months postoperatively and additional follow-up visits depending on the patients’ risk profile and number of previous cases of skin malignancies, as well as actinic degeneration. For example, patients with multiple BCCs are typically offered yearly checkups. When patients are discharged from hospital care, they are usually recommended to continue screening and checkups through community dermatology clinics or general practitioners. If recurrence is suspected, patients are recommended to contact the MMS clinics. Recurrences following MMS were histologically verified by punch biopsy if there was any sign of tumour adjacent to the surgical scar. We also screened the Danish pathology database (Patobank), which contains all pathology results nationwide, to look for possible recurrences detected outside the hospital setting.
Statistical analysis
Summary statistics were created and presented as frequencies with percentages for categorical variables and means with standard deviations (SDs) or medians with interquartile ranges (IQR), as appropriate, for continuous variables. The surface area of tumours and defects was calculated using the formula Area = π × (major axis/2) × (minor axis/2). A 5-year recurrence rate with 95% confidence intervals (CI) was estimated using survival analysis and visualized with Kaplan–Meier plots. Patients were censored at the date of death if they died during the follow-up period, or at data lock on December 1, 2022. Cox regression analyses, including multivariate analyses, were used to estimate predictors for recurrence, presented as hazard ratios (HR). Covariates were selected a priori based on clinical relevance. P-values less than 0.05 were considered statistically significant. Analyses were performed using Python version 3.9.2 (https://www.python.org/).
RESULTS
Population characteristics
The study comprised a total of 1,774 adults who underwent MMS between January 1, 2012 and December 1, 2022. There were slightly more female patients (54.2%) than male patients. The median age at baseline was 69.6 (IQR 57.8–76.3) years (Table I). The majority of patients had undergone only 1 MMS procedure during the study period; however, 32% of the patients had received 2 or more MMS operations. Information regarding BCC risk factors, Fitzpatrick skin type, level of sun damage, and smoking status was missing in more than half of patients (information obtained from retrospective chart reviews), and therefore not reported in the current paper. Of the included patients, 20.9% (n = 370) received anticoagulant therapy at baseline, while 62.0% (n = 1,099) did not, and in 17.2% the information was missing. The most commonly used anticoagulants were acetyl salicylic acid (7.1%) and fish oil (4.6%).
| Total, n | 1,774 |
| Gender, n (%) | |
| Male | 812 (45.8) |
| Female | 962 (54.2) |
| Age (at first Mohs operation), median (IQR) | 69.6 (57.8–76.3) |
| Total number of MMS operations, n (%) | |
| 1 | 1,497 (68.0) |
| 2 | 187 (8.5) |
| 3 | 56 (2.5) |
| 4 | 21 (1.0) |
| 5+ | 13 (0.6) |
| Anticoagulant therapy, n (%) | |
| Yes | 370 (20.9) |
| No | 1,099 (62.0) |
| Missing | 305 (17.2) |
| Anticoagulant therapy, n (%) | |
| NSAID | 3 (0.2) |
| Warfarin | 44 (2.5) |
| Clopidogrel | 52 (2.9) |
| NOAC | 73 (4.1) |
| Heparin | 5 (0.3) |
| Acetyl salicylic acid | 127 (7.1) |
| Ticagrelor | 1 (0.1) |
| Fish oil and other supplements* | 82 (4.6) |
| IQR: interquartile range; MMS: Mohs micrographic surgery; NSAID: non-steroidal anti-inflammatory drug; NOAC: novel oral anti-coagulants. *Supplements with blood-thinning properties included products containing ginseng, garlic, and ginger. |
|
Basal cell carcinoma characteristics
A total of 2,203 BCCs treated with MMS were included, based on retrospective chart reviews. The number of BCCs treated with MMS increased substantially during the study period, from 41 in 2013 to 375 in 2022 (Fig. 1). A total of 1,316 and 694 of the cases were pBCCs and rBCCs, respectively, and 193 were categorized as unknown (Table II). There was an over-representation of BCCs in the nasal region, followed by the cheek, forehead, and temple regions (Fig. 2). The most common histological type was nodular (59.4% and 48.4% for pBCC and rBCC, respectively), followed by infiltrative (7.2% and 4.0%), and superficial (4.5% and 4.9%). Information on histological type was unavailable in 25.5% and 40.1% of pBCCs and rBCCs, respectively. Of the rBCCs, the most common previous treatment modality was curettage (50.1%), followed by photodynamic therapy (PDT) (13.5%), radiation therapy (11.2%), and cryotherapy (6.6%). The median (IQR) follow-up time for all included BCCs was 3.41 (1.54–5.53) years.
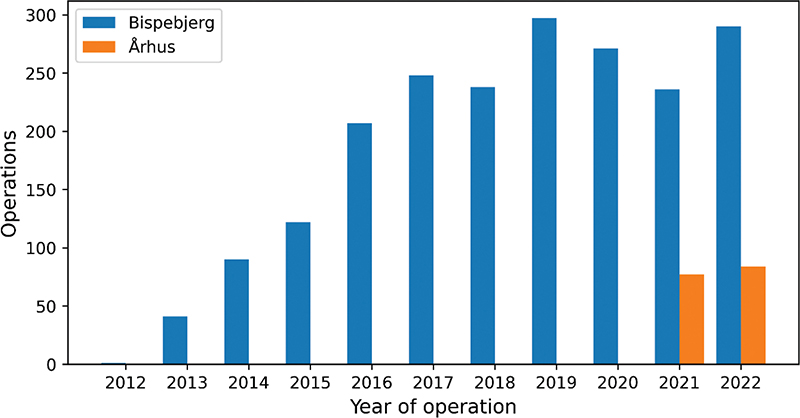
Fig. 1. Mohs surgery in Denmark over time. Number of basal cell carcinomas treated over the years since Mohs surgery was introduced in Denmark. The figure shows a rapid expansion during the first few years.
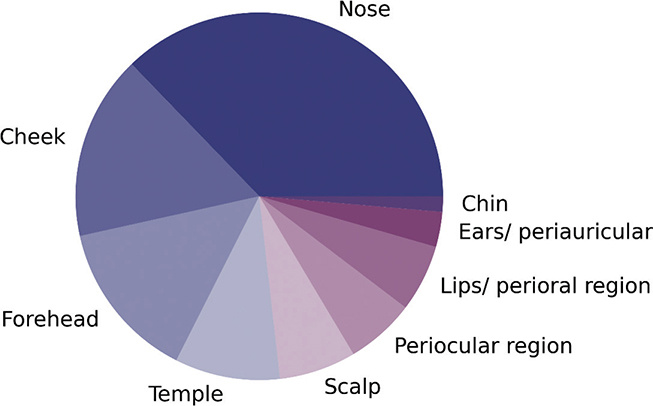
Fig. 2. Anatomical regions of basal cell carcinomas treated with Mohs surgery. A pie-chart visualizing the distribution of treated facial areas. The figure represents the total number of treated cases in the study period.
Mohs surgery characteristics
The median size of the studied BCCs was 66.0 (IQR 33.0–112.3) mm2 and 78.5 (IQR 42.8–144.9) mm2 for pBCC and rBCC, respectively (Table III). Defect sizes were similarly smaller for pBCCs (median 132.7 [IQR 78.5–251.3] mm2) than for rBCCs (median 188.5 [IQR 102.1–361.3] mm2). The difference between the measured area of defects and tumours was correspondingly smaller for pBCCs (median 62.0 [IQR 31.4–129.6] mm2) than rBCCs (median 91.1 [IQR 47.1–197.5] mm2). The largest tumour and defect diameters are presented in Table III. Approximately half of all pBCCs (46.5%) were clear after 1 stage, while only one-third of rBCCs (33.4%) were managed with 1 stage. Similarly, slightly more rBCCs (54.1%) required more complex closure techniques (techniques other than simple closure) than pBCCs (50.0%).
Recurrence rate estimations
The total number of registered BCC recurrences following MMS was 25 (1.9%) for pBCC and 28 (4.1%) for rBCCs. The overall 5-year recurrence rate for BCCs following MMS was 3.8% (95% CI 2.8–5.0), and 3.1% (95% CI 2.1–4.7) for pBCCs and 5.3% (95% CI 3.6–7.8) for rBCCs. When estimating the incidence of recurrence per person-years (only considering patients with at least 5 years of follow-up), the incidence rate was 0.92 (95% CI 0.84–1.00), 0.69 (95% CI 0.53–0.90), and 1.28 (95% CI 1.02–1.61) for overall, pBCCs, and rBCCs, respectively. Kaplan Meyer plots of recurrence rates for pBCCs and rBCCs are visualized in Fig. 4.
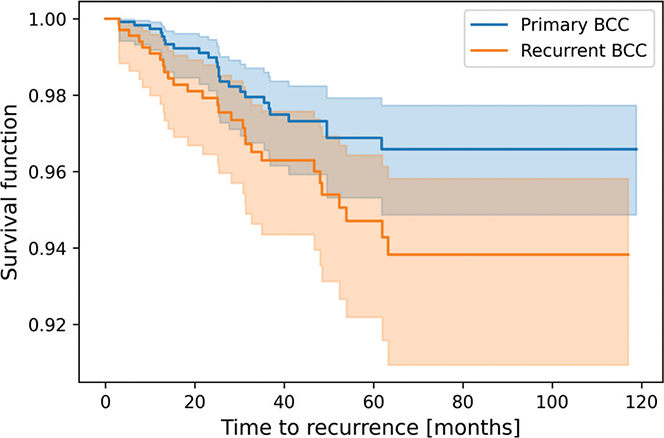
Fig. 3. Kaplan–Meier survival analysis of recurrence after Mohs surgery. Kaplan–Meier plots of recurrence after Mohs surgery in primary and recurrent basal cell carcinomas (BCC), respectively.
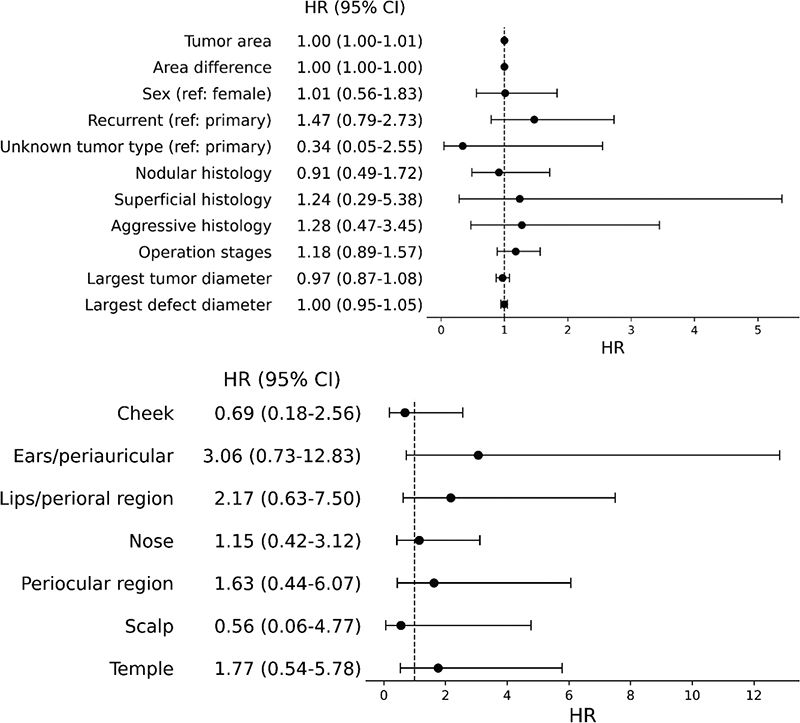
Fig. 4. Forest plots of predictors for recurrence of basal cell carcinomas after Mohs surgery. (A) Clinical predictors for basal cell carcinoma (BCC) recurrence after Mohs micrographic surgery (MMS). (B) Anatomical predictors for BCC recurrence after MMS. Variable “nodular” in (A) is coded as a binary variable (nodular vs non-nodular), as histological types other than nodular were few; (B) is presented using forehead as reference. CI: confidence interval; HR: hazard ratio.
Predictors for recurrence after MMS
In multivariate models, no significant predictor for tumour recurrence after MMS could be identified (Fig. 4). Included covariates were sex, histological type, recurrence status, defect-tumour area difference, and Mohs stages. Recurrent BCCs appeared to show a tendency for an increased risk of recurrence after MMS; however, this did not reach statistical significance (HR 1.42 [95% CI 0.77–2.62]). Similarly, we found no significant difference in recurrence rates according to different anatomical regions of the face.
DISCUSSION
The estimated 5-year recurrence rate following MMS in our study corresponds to previously reported estimates from other countries. In a Swedish Mohs clinic, the 5-year recurrence rate was 2.1% for pBCCs, 5.2% for rBCCs, and 3.3% overall (14), while a Dutch retrospective study found 5-year recurrence rates of 3.2% and 6.7% for pBCCs and rBCCs, respectively (15). Reports from the United States indicate recurrence rates as low as 1.0% for pBCCs (16) and 5.6% for rBCCs. An Australian study of more than 3,000 patients reported 5-year recurrence rates of 1.4% in pBCCs and 4% in rBCCs (17). In Spain, incidence rates of recurrence were 1.3 (95% CI 1.1–1.5) per 100 person-years for BCC (18), which is similar to our estimates. Variations in recurrence rate estimates may occur due partially to differences in patient characteristics, clinical practice, and guidelines. Narrower indication for MMS (e.g., including only aggressive subtypes or recurrent BCCs) may in general lead to higher recurrence rates, as these tumours are more difficult to treat regardless of treatment modality.
We observed that the area most commonly operated on was the nose, which is in line with the recommendations based on European consensus-based interdisciplinary guidelines for BCC (19). When comparing pBCCs with rBCCs, we observed an overall pattern of better outcomes of MMS in terms of procedural complexity (fewer stages, smaller defects, etc.). Furthermore, although not statistically significant, rBCCs tended to have a higher risk of recurrence after MMS. In previous studies, rBCCs, aggressive histological types, more than 4 stages of MMS, and skin defects larger than 4 cm in diameter have been reported as predictors for recurrence after MMS (15). These observations may support the recommendation of MMS as a first-line treatment for high-risk tumours, especially those adjacent to important facial structures. Clinical decision- making in treatment of BCC can be complex, and patients’ preference must be taken into consideration. In some elderly patients with a short life expectancy, watchful waiting or palliative strategies may be considered to spare them from hospital visits and pain. However, the overall life expectancy of the population is increasing (20), suggesting that radical treatment options may be preferable in more cases than previously anticipated, to avoid issues with numerous recurrences and treatment attempts.
Approximately half of the rBCCs in our study were previously treated with curettage, while 13.5% had received PDT and 11.2% radiation therapy prior to MMS. Management of high-risk BCCs can vary from physician to physician, based on factors such as preference, availability, and skill set. A survey of dermatologists in Sweden, Norway, and Denmark from 2015 revealed that the treatment of choice for BCCs on the face differed across the Scandinavian countries. While Swedish dermatologists were more likely to choose surgical strategies, Danish and Norwegian dermatologists preferred curettage, PDT, and/or radiotherapy more often (20). These differences may be based on variations in national guidelines and a tendency to pass on certain skills and practices. Despite the survey findings, it is our understanding that the clinical practice in the Danish dermatological community is changing, and that more patients are being referred for surgical treatments than previously. This newly established registry may facilitate the development of new interdisciplinary, evidence-based guidelines in the future.
The use of MMS has repeatedly been subject to discussion, as the procedure is considered costly and more time-consuming than standard excisions due to the staged excision in MMS. Studies have shown that MMS is not substantially more costly than standard excision (21, 22). Importantly, we found that the majority of cases were managed with 1 or 2 MMS stages, supporting the notion that the time factor should not play a crucial role in clinical decision-making. Although we do not have comparable data regarding the efficacy of standard excisions for high-risk BCCs in Denmark, a prospective randomized trial in the Netherlands found a 10-year cumulative probability of recurrence of 4.4% after MMS and 12.2% after standard excision (23). Furthermore, a systematic review and meta-analysis found that MMS is superior to standard excision in terms of reducing the defect size and lowering the recurrence rate for pBCC (OR 0.27, 95% CI 0.15–0.46) and rBCC (OR 0.26, 95% CI 0.09–0.78), respectively (24). Interestingly, incomplete excision (cases where the surgical margins contain neoplastic cells) occurs in 6.2–20.4% of BCCs that are treated with standard excisions, resulting in the need for re-treatments for many patients (25). Taking both the recurrence rates and cases of incomplete excision into consideration, patients and healthcare providers could potentially save costs on follow-up visits and re-treatment by choosing MMS when relevant. Future investigation into cost analysis in the Danish hospital setting will be helpful to further elucidate the matter.
Strengths and limitations
Several strengths and limitations apply to this study. Although the study was a retrospective review, the cohort was large, and without risk of participation bias, as all retrospective cases were included. Additionally, we had a long follow-up period of up to 10 years. Due to the retrospective entry of data, the quality of data may be partially compromised. There may be unmeasured recurrences, for example in patients who were lost to follow-up for different reasons. Furthermore, some patients may have visited a private dermatologist or surgeon to manage rBCCs. To compensate for this, we checked the pathology database for each patient to detect any possible missed recurrences. We observed that the information regarding the histological type of BCC was often missing due to a lack of documentation in the pathology description (Patobank). The background of this deficiency is not fully known; however, it is partly due to the fact that histological data collection has not been part of the standard procedure. A retrospective review of previous Mohs slides was not possible for the current study.
Conclusion
Since MMS was introduced in Denmark, this study is the first to report long-term outcomes of BCCs treated with MMS in Denmark. We found that the 5-year recurrence rate of BCC following MMS was 3.1% (95% CI 2.1–4.7) for pBCCs and 5.3% (95% CI 3.6–7.8) for rBCCs, similar to previously reported estimates internationally. This indicates that MMS may be recommended as a treatment option in Danish dermatological care and further development and expansion of MMS practice should be supported through collaboration between sectors and specialties. Importantly, the current paper also serves as a cohort description of the newly established Danish Registry for Mohs Surgery, which provides opportunities for future research in this field.
ACKNOWLEDGEMENTS
The authors would like to thank Dr Carsten Hamann for valuable comments on the manuscript.
IRB approval status: The study was approved by the Capital Region’s Research Inventory, Denmark.
REFERENCES
- Miller DL, Weinstock MA. Nonmelanoma skin cancer in the United States: incidence. J Am Acad Dermatol 1994; 30: 774–778. https://doi.org/10.1016/S0190-9622(08)81509-5
- Bentzen J, Kjellberg J, Thorgaard C, Engholm G, Phillip A, Storm HH. Costs of illness for melanoma and nonmelanoma skin cancer in Denmark. Eur J Cancer Prev 2013; 22: 569–576. https://doi.org/10.1097/CEJ.0b013e328360150c
- Housman TS, Feldman SR, Williford PM, Fleischer AB, Goldman ND, Acostamadiedo JM, et al. Skin cancer is among the most costly of all cancers to treat for the Medicare population. J Am Acad Dermatol 2003; 48: 425–429. https://doi.org/10.1067/mjd.2003.186
- Madan V, Hoban P, Strange RC, Fryer AA, Lear JT. Genetics and risk factors for basal cell carcinoma. Br J Dermatol 2006; 154: 5–7. https://doi.org/10.1111/j.1365-2133.2006.07229.x
- Preston DS, Stern RS. Nonmelanoma cancers of the skin. New Engl J Med 1992; 327: 1649–1662. https://doi.org/10.1056/NEJM199212033272307
- Basset-Seguin N, Herms F. Update on the management of basal cell carcinoma. Acta Derm Venereol 2020; 100: 284–290. https://doi.org/10.2340/00015555-3495
- Peris K, Concetta M, Garbe C, Kaufmann R, Bastholt L, Seguin N, European Dermatology Forum (EDF), the European Association of Dermato-Oncology (EADO) and the European Organization for Research and Treatment of Cancer (EORTC). European consensus-based interdisciplinary guideline for diagnosis and treatment of basal cell carcinoma—update 2023. Eur J Cancer 2023: 192: 113254 https://doi.org/10.1016/j.ejca.2023.113254
- Puig S, Berrocal A. Management of high-risk and advanced basal cell carcinoma. Clin Transl Oncol 2015; 17: 497–503. https://doi.org/10.1007/s12094-014-1272-9
- Shriner DL, McCoy DK, Goldberg DJ, Wagner J. Mohs micrographic surgery. J Am Acad Dermatol 1998; 39: 79–97. https://doi.org/10.1016/S0190-9622(98)70405-0
- Muller FM, Dawe RS, Moseley H, Fleming CJ. Randomized comparison of Mohs micrographic surgery and surgical excision for small nodular basal cell carcinoma: tissue-sparing outcome. Dermatol Surg 2009; 35: 1349–1354. https://doi.org/10.1111/j.1524-4725.2009.01240.x
- van Kester MS, Goeman JJ, Genders RE. Tissue-sparing properties of Mohs micrographic surgery for infiltrative basal cell carcinoma. J Am Acad Dermatol 2019; 80: 1700–1703. https://doi.org/10.1016/j.jaad.2019.01.057
- Smeets NWJ, Krekels GAM, Ostertag JU, Essers BAB, Dirksen CD, Nieman FHM, et al. Surgical excision vs Mohs’ micrographic surgery for basal-cell carcinoma of the face: randomised controlled trial. Lancet 2004; 364: 1766–1772. https://doi.org/10.1016/S0140-6736(04)17399-6
- Gniadecki R, Glud M, Mortensen K, Bang B, Biskup E, Omland SH, et al. Favourable results of Mohs micrographic surgery for basal cell carcinoma. Dan Med J 2015; 62: A5171.
- Paoli J, Daryoni S, Wennberg AM, Mölne L, Gillstedt M, Miocic M, et al. 5-year recurrence rates of Mohs micrographic surgery for aggressive and recurrent facial basal cell carcinoma. Acta Derm Venereol 2011; 91: 689–693. https://doi.org/10.2340/00015555-1134
- Smeets NWJ, Kuijpers DIM, Nelemans P, Ostertag JU, Verhaegh MEJM, Krekels GAM, et al. Mohs’ micrographic surgery for treatment of basal cell carcinoma of the face: results of a retrospective study and review of the literature. Br J Dermatol 2004; 151: 141–147. https://doi.org/10.1111/j.1365-2133.2004.06047.x
- Rowe DE, Carroll RH, Day CL. Long-term recurrence rates in previously untreated (primary) basal cell carcinoma: implications for patient follow-up. J Dermatol Surg Oncol 1989; 15: 315–328. https://doi.org/10.1111/j.1524-4725.1989.tb03166.x
- Leibovitch I, Huilgol SC, Selva D, Richards S, Paver R, Adelaide F, et al. Basal cell carcinoma treated with Mohs surgery in Australia II. Outcome at 5-year follow-up. J Am Acad Dermatol 2005; 53: 452–457. https://doi.org/10.1016/j.jaad.2005.04.087
- Tomás-Velázquez A, Sanmartin-Jiménez O, Garcés JR, Rodríguez-Prieto MA, Ruiz-Salas V, Eusebio-Murillo E De, et al. Risk factors and rate of recurrence after Mohs surgery in basal cell and squamous cell carcinomas: a nationwide prospective cohort (REGESMOHS, Spanish Registry of Mohs Surgery). Acta Derm Venereol 2021; 101: 544. https://doi.org/10.2340/actadv.v101.544
- WHO. World Health Statistics 2023. Monitoring health for the SDGs: Sustainable Development Goals. Geneva: WHO: 2023; 119.
- Helsing P, Gjersvik P, Tarstedt M. Treatment of basal cell carcinoma in Scandinavia: evidence or eminence based? Int J Dermatol 2015, 54: p. 1030–1033. https://doi.org/10.1111/ijd.12599
- Cook J, Zitelli JA. Mohs micrographic surgery: a cost analysis. J Am Acad Dermatol 1998; 39: 698–703. https://doi.org/10.1016/S0190-9622(98)70041-6
- Sebaratnam DF, Choy B, Lee M, Paver R, Fernández Peñas P. Direct cost-analysis of Mohs micrographic surgery and traditional excision for basal cell carcinoma at initial margin clearance. Dermatol Surg 2016; 42: 633–638. https://doi.org/10.1097/DSS.0000000000000756
- Van Loo E, Mosterd K, Krekels GAM, Roozeboom MH, Ostertag JU, Dirksen CD, et al. Clinical trial surgical excision versus Mohs’ micrographic surgery for basal cell carcinoma of the face: a randomised clinical trial with 10 year follow-up. Eur J Cancer 2014; 50: 3011–3020. https://doi.org/10.1016/j.ejca.2014.08.018
- Alsaif A, Hayre A, Karam M, Rahman S, Abdul Z, Matteucci P. Mohs micrographic surgery versus standard excision for basal cell carcinoma in the head and neck: systematic review and meta-analysis. Cureus 2021; 13: 19981. https://doi.org/10.7759/cureus.19981
- Nolan GS, Kiely AL, Totty JP, Wormald JCR, Wade RG, Arbyn M, et al. Incomplete surgical excision of keratinocyte skin cancers: a systematic review and meta-analysis Br J Dermatol 2021; 184: 1033–1044. https://doi.org/10.1111/bjd.19660