ORIGINAL REPORT
Clinical and Pathological Characteristics of Skin Cancer Patients at Southern Medical University, Guangzhou, China
Siqi LI#, Jingwen ZOU#, Rongyi CHEN and Lian ZHANG
Dermatology Hospital, Southern Medical University, Guangzhou, Guangdong Province, China
#These authors contributed equally to this work.
The incidence of skin cancer is increasing in China, yet large-scale clinical and pathological data from China remain scarce. A retrospective analysis was conducted of 648 histopathologically confirmed skin cancer cases diagnosed at the Dermatology Hospital, Southern Medical University from January 2020 to July 2023. Patient demographics, tumour distribution, histological subtypes, and treatment approaches were evaluated. Squamous cell carcinoma (31.9%) and basal cell carcinoma (31.2%) were the most prevalent, followed by cutaneous lymphomas (13.9%), dendritic cell and histiocytic neoplasms (13.3%), and melanoma (9.7%). Over half of the patients (53.5%) were aged ≥ 60 years. basal cell carcinoma and squamous cell carcinoma primarily involved the head and neck, while acral sites were dominant in melanoma (79.4%). Most cases of both basal cell carcinoma and squamous cell carcinoma were managed surgically, with surgical treatment performed in 95.5% of basal cell carcinomas and 94.2% of squamous cell carcinomas. Skin cancers in China predominantly affect older adults and commonly involve sun-exposed areas. Basal cell carcinoma is typically managed surgically due to its low-risk nature, while squamous cell carcinoma demonstrates clinical heterogeneity, warranting varied treatment strategies. The higher proportion of cutaneous lymphomas among younger patients highlights the need for age-specific diagnostic and therapeutic considerations.
SIGNIFICANCE
Skin cancer is increasingly affecting people in China, but large clinical studies have been scarce. By reviewing 648 patients from a specialised dermatology hospital, this study shows clear differences in how various skin cancers occur across age groups and body sites. While most cases affect older adults, some cancers appear much earlier in life, calling for earlier attention and screening. These results improve understanding of skin cancer patterns in China and may guide prevention, early detection, and treatment strategies to reduce disease burden.
Key words: skin tumour; basal cell carcinoma; squamous cell carcinoma; cutaneous lymphoma; melanoma.
Citation: Acta Derm Venereol 2026; 106: adv44550. DOI: https://doi.org/10.2340/actadv.v106.44550.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Aug 5, 2025. Accepted after revision: Jan 9, 2026. Published: Feb 17, 2026.
Corr: Rongyi Chen and Lian Zhang, Dermatology Hospital, Southern Medical University, Guangzhou, China. E-mails: Rongyichen_smu@smu.edu.cn; zhanglian1@smu.edu.cn
Competing interests and funding: The authors have no conflicts of interest to declare.
This work was supported by the National Natural Science Foundation of China (82303064), Young Talent Project of Guangzhou Association for Science and Technology (QT202440), Guangdong Medical Science and Technology Research Foundation (A2023491), and Guangzhou Science and Technology Foundation (SL2024A04J02622).
INTRODUCTION
The incidence of skin cancer has been steadily increasing worldwide, driven largely by population ageing and environmental factors such as ultraviolet (UV) radiation exposure (1–4). In China, a similar upward trend is evident, especially in regions with high UV intensity (5, 6). Non-melanoma skin cancers (NMSC), including basal cell carcinoma (BCC) and squamous cell carcinoma (SCC), account for the majority of cases. However, cutaneous melanoma and cutaneous lymphomas, though less common, carry significant clinical burden due to their aggressive behaviour and potential for poor outcomes.
To date, most epidemiological data on skin cancers originate from studies in Western countries (2, 7), primarily involving White populations. Large-scale, clinicopathological studies focusing on Asian patients – particularly from mainland China – remain limited. Given the differences in genetic background, environmental exposure, and healthcare access, it is crucial to establish region-specific data to improve diagnosis, treatment strategies, and public health awareness.
In this study, we conducted a retrospective analysis of 648 patients diagnosed with skin malignancies at our hospital, one of the leading academic and research-oriented dermatology institutions in China (Table I). This study aimed to provide a comprehensive clinicopathological analysis of skin cancer patients in China, focusing on tumour distribution, pathological subtypes, and treatment strategies, in order to address the current gap in large-scale data from Asian populations.
MATERIALS AND METHODS
Study design and setting
This was a single-centre, retrospective observational study conducted at the Dermatology Hospital, Southern Medical University. The study was designed to assess the clinical and pathological characteristics of patients diagnosed with skin malignancies over a 3.5-year period, from January 2020 to July 2023.
Patient selection
A total of 648 patients were included in the final analysis. Inclusion criteria were: (i) patients of any age and sex diagnosed with malignant cutaneous tumours; (ii) diagnosis confirmed by histopathological examination performed by 2 board-certified dermatopathologists; and (iii) complete clinical and pathological records available for review. Exclusion criteria included: (i) uncertain or provisional diagnoses without histological confirmation; and (ii) incomplete medical records or biopsy samples insufficient for accurate classification.
Patient population and referral system
The Dermatology Hospital, Southern Medical University (Guangdong Provincial Dermatology Hospital) is a tertiary referral centre and a leading specialized hospital for dermatological diseases in South China. It serves as the “Guangdong Provincial Center for Dermatology and Venereology Control” and the coordinating hospital of the “Guangdong Integrated Dermatology Prevention and Treatment Alliance”. As a designated referral centre, the hospital receives patients not only from Guangdong Province but also from other parts of South China, including both common and complex dermatologic conditions.
Annually, the hospital manages more than 1 million outpatient visits, among which the skin tumour subspecialty clinic (1 of the 14 key specialty clinics in the hospital) accounts for over 63,000 visits, covering more than 30 types of common and rare skin tumours. Therefore, the study cohort includes both routine and complicated cases, ensuring broad representativeness of skin cancer patients in the region. Potential referral-related selection bias was acknowledged and discussed in the limitations.
Data collection
Demographic and clinical data were extracted from the hospital’s electronic medical records and pathology archives. The variables collected included sex, age at diagnosis, anatomic site of the lesion, clinical presentation, tumour subtype (based on pathology), treatment modality (surgical vs non-surgical), and, where applicable, histopathologic grading (e.g., Clark level and Breslow thickness for melanoma; WHO classification for cutaneous lymphomas).
For basal cell carcinoma, histological subtypes were stratified into low-risk (e.g., nodular, superficial, infundibulocystic) and high-risk (e.g., infiltrative, micronodular, basosquamous, morpheaform) categories. In patients with multifocal tumours or mixed histological features, classification was based on the highest-risk subtype present. For cutaneous lymphomas, diagnostic classification followed the WHO-EORTC 2018 guidelines (8).
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics, version 27.0 (IBM Corp, Armonk, NY, USA). Descriptive statistics were used to summarize patient demographics and tumour characteristics. Continuous variables were expressed as mean ± standard deviation (SD), and categorical variables were presented as frequencies and percentages. No inferential or comparative statistical tests were conducted, as the study was descriptive in nature.
Ethical considerations
The study protocol was approved by the Institutional Review Board of the Dermatology Hospital, Southern Medical University. This research was conducted in accordance with the Declaration of Helsinki. Patient data were anonymized and de-identified prior to analysis to ensure confidentiality.
RESULTS
Non-melanoma skin cancer: basal cell carcinoma and squamous cell carcinoma
A total of 648 patients with skin cancers were included in this study, comprising 360 males (55.6%) and 288 females (44.4%), with an overall male-to-female ratio of approximately 1.25:1. Among them, non-melanoma skin cancer (NMSC) accounted for the majority, with 202 cases of basal cell carcinoma and 207 cases of squamous cell carcinoma (accounting for 31.2% and 31.9%, respectively, Fig. 1). The mean age at diagnosis was 64.49 years (SD 13.45) for BCC and 70.53 years (SD 13.22) for SCC, consistent with the global trend of higher incidence in the elderly population (3, 4, 9, 10) (Table I). Both BCC and SCC predominantly affected sun-exposed areas, particularly the head and neck, with 91.1% of BCC and 48.8% of SCC lesions located in these regions (Table II).
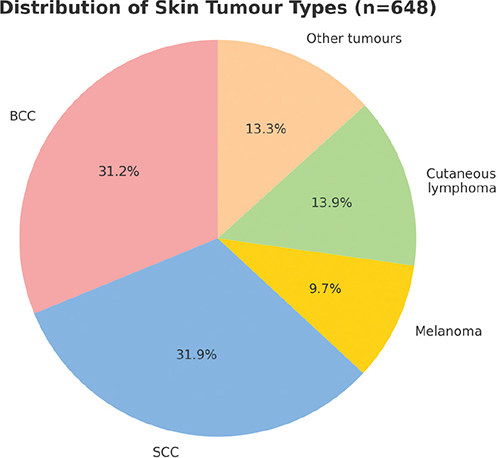
Fig. 1. Distribution of skin tumour types (n = 648).
The pathological subtypes of BCC were primarily low-risk, with nodular BCC comprising 74.8% of cases. High-risk subtypes, such as infiltrative and basosquamous variants, were less common (21.8%). Most BCC patients (95.5%) underwent surgical treatment (see Table I). In contrast, SCC showed a more heterogeneous clinical course, with 91.8% classified as invasive SCC and 8.2% SCC in situ. Regarding management, 95.2% of SCC patients underwent surgical interventions, and 4.8% received immunotherapy. Two patients had immunosuppressive conditions (renal transplantation and post-chemotherapy for haematologic malignancy). Among the 27 patients with genital SCC, 5 were HPV-positive, 6 HPV-negative, and 16 had unknown HPV status (see Table II).
Compared with Western data, where BCC is more prevalent than SCC (11), our cohort showed a near-equal distribution. This may be attributed to our inclusion of SCC in situ, which is less consistently reported in other datasets. It may also reflect selection bias from the single-centre design and limited study period. In addition, Asian populations are known to engage more consistently in physical sun-protective behaviours than Western populations (12, 13), which may contribute to the relatively lower prevalence of BCC observed in our cohort.
Malignant melanoma
A total of 63 patients (9.7%) were diagnosed with malignant melanoma (MM) (Table III). Unlike White populations where superficial spreading melanoma is most common, 79.4% of our cases were acral lentiginous melanoma, consistent with patterns observed in Asian populations (14–19). The mean age at diagnosis was 55.7 years (SD 17.23), with a higher prevalence in females (M:F ratio 1:1.74), a trend that diverges from male-dominant patterns reported in Western cohorts (16, 20).
Pathological staging showed that Clark level IV was most frequent (33.3%), while 30.2% of tumours had a Breslow thickness ≤ 1 mm. However, a significant proportion (35%) exhibited Breslow thickness > 2 mm, suggesting delayed diagnosis. Notably, acral sites – typically hidden and less likely to be screened – accounted for the majority of lesions, highlighting a need for improved public and physician awareness in non-sun-exposed anatomical regions (see Table III).
Cutaneous lymphoma
Cutaneous lymphomas were identified in 90 patients (13.9%), an unusually high proportion compared with both domestic and international studies (5, 6, 21). The overall mean age at onset was 42.79 years (SD 21.8). T- and NK-cell lymphomas were predominant (93.3%), with mycosis fungoides (MF) as the most frequent subtype (55.6%), followed by lymphomatoid papulosis and primary cutaneous anaplastic large-cell lymphoma. EBV-associated T/NK-Cell lymphoproliferative diseases accounted for 10% of cutaneous lymphoma, which included hydroa vacciniforme-like lymphoproliferative disorder, hydroa vacciniforme-like T-cell lymphoma, extranodal NK/T-cell lymphoma, nasal type, and EBV-positive T/NK-cell lymphoproliferative disorders. All 9 patients were EBV-positive (Tables IV–VI). Compared with existing literature, particularly from Western registries (21–23), our data suggest a younger age of onset and a broader spectrum of lymphoma subtypes. These findings indicate potential regional or ethnic differences in pathogenesis, diagnosis, or referral patterns.
Other tumours
In addition to skin cancers and cutaneous lymphomas, 86 patients (13.3% of the total cohort) were diagnosed with dendritic cell and histiocytic neoplasms. The most frequent subtype was juvenile xanthogranuloma (82.6%), followed by Rosai–Dorfman disease (11.6%), blastic plasmacytoid dendritic cell neoplasm (BPDCN) (2.3%), and several rare entities, including multicentric reticulohistiocytosis and Langerhans cell histiocytosis (LCH) (Table VII). The mean age at onset was 19.62 years (SD 18.03), indicating a marked predominance among children and young adults. Lesions were most commonly located on the head and neck (50.0%). Most of these tumours represented benign or indolent proliferations, whereas truly malignant dendritic cell neoplasms such as BPDCN and LCH were rare. According to the WHO–EORTC 2018 classification, these tumours are not categorized as lymphomas and were therefore analysed separately in our study.
DISCUSSION
The global incidence of skin malignancies has been increasing steadily over the past several decades, largely driven by factors such as population ageing, heightened UV exposure (2, 24), and improved diagnostic awareness. However, most of the available epidemiological data originate from developed countries, particularly those in Europe, North America, and Australia, where the majority of patients are of White ethnicity (9). Consequently, there is a significant gap in data representing Asian populations. In our study, we found that the distribution and characteristics of non-melanoma skin cancers (NMSC), including basal cell carcinoma and squamous cell carcinoma, were generally consistent with those reported in Western cohorts, especially in terms of anatomical distribution and age prevalence. A small subset of SCC cases in our cohort occurred in immunocompromised individuals, such as those with a history of organ transplantation or chemotherapy, highlighting the increased susceptibility to skin cancer under systemic immunosuppression. Among patients with genital SCC, a proportion were HPV-positive, supporting the aetiologic role of high-risk HPV infection in mucocutaneous carcinogenesis. However, HPV testing was not routinely performed, resulting in many cases with unknown status. These findings emphasize the need for greater attention to viral and immune-related factors in the management and prevention of SCC in Asian populations.
In contrast to studies from Western countries, where cutaneous lymphomas predominantly affect older adults (21–23), the patients in our cohort showed a markedly younger age of onset, with the peak incidence occurring between 20 and 49 years of age. Similar age trends have been reported in other Asian studies, likely reflecting the higher frequency of EBV-positive T/NK-cell lymphoproliferative disorders and MF among Asian patients (25–27). Dendritic cell and histiocytic neoplasms represented a small but distinct subset of cutaneous tumours in our series. These lesions encompass both benign proliferations, such as juvenile xanthogranuloma, and rare malignant entities, including BPDCN and LCH. The age distribution and clinicopathologic spectrum of dendritic cell and histiocytic neoplasms in our cohort are broadly consistent with previous observational studies, in which juvenile xanthogranuloma predominate and typically present in children or young adults (28, 29). Given the limited epidemiological data from Asian populations, our findings provide a valuable reference for the spectrum and age distribution of dendritic cell and histiocytic neoplasms in China.
Our study offers several unique strengths compared with previously published data. First, it represents one of the largest single-centre datasets of skin cancer patients from China, encompassing both common and rare malignant skin tumours. Second, we provided detailed pathological subclassification, risk stratification, and treatment patterns for each tumour type, which are often underreported in prior Chinese studies. Third, we included a high number of cutaneous lymphoma cases – particularly MF – which are rarely the focus of skin cancer epidemiology studies but appear to be of growing relevance in younger Asian populations. The inclusion of Breslow thickness and Clark-level data for melanoma cases, along with tumour subtype and anatomic localization, further strengthens the depth and clinical relevance of our findings. In addition, our cohort included 2 cases of melanoma in situ. However, due to the very small number, these cases were not analysed separately, which we acknowledge as a limitation of this study.
Despite its strengths, this study has several limitations. As a single-centre retrospective analysis conducted in a tertiary referral hospital, selection bias is inevitable and the findings may not be fully generalizable to the broader population, as our hospital serves as a major referral centre for complex dermatologic conditions in South China. The retrospective design may also have led to incomplete clinical information in some cases, and some staging data, such as TNM classification for melanoma and lymphoma, were incomplete or unavailable, limiting our ability to fully assess prognosis or compare outcomes. Furthermore, the observational nature of the study precludes conclusions concerning causality or temporal trends. Finally, very rare tumours, such as Merkel cell carcinoma and adnexal tumours, were not represented due to their extremely low frequency in our centre. Future multicentre, population-based studies with standardized data collection are needed to validate and expand upon these findings. Nonetheless, our data provide a valuable regional supplement to the global understanding of skin cancer epidemiology. The insights gained from this study may help clinicians in Asia recognize local patterns of disease and tailor treatment strategies accordingly, and also support public health officials and policymakers in developing targeted prevention programmes, early screening initiatives, and educational campaigns that reflect the specific needs of Asian populations.
ACKNOWLEDGEMENTS
Ethics approval and consent to participate: This study was approved by the Institutional Review Board of the Dermatology Hospital, Southern Medical University. All procedures involving human participants were conducted in accordance with the ethical standards of the institutional and national research committees and with the Declaration of Helsinki. Written informed consent was obtained from all patients prior to inclusion in the study.
Data availability statement: The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- Leiter U, Garbe C. Epidemiology of melanoma and nonmelanoma skin cancer: the role of sunlight. Adv Exp Med Biol 2020; 89–103. https://doi.org/10.1007/978-0-387-77574-6_8
- Leiter U, Keim U, Garbe C. Epidemiology of skin cancer: update 2019. Adv Exp Med Biol 2020; 1268: 123–139. https://doi.org/10.1007/978-3-030-46227-7_6
- Wang R, Chen Y, Shao X, Chen T, Zhong J, Ou Y, et al. Burden of skin cancer in older adults from 1990 to 2021 and modelled projection to 2050. JAMA Dermatol 2025; e251276. https://doi.org/10.1001/jamadermatol.2025.1276
- Schmitt J, Seidler A, Diepgen TL, Bauer A. Occupational ultraviolet light exposure increases the risk for the development of cutaneous squamous cell carcinoma: a systematic review and meta-analysis: occupational UV exposure and cutaneous SCC. Br J Dermatol 2011; 164: 291–307. https://doi.org/10.1111/j.1365-2133.2010.10118.x
- Liu Y, Cen Y, Xu X, Li Z. Clinical analysis of 236 cases of skin cancer. West China Med J 2007; 22: 368–369.
- Huang Y, Li H, Tu P, Chen X, Yang S, Wu L, et al. Skin cancer and precancerous skin lesions: clinical analysis of 632 cases. Chinese J Dermatol 2010; 43: 452–454. https://doi.org/10.3760/cma.j.issn.0412-4030.2010.07.002
- Chahal HS, Rieger KE, Sarin KY. Incidence ratio of basal cell carcinoma to squamous cell carcinoma equalizes with age. J Am Acad Dermatol 2017; 76: 353–354. https://doi.org/10.1016/j.jaad.2016.08.019
- Willemze R, Cerroni L, Kempf W, Berti E, Facchetti F, Swerdlow SH, et al. The 2018 update of the WHO-EORTC classification for primary cutaneous lymphomas. Blood 2019; 133: 1703–1714. https://doi.org/10.1182/blood-2018-11-881268
- Lomas A, Leonardi-Bee J, Bath-Hextall F. A systematic review of worldwide incidence of nonmelanoma skin cancer. Br J Dermatol 2012; 166: 1069–1080. https://doi.org/10.1111/j.1365-2133.2012.10830.x
- Rogers HW, Weinstock MA, Feldman SR, Coldiron BM. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population, 2012. JAMA Dermatol 2015; 151: 1081. https://doi.org/10.1001/jamadermatol.2015.1187
- Madan V, Lear JT, Szeimies R-M. Non-melanoma skin cancer. Lancet 2010; 375: 673–685. https://doi.org/10.1016/s0140-6736(09)61196-x
- Morita A, Lim HW, Passeron T, Goh CL, Kang HY, Ly F, et al. Attitudes and behaviors regarding sun exposure in Japan compared to Europe and North America. J Dermatol 2024; 51: 1004–1009. https://doi.org/10.1111/1346-8138.17217
- Lau WC, Yang K, Lau CB, Patel S, Nambudiri VE. Characterization of sun-protective behaviours among Asian people in the USA stratified by geographic location of birth. Clin Exp Dermatol 2024; 49: 282–284. https://doi.org/10.1093/ced/llad363
- Chen Z, Lin Y, Qin Y, Qu H, Zhang Q, Li Y, et al. Prognostic factors and survival outcomes among patients with mycosis fungoides in China: a 12-year review. JAMA Dermatol 2023; https://doi.org/10.1001/jamadermatol.2023.2634
- Suh KS, Jang MS, Jung JH, Kwon DI, Seong SH, Lee KH, et al. Clinical characteristics and long-term outcome of 223 patients with mycosis fungoides at a single tertiary center in Korea: a 29-year review. J Am Acad Dermatol 2022; 86: 1275–1284. https://doi.org/10.1016/j.jaad.2021.06.860
- Bradford PT, Goldstein AM, McMaster ML, Tucker MA. Acral lentiginous melanoma: incidence and survival patterns in the United States, 1986–2005. Arch Dermatol 2009; 145. https://doi.org/10.1001/archdermatol.2008.609
- Chang J W-C, Yeh K-Y, Wang C-H, Yang T-S, Chiang H-F, Wei F-C, et al. Malignant melanoma in Taiwan: a prognostic study of 181 cases. Melanoma Res 2004; 14: 537–541. https://doi.org/10.1097/00008390-200412000-00016
- Cormier JN, Xing Y, Ding M, Lee JE, Mansfield PF, Gershenwald JE, et al. Ethnic differences among patients with cutaneous melanoma. Arch Intern Med 2006; 166: 1907. https://doi.org/10.1001/archinte.166.17.1907
- Ossio R, Roldán-Marín R, Martínez-Said H, Adams DJ, Robles-Espinoza CD. Melanoma: a global perspective. Nat Rev Cancer 2017; 17: 393–394. https://doi.org/10.1038/nrc.2017.43
- Behbahani S, Maddukuri S, Cadwell JB, Lambert WC, Schwartz RA. Gender differences in cutaneous melanoma: demographics, prognostic factors, and survival outcomes. Dermatol Ther 2020; 33. https://doi.org/10.1111/dth.14131
- Bradford PT, Devesa SS, Anderson WF, Toro JR. Cutaneous lymphoma incidence patterns in the United States: a population-based study of 3884 cases. Blood 2009; 113: 5064–5073. https://doi.org/10.1182/blood-2008-10-184168
- Bekkenk MW, Geelen FAMJ, Vader PCVV, Heule F, Geerts M-L, Van Vloten WA, et al. Primary and secondary cutaneous CD30+lymphoproliferative disorders: a report from the Dutch Cutaneous Lymphoma Group on the long-term follow-up data of 219 patients and guidelines for diagnosis and treatment. Blood 2000; 95: 3653–3661. https://doi.org/10.1182/blood.v95.12.3653
- Shi H-Z, Liu Y-X, Jiang Y-Q, Chen S-L, Xu X-L, Lu X-M, et al. Clinical characteristics of primary cutaneous lymphoma: analysis from two centres in China. Br J Dermatol 2019; 181: 1332–1333. https://doi.org/10.1111/bjd.18266
- Apalla Z, Nashan D, Weller RB, Castellsagué X. Skin cancer: epidemiology, disease burden, pathophysiology, diagnosis, and therapeutic approaches. Dermatol Ther (Heidelb) 2017; 7: 5–19. https://doi.org/10.1007/s13555-016-0165-y
- Kimura H, Fujiwara S. Overview of EBV-associated T/NK-cell lymphoproliferative diseases. Front Pediatr 2019; 6: 417. https://doi.org/10.3389/fped.2018.00417
- Lai W, Wang Y, Wang J, Wu L, Jin Z, Wang Z. Epstein-Barr virus-associated hemophagocytic lymphohistiocytosis in adults and adolescents – a life-threatening disease: analysis of 133 cases from a single center. Hematology 2018; 23: 810–816. https://doi.org/10.1080/10245332.2018.1491093
- Kimura H, Ito Y, Kawabe S, Gotoh K, Takahashi Y, Kojima S, et al. EBV-associated T/NK-cell lymphoproliferative diseases in nonimmunocompromised hosts: prospective analysis of 108 cases. Blood 2012; 119: 673–686. https://doi.org/10.1182/blood-2011-10-381921
- Salari B, Dehner LP. Juvenile and adult xanthogranuloma: a 30-year single-center experience and review of the disorder and its relationship to other histiocytoses. Ann Diagn Pathol 2022; 58: 151940. https://doi.org/10.1016/j.anndiagpath.2022.151940
- Kundak S, Çakır Y. Juvenile xanthogranuloma: retrospective analysis of 44 pediatric cases (single tertiary care center experience). Int J Dermatol 2021; 60: 564–569. https://doi.org/10.1111/ijd.15223