REVIEW ARTICLE
Health-related Quality of Life Measurements in Patients with Actinic Keratosis: A Systematic Review
Zohra F. AHMADZAY1 , Mattias A. S. HENNING2
, Mattias A. S. HENNING2 , Gregor B. E. JEMEC2,3
, Gregor B. E. JEMEC2,3 and Gabrielle R. VINDING2–4
and Gabrielle R. VINDING2–4
1Department of Dermatology, Aarhus University Hospital, Aarhus, 2Department of Dermatology and Allergy, Copenhagen University Hospital – Herlev and Gentofte, Copenhagen, 3Institute for Clinical Medicine, Faculty of Health and Medical Sciences, University of Copenhagen, Copenhagen, Denmark, and 4Department of Dermatology, Zealand University Hospital, Roskilde, Denmark
Actinic keratosis is a common, chronic, and potentially pre-cancerous skin disease that can negatively impact patients’ health-related quality of life. This systematic review aimed to identify and describe the questionnaires used to assess health-related quality of life in patients with actinic keratosis and summarize reported impacts. Four databases (EMBASE, PsycINFO, PubMed, and Cochrane Library) were searched up to February 2025. Two reviewers independently screened articles, and risk of bias was assessed using tools appropriate to study design. Of 639 records, 37 studies including 9,326 patients met inclusion criteria. Most studies were interventional (59%) and of moderate to high quality (64%). Health-related quality of life was assessed using 3 disease-specific, 3 dermatology-specific, and 2 generic questionnaires. The most frequently used instruments were the Actinic Keratosis Quality of Life (n = 13) and the Dermatology Life Quality Index (n = 16). All questionnaires detected health-related quality of life impairment, especially during treatment. Actinic Keratosis Quality of Life identified patient subgroups with greater health-related quality of life reduction, including females, younger individuals, patients with comorbidities, and those with a history of skin cancer. All health-related quality of life questionnaires captured impairment in patients with actinic keratosis. The Actinic Keratosis Quality of Life and Dermatology Life Quality Index questionnaires were widely used. Actinic Keratosis Quality of Life identified subpopulations with health-related quality of life impairment. Among studies reporting mean health-related quality of life scores, the Dermatology Life Quality Index ranged from 1.55 to 24 and the Actinic Keratosis Quality of Life from 4.22 to 17, reflecting very broad variability and underlying differences in actinic keratosis severity across study populations.
SIGNIFICANCE
Actinic keratosis lesions can be painful, itchy, cosmetically concerning, and may cause anxiety about skin cancer. Thus, actinic keratosis can affect health-related quality of life. We reviewed 37 studies involving 9,326 patients with actinic keratosis that assessed their health-related quality of life using questionnaires. Nearly half of the included studies measured health-related quality of life before and after treatment. Most used tools developed for other conditions. Only one, the Actinic Keratosis Quality of Life questionnaire, was actinic keratosis-specific tool and used in 13 studies. Three studies identified subgroups with most affected health-related quality of life: females, younger patients, and those with comorbidities or prior skin cancer.
Key words: health-related quality of life; quality of life; patient-reported outcomes; actinic keratosis; systematic review.
Citation: Acta Derm Venereol 2026; 106: adv44163. DOI: https://doi.org/10.2340/actadv.v106.44163.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
Submitted: Jun 17, 2025. Accepted after revision: Feb 18, 2026. Published: Mar 12, 2026.
Corr: Zohra Faizy Ahmadzay, MD, PhD, Department of Dermatology, Aarhus University Hospital, Aarhus, Denmark. E-mail: zohra.ahmadzay@gmail.com
Competing interests and funding: ZFA: None. MASH: Support from Pfizer for attending conference. GBJ: Copyright holder of AKQoL. Grants from AbbVie, AstraZeneca, Inflarx, Janssen-Cilag, Leo Pharma, Novartis, Regeneron, and Sanofi. Received speaker honoraria from AbbVie, Boehringer Ingelheim, Galderma, and MSD. Received honoraria from AbbVie, Chemocentryx, Coloplast, Incyte, Inflarx, Novartis, Pierre Fabre, and UCB for participation on advisory boards. Received unrestricted departmental grants from AbbVie, Leo Pharma, and Novartis. GRV: Copyright holder of AKQoL. Toosonix through department for conducting a research study regarding BCC. Support from Pfizer for attending conference.
Actinic keratosis (AK) is a common dysplastic, ery-thematous, and hyperkeratotic lesion found mainly among fair-skinned individuals with sun-damaged skin (1). AK is considered a precursor to squamous cell carcinoma (SCC), and the presence of multiple AK lesions may reflect chronic ultraviolet radiation exposure, leading to field cancerization, an area predisposed to malignancy (1).
It is obvious that a tumour that occurs in sun-exposed sites is likely in a visible site. Consequently, adverse effects of the treatment are also likely to be visible, and therefore cosmetically more sensitive (2). This naturally encompasses both disease and the treatment used. AKs intrinsically cause morbidity due to pain, itch, disfigurement, or because of their association with SCC through apprehension and fear of possible future malignancies (3, 4). In any case, AKs affect the patient’s health-related quality of life (HRQoL) (4).
The European Academy for Dermatology and Venereology (EADV) Task Force on QoL recommends that patients are included in the assessment of treatment outcome of interventions and the provided care (5). Special patient subpopulations may be in greater need of attention than others, e.g., solid organ transplant recipients. It has been suggested that general skin tumour surveillance can be done through appropriate HRQoL tools (5, 6). These are mostly patient-reported outcome measures (PROMs), i.e., validated and standardized questionnaires. We therefore aimed to conduct a systematic review of the currently available questionnaires used to assess HRQoL in patients diagnosed with AK and to describe the measured impact of HRQoL, including whether these were sensitive to change during the disease course and if each could identify subpopulations with greater impairment of HRQoL.
Methods
Search strategy and study selection
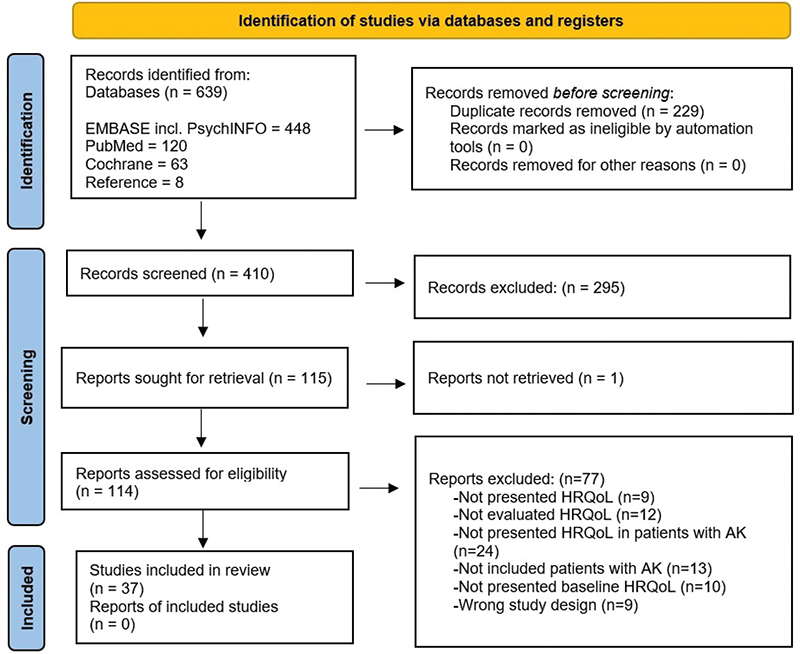
A systematic literature search was conducted on EMBASE (including PsycINFO), PubMed, and Cochrane Library from inception to February 2025. The search strings for each database are provided in Table SI. No language restriction was applied, but only human studies were included. Observational studies and clinical trials reporting HRQoL in patients with AK were eligible while studies with fewer than 5 participants were excluded. Two investigators (ZA and MH) independently screened titles, abstracts, and full-text for eligibility. Reference lists of relevant studies were also assessed for additional studies. Any disagreements were resolved through discussion internally among the investigators. The search strategy followed the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) flow diagram (7), and the protocol of this systematic review was registered on PROSPERO (CRD42021224932, 7 December 2020).
Data extraction
Data were extracted independently by ZA and MH into a pre-defined Microsoft Excel sheet (Microsoft Corp, Armonk, NY, USA) and then compared. For each study, the following items were recorded: (i) the study design and country of origin; (ii) the study’s population: number of patients with AK, age, sex, comorbidities, AK lesion location, treatment, and AK lesion quantity and severity before and after treatment; (iii) HRQoL questionnaire used and the outcome overall, before, and after treatment.
Risk of bias assessment
The risk of bias was assessed independently by ZA and MH using appropriate tools: the Newcastle-Ottawa Scale (NOS) for observational studies and adapted NOS for cross-sectional studies, the Joanna Briggs Institute Critical Appraisal Checklist (JBI) for case series, the National Institutes of Health quality assessment tool (NIH) for before–after (Pre–Post) studies with no control group, and the Cochrane Risk of Bias tool 2 (RoB 2) for randomized controlled trials (8).
NOS and adapted NOS assess 3 domains: selection, comparability, and outcome/exposure, with scores ≥ 7 indicating a high-quality study. The JBI uses 10 items to assess internal validity, with studies rated high quality if > 80% of items are endorsed and moderate quality if 50–80% are endorsed. NIH has 12 items categorizing studies as having poor, fair, or good quality. RoB 2 assesses 5 domains: random sequence generation, allocation, blinding, incomplete data, and selective reporting as low, unclear, or high risk.
Results
Literature search
The search strategy yielded 639 articles (EMBASE including PsychINFO: 448; PubMed: 120; Cochrane: 63) (Fig. 1, Table SI). After removing duplicates, 410 papers were screened, 114 full-texts were assessed, with 37 studies meeting the inclusion criteria. The included studies comprised 37 unique study populations with a total of 9,326 participants, predominantly males. Most studies were based in Europe (70%); others were from North America (14%), South America (11%), Australia (5%), and Asia (4%), while none were from Africa. AK severity was characterized by the amount, size, and type (macule, hyperkeratotic, or hypertrophic), and often graded using Olson’s clinical classification scheme (O I–III) according to their thickness and degree of hyperkeratosis (9). Nevertheless, most of the lesions were O I/II and were mainly located on the face or scalp. Extracted data from all included studies are presented in Tables I and II, with full study characteristics and references provided in Table SII.
Disease-specific questionnaires
The AKQoL tool was the second most used tool (35%, 13 out of 37 included studies) to measure the HRQoL in patients with AK (Table III). The tool is a disease-specific questionnaire validated for AK and comprises 3 domains: (i) Emotion, (ii) Function, and (iii) Control. It has a maximum score of 27 in which a higher score represents a more severe impairment on HRQoL (10). Among studies that reported the overall mean score of AKQoL, the lowest score was 4.22 and the highest score was 17 (Fig. 2A, Table III). The tool identified a significant association between being female and a worse HRQoL impairment (11–13). Additionally, older patients had a lower AKQoL-score compared with younger patients with AK (12, 13). A correlation between a higher AKQoL score and a history of skin cancer, cardiovascular diseases, or the number of comorbidities was significant according to Neri et al. and showed a trend in Tennvall et al. (12, 13). AKQoL identified a significant impact on HRQoL in individuals with severe actinic damage (12, 14, 15).
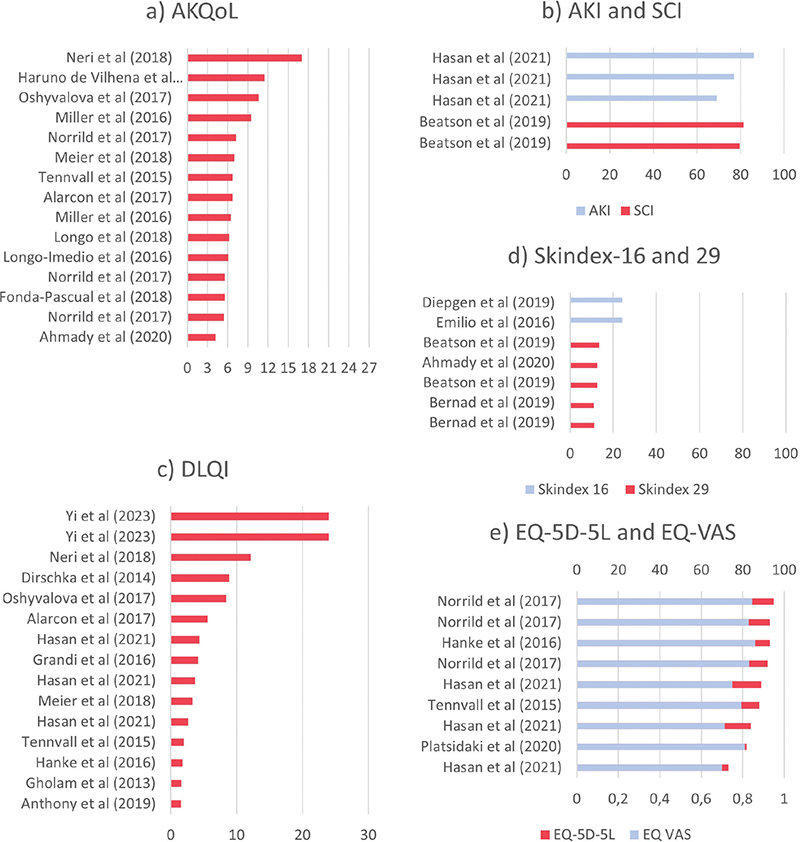
Fig. 2. Studies that reported HRQoL mean scores in their overall population by disease-specific questionnaires. (A) AKQoL, (B) AKI and SCI; dermatology-specific questionnaires:(C) DLQI, (D) Skindex-16/29; generic questionnaire: (E) EQ-5D-5L and EQ-VAS. Multiple bars for the same study represent different subpopulations reported within the study.
The Skin Cancer Index (SCI) is a disease-specific validated questionnaire designed to assess HRQoL in patients with skin cancer (16). It has been used in 3 studies (8%, 3 out of 37 included studies) to assess HRQoL in patients with AK. The tool comprises 3 domains: (i) Emotion, (ii) Social, and (iii) Appearance. The scoring system is according to a 5-point Likert scale with a score ranging from 0–100, where the lower scores represent a severe impairment in HRQoL. Among studies that reported the overall mean score of SCI, the lowest score was 79.46 and the highest score was 81.28 (Fig. 2B).
The Actinic Keratosis Index (AKI) is an exploratory questionnaire based on the SCI. It was developed and piloted for 1 clinical study (17). The lowest overall mean score was 69.07 and the highest score was 86.2 (Fig. 2B).
Dermatology-specific questionnaires
The DLQI was used in 16 studies, thus making it the most frequent tool to measure HRQoL in patients with AK (43%, 16 out of 37 included studies) (Table III). The tool is a dermatology-specific questionnaire that quantifies the burden a dermatosis has on the patient’s HRQoL for the past 7 days (18). DLQI consists of 10 items covering 6 domains: (i) Symptoms and feelings, (ii) Daily activities, (iii) Leisure, (iv) Work and school, (v) Personal relationships, and (vi) Treatments. The score ranges from 0 to 30, where a higher score denotes a greater impairment in HRQoL. The interpretation of DLQI scores on HRQoL is as follows: none (score 1), small (scores 2–5), moderate (score 6–10), very large (scores 11–20), and extremely large (scores 21–30) (19).
Among studies that reported the overall mean score of DLQI, the lowest mean score was 1.55 and the highest score was 24 (Fig. 2C, Table III). The tool found a small impairment in HRQoL in 8 out of the 16 included studies, (Fig. 3A). Two out of 3 studies found no significant differences between female and male sexes regarding HRQoL (12, 13, 20). A study by Tennvall et al. found that severe actinic damage, defined by the presence of multiple lesions or by dominantly consisting of O II/III AK lesions, previous SCC, or immunosuppressive treatment had a significant negative impact on HRQoL when measured using DLQI (12). However, they found that DLQI was not sensitive to sex and age.
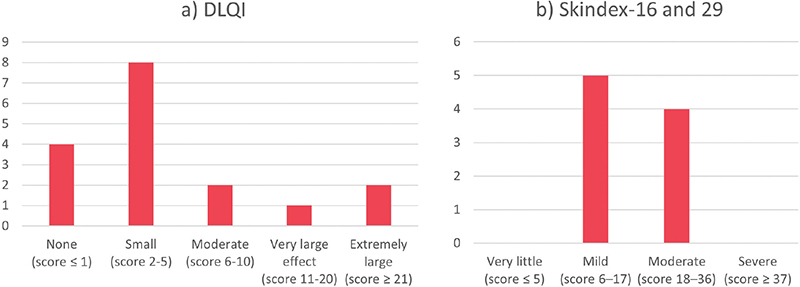
Fig. 3. Categorization of the overall mean scores for HRQoL in patients with actinic keratosis. By (A) DLQI total score interpretation: 0–1, no effect at all on patient’s life; 2 –5, small effect on patient’s life; 6–10, moderate effect on patient’s life; 11–20, very large effect on patient’s life; 21–30, extremely large effect on patient’s life; (B) Skindex-16 and 29 tool total score interpretation: ≤5, very little effect; 6–17, mild effect; 18–36, moderate effect; ≥ 37, severe effect.
The Skindex-16, -17 or -29 has been selected to assess HRQoL in patients with AK in 3, 1, and 6 studies, respectively (overall in 37%, 10 out of 37 included studies). The tools are dermatology-specific, evaluating HRQoL impairment up to the last 4 weeks of the patient’s life. Skindex-16 and -29 covers 3 domains: (i) Symptom, (ii) Emotion, and (iii) Functioning (21, 22), while Skindex-17 covers 2 domains: (i) Symptom and (ii) Psychosocial (23). Skindex-16 has a score scale ranging from 0 to 100; the higher the score the greater the impact on HRQoL. In Skindex-17 and -29 each item is rated according to a 3- and 5-point Likert scale, respectively; a higher point indicates a greater impact on HRQoL. The Skindex scores can be interpreted as the following effect on HRQoL: very little (score 5), mild (scores 6–17), moderate (scores 18–36), and severe (scores ≥ 37) (24, 25).
Among studies reporting the overall mean scores of Skindex-16 and Skindex-29, the lowest mean score on Skindex-16 was 24.25 and the highest score was 24.3 and for Skindex-29 the lowest mean score was 10.5 and the highest score was 13.43 (Fig. 2D, Table III). The Skindex-16 and 29 tools found mild to moderate HRQoL impairment, (Fig. 3B). Skindex-16 captured impairment in HRQoL when comparing patients with and without scarring (26). In Skindex-17, the HRQoL impairment was dominantly captured by the “symptom” domain and there was no correlation between age, sex, or level of education (27).
Generic questionnaires
The EuroQoL 5-dimensions 5-levels (EQ-5D-5L) and EuroQol Visual Analogue Scale (EQ-VAS) are generic standardized questionnaires that measure HRQoL (28). The tools have been included in 7 studies (19%, 7 out of 37 included studies) to assess the HRQoL in patients with AK. The EQ-5D-5L covers the following 5 dimensions: (i) Mobility, (ii) Self-care, (iii) Usual activities, (iv) Pain/discomfort, and (v) Anxiety/depression. Responses are converted into a single index value using country-specific value sets derived from general population preferences. The index ranges from 1 (full health) to 0 (death) and to negative values for health states considered worse than death. The index score is commonly used to calculate the quality-adjusted life years to give a health-economical evaluation of a healthcare intervention. The EQ-VAS lets the patient rate their current health state from 0–100, where 100 is the best imaginable health state and 0 is the worst.
Among studies that reported the overall mean scores the lowest EQ-5D-5L mean score was 0.73, the highest score was 0.95, the lowest mean score of EQ-VAS was 69.92 and the highest score was 86 (Fig. 2E, Table III). The EQ-5D-5L and EQ-VAS tools found a significant increase in HRQoL impairment in patients with AK that had comorbidities, current, or a history of SCC (12,29). However, there were no significant differences found between female and male sexes (12, 14, 29), other than for females with AK and SCC (29). The EQ-VAS recognized a change in HRQoL impairment in patients who had multiple and severe AK lesions (12, 14). Not all studies reported which EQ-5D value sets they used and, among those that did, the value sets differed across studies (Tables I and II).
The 20-Item Short Form Survey (SF-20) is likewise a generic questionnaire originally developed for the study of patients with chronic conditions (30). It consists of 6 domains: (i) Physical functioning, (ii) Role functioning, (iii) Social functioning, (iv) Mental health, (v) Current health perceptions, and (vi) Pain. The overall score is transformed to a 0 to 100 scale.
One study used SF-20 to measure HRQoL in patients with AK and field cancerization. The questionnaire found that with more severe field cancerization there was a decrease in physical, role, and social functioning and current health (31).
Impact of treatments on HRQoL
Of the 37 studies, 22 (59%) were interventional. Treatments included topical therapies (16 out of 22 studies, 73%: 5-Fluoruracil ± salicylic acid; diclofenac ± hyaluronic acid; ingenol mebutate; imiquimod; tretinoin; sunscreen), light therapy (8 out of 22 studies, 36%: photodynamic therapy ± methyl amino levulinate or indole-3 acetic acid), surgical (4 out of 22 studies, 18%: excision; cryosurgery ± ingenol mebutate, surgery ± photodynamic therapy), and systemic therapy (1 out of 22 studies, 0.05%: isotretinoin) (Table II). The HRQoL was initially more impaired immediately post-treatment, regardless of treatment type or PROM used, but improved after weeks of follow-up, often exceeding baseline levels (13, 27, 32–35) (Fig. S1). AK lesion counts also decreased (Table II).
Risk of bias
The number of quality studies were classified according to risk of bias assessment and showed 1/3 cohort studies were high quality (NOS), 4/8 cross-sectional studies were high quality (adapted NOS), 1/15 case series were high quality and 7/15 moderate quality (JBI), and interventional studies without control scored > 66% (NIH tool). Among RCTs, 3/7 had low risk, 3/7 some risk, and 1/7 high risk (RoB 2). Overall, 64% of studies were moderate to high quality. Full details are given in Tables SIII–SVII.
DISCUSSION
This systematic review provides a comprehensive overview of 37 studies that reported HRQoL measures in patients with AK. More than half of the studies were interventional and provided the HRQoL measures before and after intervention. The PROMs used in the studies to assess HRQoL in patients with AK were disease-specific (3 tools), dermatology-specific (3 tools), and generic questionnaires (2 tools).
Among the disease-specific questionnaires, only the AKQoL was a validated AK-specific HRQoL tool, as the SCI was developed and validated for measuring HRQoL in patients with skin cancer and not AK, and the AKI was an exploratory non-validated questionnaire based on the SCI. The AKQoL was also the most frequently used disease-specific questionnaire. It was able to differentiate between age, sex, severity of AK, and those who had comorbidities or a history of skin cancer. The dermatology-specific and generic questionnaires supported the findings, but they could not necessarily distinguish between all the mentioned subpopulations. However, it should be highlighted that only a few studies investigated whether an HRQoL tool could identify subpopulations.
Compared with our results, a literature review by Chernyshov et al. on the HRQoL in patients with melanoma and NMSC found that most studies used the disease-specific questionnaires, while the most frequently used dermatology-specific and generic questionnaires were the DLQI and SF-36, respectively (6). However, unlike our findings, they found only 1 study that used multiple questionnaires simultaneously. This discrepancy may be explained by the fact that melanoma and NMSC have a larger number of validated disease-specific HRQoL tools available, whereas only 1 such instrument exists for AK. Consequently, AK studies are more likely to include multiple questionnaires simultaneously in order to capture different HRQoL domains. Despite this limitation, the AKQoL was used in nearly as many studies as the DLQI in our review. Overall, the pattern across studies suggests that disease-specific HRQoL instruments are generally preferred when available. The EADV Task Forces on QoL and Patient-Oriented Outcomes, Melanoma and NMSC recommend using validated disease-specific questionnaires to unveil any fear related to having skin cancer combined with dermatology-specific questionnaires to cover any factors related to the symptoms of other major skin issues (6). This is also supported by correlation analysis, which suggests that the disease-specific AKQoL, dermatology-specific DLQI, and generic questionnaire EQ-5D-5L/EQ-VAS can be complementary as they focus on different aspects (12). Each category of instrument serves a distinct purpose: disease-specific tools tend to be the most sensitive to clinical change, dermatology-specific tools such as the DLQI allow comparisons across skin conditions but may not fully reflect AK-specific burden, as AK patients were not included in its original development, and generic instruments like the EQ-5D facilitate health-economic evaluations and cross-disease comparisons. While EQ-5D has been shown to detect differences between patient subgroups, it is prone to ceiling effects in populations with mild disease. Ultimately, the choice of instrument should be guided by the research question and target population (36).
The measurement of HRQoL in daily clinical practice can be valuable for identifying specific patient subpopulations, assessing the outcome of interventions, monitoring patient care, enhancing clinician–patient communication, and raising awareness of the skin disease burden (5). As with any other chronic condition, the patients with AK HRQoL differ with time and in response to treatment. Overall, we found that in interventional studies all PROMs captured changes in HRQoL due to local skin reactions as well as improvements in disease after treatment, demonstrating their responsiveness to both adverse events and treatment effectiveness. In a recent systematic review, Grada et al. found that simpler and shorter topical therapies were associated with an overall improvement in HRQoL (37).
This systematic review has some limitations. Most studies were interventional and very few compared HRQoL in patients with and without AK. This hindered the conduction of a meta-analysis and whether an HRQoL measurement is different in a matched control group. Few studies explored potential factors that could influence the HRQoL in patients with AK, such as age, sex, disease duration, socioeconomic status, and comorbidities. The strength of our study was the extensive data extraction and inclusion of all questionnaires used to measure HRQoL to give a comprehensive overview of HRQoL impairment in patients with AK. Furthermore, our review covered all geographical continents where AK is most prevalent: Europe, Australia, North and South America, and Asia. Future data should focus on relative HRQoL (AK vs other skin diseases), and on validating appropriate cut-off values for the HRQoL impairment severity.
Conclusion
The DLQI and AKQoL were widely used to measure HRQoL in AK patients. The validated disease-specific questionnaire for AK is the AKQoL, which was reported to identify subpopulations with reduced HRQoL: females, younger patients, comorbid patients, and those who have developed skin cancer. Generally, all HRQoL tools were able to measure changes in HRQoL during treatment. The wide range of mean DLQI and AKQoL scores across studies suggests substantial heterogeneity in AK severity among study populations.
ACKNOWLEDGEMENTS
Data availability statement: The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- Malvehy J, Stratigos AJ, Bagot M, Stockfleth E, Ezzedine K, Delarue A. Actinic keratosis: current challenges and unanswered questions. J Eur Acad Dermatol Venereol 2024; 38(Suppl. 5): 3–11. https://doi.org/10.1111/jdv.19559
- de Oliveira ECV, da Motta VRV, Pantoja PC, Ilha CS de O, Magalhães RF, Galadari H, Leonardi GR. Actinic keratosis – review for clinical practice. Int J Dermatol 2019; 58(4): 400–407. https://doi.org/10.1111/ijd.14147
- Esmann S. Patients’ perspectives on actinic keratosis. Curr Probl Dermatol (Switzerland) 2015; 46: 8–13. https://doi.org/10.1159/000366530
- Esmann S, Jemec GBE. Management of actinic keratosis patients: a qualitative study. J Dermatol Treat 2007; 18(1): 53–58. https://doi.org/10.1080/09546630601028737
- Finlay AY, Salek MS, Abeni D, Tomás-Aragonés L, van Cranenburgh OD, Evers AWM, et al. Why quality of life measurement is important in dermatology clinical practice. J Eur Acad Dermatol Venereol 2017; 31: 424–431. https://doi.org/10.1111/jdv.13985
- Chernyshov PV, Lallas A, Tomas-Aragones L, Arenbergerova M, Samimi M, Manolache L, et al. Quality of life measurement in skin cancer patients: literature review and position paper of the European Academy of Dermatology and Venereology Task Forces on Quality of Life and Patient Oriented Outcomes, Melanoma and Non-Melanoma Skin Cancer. J Eur Acad Dermatol Venereol 2019; 33: 816–827. https://doi.org/10.1111/jdv.15487
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021; 372. https://doi.org/10.1136/bmj.n71
- Ma LL, Wang YY, Yang ZH, Huang D, Weng H, Zeng XT. Methodological quality (risk of bias) assessment tools for primary and secondary. Mil Med Res. Military Medical Research 2020; 7: 1–11. Available from: https://mmrjournal.biomedcentral.com/track/pdf/10.1186/s40779-020-00238-8.pdf
- Olsen EA, Lisa Abernethy M, Kulp-Shorten C, Callen JP, Glazer SD, Huntley A, et al. A double-blind, vehicle-controlled study evaluating masoprocol cream in the treatment of actinic keratoses on the head and neck. J Am Acad Dermatol; 1991; 24: 738–743. https://doi.org/10.1016/0190-9622(91)70113-G
- Esmann S, Vinding GR, Christensen KB, Jemec GBE. Assessing the influence of actinic keratosis on patients’ quality of life: the AKQoL questionnaire. Br J Dermatol 2013; 168(2): 277–283. https://doi.org/10.1111/bjd.12036
- Longo I, Serra-Guillén C. Quality of life, behaviour and attitudes towards actinic keratosis in SpaIn: the PIQA study. Actas Dermosifiliogr 2018; 109: 331–339. https://doi.org/10.1016/j.ad.2018.01.004
- Tennvall GR, Norlin JM, Malmberg I, Erlendsson AM, Hædersdal M. Health related quality of life in patients with actinic keratosis: an observational study of patients treated in dermatology specialist care in Denmark. Health Qual Life Outcomes 2015; 13: 1–9. https://doi.org/10.1186/s12955-015-0295-4
- Neri L, Peris K, Longo C, Calvieri S, Frascione P, Parodi A, Actinic Keratosis – TReatment Adherence INitiative (AK-TRAIN) study group. Physician–patient communication and patient-reported outcomes in the actinic keratosis treatment adherence initiative (AK-TRAIN): a multicenter, prospective, real-life study of treatment satisfaction, quality of life and adherence to topical field-directed therapy or the treatment of actinic keratosis in Italy. J Eur Acad Dermatol Venereol 2019; 33: 93–107. https://doi.org/10.1111/jdv.15142
- Norrlid H, Norlin JM, Holmstrup H, Malmberg I, Sartorius K, Thormann H, et al. Patient-reported outcomes in topical field treatment of actinic keratosis in Swedish and Danish patients. J Dermatolog Treat 2018; 29: 68–73. https://doi.org/10.1080/09546634.2017.1329514
- Vis K, Waalboer-Spuij R, Snels DGCTM, Hollestein LM. Validity and reliability of the Dutch adaptation of the Actinic Keratosis Quality of Life Questionnaire (AKQoL). Dermatology 2018; 234: 60–65. https://doi.org/10.1159/000489118
- Rhee JS, Matthews BA, Neuburg M, Logan BR, Burzynski M, Nattinger AB. The skin cancer index: clinical responsiveness and predictors of quality of life. Laryngoscope 2007; 117: 399–405. https://doi.org/10.1097/MLG.0b013e31802e2d88
- Hasan ZU, Ahmed I, Matin RN, Homer V, Lear JT, Ismail F, et al. Topical treatment of actinic keratoses in organ transplant recipients: a feasibility study for SPOT (Squamous cell carcinoma Prevention in Organ transplant recipients using Topical treatments). Br J Dermatolo 2022; 18: 324–337. https://doi.org/10.1111/bjd.20974
- Finlay AY, Khan GK. Dermatology Life Quality Index (DLQI): a simple practical measure for routine clinical use. Clin Exp Dermatol 1994; 19: 210–216. https://doi.org/10.1111/j.1365-2230.1994.tb01167.x
- Hongbo Y, Thomas CL, Harrison MA, Sam Salek M, Finlay AY. Translating the science of quality of life into practice: what do dermatology life quality index scores mean? J Investig Dermatol 2005; 125: 659–664. https://doi.org/10.1111/j.0022-202X.2005.23621.x
- Tejada C dos S, Mendoza-Sassi RA, Almeida Jr HL de, Figueiredo PN, Tejada VF dos S. Impact on the quality of life of dermatological patients in southern Brazil. An Bras Dermatol 2011; 86: 1113–1121. https://doi.org/10.1590/s0365-05962011000600008
- Chren MM, Lasek RJ, Sahay AP, Sands LP. Measurement properties of Skindex-16: a brief quality-of-life measure for patients with skin diseases. J Cutan Med Surg 2001; 5: 105–110. https://doi.org/10.1007/BF02737863
- Chren MM, Lasek RJ, Flocke SA, Zyzanski SJ. Improved discriminative and evaluative capability of a refined version of Skindex, a quality-of-life instrument for patients with skin diseases. Arch Dermatol 1997; 133: 1433–1440.
- Nijsten TEC, Sampogna F, Chren MM, Abeni DD. Testing and reducing Skindex-29 using Rasch analysis: Skindex-17. J Invest Dermatol 2006; 126: 1244–1250. https://doi.org/10.1038/sj.jid.5700212
- Chren MM. The Skindex instruments to measure the effects of skin disease on quality of life. Dermatol Clin 2012; 30: 231–236. https://doi.org/10.1016/j.det.2011.11.003
- Nijsten T, Sampogna F, Abeni D. Categorization of Skindex-29 scores using mixture analysis. Dermatology 2009; 218(2): 151–154. https://doi.org/10.1159/000182253
- Shergill B, Zokaie S, Carr AJ. Non-adherence to topical treatments for actinic keratosis. Patient Prefer Adherence 2013; 8: 35–41. https://doi.org/10.2147/PPA.S47126
- Waalboer-Spuij R, Holterhues C, Van Hattem S, Schuttelaar MLA, Gaastra MTW, Kuijpers DIM, et al. Patient perception of imiquimod treatment for actinic keratosis and superficial basal cell carcinoma in 202 patients. Dermatology 2015; 231: 56–62. https://doi.org/10.1159/000381420
- EuroQol Research Foundation. EQ-5D-5L User Guide 2019 (Version 3.0). Available from: EuroQol Research Foundation. EQ-5D-5L User Guide 2019 (Version 3.0).
- Philipp-Dormston WG, Müller K, Novak B, Strömer K, Termeer C, Hammann U, NMSC-QoL Study Group. Patient-reported health outcomes in patients with non-melanoma skin cancer and actinic keratosis: results from a large-scale observational study analysing effects of diagnoses and disease progression. J Eur Acad Dermatol Venereol 2018; 32: 1138–1146. https://doi.org/10.1111/jdv.14703
- RAND Health Care 20-Item Short Form Survey (SF-20) [Internet] [accessed 2025-01-29]. Available from: https://www.rand.org/health-care/surveys_tools/mos/20-item-short-form.html
- Thomas SI, Taylor MA, Plampton K, Sharma D, Samson KK, Wysong A, et al. Assessing quality of life in field cancerization: an institutional cohort analysis. Dermatol Surg 2024; 50: 1202–1203. https://doi.org/10.1097/DSS.0000000000004311
- Anthony E, Lun K, Smith SD. Full face ingenol mebutate for actinic keratosis: patient perspective. Int J Dermatol 2019; 58: e6–e9. https://doi.org/10.1111/ijd.14263
- Hanke WC, Norlin JM, Mark Knudsen K, Larsson T, Stone S. Quality of life in treatment of AK: treatment burden of ingenol mebutate gel is small and short lasting. J Dermatol Treat 2016; 27: 450–455. https://doi.org/10.3109/09546634.2016.1160024
- Gholam P, Kroehl V, Enk AH. Dermatology life quality index and side effects after topical photodynamic therapy of actinic keratosis. Dermatology 2013; 226: 253–259. https://doi.org/10.1159/000349992
- Răducu L, Avino A, Purtan RP, Balcangiu-Stroescu AE, Balăn DG, Timofte D, et al. Quality of life in patients with surgically removed skin tumors. Medicina (Lithuania) 2020; 56: 3–9. https://doi.org/10.3390/medicina56020066
- Prinsen CAC, de Korte J, Augustin M, Sampogna F, Salek SS, Basra MKA, EADV Taskforce on Quality of Life. Measurement of health-related quality of life in dermatological research and practice: outcome of the EADV Taskforce on Quality of Life. J Eur Acad Dermatol Venereol 2013; 27: 1195–203. https://doi.org/10.1111/jdv.12090
- Grada A, Feldman SR, Bragazzi NL, Damiani G. Patient-reported outcomes of topical therapies in actinic keratosis: a systematic review. Dermatol Ther 2021; 34: 1–10. https://doi.org/10.1111/dth.14833