SHORT COMMUNICATION
Non-protuberant Lesions and Diagnostic Delays in Dermatofibrosarcoma Protuberans: A Single Institutional Retrospective Analysis
Jun AKOME1, Yosuke ISHITSUKA1*, Atsushi TANEMURA1, Tateki KUBO2 and Manabu FUJIMOTO1
1Department of Dermatology of Osaka University Graduate School of Medicine, Suita, Osaka, and 2Department of Plastic Surgery of Osaka University Graduate School of Medicine, Suita, Osaka, Japan. *E-mail: ishitsuka@derma.med.osaka-u.ac.jp
Citation: Acta Derm Venereol 2024; 104: adv41234. DOI https://doi.org/10.2340/actadv.v104.41234.
Copyright: © 2024 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Jul 24, 2024; Accepted after revision: Sep 5, 2024; Published: Sep 24, 2024
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
Dermatofibrosarcoma protuberans (DFSP) is a slow-growing, low-grade cutaneous mesenchymal neoplasm affecting young and middle-aged adults (20s to 50s). However, the fibrosarcomatous variant is associated with poorer clinical outcomes (1). While traditionally perceived as multinodular, DFSP can present with various skin changes similar to morphea, atrophoderma, or angioma (2), or even as a purely subcutaneous mass (3). Low disease incidence rates and innocuous-looking clinical variants can significantly delay definitive diagnosis (2). Therefore, evidence guiding clinicians to early and precise diagnosis is crucial, as complete surgical removal remains the primary treatment for the disease. Non-protuberant variants, especially the flat stage that reportedly precedes noticeable tumescence in over 40% of cases (2, 4), can lead even trained dermatologists to misdiagnose (4). This evidence challenges the term “protuberans”, and “dermatofibrosarcoma, often protuberant” has been proposed as an alternative (4). The enigmatic nature of DFSP’s clinical trajectory prompted us to conduct a retrospective analysis.
MATERIALS AND METHODS
A retrospective review of medical records was conducted with ethics committee approval (No. 23486), involving 30 patients with 31 lesions (1 case with a second lesion) histopathologically diagnosed with DFSP (uniform proliferation of CD34+/Factor XIII– spindle cells) at Osaka University Hospital from July 2011 to October 2023. Statistical analysis was performed using GraphPad Prism 10 software (GraphPad, La Jolla, CA), with p < 0.05 considered statistically significant.
RESULTS
The sex ratio was 1:1 (n = 15 for each), and the mean (SD) ages at initial disease perception and DFSP diagnosis were 34.19 (10.64) and 43.42 (10.63) years, respectively. No fibrosarcomatous DFSPs were identified. The mean (SD) largest lesional diameter was 35.68 (16.56) mm (n = 19). Disease locations were: head/neck (4 ), anterior torso (16), posterior torso (4), and limbs (7). Clinical appearances at initial disease perception included protuberant (papules [n = 3], nodules [n = 3], and tumors [n = 8]), subcutaneous (tumors [n=6] and induration [n = 5]), discolored (pigmented macules [n = 2]), and scar-like (n = 1) (Fig. 1). Prebiopsy clinical diagnoses, excluding general terms like “tumor” or “malignancy”, were made by dermatologists (n = 6), plastic surgeons (n = 3), or general practitioners (n = 2): DFSP (n = 4), hemangioma (n = 2), epidermal cyst/lipoma (n = 4), fibroma (n = 1), or scar (n = 2). Expectedly, the non-protuberant group had a higher mean (SD) interval in years between disease perception and histopathological diagnosis (11.21 [11.58]; n = 14) compared with the protuberant group (7.79 [7.08]; n = 14). A similar trend was observed in the mean (SD) largest lesional diameter (mm): 42.71 (21.91; n = 7) vs 34.09 (8.24; n = 11), suggesting that the non-protuberant group tend to have larger lesions at the time of definitive diagnosis. Welch’s t-test did not identify statistically significance either in the interval (p = 0.3550) (2) or the diameter (p = 0.3514) (4). However, a significant interaction between clinical appearances and age was identified through two-way analysis of variance (Fig. 2), indicating that initial clinical appearance strongly influenced the diagnostic interval (2, 4).
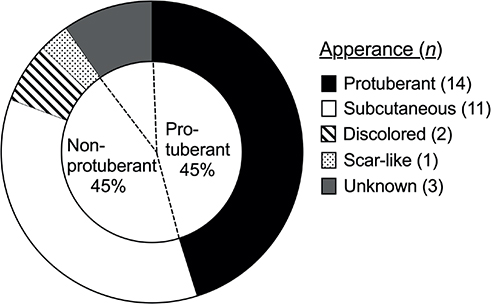
Fig. 1. Prevalence of non-protuberant subtypes in dermatofibroma protuberans. Patients classified according to clinical appearances.
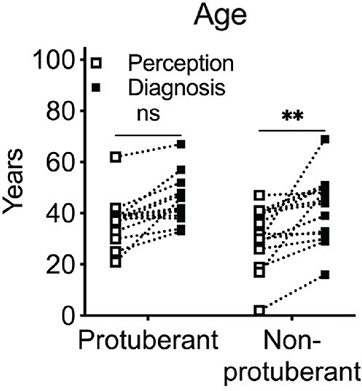
Fig. 2. Diagnostic delays in non-protuberant dermatofibroma protuberans. Age (in years) at initial visit and defenitive diagnosis classified according to protuberant or non-protuberant diseases. ns: not significnt; **p<0.01 as per two-way analysis of variance.
DISCUSSION
Further investigations are needed to determine whether non-protuberant lesions represent an early, transient stage (2, 4) or reflect a distinctive aetiology influenced by host susceptibility factors, such as adenosine deaminase-deficient severe combined immunodeficiency (5). Nonetheless, it should be noted that full-blown DFSPs are histopathologically described as occult pseudopodal infiltration (2). This locally invasive nature sometimes necessitates repetitive surgical procedures for disease clearance. In accordance with previous studies (2, 4), we found that various skin changes presented as “non-protuberant” in 45% of DFSPs (see Fig. 1). Thus, we propose that clinicians need training to recognize clinical appearances of lesions that are not necessarily elevated, contrary to what the term “protuberance” suggests (2, 4), to avoid delays in definitive diagnosis (4). Despite being based on a small sample from a single institution, our findings underscore the critical need for accurate and comprehensive understanding of DFSP’s developmental trajectory from a clinical perspective. We advocate for the adoption of the alternative terminology “dermatofibrosarcoma, often protuberant (DFSoP)”.
ACKNOWLEDGEMENTS
Funding sources: JSPS KAKENHI Grant, Grant-in-Aid for Scientific Research (C) (22K08404 to Y.I.), Grant Aid from Japan Foundation for Applied Enzymology to Y.I.
REFERENCES
- Hoesly PM, Lowe GC, Lohse CM, Brewer JD, Lehman JS. Prognostic impact of fibrosarcomatous transformation in dermatofibrosarcoma protuberans: a cohort study. J Am Acad Dermatol 2015; 72: 419–425. https://doi.org/10.1016/j.jaad.2014.11.020
- Martin L, Piette F, Blanc P, Mortier L, Avril MF, Delaunay MM, et al. Clinical variants of the preprotuberant stage of dermatofibrosarcoma protuberans. Br J Dermatol 2005; 153: 932–936. https://doi.org/10.1111/j.1365-2133.2005.06823.x
- Llombart B, Serra-Guillen C, Rubio L, Nagore E, Requena C, Traves V, et al. Subcutaneous dermatofibrosarcoma protuberans, a rare subtype with predilection for the head: a retrospective series of 18 cases. J Am Acad Dermatol 2017; 77: 503–511 e501. https://doi.org/10.1016/j.jaad.2017.02.046
- David MP, Funderburg A, Selig JP, Brown R, Caliskan PM, Cove L, et al. Perspectives of patients with dermatofibrosarcoma protuberans on diagnostic delays, surgical outcomes, and nonprotuberance. JAMA Netw Open 2019; 2: e1910413. https://doi.org/10.1001/jamanetworkopen.2019.10413
- Kesserwan C, Sokolic R, Cowen EW, Garabedian E, Heselmeyer-Haddad K, Lee CC, et al. Multicentric dermatofibrosarcoma protuberans in patients with adenosine deaminase-deficient severe combined immune deficiency. J Allergy Clin Immunol 2012; 129: 762–769 e761. https://doi.org/10.1016/j.jaci.2011.10.028