SHORT COMMUNICATION
Case Report Concerning a Varicella Zoster Virus Infection in a Six-year-old Child Receiving Dupilumab for Atopic Dermatitis
Jens K. RANGEN1* and Maiken G. DALAGER1
1Department of dermato- and venereology, Aalborg University Hospital, Aalborg, Denmark. *Email: jenjen@rn.dk
Citation: Acta Derm Venereol 2026; 106: adv-2025-0045. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0045.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Sept 22, 2025. Accepted after revision: Feb 2, 2026.
Published: Feb 25, 2026.
Competing interests and funding: The data that support the findings of this study are available from the corresponding author upon reasonable request.
MG Dalager has the following conflicts of interests: Consultant: Abbvie, Leo Pharma, Eli Lilly, BMS, Boehringer Ingelheim, UCB, Almirall, Novartis, Sanofi, GaldermaInvited speaker: Astra Zenaca, Pfizer, Abbvie, GaldermaClinical investigator; Sanofi, Almirall, Johnson & Johnson, UCB, BMS.
To our knowledge, no case report exists of a first-time infection with varicella zoster virus (VZV) in a child with atopic dermatitis (AD) who was receiving treatment with dupilumab at the recommended dosage without prior vaccination (1, 2).
We present a case and a 4-month follow-up of a 6-year-old boy who developed VZV infection during dupilumab treatment. The patient had not been immunized against VZV prior to initiating the dupilumab treatment.
CASE
A 6-year-old boy with AD, asthma, chronic urticaria and multiple allergies, who had previously been treated with omalizumab for chronic urticaria and topical corticosteroids for AD. The patient was switched from omalizumab to dupilumab due to persistent dermatitis and urticaria despite ongoing treatment. A loading dose of dupilumab at 300 mg was administered and followed by a treatment dose of 300 mg every 28 days. The treatment had a remarkable effect on the patient’s urticaria and AD, reducing both the itching sensation, hives and eczema on the body.
Prior to the initiation of dupilumab treatment, the patient underwent a battery of blood tests, which showed no contraindication for IL4/IL13 treatment, except that the patient had a negative IgG for VZV. Informed of the risks of potentially prolonged and severe infection during treatment with dupilumab, the patient’s family agreed to start the treatment without a vaccine for VZV. After 4.5 months of regular dupilumab treatment, the patient presented symptoms of a VZV infection. Two samples were taken from a vesicle and erosion, both confirming the diagnosis of a VZV infection. See photos.
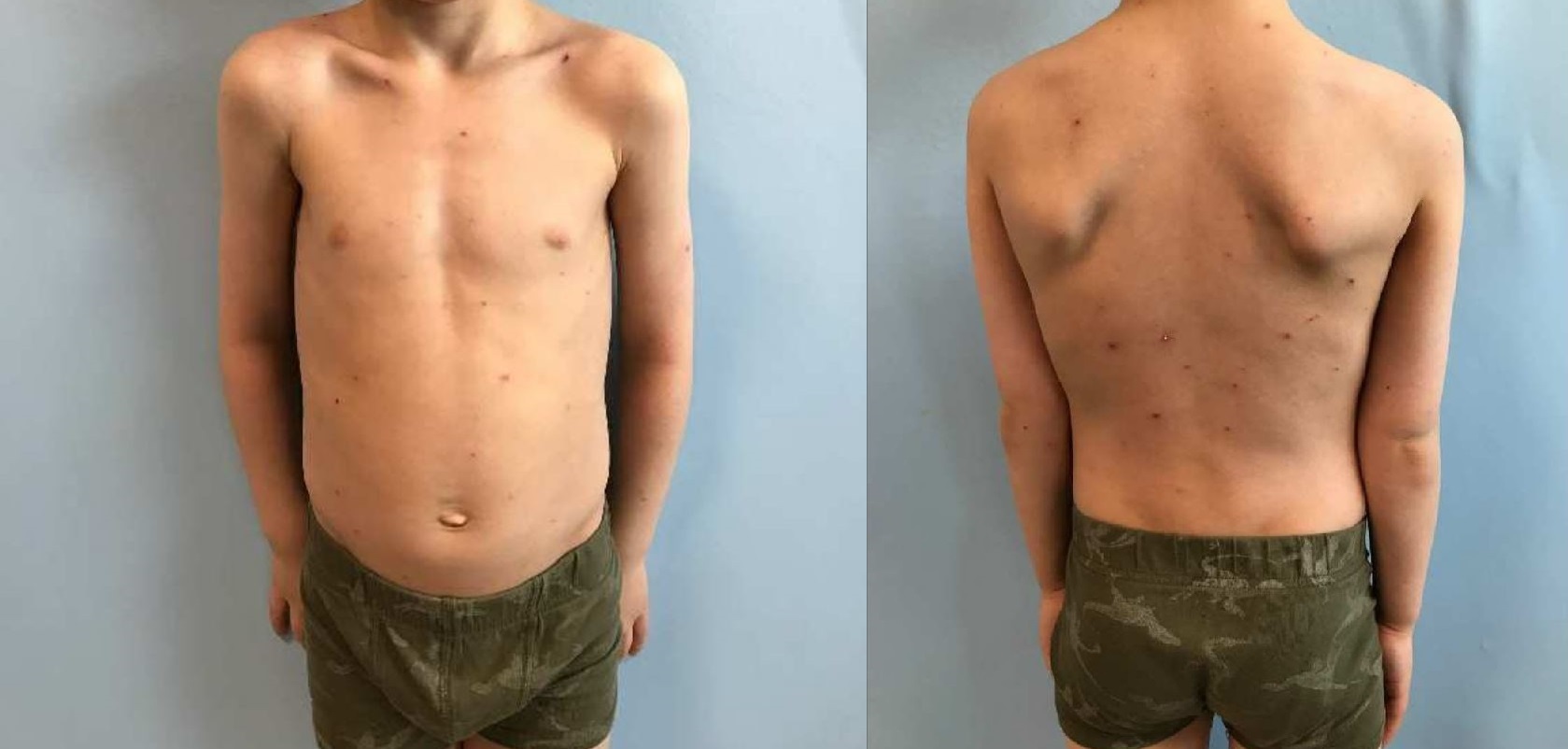
Fig. 1. A six-year old boy with several vesicles and erosion from a varicella zoster infection.
During the infection, the patient did not feel unwell and remained in good general condition. After 5 days, no vesicles were left, and the child was ready to resume day care. At a follow-up 4.5 months later, the patient reported no adverse effects from the infection.
DISCUSSION
Since dupilumab has been approved to treat children from the age of 6 months for AD, common childhood diseases are expected to occur during treatment. Previously, it has been reported that patients treated with other immunosuppressants (e.g. anti-TNF or JAK-inhibitors) have a higher risk of infection (3). In contrast, clinical trials have reported lower infection rates in adults and children treated with dupilumab, although none of the included clinical trials report a first-time infection with VZV (1, 3).
Age, skin barrier dysfunction and skin barrier damage from scratching are known risk factors that increase susceptibility to viral skin infections, which may raise concern for a more prolonged infection of, for example, VZV in patients with significant dermatitis.
Although it is not standard to recommend vaccination before initiating dupilumab treatment in children, it may be considered in some cases. Furthermore, studies covering administration of live vaccines after initiating treatment with dupilumab did not find adverse effects, including vaccine-related infection, in children receiving dupilumab (4).
On account of this case, describing an uncomplicated VZV infection in a child treated with dupilumab, it may be argued that concerns about initiating dupilumab in patients who are naive to VZV infection are less warranted. Furthermore, it is debatable whether the treatment for VZV infection could be milder in a child with well-treated eczema compared to an insufficiently treated eczema.
Consequently, we argue that it may not be absolutely necessary to wait for a VZV vaccination if there is an indication to initiate biological treatment with dupilumab for eczema.
REFERENCES
- Paller AS, Beck LA, Blauvelt A, Siegfried EC, Cork MJ, Wollenberg A, et al. Infections in children and adolescents treated with dupilumab in pediatric clinical trials for atopic dermatitis-A pooled analysis of trial data. Pediatr Dermatol 2022; 39: 187–196. https://doi.org/10.1111/pde.14909
- Rimmer S, Culotta N, Hill I. Disseminated varicella zoster virus and herpes zoster virus infection in a pediatric patient with atopic dermatitis. Pract Dermatol 2024; 14: 29–31.
- Eichenfield LF, Bieber T, Beck LA, Simpson EL, Thaçi D, de Bruin-Weller M, et al. Infections in dupilumab clinical trials in atopic dermatitis: a comprehensive pooled analysis. Am J Clin Dermatol 2019; 20: 443–456. https://doi.org/10.1007/s40257-019-00445-7
- Siegfried EC, Wine Lee L, Spergel JM, Prescilla R, Uppal S, Coleman A, et al. A case series of live attenuated vaccine administration in dupilumab-treated children with atopic dermatitis. Pediatr Dermatol 2024; 41: 204–209. https://doi.org/10.1111/pde.15518