ORIGINAL REPORT
Poor Outcome of Metastatic Cutaneous Squamous Cell Carcinoma: A Swedish Retrospective Study
Charlotta L. PALMQVIST1,2 and Mathias VON BECKERATH2
and Mathias VON BECKERATH2
1Visby Regional Hospital, Visby, and 2Karolinska institute, Stockholm, Sweden
The incidence of cutaneous squamous cell carcinoma is increasing rapidly, and 1.5–5% of cases develop metastatic disease, which is associated with a markedly worse prognosis. This study aimed to describe the characteristics of the primary tumour, the patients, and the metastases, as well as treatment and outcomes in patients with metastatic cutaneous squamous cell carcinoma. A total of 137 patients with a diagnosis of metastatic cutaneous squamous cell carcinoma within the Stockholm region during 2012–2020 were identified and included. Median age was 81; 79% were male; 70% had a WHO performance status of 0–1. 50% of the primary tumours were classified as high-risk tumours according to the Brigham and Women’s Hospital system. The median time between diagnosis of the primary tumour and the diagnosis of regional metastases was 7 months and 90% of the metastases were diagnosed within 2 years. Most patients were treated with a combination of surgery and radiotherapy. Overall survival at 2 years was 56%. This study shows that metastatic cutaneous squamous cell carcinoma occurs in elderly but otherwise generally healthy patients, that primary tumours with a high risk of causing metastases seem hard to identify, and that metastases occur within 2 years after primary tumour diagnosis. The patient group overall has a high mortality rate, particularly among those who do not undergo surgery.
SIGNIFICANCE
Cutaneous squamous cell carcinoma is the second most common cancer in Sweden, its incidence is increasing, and metastasized disease has a high mortality rate. This is the first published study on metastatic cutaneous squamous cell carcinoma in Sweden and reports descriptions of this disease.
Key words: metastasis; cutaneous squamous cell carcinoma; skin; SCC; metastatic.
Citation: Acta Derm Venereol 2026; 106: adv44057. DOI: https://doi.org/10.2340/actadv.v106.44057.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
Submitted: Jun 5, 2025. Accepted after revision: Jan 9, 2026. Published: Feb 23, 2026.
Corr: Mathias von Beckerath, Karolinska Institute, Stockholm, Sweden. E-mail: mathias.von.beckerath@ki.se
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
Cutaneous squamous cell carcinoma (cSCC) is the second most common cancer in Sweden and its incidence is increasing rapidly. In Sweden during 2023, over 12,000 new cases of cSCC were reported and its incidence is calculated at 137 new cases per 100,000 people for men and 107 for women (1). Cumulative UV radiation, old age, and male sex are the most important risk factors for developing cSCC (2–8). The increasing incidence is believed to be caused by an ageing population and increased exposure to UV radiation. Approximately 80% of cSCC cases are localized to the head and neck region, accounting for a large proportion of consultations in the ENT department (5, 8–10).
For the majority of patients, the disease is easily treated by surgical excision. Unfortunately, 1.5–5% of cSCC metastasize and in these cases the patients’ prognosis worsens radically (11–22).
Risk factors for developing metastatic cSCC (mcSCC) include tumour size ≥ 2 cm, invasion depth, degree of differentiation, perineural invasion, intravascular invasion, subcutaneous invasion, immunosuppression, tumour relapse, and tumour location (11, 14, 15, 19, 23–30). In the national guidelines of Sweden, the Brigham and Women’s Hospital (BWH) staging system is used to identify high-risk tumours postoperatively (16, 31). For mcSCC the TNM-system (UICC 8) is used.
The great majority of metastases occur within the first 2 years after the diagnosis of the primary tumour and most often affect the parotid gland or the lymph nodes of the neck (14, 15, 18–20, 26, 32, 33). A literature review from 2018 stated that disease-specific 5-year survival was between 58% and 83% and a large Australian retrospective cohort from 2024 showed a 5-year Disease Specific Survival (DSS) of 77.6% (22, 34). There is currently no reported survival rate in Sweden, but unpublished Swedish data point towards a 52% overall survival within 18 months, which is similar to the results of a recent Finnish study by Knuuttila et al. (18). Little is known about mcSCC in Sweden as it is not recorded in a national registry as a unique diagnosis and no recent studies have been published.
As we expect an even higher incidence of cSCC in the future we can assume that mcSCC will cause a significant patient burden in ENT cancer departments. In this study we aim to describe the patient characteristics, tumour characteristics, treatment, and outcome of Swedish patients with mcSCC in Stockholm region during the study period 2012–2020.
MATERIALS AND METHODS
The study was approved by the Swedish ethical committee. Stockholm region’s charting system Take Care was searched for adult patients who were diagnosed with metastasized or locally advanced cSCC between 2012 and 2020. The charting system was searched for the ICD-10 diagnostic code C44 (skin tumour) in combination with C77 (lymph node metastases) and C44 in combination with surgical codes ELB40 (parotic resection), ELB50 (parotidectomy), or PJD51 (neck dissection). Patients were included if they had cSCC in the head and neck area and locally advanced or metastatic disease. Their medical records were reviewed, and data (see list of variables in Table I) were collected.
RESULTS
The search identified 309 patients of whom 172 were excluded because they had cancer diagnoses other than cSCC, their primary tumours were located outside the head and neck area, or they were treated for their metastasized or locally advanced cSCC at a hospital outside of the study period or outside of the Stockholm region. This resulted in 137 patients who were included in the study.
Patient characteristics
The average age at the time of the primary tumour diagnosis was 80 years (81 median). On average each patient had 3.4 comorbidities; 70% had a WHO performance status of 0–1 and 30% 2–4. The remaining patient characteristics are summarized in Table I.
Primary tumour characteristics
In 2/137 cases the primary tumour was unknown, resulting in 135 primary tumours to analyse. According to the BWH system 50% were classified as high-risk tumours after the primary surgery. The remaining tumour characteristics are summarized in Table II.
| Variable | n (%) |
| Number of known primary tumours | 135 (100) |
| Site | |
| Ear | 28 (21) |
| Temple | 26 (19) |
| Cheek | 21 (16) |
| Scalp | 18 (13) |
| Periauricular | 14 (10) |
| Forehead | 9 (7) |
| Lip (vermilion) | 7 (5) |
| Neck | 6 (4) |
| Nose | 5 (4) |
| Chin | 1 (1) |
| Size | |
| < 2 cm | 51 (39)a |
| ≥ 2 cm | 80 (61)a |
| unknown | 4 (3) |
| Degree of pathological differentiation | |
| Low | 52 (41)a |
| Moderate | 66 (52)a |
| High | 9 (7)a |
| Unknown | 8 (6) |
| Perineural invasion | |
| Yes | 21 (16) |
| No/unknown | 114 (84) |
| Growth beyond fat | |
| Yes | 51 (38) |
| No/unknown | 84 (62) |
| BWHb | |
| T1 | 27 (21)a |
| T2a | 38 (30)a |
| T2b | 57 (45)a |
| T3 | 5 (4)a |
| Unknown | 3 |
| Radical margins | |
| Positive | 49 (39)a |
| Negative | 76 (61)a |
| Unknown | 2 (1) |
| Non-surgical treatment of primary tumour | 8 (6) |
| aPercentage excludes the unknown data. b7 lip tumours excluded. | |
Metastases characteristics
The average time between diagnosis of the primary tumour and the diagnosis of regional metastases was 11 months and median time 7 months; 90% of the metastases were diagnosed within 2 years. The average time between the diagnosis of the primary tumour and the diagnosis of distant metastases was 26 months and median time 16 months. The average and median age at the diagnosis of regional metastases was 82 years. The average and median age at the time of distant metastases was 81 and 82 years respectively. The average and median disease-free survival (DFS) was 11 and 6 months respectively. The remaining characteristics of the metastases are summarized in Table III.
| Variables | n (%) |
| Type of advanced cSCC | |
| Metastases | 128 (93) |
| Regional metastases | 123 |
| Distant metastases only | 5 |
| Distant metastases | 29 |
| Locally advanced disease without metastases | 9a (7) |
| cTNMb | |
| T1 | 53 (39) |
| T2 | 19 (14) |
| T3 | 60 (44) |
| T4 | 1 (1) |
| Tx | 4 (3) |
| N0 | 16 (12) |
| N1 | 21 (15) |
| N2a | 4 (3) |
| N2b | 23 (17) |
| N2c | 5 (4) |
| N3a | 0 (0) |
| N3b | 68 (50) |
| M0 | 119 (87) |
| M1 | 14 (10) |
| Mx | 4 (3) |
| Stage (excluding lip) | |
| I | 0 (0) |
| IIa | 12 (9) |
| IIb | 0 (0) |
| IIIa | 16 (12) |
| IIIb | 27 (21) |
| IV | 75 (58) |
| Stage lip cancer | |
| I | 0 |
| II | 0 |
| III | 3 (43) |
| IVa | 2 (29) |
| IVb | 2 (29) |
| aLocally advanced disease was defined as at least T3 tumours that needed extensive surgery in the ENT department or radiotherapy in the oncology department. bClassified at the multidisciplinary cancer conference (MCC) after the metastases surgery, or at the primary MCC if the patient did not undergo surgery, or at the MCC after the major surgery if locally advanced tumour without metastases. | |
Treatment
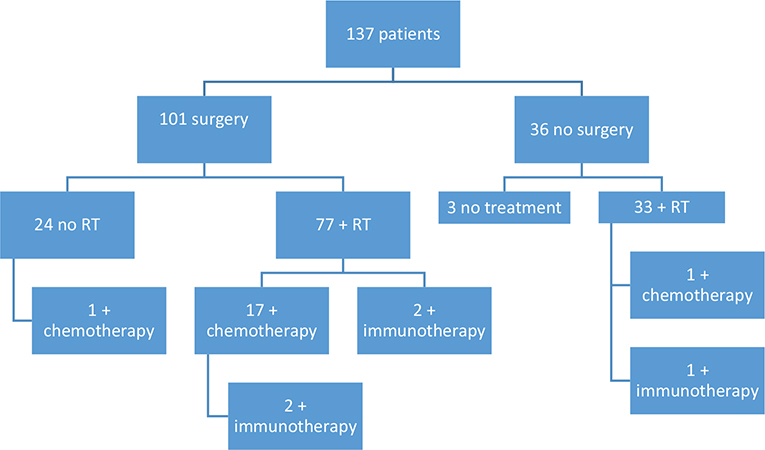
A total of 101 patients received surgery due to regional metastases or locally advanced disease, 76% (94/123) of those with regional metastases and 78% (7/9) of those with locally advanced tumours. Radical excisions were performed in 45% (45/101) of the surgeries. 80% (110/137) of the patients had radiotherapy (RT); 14% (19/137) had chemotherapy; 4% (5/137) had immunotherapy. The patients’ treatments are shown in Fig. 1. The rest of the treatment data are summarized in Table IV.
| Variable | n (%) |
| Type of treatment | |
| Surgery only | 23 (17) |
| Surgery + RT | 58 (42) |
| Surgery + RT + chemotherapy | 15 (11) |
| Surgery + RT + chemotherapy + immunotherapy | 2 (1) |
| Surgery + RT + immunotherapy | 2 (1) |
| Surgery + chemotherapy | 1 (1) |
| RT only | 31 (23) |
| RT + chemotherapy | 1 (1) |
| RT + immunotherapy | 1 (1) |
| None | 3 (2) |
| Number of patients who underwent surgery | 101 (74) |
| Type of surgery | |
| Parotidectomy + neck dissectiona | 48 (48) |
| Parotidectomy + lumpectomya | 13 (13) |
| Neck dissection only | 21 (21) |
| Parotidectomy only | 7 (7) |
| Other surgery | 12 (12) |
| Location site of the metastasisb | |
| Parotic gland | 49 (49) |
| Region I | 20 (20) |
| Region II | 32 (32) |
| Region III | 12 (12) |
| Region IV | 3 (3) |
| Region V | 15 (15) |
| aNeck dissection is defined as removal of 3 or more regions. Lumpectomy is defined as removal of 2 or fewer regions. Percentage is calculated from the patients who underwent surgery. bFrom the pathological records of the patients who underwent surgery (total 101 patients). The same patient can appear in multiple categories. | |
Outcome
At the end of the study period (July 2023) an 80% (109/137) mortality rate was seen in the study population. Among the immunosuppressed an 88% (30/34) mortality rate was seen. Average time between the primary diagnosis of cSCC and death was 33 months and median 22 months. Average time between the identification of regional metastases and death was 21.5 months and median 13 months. Average time between diagnosis of distant metastases and death was 14 months and median 5 months; 51% (56/109) of the deaths were determined to be directly related to the cSCC diagnosis. In 18 cases the cause of death was unknown. Excluding unknown causes of deaths, 62% (56/91) of the deaths were assessed to be caused by cSCC. Other outcomes are summarized in Table V.
| n (%) | |
| Overall survival 2 years | |
| After primary tumour | 76/135a (56) |
| After regional metastases | 51/123 (41) |
| After distant metastases | 7/29 (24) |
| Overall survival 2 years after primary diagnosis | |
| Surgery, no RT | 12/23b (52) |
| Surgery + RT | 54/77 (70) |
| RT, no surgery | 10/32c (31) |
| No treatment | 0/3 (0) |
| Overall survival 2.5 years | |
| After primary tumour | 68 /135a (50) |
| After regional metastases | 45/123 (37) |
| After distant metastases | 5/29 (17) |
| Overall survival 2.5 years among immunosuppressed | |
| After primary tumour | 14/34 (41) |
| After regional metastases | 9/30 (30) |
| After distant metastases | 2/8 (25) |
| Overall survival 2.5 years after primary diagnosis | |
| Surgery, no RT | 11/23b(48) |
| Surgery + RT | 49/77 (64) |
| RT, no surgery | 9/32c (28) |
| No treatment | 0/3 (0) |
| Dead in July 2023 | 109/137 (80) |
| Dead in July 2023 among immunosuppressed | 30/34 (88) |
| a2 patients excluded due to unknown data concerning primary diagnosis. b1 excluded due to unknown primary. c1 excluded due to unknown primary. | |
DISCUSSION
Cutaneous SCC is the second most common cancer in Sweden, its incidence is increasing, and metastasized disease has a high mortality rate. This is the first published study on mcSCC in Sweden and reports descriptions of this disease.
The most common sites of primary tumours among mcSCC in Sweden were ear and temple. This is similar to studies from other countries (11, 18).
The median age of the patient group was 81 but 70% were assessed as WHO-performance status 0-1, indicating that these patients are elderly but relatively healthy at the time prior of the treatment. Despite this, overall survival after 2.5 years from diagnosis of regional metastases was only 37%, which suggests the aggressiveness of the disease and/or the lack of effective treatment. With immunosuppression the prognosis is even worse.
In Sweden the BWH system is used to identify high-risk primary tumours. Other studies have reported a low sensitivity but a high specificity of the BWH system (16, 35–37). Knuuttila et al. could not find a correlation between BWH and prognosis (18). In our study only 50% of the primary tumours were assessed as BWH high-risk, which supports these other studies’ results reporting its low sensitivity. We encourage the development of a more sensitive risk stratification system to find high-risk primary cSCC.
Median time between primary tumour and regional metastases was 7 months and 90% of the metastases were diagnosed within 2 years. This data is in line with earlier studies, including Knuuttila et al., which reported 6.6 months and 85% respectively (13, 14, 18, 21, 26, 32, 38). This suggests that close monitoring of patients during the first 2 years following a high-risk cSCC is essential for the early detection of metastases.
Most metastases were located in the parotic gland and region I–II, which concurs with earlier studies (13). 58% of our patients were classified as TNM stage IV after their surgery. The majority (87%) were due to clinically extracapsular growth of the metastases. Ebrahimi et al. found that 81% of their study population had metastases with extracapsular growth and that this significantly affected the patient outcome. However, they suggested that the presence of extracapsular growth upstaged too many patients to TNM stage IV, despite many having curable disease (34).
The patients received different combinations of treatment, mostly surgery with adjuvant radiotherapy. Chemotherapy and immunotherapy were never given as single therapy, but were given as an addition to surgery and/or radiotherapy. The highest overall survival was among the patients receiving surgery + radiotherapy, where 64% were alive 2.5 years after the primary diagnosis. The overall survival was 48% for patients who received only surgery and 28% for those who received only radiotherapy. Obviously, the patient’s comorbidities and level of functioning are taken into consideration when choosing which treatment a patient will receive, but our results suggest a higher mortality rate without surgery. Earlier studies have also shown better prognosis if patients receive both surgery and radiotherapy compared with surgery alone (21, 34, 39).
Our overall survival rate (OS) at 2 years was 56% from primary tumour diagnosis and 41% from metastases diagnosis. These numbers are similar to the results of Knuuttila et al. where 2-year OS from primary tumour diagnosis and metastases diagnosis was 63.8% and 43.4% respectively (18). Other studies have shown OS at 2 years of 50–66% from time of metastases (32, 38).
Study limitations are mostly related to those expected in a retrospective cohort study. Some data were missing, and patients may have not been identified due to incorrect ICD coding of patients during treatment. Some patients may never have been referred to a hospital for treatment but rather received palliative treatment at home. Cause of death was rarely confirmed by autopsy and was therefore difficult to relate to the cSCC diagnosis. Another limitation is that only 4% of our patients received immunotherapy as this was not part of the European guidelines during the study period. Given that 32% of the patients had a medical history of a previous cSCC, the tumour defined as the primary source of metastasis may have been incorrectly identified.
Conclusion
Our study, which is the first reported in the Swedish population, shows that mcSCC occurs in elderly but otherwise generally healthy patients, that primary tumours with a high risk of causing mcSCC seem hard to identify, and that metastases occur within 2 years after primary tumour diagnosis. The patient group overall has a high mortality rate, particularly among those who do not undergo surgery. We encourage the development of a more sensitive risk stratification system to find high-risk primary cSCC, and close monitoring of patients during the first 2 years following a high-risk primary cSCC.
REFERENCES
- Socialstyrelsen. Statistikdatabas för cancer 2023. Available from: https://sdb.socialstyrelsen.se/if_can/resultat.aspx
- de Gruijl FR. Skin cancer and solar UV radiation. Eur J Cancer 1999; 35: 2003–2009. https://doi.org/10.1016/S0959-8049(99)00283-X
- Rigel DS. Cutaneous ultraviolet exposure and its relationship to the development of skin cancer. J Am Acad Dermatol 2008; 58: S129–S132. https://doi.org/10.1016/j.jaad.2007.04.034
- Armstrong BK, Kricker A. The epidemiology of UV induced skin cancer, J Photochem Photobiol B 2001; 63: 8–18. https://doi.org/10.1016/S1011-1344(01)00198-1
- Xiang F, Lucas R, Hales S, Neale R. Incidence of nonmelanoma skin cancer in relation to ambient UV radiation in white populations, 1978–2012: empirical relationships. JAMA Dermatol 2014; 150: 1063–1071. https://doi.org/10.1001/jamadermatol.2014.762
- Green AC, Olsen CM. Cutaneous squamous cell carcinoma: an epidemiological review. Br J Dermatol 2017; 177: 373–381. https://doi.org/10.1111/bjd.15324
- Marks R, Staples M, Giles GG. Trends in non-melanocytic skin cancer treated in Australia: the second national survey. Int J Cancer 1993; 53: 585–590. https://doi.org/10.1002/ijc.2910530410
- Dal H, Boldemann C, Lindelof B. Trends during a half century in relative squamous cell carcinoma distribution by body site in the Swedish population: support for accumulated sun exposure as the main risk factor. J Dermatol 2008; 35: 55–62. https://doi.org/10.1111/j.1346-8138.2008.00416.x
- Franceschi S, Levi F, Randimbison L, La Vecchia C. Site distribution of different types of skin cancer: new aetiological clues. Int J Cancer 1996; 67: 24–28. https://doi.org/10.1002/(SICI)1097-0215(19960703)67:1<24::AID-IJC6>3.0.CO;2-1
- Robsahm TE, Helsing P, Veierod MB. Cutaneous squamous cell carcinoma in Norway 1963–2011: increasing incidence and stable mortality. Cancer Med 2015; 4: 472–480. https://doi.org/10.1002/cam4.404
- Schmults CD, Karia PS, Carter JB, Han J, Qureshi AA. Factors predictive of recurrence and death from cutaneous squamous cell carcinoma: a 10-year, single-institution cohort study. JAMA Dermatol 2013; 149: 541–547. https://doi.org/10.1001/jamadermatol.2013.2139
- Brougham ND, Dennett ER, Cameron R, Tan ST. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J Surg Oncol 2012; 106: 811–815. https://doi.org/10.1002/jso.23155
- Mourouzis C, Boynton A, Grant J, Umar T, Wilson A, Macpheson D, et al. Cutaneous head and neck SCCs and risk of nodal metastasis: UK experience. J Craniomaxillofac Surg 2009; 37: 443–447. https://doi.org/10.1016/j.jcms.2009.07.007
- Venables ZC, Autier P, Nijsten T, Wong KF, Langan SM, Rous B, et al. Nationwide incidence of metastatic cutaneous squamous cell carcinoma in England. JAMA Dermatol 2019; 155: 298–306. https://doi.org/10.1001/jamadermatol.2018.4219
- Brantsch KD, Meisner C, Schonfisch B, Trilling B, Wehner-Caroli J, Rocken M, et al. Analysis of risk factors determining prognosis of cutaneous squamous-cell carcinoma: a prospective study. Lancet Oncol 2008; 9: 713–720. https://doi.org/10.1016/S1470-2045(08)70178-5
- Roscher I, Falk RS, Vos L, Clausen OPF, Helsing P, Gjersvik P, et al. Validating 4 staging systems for cutaneous squamous cell carcinoma using population-based data: a nested case-control study. JAMA Dermatol 2018; 154: 428–434. https://doi.org/10.1001/jamadermatol.2017.6428
- Karia PS, Han J, Schmults CD. Cutaneous squamous cell carcinoma: estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J Am Acad Dermatol 2013; 68: 957–966. https://doi.org/10.1016/j.jaad.2012.11.037
- Knuutila JS, Riihila P, Kurki S, Nissinen L, Kahari VM. Risk factors and prognosis for metastatic cutaneous squamous cell carcinoma: a cohort study. Acta Derm Venereol 2020; 100: adv00266. https://doi.org/10.2340/00015555-3628
- Rowe DE, Carroll RJ, Day CL Jr. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip: implications for treatment modality selection. J Am Acad Dermatol 1992; 26: 976–990. https://doi.org/10.1016/0190-9622(92)70144-5
- Joseph MG, Zulueta WP, Kennedy PJ. Squamous cell carcinoma of the skin of the trunk and limbs: the incidence of metastases and their outcome. Aust N Z J Surg 1992; 62: 697–701. https://doi.org/10.1111/j.1445-2197.1992.tb07065.x
- Givi B, Andersen PE, Diggs BS, Wax MK, Gross ND. Outcome of patients treated surgically for lymph node metastases from cutaneous squamous cell carcinoma of the head and neck. Head Neck 2011; 33: 999–1004. https://doi.org/10.1002/hed.21574
- Rotman A, Kerr SJ, Giddings CEB. Elective neck dissection in metastatic cutaneous squamous cell carcinoma to the parotid gland: a systematic review and meta-analysis. Head Neck 2019; 41: 1131–1139. https://doi.org/10.1002/hed.25561
- Oddone N, Morgan GJ, Palme CE, Perera L, Shannon J, Wong E, et al. Metastatic cutaneous squamous cell carcinoma of the head and neck: the Immunosuppression, Treatment, Extranodal spread, and Margin status (ITEM) prognostic score to predict outcome and the need to improve survival. Cancer 2009; 115: 1883–1891. https://doi.org/10.1002/cncr.24208
- Eigentler TK, Leiter U, Hafner HM, Garbe C, Rocken M, Breuninger H. Survival of patients with cutaneous squamous cell carcinoma: results of a prospective cohort study. J Invest Dermatol 2017; 137: 2309–2315. https://doi.org/10.1016/j.jid.2017.06.025
- Dinehart SM, Pollack SV. Metastases from squamous cell carcinoma of the skin and lip: an analysis of twenty-seven cases. J Am Acad Dermatol 1989; 21: 241–248. https://doi.org/10.1016/S0190-9622(89)70168-7
- Cherpelis BS, Marcusen C, Lang PG. Prognostic factors for metastasis in squamous cell carcinoma of the skin. Dermatol Surg 2002; 28: 268–273. https://doi.org/10.1046/j.1524-4725.2002.01169.x
- Goepfert H, Dichtel WJ, Medina JE, Lindberg RD, Luna MD. Perineural invasion in squamous cell skin carcinoma of the head and neck. Am J Surg 1984; 148: 542–547. https://doi.org/10.1016/0002-9610(84)90385-4
- Galloway TJ, Morris CG, Mancuso AA, Amdur RJ, Mendenhall WM. Impact of radiographic findings on prognosis for skin carcinoma with clinical perineural invasion. Cancer 2005; 103: 1254–1257. https://doi.org/10.1002/cncr.20913
- Thompson AK, Kelley BF, Prokop LJ, Murad MH, Baum CL. Risk factors for cutaneous squamous cell carcinoma recurrence, metastasis, and disease-specific death: a systematic review and meta-analysis. JAMA Dermatol 2016; 152: 419–428. https://doi.org/10.1001/jamadermatol.2015.4994
- Moore BA, Weber RS, Prieto V, El-Naggar A, Holsinger FC, Zhou X, et al. Lymph node metastases from cutaneous squamous cell carcinoma of the head and neck. Laryngoscope 2005; 115: 1561–1567. https://doi.org/10.1097/01.mlg.0000173202.56739.9f
- Karia PS, Jambusaria-Pahlajani A, Harrington DP, Murphy GF, Qureshi AA, Schmults CD. Evaluation of American Joint Committee on Cancer, International Union Against Cancer, and Brigham and Women’s Hospital tumor staging for cutaneous squamous cell carcinoma. J Clin Oncol 2014; 32: 327–334. https://doi.org/10.1200/JCO.2012.48.5326
- Bobin C, Ingrand P, Dreno B, Rio E, Malard O, Espitalier F. Prognostic factors for parotid metastasis of cutaneous squamous cell carcinoma of the head and neck. Eur Ann Otorhinolaryngol Head Neck Dis 2018; 135: 99–103. https://doi.org/10.1016/j.anorl.2017.09.006
- Veness MJ, Porceddu S, Palme CE, Morgan GJ. Cutaneous head and neck squamous cell carcinoma metastatic to parotid and cervical lymph nodes. Head Neck 2007; 29: 621–631. https://doi.org/10.1002/hed.20576
- Ebrahimi A, Gupta R, McDowell L, Magarey MJR, Smith PN, Schulte KM, et al. Determinants of prognosis in head and neck cutaneous squamous cell carcinoma with nodal metastases. JAMA Otolaryngol Head Neck Surg 2024;150: 986–994. https://doi.org/10.1001/jamaoto.2024.3103
- Venables ZC, Tokez S, Hollestein LM, Mooyaart AL, van den Bos RR, Rous B, et al. Validation of four cutaneous squamous cell carcinoma staging systems using nationwide data. Br J Dermatol 2022; 186: 835–842. https://doi.org/10.1111/bjd.20909
- Girardi FM, Wagner VP, Machado CDC, Wysong A, Ran NA, Granger EE, et al. Validation of current staging systems in HNCSCC: a multinational cohort study. Head Neck 2025; 47: 3385–3393. https://doi.org/10.1002/hed.28259
- Elaldi R, Chamorey E, Schiappa R, Sudaka A, Anjuere F, Villarme A, et al. Comparative performance of four staging classifications to select “high-risk” head and neck cutaneous squamous cell carcinomas. J Clin Med 2023; 12: 3929. https://doi.org/10.3390/jcm12123929
- Hirshoren N, Danne J, Dixon BJ, Magarey M, Kleid S, Webb A, et al. Prognostic markers in metastatic cutaneous squamous cell carcinoma of the head and neck. Head Neck 2017; 39: 772–778. https://doi.org/10.1002/hed.24683
- Hirshoren N, Ruskin O, McDowell LJ, Magarey M, Kleid S, Dixon BJ. Management of parotid metastatic cutaneous squamous cell carcinoma: regional recurrence rates and survival. Otolaryngol Head Neck Surg 2018; 159: 293–299. https://doi.org/10.1177/0194599818764348