SHORT COMMUNICATION
Ultrasound-detected Hypervascular Proliferating Trichilemmal Tumour
Kazutoshi NISHIMURA1, Kazuyasu FUJII1* , Yuto YAMAMURA1
, Yuto YAMAMURA1 , Shunya USUI1 and Atsushi OTSUKA1
, Shunya USUI1 and Atsushi OTSUKA1
1Department of Dermatology, Kindai University Hospital, Osaka, Japan. *Email: kazuyasu.fujii@med.kindai.ac.jp
Citation: Acta Derm Venereol 2026; 106: adv-2025-0309. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0309.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Dec 30, 2025. Accepted after revision: Feb 3, 2026.
Published: Mar 2, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Proliferating trichilemmal tumours (PTTs), also known as proliferating pilar tumours, are uncommon adnexal neoplasms derived from the outer root sheath of the hair follicle (1). They typically arise on the scalp of elderly women and are often associated with pre-existing trichilemmal cysts (2). Although most PTTs are benign, malignant proliferating trichilemmal tumours (MPTTs) have been reported (3, 4).
Ultrasonography has become a valuable tool for noninvasive evaluation of cutaneous and subcutaneous tumours, including cystic adnexal lesions. Previous reports have described PTTs as well-defined, encapsulated, heterogeneous lesions on B-mode imaging, often with internal calcifications and posterior acoustic enhancement, but lacking internal vascularity on colour Doppler studies (5, 6, 7).
Here, we report a case of a scalp PTT that demonstrated abundant intralesional vascular flow on colour Doppler ultrasonography – an imaging feature that is atypical for benign PTTs and has been scarcely described in the literature.
CASE REPORT
A 64-year-old man presented with a several-year history of a slowly enlarging mass on the right occipital scalp. Dermatological examination revealed a mobile, dome-shaped subcutaneous nodule approximately 3 cm in diameter, without tenderness or ulceration (Fig. 1A).
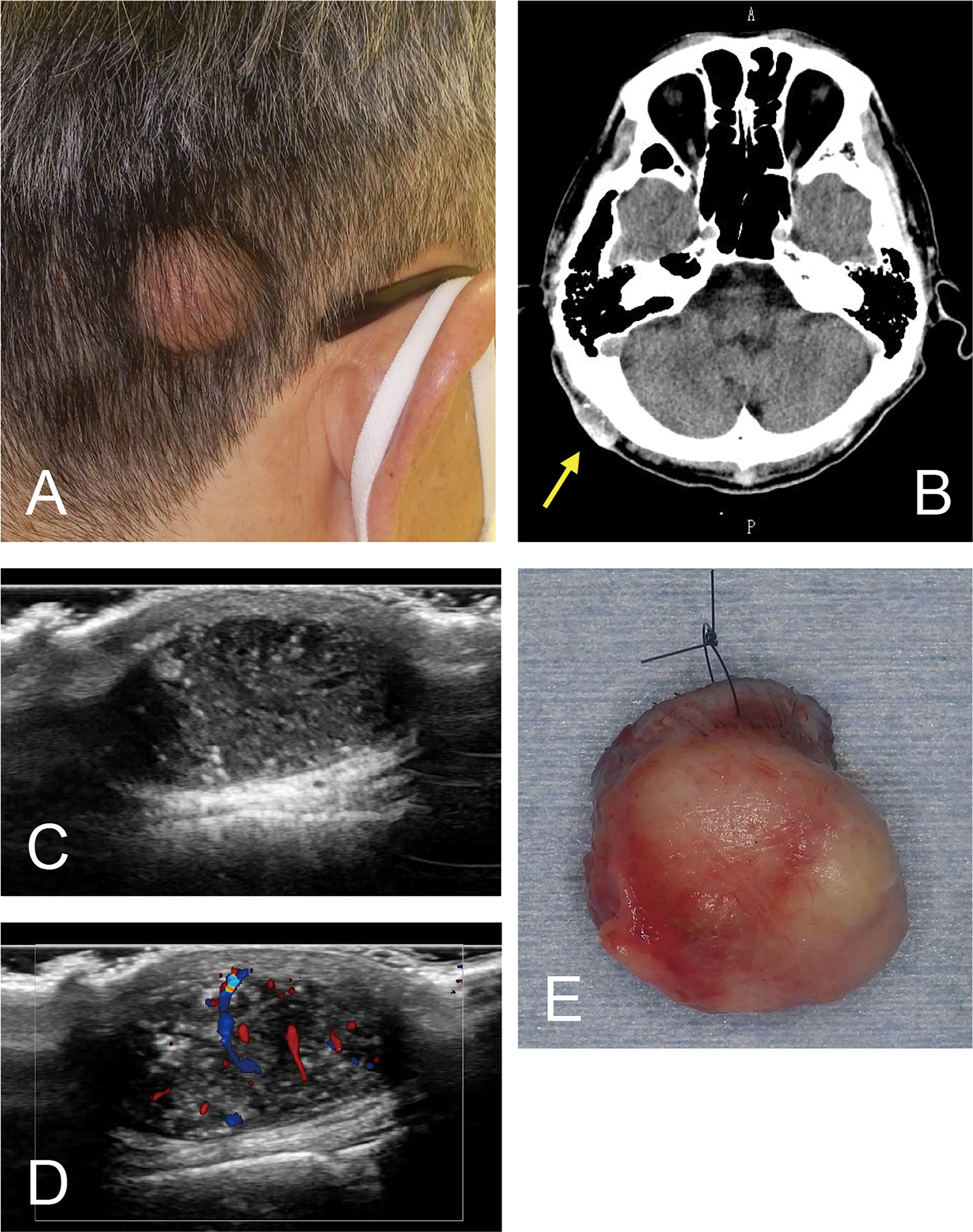
Fig. 1. Clinical and imaging findings of the lesion. (A) Clinical appearance of the tumour. (B) Computed tomography finding of the tumour (indicated by an arrow). (C) Grey-scale and (D) colour Doppler ultrasonographic findings. (E) Gross appearance of the excised lesion, viewed from the deep aspect.
Computed tomography revealed a subcutaneous mass in the right occipital region without continuity with the outer table of the skull. The lesion showed mixed low- and high-attenuation areas with a markedly hyperattenuating rim (Fig. 1B). Ultrasound examination revealed a well-circumscribed, heterogeneous mass measuring 21 × 27 × 12 mm in the subcutaneous layer (Fig. 1C). The lesion exhibited posterior acoustic enhancement and a heterogeneous internal echo pattern. Notably, colour Doppler imaging demonstrated marked internal vascularity with pulsatile flow signals, an atypical feature for trichilemmal cysts or benign PTTs (Fig. 1D). Based on these findings, an epidermal cyst was excluded, and a vascularized adnexal tumour, including a malignant lesion, was considered in the preoperative differential diagnosis. However, intraoperative examination revealed a well-circumscribed, cystic mass with a smooth surface and a well-formed capsule, without evidence of invasion or adhesion to the surrounding tissue. Given these intraoperative findings suggestive of a benign, encapsulated lesion, the tumour was excised completely along the capsule without a wide surgical margin. No dominant feeding vessel or identifiable feeder artery was observed during dissection. The lesion was easily dissected from the subcutaneous plane and removed in its entirety (Fig. 1E).
Histopathological examination revealed a well-circumscribed dermal tumour composed of multiple lobulated epithelial proliferations separated by fibrous stroma (Fig. 2A). The tumour lobules consisted of squamous epithelial cells showing characteristic trichilemmal keratinization without an intervening granular layer (Fig. 2B). Peripheral cells were basaloid to polygonal, while central areas demonstrated compact eosinophilic keratinous material, focally accompanied by calcification. Foci of nuclear pleomorphism, scattered mitotic figures and limited necrotic changes were observed; however, the overall lobular architecture was preserved, and no infiltrative growth pattern, stromal destruction or vascular invasion was identified. Based on the maintained architectural organization despite focal cytologic atypia, the lesion was interpreted as a benign PTT. Surgical margins were tumour-free. The final diagnosis was PTT of the right occipital region. No local recurrence or complications were observed during 6 months of follow-up.
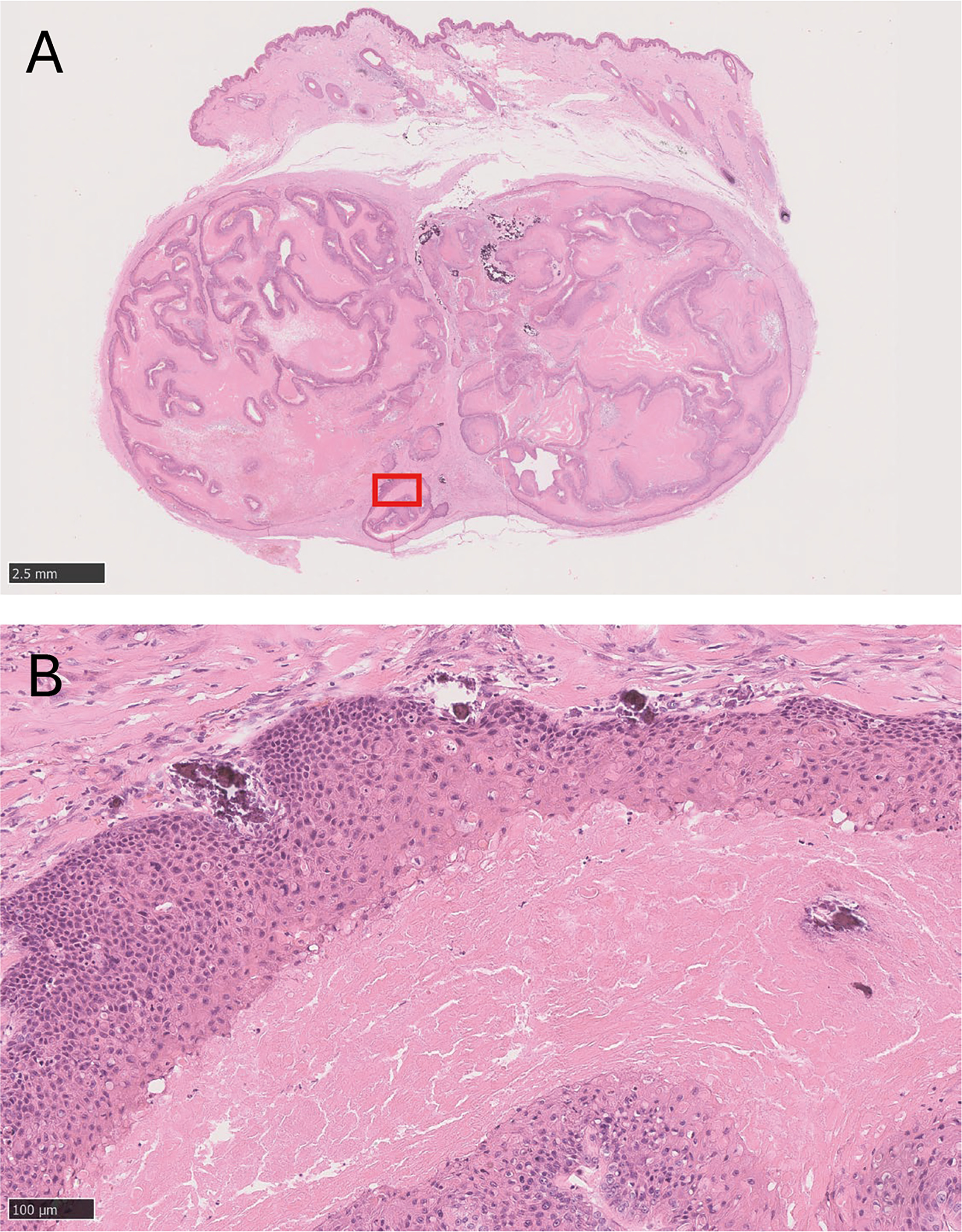
Fig. 2. Histopathologic findings of the tumour. (A) Low-power view showing a well-circumscribed, lobulated dermal tumour. (B) Higher magnification of the area indicated in (A), demonstrating lobules of squamous epithelial cells with characteristic trichilemmal keratinization lacking an intervening granular layer (haematoxylin-eosin stain). Scale bars: 2.5 mm in (A) and 100 μm in (B).
DISCUSSION
The present case highlights the diagnostic challenges in distinguishing PTTs from their malignant counterparts. Historically, the histopathologic criteria distinguishing PTT from MPTT have been inconsistent across studies. Earlier reports variably regarded any degree of cytological atypia as malignant, whereas others emphasized that atypia alone was insufficient for this diagnosis. Well-circumscribed lesions with nuclear pleomorphism but without infiltrative growth, high mitotic activity or necrosis were later shown to behave benignly, and the term “malignant PTT” was proposed to be reserved for tumours showing a combination of infiltrative architecture, marked atypia and mitotic figures (1). These criteria were subsequently expanded based on a large series of cases, establishing a three-tiered classification – benign, low-grade malignant and high-grade malignant – defined by both cytologic and architectural features (8). Within this framework, MPTTs are regarded not as a distinct entity, but as part of a biologic continuum characterized by increasing degrees of cytologic and architectural atypia. In contrast, morphologic overlap between PTT and squamous cell carcinoma has also been described, further blurring diagnostic boundaries (9). Recent series (4, 10, 11) have largely reconciled these discrepancies by framing TTs as a biologic spectrum – from benign to atypical to malignant – where the diagnosis of MPTT requires integration of cytologic, architectural and clinical indicators such as rapid growth, >5 cm diameter and extracephalic location. According to these criteria, the present lesion was classified as a benign PTT based on its overall histopathologic architecture and clinical features.
PTTs generally appear on ultrasound as heterogeneous, well-circumscribed, cystic-solid masses located in the subcutaneous tissue with posterior enhancement and occasional calcifications (12). Importantly, colour Doppler studies typically reveal no internal blood flow (6, 7), reflecting the cystic and keratinous nature of the lesion. The absence of internal vascularity has therefore been considered a hallmark of benign PTTs.
In contrast to the typically avascular appearance of benign PTTs on colour Doppler ultrasonography (6, 7), the present lesion demonstrated marked intralesional vascularity with pulsatile flow signals. Histopathologic examination, however, confirmed a well-circumscribed PTT with preserved lobular architecture and without infiltrative growth or vascular invasion. This clinicoradiologic discrepancy highlights an important diagnostic pitfall: prominent Doppler flow can be observed even in histologically benign PTTs and should not be interpreted as definitive evidence of malignancy (13). In the present case, numerous intratumoural vascular spaces were identified histologically, likely accounting for the marked Doppler signals observed preoperatively. Accordingly, Doppler hypervascularity should be interpreted in conjunction with overall architectural and cytological features and regarded as a nonspecific but clinically useful finding that may prompt excision for definitive diagnosis and alert surgeons to the potential for increased intraoperative bleeding.
The differential diagnosis of a well-circumscribed subcutaneous scalp mass with heterogeneous internal echoes and prominent Doppler flow includes epidermal cysts, pilomatricoma and cutaneous squamous cell carcinoma. Uncomplicated epidermal cysts are typically avascular, whereas pilomatricomas may show internal calcifications with variable vascularity, and squamous cell carcinoma more often presents as an irregular, ill-defined, hypervascular lesion. In this context, unexpected hypervascularity within an otherwise well-defined cystic–solid mass should prompt consideration of adnexal tumours such as PTT rather than being interpreted as direct evidence of malignancy (13).
In summary, this case suggests that even when increased vascularity is detected on ultrasonography in lesions initially suspected to be trichilemmal cysts or epidermal cysts, the possibility of a benign PTT – as well as MPTT – should be considered. In such cases, treatment decisions should be based on an integrated assessment of clinical, imaging, intraoperative and histopathologic findings. This approach may help avoid overtreatment, as demonstrated in the present case, in which complete excision along the capsule was sufficient.
REFERENCES
- Folpe AL, Reisenauer AK, Mentzel T, Rütten A, Solomon AR. Proliferating trichilemmal tumors: clinicopathologic evaluation is a guide to biologic behavior. J Cutan Pathol 2003; 30: 492–498. https://doi.org/10.1034/j.1600-0560.2003.00041.x
- Singh P, Usman A, Motta L, Khan I. Malignant proliferating trichilemmal tumour. BMJ Case Rep 2018; 2018: bcr2018224460. https://doi.org/10.1136/bcr-2018-224460
- Kim UG, Kook DB, Kim TH, Kim CH. Trichilemmal carcinoma from proliferating trichilemmal cyst on the posterior neck. Arch Craniofac Surg 2017; 18: 50–53. https://doi.org/10.7181/acfs.2017.18.1.50
- Abdelhammed MH, Siatecka H, Diwan AH, Finch CJ, Haskins AD, Hernandez DJ, et al. A rare case of a malignant proliferating trichilemmal tumor: a molecular study harboring potential therapeutic significance and a review of literature. Dermatopathology 2024; 11: 354–363. https://doi.org/10.3390/dermatopathology11040038
- Miyachi H, Togawa Y, Yamamoto Y, Oguma R, Suehiro K, Matsue H. Proliferating trichilemmal tumour: a comparison of dermoscopic, ultrasonographic and histopathological features. Eur J Dermatol 2016; 26: 400–402. https://doi.org/10.1684/ejd.2016.2795
- Kwon KE, Kim SJ, Kim JH, Jung YY, Park NH, Park JY, et al. Imaging sonographic findings of in a case of proliferating trichilemmal tumor of a finger: A case report. J Clin Ultrasound 2018; 46: 215–217. https://doi.org/10.1002/jcu.22500
- He P, Cui LG, Wang JR, Zhao B, Chen W, Xu Y. Trichilemmal cyst: clinical and sonographic features. J Ultrasound Med 2019; 38: 91–96. https://doi.org/10.1002/jum.14666
- Ye J, Nappi O, Swanson PE, Patterson JW, Wick MR. Proliferating pilar tumors: a clinicopathologic study of 76 cases with a proposal for definition of benign and malignant variants. Am J Clin Pathol 2004; 122: 566–574. https://doi.org/10.1309/0XLEGFQ64XYJU4G6
- López-Ríos F, Rodríguez-Peralto JL, Aguilar A, Hernández L, Gallego M. Proliferating trichilemmal cyst with focal invasion: report of a case and a review of the literature. Am J Dermatopathol 2000; 22: 183–187. https://doi.org/10.1097/00000372-200004000-00018
- Kiel CM, Homøe P. Bleeding and ulcerating proliferating trichilemmal cyst, with delayed treatment due to coronavirus outbreak: a case report and review of the literature. Front Surg 2021; 8: 680160. https://doi.org/10.3389/fsurg.2021.680160
- Kearns-Turcotte S, Thériault M, Blouin MM. Malignant proliferating trichilemmal tumors arising in patients with multiple trichilemmal cysts: A case series. JAAD Case Rep 2022; 22: 42–46. https://doi.org/10.1016/j.jdcr.2022.01.033
- Kawaguchi M, Kato H, Suzui N, Miyazaki T, Tomita H, Hara A, et al. Imaging findings of trichilemmal cyst and proliferating trichilemmal tumour. Neuroradiol J 2021; 34: 615–621. https://doi.org/10.1177/19714009211017789
- Wortsman X. Ultrasound in dermatology: why, how, and when? Semin Ultrasound CT MR 2013; 34: 177–195. https://doi.org/10.1053/j.sult.2012.10.001