RESEARCH LETTER
Low Subcutaneous Adipose Tissue is Associated with Poor Prognosis for Aged Patients with Cutaneous Angiosarcoma: A Retrospective Cohort Study
Akira MIYAZAKI1, Mariko OGAWA-MOMOHARA1*, Tomoki TAKI1, Shoichiro MORI1 and Masashi AKIYAMA1
1Department of Dermatology, Nagoya University Graduate School of Medicine, Nagoya, Japan. *Email: marikkori0910@gmail.com
Citation: Acta Derm Venereol 2026; 106: adv-2025-0058. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0058.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Sept 28, 2025. Accepted after revision: Dec 8, 2025.
Published: Feb 11, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
This work was financially supported by JST SPRING under Grant Number JPMJSP2125.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
To the Editor,
Cutaneous angiosarcoma is a highly malignant sarcoma derived from vascular endothelium. It frequently develops on the scalp of the elderly, and the outcomes remain dismal. Low body weight adversely affects the prognosis of cancer patients (1). To investigate which components of body mass index (BMI) are associated with prognosis, we addressed body composition. Adipose tissue index (ATI) and skeletal muscle index (SMI) can be measured non-invasively using routine abdominal computed tomography (CT). We investigated ATI as a prognostic predictor in angiosarcoma, a representative malignant tumour in the elderly.
Thirty six patients were extracted based on the Japanese Diagnosis Procedure Combination database of our hospital from January 2011 to December 2023. The primary composite end-point was event-free survival (EFS): the time from CT at the initial visit to death/transfer to best supportive care. Areas of adipose tissue (AT) and skeletal muscle were measured using AI-assisted SYNAPSE VINCENT software (FUJIFILM Corporation, Tokyo, Japan) (Appendix S1). Values were normalized for height (cm2/m2) (See Appendix S2 for detailed materials and methods).
The baseline characteristics are shown in Table SI. Twenty-seven events had occurred at the time of data analysis. The univariate and multivariate analyses are shown in Table I. Among the previously defined factors, ATI [hazard ratio (HR)=0.9830, 95% confidence interval (95% CI)=0.9686–0.9966, p=0.0187], subcutaneous ATI (HR=0.9735, 95% CI=0.9502–0.9954, p=0.0228), BMI (HR=0.8155, 95% CI=0.6647–0.9861, p=0.0429), stage (HR=4.604, 95% CI=1.590–13.33, p=0.0049), and radiation therapy (RT) status (HR=0.1578, 95% CI=0.04317–0.5765, p=0.0052) were significantly associated with EFS in univariate analysis (Table I). Visceral ATI and SMI were not significantly associated with EFS. In the multivariate analysis of subcutaneous ATI, BMI, stage and RT, subcutaneous ATI was significantly associated with EFS (HR=0.9696, 95% CI=0.9423–0.9943, p=0.0230) (Table I). BMI was not significantly associated with EFS (HR=0.8596, 95% CI=0.6935–1.053, p=0.1524). Stratification by subcutaneous ATI found a poorer prognosis for subcutaneous ATI<35 cm2/m2 than for subcutaneous ATI>35 cm2/m2 (p=0.0313) (Fig. 1).
Table I. Univariate and multivariate analyses
| Univariate analyses | Multivariate analyses | ||||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | ||
| Age | 1.019 | 0.9722–1.072 | 0.4413 | ||||
| Sex (female, ref=male) | 0.8466 | 0.3599–1.991 | 0.7028 | ||||
| ECOG PS (1–2, ref=0) | 1.153 | 0.4590–2.897 | 0.7617 | ||||
| Stage (3–4, ref=1–2) | 4.604 | 1.590–13.33 | 0.0049 | 6.257 | 1.839–21.29 | 0.0033 | |
| Chemotherapy (paclitaxel, ref=other) | 0.7198 | 0.3120–1.661 | 0.4409 | ||||
| Radiation therapy (yes, ref=no) |
0.1578 | 0.04317–0.5765 | 0.0052 | 0.2067 | 0.05240–0.8154 | 0.0244 | |
| BMI (kg/m2) | 0.8155 | 0.6647–0.9861 | 0.0429 | 0.8593 | 0.6935–1.053 | 0.1524 | |
| SMI (cm2/m2) | 0.9871 | 0.9401–1.036 | 0.5950 | ||||
| ATI (cm2/m2) | 0.9830 | 0.9686–0.9966 | 0.0187 | ||||
| Subcutaneous ATI (cm2/m2) | 0.9735 | 0.9502–0.9954 | 0.0228 | 0.9696 | 0.9423–0.9943 | 0.0230 | |
| Visceral ATI (cm2/m2) | 0.9860 | 0.9632–1.005 | 0.1883 | ||||
| ATI:adipose tissue index; BMI:body mass index; CI:confidence interval; ECOG PS:Eastern Cooperative Oncology Group Performance Status; HR:hazard ratio; SMI:skeletal muscle index. | |||||||
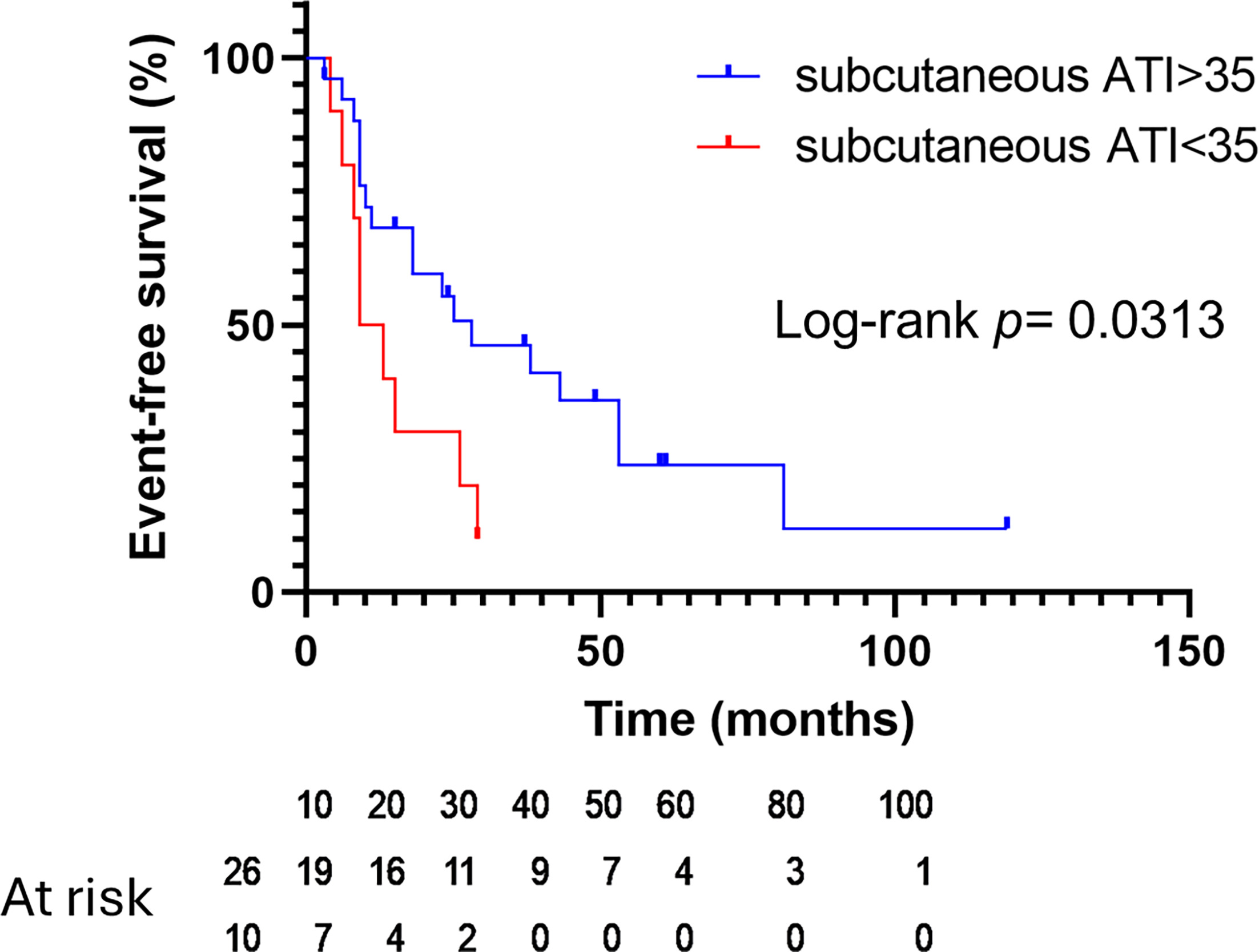
Fig. 1. Kaplan–Meier estimates of event-free survival (EFS) stratified on subcutaneous adipose tissue index (ATI). Red line, subcutaneous ATI<35; blue line, subcutaneous ATI>35. Censored data points are marked with crosses.
ATI and subcutaneous ATI predicted the prognosis more accurately than BMI or even performance status. Within the BMI range of 20–25, subcutaneous ATI values span 23–84. It is difficult to directly estimate subcutaneous ATI from BMI. Generally, people with pear-shaped bodies are likely to have more subcutaneous AT. Abdominal skinfold is known to correlate with subcutaneous AT measured by CT (2). Using these body features may help in estimating subcutaneous AT volume easily, although there is a limitation in accuracy. Ebadi et al. reported that low subcutaneous ATI is associated with shorter survival in gastrointestinal and respiratory cancers and in metastatic renal cell carcinoma in Canada (3). Mengoni et al. reported that abundant subcutaneous AT is associated with enhanced objective response rate to immune checkpoint inhibitors and prolonged progression-free survival in metastatic melanoma (4). Evidence of a link between subcutaneous AT and prognosis has been steadily accumulating. Our study clarifies this association in an elderly Asian cohort with rapidly progressive cutaneous malignancies. ATI primarily reflects a patient’s nutritional status. Consistent with our univariate analysis, low BMI is associated with poor prognosis in soft-tissue sarcoma among patients aged ≥60 years (5). However, low body weight might be a consequence rather than a cause of that prognosis. Cancer-related systemic inflammatory response contributes to malnutrition (6). Subcutaneous AT produces the anti-inflammatory cytokine adiponectin, whose expression is higher in subcutaneous AT than in visceral AT in human subjects (7). Serum adiponectin is positively associated with subcutaneous AT volume but inversely associated with visceral AT volume in Japanese men (8). These findings suggest that subcutaneous AT not only reflects nutritional status but also actively participates in the inflammatory response and cancer progression. This condition may be shared among malignant tumours associated with poor prognosis in elderly patients.
REFERENCES
- Tu H, McQuade JL, Davies MA, Huang M, Xie K, Ye Y, et al. Body mass index and survival after cancer diagnosis: a pan-cancer cohort study of 114 430 patients with cancer. Innovation 2022; 3: 100344. https://doi.org/10.1016/j.xinn.2022.100344
- Orphanidou C, McCargar L, Birmingham CL, Mathieson J, Goldner E. Accuracy of subcutaneous fat measurement: comparison of skinfold calipers, ultrasound, and computed tomography. J Am Diet Assoc 1994; 94: 855–858. https://doi.org/10.1016/0002-8223(94)92363-9
- Ebadi M, Martin L, Ghosh S, Field CJ, Lehner R, Baracos VE, et al. Subcutaneous adiposity is an independent predictor of mortality in cancer patients. Br J Cancer 2017; 117: 148–155. https://doi.org/10.1038/bjc.2017.149
- Mengoni M, Braun AD, Hinnerichs MS, Tüting T, Surov A. Subcutaneous fat abundance and density are associated with an enhanced response to immunotherapy in metastatic melanoma: a retrospective cohort study. Acad Radiol 2023; 30 Suppl 1: S257–S267. https://doi.org/10.1016/j.acra.2023.05.007
- Tamiya H, Imura Y, Wakamatsu T, Takenaka S. Comorbidity, body mass index, and performance status as prognostic factors in older patients with soft-tissue sarcoma. J Geriatr Oncol 2022; 13: 673–681. https://doi.org/10.1016/j.jgo.2022.02.011
- Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol 2011; 12: 489–495. https://doi.org/10.1016/S1470-2045(10)70218-7
- Lihn AS, Bruun JM, He G, Pedersen SB, Jensen PF, Richelsen B. Lower expression of adiponectin mRNA in visceral adipose tissue in lean and obese subjects. Mol Cell Endocrinol 2004; 219: 9–15. https://doi.org/10.1016/j.mce.2004.03.002
- Nakamura Y, Sekikawa A, Kadowaki T, Kadota A, Kadowaki S, Maegawa H, et al. Visceral and subcutaneous adiposity and adiponectin in middle-aged Japanese men: the ERA JUMP study. Obesity 2009; 17: 1269–1273. https://doi.org/10.1038/oby.2009.3