SHORT COMMUNICATION
A Case of Pemphigus Herpetiformis Strongly Associated with Gastric Cancer
Masakazu KAKURAI1*, Hana TAKESHITA1, Hiroshi KOGA2, Norito ISHII2 and Yoshihiro MORIYAMA1
1Department of Dermatology, Tsuchiura Kyodo General Hospital, 4-1-1 Otsuno, Tsuchiura, Ibaraki 300-0028, and 2Department of Dermatology, Kurume University School of Medicine, and Kurume University Institute of Cutaneous Cell Biology, Kurume, Japan. *E-mail: kakurai.masakazu.or@ms.hosp.tsukuba.ac.jp
Citation: Acta Derm Venereol 2024; 104: adv41319. DOI https://doi.org/10.2340/actadv.v104.41319.
Copyright: © 2024 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Aug 22, 2024. Accepted after revision: Nov 5, 2024. Published: Dec 5, 2024.
INTRODUCTION
Pemphigus herpetiformis (PH) is a rare type of pemphigus with a unique clinical and histological presentation (1). PH has been reported to be associated with some malignancies, including lung cancer, prostate cancer, lymphoma, oesophageal carcinoma, cutaneous angiosarcoma, and extramammary Paget’s disease (2–10). However, as these patients responded well to prednisolone therapy before cancer treatment and had variable immunologic profiles, the association between PH and malignancies remains unclear. Herein, we present a case of PH strongly associated with gastric cancer in which the skin lesions healed rapidly and anti-desmoglein (Dsg) 3 antibodies became negative after total gastrectomy without treatment for PH.
CASE REPORT
An 80-year-old Japanese man presented with a 10-month history of a pruritic rash beginning on the neck and gradually spreading to the trunk and extremities. Medical history included hypertension and cerebral infarction, and medications included nifedipine and clopidogrel sulphate. Physical examination revealed multiple urticarial erythematous plaques up to 3 cm in diameter, with vesicles, erosions, and crusts on the head, face, trunk, and extremities (Fig. 1A–D). The patient complained of severe itching. The mucous membranes were clear. Blood tests showed elevated white blood cell count of 9.07 × 103/μL (normal; 4,000–9,000/μL) with eosinophilia (7.7%) and elevated C-reactive protein level of 1.15 mg/dL (normal; < 0.30 mg/dL); other laboratory findings were unremarkable. A skin biopsy of the erythematous lesion revealed eosinophilic and neutrophilic spongiosis, with infiltration of some eosinophils and lymphocytes in the dermis (Fig. 1E). Another skin biopsy of the vesicle revealed an intraepidermal vesicle formed from the lower layers of the epidermis, containing some eosinophils and neutrophils, without acantholysis, and an infiltration of some eosinophils and lymphocytes, with a few neutrophils observed in the dermis (Fig. 1F). Direct immunofluorescence showed epidermal intercellular deposition of IgG (Fig. 1G) and C3, but no IgA or IgM. Enzyme-linked immunosorbent assay (ELISA) showed elevated serum levels of IgG antibodies against Dsg3 (94.20 U/mL: cut-off < 20 U/mL) but was negative for Dsg1. Additionally, we performed ELISA using recombinant proteins expressed by mammalian cells, which was positive for IgG and IgA anti-desmocollin (Dsc) 1 (0.238: cut-off < 0.200 and 1.559: cut-off < 0.091, respectively) and negative for IgG and IgA anti-Dsc2 and anti-Dsc3. Immunoblot analysis using epidermal extracts as antigens was negative for envoplakin, periplakin, bullous pemphigoid (BP) 180 and BP230.
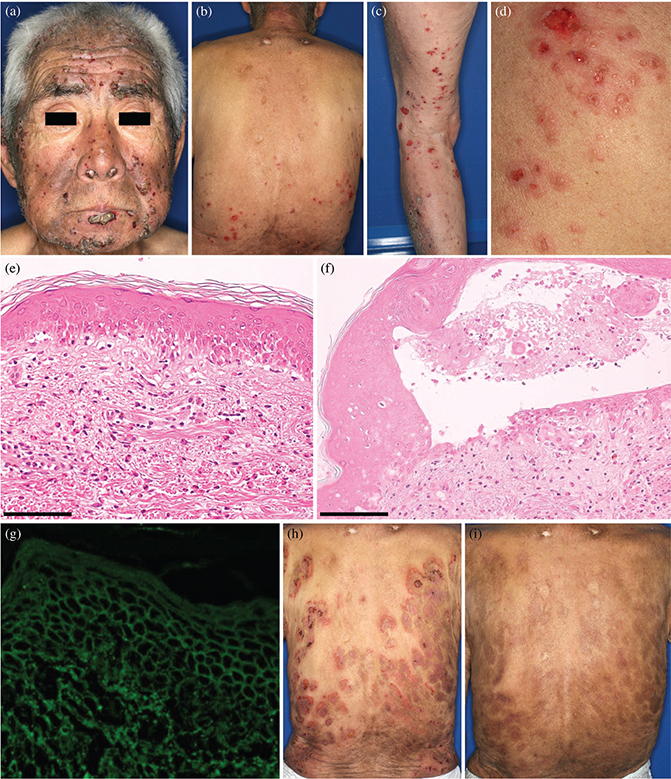
Fig. 1. Clinical and histological findings. (A–D) Clinical images of multiple urticarial erythematous plaques with vesicles, erosions, and crusts on the head, face, trunk, and extremities. (E) A haematoxylin-eosin-stained slide of the erythematous lesion revealed eosinophilic and neutrophilic spongiosis, with infiltrations of some eosinophils and lymphocytes in the dermis (bars in the image = 100 μm). (F) Another skin biopsy from the vesicle revealed an intraepidermal vesicle formed from lower layers of the epidermis, containing some eosinophils and neutrophils, without acantholysis (bars in the image = 100 μm). (G) Direct immunofluorescence showed epidermal intercellular deposition of IgG. (H) The skin lesions did not improve with topical steroids, forming erythematous plaques of annular arrangement with vesicles, erosions, and crusts. (I) The erythematous lesions, vesicles, and erosions had healed within 2 weeks after total gastrectomy, leaving pigmentation (photo shows 4 weeks postoperatively).
Because the patient also reported hoarseness during his first visit, upper gastrointestinal endoscopy was performed, which revealed no mucosal lesions in the oesophagus, but an ulcerative lesion in the body of the stomach. Biopsy and computed tomography findings led to the diagnosis of advanced gastric cancer; the histological diagnosis was well-differentiated tubular adenocarcinoma. The skin lesions did not improve with topical steroids and the urticarial erythematous plaques gradually increased in size and number, forming an annular arrangement with vesicles and erosions (Fig. 1H). Although treatment with oral prednisolone was planned, priority was given to the advanced gastric cancer. Interestingly, the intense itching rapidly improved within 3 days of total gastrectomy, and the erythematous lesions, vesicles, and erosions healed within 2 weeks postoperatively without additional treatment, leaving pigmentation (Fig. 1I). Subsequently, an oral provocation test was performed using nifedipine and clopidogrel sulphate. Neither recurrence of skin lesions nor increase in eosinophil cell count was observed, excluding the drug-induced PH. Taken together, we corroborated the diagnosis of PH. The Dsg3 ELISA index value decreased below the cut-off value at 3 months postoperatively. Seven months postoperatively, ELISA on the patient’s serum was negative for IgG antibodies against Dsg3 and positive for IgG and IgA anti-Dsc1 (0.278 and 1.126, respectively) antibodies. The patient was followed up for 1 year, during which no recurrence of PH or gastric cancer was observed.
DISCUSSION
PH is characterized by a combination of pruritic annular or urticarial erythematous plaques and grouped vesiculobullous lesions, often in a herpetiform configuration; histological eosinophilic and/or neutrophilic spongiosis, and intraepidermal vesicles with minimal acantholysis; and the immunological features of pemphigus (1). Although rare, concomitant PH and malignancies have been reported (2–10). Among the reported cases, 3 patients had lung cancer (2–4), 2 had prostate cancer (5, 6), 1 had lymphoma (7), 1 had oesophageal carcinoma (8), 1 had cutaneous angiosarcoma (9), and 1 had extramammary Paget’s disease (10). To our knowledge, this report is the first to detail a case of PH associated with gastric cancer. The course of treatment for PH and malignancy was documented in 7 cases, of which 5 were started on prednisolone prior to cancer treatment, and the skin lesions improved in all cases (3, 4, 5, 9, 10), 1 developed PH during regular hormone therapy for prostate cancer and responded well to low-dose prednisolone (6), and the remaining case was treated with dexamethasone for lymphoma and the skin lesions quickly improved (7). Therefore, proving a correlation between PH and malignancies is challenging because prednisolone therapy was effective. In the current case, the symptoms of PH healed rapidly and anti-Dsg3 antibodies became negative after total gastrectomy without additional treatment, and PH did not recur; this suggests that PH onset and progression possibly occurred in association with gastric cancer.
Target antigens for PH are usually Dsg1 and less commonly Dsg3 (1, 11). Furthermore, anti-Dsc antibodies have been detected in several cases (1, 11). In cases of PH with malignancy (2–10), anti-Dsg1 and/or anti-Dsg3 antibodies were positive in most cases, and a few exhibited reactivity against other antigens, such as Dscs. Given the variable combination of autoantibodies in these cases, and the fact that PH differs from the clinical and histological features of pemphigus vulgaris and pemphigus foliaceus, skin lesions in PH may be caused by pathophysiological mechanisms other than autoantibodies that directly attack epidermal antigens. Several hypotheses have been proposed. One is that IgG autoantibodies in PH attract eosinophils and neutrophils to the epidermis by inducing chemical mediator secretion, including keratinocyte interleukin (IL)-8, leading to protease release and blister formation with minimal acantholysis (1, 12). Zhai et al. reported that serum IL-8 levels in patients with advanced gastric cancer decreased after radical surgery (13). In the current case, anti-Dsg3 antibody levels decreased after total gastrectomy, suggesting that gastric cancer may have been involved in the production of anti-Dsg3 antibodies.
Many malignancies have a tumour microenvironment (TME) comprising several cell types, including cancer-associated fibroblasts (CAFs), immune cells, and endothelial cells (13, 14). CAFs are the major cell types in the TME; several studies have reported that CAFs produce various cytokines, such as IL-6 and IL-8, in patients with gastric cancer (13, 14). In the current case, we speculated that cytokines (e.g., IL-6 and IL-8) produced by CAFs or gastric cancer-induced IgG autoantibodies might attract eosinophils and neutrophils to the epidermis, resulting in spongiosis, followed by blister formation without acantholysis.
In summary, PH can rarely be complicated by malignancies. In the current case, gastric cancer may have been involved in PH onset and progression and antibody production. Although concomitant PH and malignancy has been proposed as paraneoplastic PH in the cutaneous paraneoplastic syndromes (3), their immunologic profiles and the tendency for associated malignancies remain unestablished. Further accumulation of PH cases associated with malignancies is warranted to clarify the causal link between these 2 diseases and to comprehensively understand their pathophysiological mechanisms.
REFERENCES
- Costa LMC, Cappel MA, Keeling JH. Clinical, pathologic, and immunologic features of pemphigus herpetiformis: a literature review and proposed diagnostic criteria. Int J Dermatol 2019; 58: 997–1007. https://doi.org/10.1111/ijd.14395
- Kubota Y, Yoshino Y, Mizoguchi M. A case of herpetiform pemphigus associated with lung cancer. J Dermatol 1994; 21: 609–611. https://doi.org/10.1111/j.1346-8138.1994.tb01803.x
- Palleschi GM, Giomi B. Herpetiformis pemphigus and lung carcinoma: a case of paraneoplastic pemphigus. Acta Derm Venereol 2002; 82: 304–305. https://doi.org/10.1080/000155502320323333
- Prado R, Brice SL, Fukuda S, Hashimoto T, Fujita M. Paraneoplastic pemphigus herpetiformis with IgG antibodies to desmoglein 3 and without mucosal lesions. Arch Dermatol 2011; 147: 67–71. https://doi.org/10.1001/archdermatol.2010.362
- Marzano AV, Tourlaki A, Cozzani E, Gianotti R, Caputo R. Pemphigus herpetiformis associated with prostate cancer. J Eur Acad Dermatol Venereol 2007; 21: 696–698. https://doi.org/10.1111/j.1468-3083.2006.01992.x
- Huang C, Wu Y, Kao C. Localized pemphigus herpetiformis: two case reports. Dermatologica Sinica 2010; 28: 36–39. https://doi.org/10.1016/S1027-8117(10)60006-7
- Schoch JJ, Boull CL, Camilleri MJ, Tollefson MM, Hook KP, Polcari IC. Transplacental transmission of pemphigus herpetiformis in the setting of maternal lymphoma. Pediatr Dermatol 2015; 32: e234–e237. https://doi.org/10.1111/pde.12649
- Arranz D, Corral M, Prats I, López-Ayala E, Castillo C, Vidaurrázaga C, et al. Herpetiform pemphigus associated with esophageal carcinoma. Actas Dermosifiliogr 2005; 96: 119–121. https://doi.org/10.1016/S0001-7310(05)73050-7
- Lu Y, Zhang M. Pemphigus herpetiformis in a patient with well-differentiated cutaneous angiosarcoma: case report and review of the published work. J Dermatol 2012; 39: 89–91. https://doi.org/10.1111/j.1346-8138.2011.01287.x
- Wang FF, Bai YP, Zheng ZC, Tong J, Wu YT. Coexistence of pemphigus herpetiformis with extramammary Paget disease. Clin Exp Dermatol 2018; 43: 324–326. https://doi.org/10.1111/ced.13318
- Ishii N, Teye K, Fukuda S, Uehara R, Hachiya T, Koga H, et al. Anti-desmocollin autoantibodies in nonclassical pemphigus. Br J Dermatol 2015; 173: 59–68. https://doi.org/10.1111/bjd.13711
- O’Toole EA, Mak LL, Guitart J, Woodley DT, Hashimoto T, Amagai M, et al. Induction of keratinocyte IL-8 expression and secretion by IgG autoantibodies as a novel mechanism of epidermal neutrophil recruitment in a pemphigus variant. Clin Exp Immunol 2000; 119: 217–224. https://doi.org/10.1046/j.1365-2249.2000.01104.x
- Zhai J, Shen J, Xie G, Wu J, He M, Gao L, et al. Cancer-associated fibroblasts-derived IL-8 mediates resistance to cisplatin in human gastric cancer. Cancer Lett 2019; 454: 37–43. https://doi.org/10.1016/j.canlet.2019.04.002
- Ham IH, Oh HJ, Jin H, Bae CA, Jeon SM, Choi KS, et al. Targeting interleukin-6 as a strategy to overcome stroma-induced resistance to chemotherapy in gastric cancer. Mol Cancer 2019; 18: 68. https://doi.org/10.1186/s12943-019-0972-8