SHORT COMMUNICATION
A Novel Presentation of Mycobacterium haemophilum Cutaneous Infection: A Diagnostic Challenge
Masakazu KAKURAI1* , Shigemi HITOMI2, Takumi AKASHI3 and Yoshihiro MORIYAMA1
, Shigemi HITOMI2, Takumi AKASHI3 and Yoshihiro MORIYAMA1
1Department of Dermatology, Tsuchiura Kyodo General Hospital, Ibaraki, Japan, 2Department of Infectious Diseases, University of Tsukuba Hospital, Ibaraki, Japan, and 3Department of Pathology, Tsuchiura Kyodo General Hospital, Ibaraki, Japan. *Email: kakurai.masakazu.or@ms.hosp.tsukuba.ac.jp
Citation: Acta Derm Venereol 2026; 106: adv-2025-0233. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0233.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Nov 27, 2025. Accepted after revision: Jan 19, 2026.
Published: Feb 11, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Mycobacterium haemophilum is an acid-fast bacillus belonging to the group of nontuberculous mycobacteria (NTM) that are frequently found in environmental habitats and can infect humans (1, 2, 3). The skin is the most common site of M. haemophilum infection, followed by the joints, bones, lymph nodes and lungs (1, 2, 3). Cutaneous infections may result from direct inoculation or haematogenous dissemination from other parts of the body (1). The clinical manifestations of M. haemophilum cutaneous infections include nodules, ulcers, swelling, abscesses and pustules (1, 2, 3); however, diffuse erythema has not been previously reported. We report a case of M. haemophilum cutaneous infection presenting with diffuse erythema on the trunk and extremities.
CASE REPORT
A 78-year-old Japanese man presented with a 3-month history of mild pruritic erythema on his left upper extremity without general symptoms, such as fever and arthralgia. His medical history included type 2 diabetes mellitus, chronic kidney disease and interstitial pneumonia with autoimmune features (anti-ribonucleoprotein antibody positivity). He was treated with prednisolone (currently taking 5 mg daily) and mycophenolate mofetil for 5 years. No history of water or sea-animal exposure was reported. Physical examination revealed diffuse indurated erythema with scales on the left upper extremity, sparing the left palm (Fig. 1a, b). Oedema was observed on the left upper extremity. Additionally, multiple indurated erythematous macules measuring up to 1 cm were observed from the left shoulder to the left chest (Fig. 1b). Mucosal involvement and lymph node enlargement were not observed. Blood tests showed leukocytosis (13,060/μL), slightly elevated C-reactive protein (0.95 mg/dL) and elevated soluble interleukin-2 receptor (1,579 IU/mL). The β-D-glucan level was within normal ranges. The T-SPOT.TB assay and human immunodeficiency virus antigen/antibody were negative. A biopsy of the indurated erythema on the upper arm revealed non-caseating epithelioid cell granulomas with Langhans-type giant cells surrounded by lymphocytes in the dermis (Fig. 1c). Gram, periodic acid-Schiff, Grocott and Ziehl–Neelsen staining results were all negative. Based on these clinicopathological findings, further testing was performed, with a provisional diagnosis of sarcoidosis. Contrast-enhanced computed tomography revealed no abnormal findings, such as bilateral hilar lymphadenopathy. The electrocardiographic and echocardiographic results were normal. Ophthalmological examination revealed no abnormalities. Treatment with high-potency topical steroids was ineffective. Six weeks after the initial visit, the patient developed diffuse erythema with scales on the dorsum of the right hand and multiple indurated erythematous macules with scales measuring up to 1 cm on the right lower leg (Fig. 1d–f). Oedema was observed on the dorsum of the hands and the right lower leg. A skin biopsy of the erythema on the hand revealed dilated blood vessels and infiltration of lymphocytes, histiocytes and giant cells in the dermis (Fig. 1g). Furthermore, a biopsy of the indurated erythematous macule on the right lower leg revealed non-caseating epithelioid cell granulomas, similar to the initial biopsy. Direct immunofluorescence and Ziehl–Neelsen staining yielded negative results. Two weeks after the second biopsy, a further biopsy of the indurated erythematous macule on the right lower leg revealed mainly non-caseating epithelioid cell granulomas and partial epithelioid cell granulomas with caseating necrosis in the dermis (Fig. 1h). Additionally, Ziehl–Neelsen staining confirmed the presence of acid-fast bacilli. In contrast, cultures of bacteria, fungi and acid-fast bacilli from the skin of the leg, sputum and blood were negative. These samples were incubated at 30℃ and 35℃ for 6 weeks using Middlebrook 7H11 medium for the culture of acid-fast bacilli. Fifteen weeks after the initial visit, the patient developed multiple indurated, erythematous, scaly macules on the upper back, which tended to coalesce (Fig. 1i). Skin from this region was also cultured on the Middlebrook 7H11 medium, but results were negative. A skin sample was then used to perform a polymerase chain reaction on the gene encoding the 65 kDa heat shock protein, as previously described (4). A homology search using the Basic Local Alignment Search Tool showed that the sequence of 362 base pairs was 100% identical to that of M. haemophilum strain ATCC 29548T. Culture tests on sputum and blood using chocolate agar medium containing haemin yielded negative results. Consequently, M. haemophilum cutaneous infection was diagnosed. Ciprofloxacin (800 mg daily) and clarithromycin (800 mg daily) were initiated, resulting in the resolution of all lesions within 1 month, leaving hyperpigmentation with no new skin lesions. Three months have passed since treatment began with no recurrence, and treatment is scheduled to continue for at least 6 months.
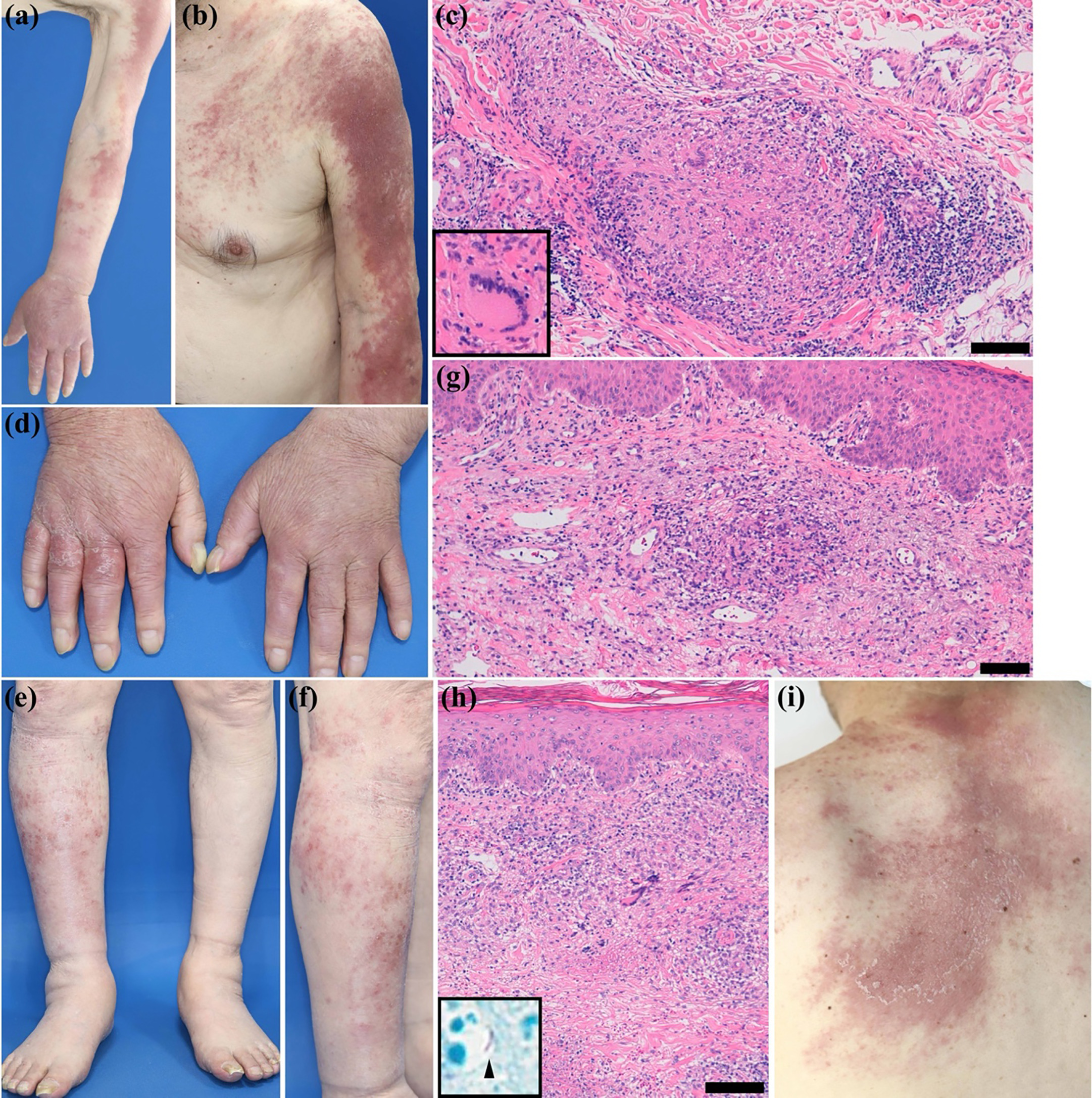
Fig. 1. Clinical and histological findings. (a, b) Diffuse indurated erythema with scales on the left upper extremity, excluding the left palm. Multiple indurated erythematous macules extending from the left shoulder to the left chest. (c) A haematoxylin-eosin-stained slide of the indurated erythema on the upper arm showing non-caseating epithelioid cell granulomas with Langhans-type giant cells in the dermis (bar in the image = 100 μm). (d–f) Diffuse erythema with scales on the dorsum of the right hand and multiple indurated erythematous macules with scales on the right lower leg. (g) A haematoxylin-eosin-stained slide of erythema on the right hand showing dilated blood vessels and infiltration of lymphocytes, histiocytes and giant cells in the dermis (bar in the image = 100 μm). (h) An additional biopsy of the indurated erythematous macule on the right lower leg showing epithelioid cell granulomas with caseating necrosis in the dermis (bar in the image = 100 μm). Positive acid-fast bacillus Ziehl-Neelsen staining (black arrow). (i) Multiple indurated, erythematous, scaly macules on the upper back, which tended to coalesce.
DISCUSSION
The lesions in NTM cutaneous infections are typically erythematous nodules that can ulcerate and form abscesses; however, their clinical manifestations vary (5). In a previous study of 79 patients with M. haemophilum cutaneous infections, the clinical manifestations included nodules (60.8%), ulcers (24.1%), swelling (22.8%), abscesses (21.5%) and pustules (21.5%) (1). Although less common, M. haemophilum infections presenting with a cellulitis-like appearance or sporotrichoid spread involving nodules and ulcers along lymphatic pathways have also been reported (2, 3). To our knowledge, this is the first reported case of diffuse erythema associated with M. haemophilum cutaneous infection. A previous report described a patient with diffuse erythema caused by Mycobacterium marinum with clinical manifestations similar to those observed in our patient (6). Considering that our patient presented with simultaneous lesions at multiple sites, the pathogen may have initially invaded the skin and subsequently spread via haematogenous dissemination secondary to bacteraemia. Furthermore, since the optimum temperature for growth of M. haemophilum and M. marinum is approximately 30℃ (1, 2, 3, 6), the infection may not have spread beneath the subcutaneous tissues and may remain in the dermis. Although diffuse erythema is rare in cutaneous infections caused by Mycobacterium species, it is important to recognize that diffuse erythema is one of the clinical manifestations of NTM cutaneous infections.
The optimal detection of M. haemophilum in biopsy specimens requires a combination of acid-fast staining, bacterial culture at a temperature of 30℃ using media with iron additives and molecular detection (1, 2, 3). The histological features of tissues infected with M. haemophilum are usually epithelioid cell granulomas with caseating necrosis; however, non-caseating epithelioid cell granulomas may be present (1, 2, 3). Importantly, even when necrosis is evident, Ziehl–Neelsen staining may be negative (1, 3). For optimal growth, M. haemophilum prefers a low growth temperature of 30℃ and requires iron supplementation, such as haemin (2). Culturing M. haemophilum is slow and can take up to 8 weeks (2). In our patient, necrosis was observed during the third biopsy, and positive acid-fast bacillus staining raised the suspicion of a mycobacterial infection. Since the tissues from our patient were cultured on a medium without iron additives, they may not have met the growth requirements of M. haemophilum. Based on these findings, the presence of a positive acid-fast bacillus stain in samples with no growth on media should be considered as M. haemophilum, and culture conditions should be adapted accordingly.
Although no optimal therapeutic regimen for M. haemophilum infections has been established, several reports recommend combination therapy with antibiotics, including quinolones (ciprofloxacin), macrolides (clarithromycin) and rifampicin (1, 2, 3). Notably, patients with M. haemophilum cutaneous infections treated with quinolones and macrolides have shown better prognoses (1). Given that the median duration of M. haemophilum infection therapy is approximately 6 months (2), treatment in our patient is planned to continue for at least 6 months, unless side-effects occur or recurrence is observed.
Our patient demonstrated that diffuse erythema could be a clinical manifestation of M. haemophilum cutaneous infection. Clinicians should be aware of the diverse clinical manifestations of M. haemophilum cutaneous infection, as well as the laboratory pitfalls associated with M. haemophilum.
REFERENCES
- Yasen D, Liu C, Mei X, Zhou H, Tang X, Chen X. Mycobacterium haemophilum infection with cutaneous involvement: two case reports and an updated literature review: Mycobacterium haemophilum skin infection. J Dtsch Dermatol Ges 2023; 21: 1291–1305. https://doi.org/10.1111/ddg.15163
- Lindeboom JA, Bruijnesteijn van Coppenraet LES, van Soolingen D, Prins JM, Kuijper EJ. Clinical manifestations, diagnosis, and treatment of Mycobacterium haemophilum infections. Clin Microbiol Rev 2011; 24: 701–717. https://doi.org/10.1128/CMR.00020-11
- Tyner HL, Wilson JW. Fifteen-year clinical experience with Mycobacterium haemophilum at the mayo clinic: A case series. J Clin Tuberc Other Mycobact Dis 2017; 8: 26–32. https://doi.org/10.1016/j.jctube.2017.06.002
- Telenti A, Marchesi F, Balz M, Bally F, Böttger EC, Bodmer T. Rapid identification of mycobacteria to the species level by polymerase chain reaction and restriction enzyme analysis. J Clin Microbiol 1993; 31: 175–178. https://doi.org/10.1128/jcm.31.2.175-178.1993
- Lamb RC, Dawn G. Cutaneous non-tuberculous mycobacterial infections. Int J Dermatol 2014; 53: 1197–1204. https://doi.org/10.1111/ijd.12528
- Park JB, Seong SH, Kwon DI, Jang JY, Suh KS, Jang MS. Mycobacterium marinum Infection spreading in a “Birds in Flocks” Pattern: All Caseating Granuloma is Not Tuberculosis. Acta Derm Venereol 2020; 100: adv00200. https://doi.org/10.2340/00015555-3538