ORIGINAL REPORT
Cutaneous Leishmaniasis in Patients under Biologic Therapies: A Spanish CLINI-AEDV Multicentre Study
Andrea CIUDAD1* , Ramón M PUJOL1, Fernándo GALLARDO1, Miguel Antonio LASHERAS-PÉREZ2, Rafael BOTELLA ESTRADA2, Miquel ARMENGOT-CARBÓ3, Verónica FERNÁNDEZ TAPIA4, Bernat MAS MATAS4, Amador SOLÁ-TRUYOLS4, Álvaro AGUADO VÁZQUEZ5, Irene BALLESTER NORTES6, Mar LLAMAS-VELASCO7, Mireia ESQUIUS RAFAT8, Irene FUERTES9, Emili MASFERRER10,11, Esther ROÉ CRESPO12, Diego DE LA VEGA-RUIZ13, Vicenç ROCAMORA DURÁN14, Adrián IMBERNÓN-MOYA15, María del Mar BLANES MARTÍNEZ16, Patricia GARBAYO-SALMONS17, Laura ALONSO NARANJO18, Pedro MERCADER-GARCÍA6, Vicente EXPÓSITO-SERRANO17, David PESQUÉ1, Miguel Angel DESCALZO19, Ignacio GARCÍA-DOVAL19 and Gemma MARTÍN-EZQUERRA1
, Ramón M PUJOL1, Fernándo GALLARDO1, Miguel Antonio LASHERAS-PÉREZ2, Rafael BOTELLA ESTRADA2, Miquel ARMENGOT-CARBÓ3, Verónica FERNÁNDEZ TAPIA4, Bernat MAS MATAS4, Amador SOLÁ-TRUYOLS4, Álvaro AGUADO VÁZQUEZ5, Irene BALLESTER NORTES6, Mar LLAMAS-VELASCO7, Mireia ESQUIUS RAFAT8, Irene FUERTES9, Emili MASFERRER10,11, Esther ROÉ CRESPO12, Diego DE LA VEGA-RUIZ13, Vicenç ROCAMORA DURÁN14, Adrián IMBERNÓN-MOYA15, María del Mar BLANES MARTÍNEZ16, Patricia GARBAYO-SALMONS17, Laura ALONSO NARANJO18, Pedro MERCADER-GARCÍA6, Vicente EXPÓSITO-SERRANO17, David PESQUÉ1, Miguel Angel DESCALZO19, Ignacio GARCÍA-DOVAL19 and Gemma MARTÍN-EZQUERRA1
1Servicio de Dermatología, Hospital del Mar, Instituto Hospital del Mar de Investigaciones Médicas (IMIM), Departament de Medicina i Ciències de la Vida (MELIS), Universitat Pompeu Fabra, Barcelona, Spain, 2Servicio de Dermatología, Hospital Universitario y Politécnico La Fe, Valencia, Spain, 3Servicio de Dermatología, Hospital General Universitario de Castellón, Castellón de la Plana, Spain, 4Servicio de Dermatología, Hospital Universitario Son Llàtzer, Palma de Mallorca, Spain, 5Servicio de Dermatología, Hospital Universitario Doctor Peset, Valencia, Spain, 6Servicio de Dermatología, Hospital Universitario Morales Meseguer, Murcia, Spain, 7Servicio de Dermatología, Hospital Universitario La Princesa, Madrid, Spain, 8Servicio de Dermatología, Hospital Universitario Sant Joan de Déu de Manresa, Manresa, Barcelona, Spain, 9Servicio de Dermatología, Hospital Universitario Clinic de Barcelona, Barcelona, Spain, 10Servicio de Dermatología, Hospital Universitario de Terrassa, Terrassa, Barcelona, Spain, 11Universitat Central de Catalunya, Vic, Spain, 12Servicio de Dermatología, Hospital Universitario Santa Creu i Sant Pau, Barcelona, Spain, 13Servicio de Dermatología, Hospital Universitario Fundación Alcorcón, Madrid, Spain, 14Servicio de Dermatología, Hospital Universitario de Manacor, Manacor, Islas Baleares, Spain, 15Servicio de Dermatología, Hospital Universitario Severo Ochoa, Leganés, Madrid, Spain, 16Servicio de Dermatología, Hospital General Universitario Dr. Balmis de Alicante, Instituto de Investigación Sanitaria 37 y Biomédica de Alicante (ISABIAL), Alicante, Spain, 17Servicio de Dermatología, Hospital Universitario del Parc Taulí, Sabadell, Barcelona, Spain, 18Servicio de Dermatología, Hospital Universitario Doctor Negrín, Las Palmas de Gran Canaria, Las Palmas, Spain, and 19Unidad de Investigación, Academia Española de Dermatología y Venereología (AEDV), Madrid, Spain
Corr: Andrea Ciudad, Department of Dermatology, Hospital del Mar, Passeig Maritim 25-29, 08003, Barcelona, Spain. *Email: andreaciudad33@gmail.com
Key words: leishmaniasis; parasitic; biological therapies; immunosuppression; TNFα antagonist; opportunistic infection.
Citation: Acta Derm Venereol 2026; 106: adv-2025-0132. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0132.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Oct 28, 2025. Accepted after revision: Feb 3, 2026.
Published: Mar 12, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
The authors received no funding for this work.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
The study was reviewed and approved by the Hospital Universitario Puerta de Hierro-Majadahonda Research Ethics Committee (file 18/2022).
Anti-TNFα agents have been associated with the reactivation of granulomatous infections, including leishmaniasis. Cases of atypical or recurrent cutaneous (CL) or mucocutaneous leishmaniasis (MCL) have been reported in patients on biologics. We conducted a retrospective analysis of a multicenter database to describe the clinical characteristics and management of CL and MCL in patients receiving biological therapy in an endemic area. Clinical features, diagnostics, therapies, and outcomes were analyzed. Seventy-one patients were included (63 CL and 8 MCL). The most common underlying condition was inflammatory bowel disease (33.8%). Lesions were multifocal in 40% and larger than 1 cm in 94.4%. Treatment failures were more frequent in MCL (50%) than CL (8.1%). Biologic therapy was discontinued in 53.5%, leading to worsening of the underlying disease in 44.7%. No significant difference in cure rates was observed between patients who continued vs. discontinued biologics (p=0.868). These findings highlight the clinical burden of CL and MCL in patients undergoing anti-TNFα therapy and suggest that discontinuation of biologic therapy did not significantly impact cure rates, emphasizing the need for standardized strategies balancing infection control and underlying inflammatory disease management.
Leishmaniasis is a chronic protozoan infection of the mononuclear phagocyte system caused by various species of Leishmania and transmitted by female sandflies of the genera Phlebotomus (Old World) and Lutzomyia (New World). An estimated 700,000 to 1 million new cases occur annually worldwide. It is endemic in more than 90 countries, predominantly affecting regions of the Mediterranean basin, the Middle East, South America and parts of Asia and Africa. The clinical manifestations of leishmaniasis depend on several factors, particularly the virulence of the causative microorganism and the host immune response. Leishmania infection can manifest as a skin disease (cutaneous leishmaniasis, CL) and/or as involvement of mucous membranes (mucosal or mucocutaneous leishmaniasis, MCL) or even internal organs (visceral leishmaniasis, VL).
Leishmania infantum is the major causative agent of leishmaniasis in Southern Europe. In Mediterranean endemic areas, CL accounts for approximately 80–90% of reported cases, MCL for 5–15%, and VL for less than 10%, with Spain being one of the European countries with the highest reported incidence. In immunocompetent individuals, the immunity of the host is capable of controlling the infection, and the natural course of CL is often benign and tends to heal spontaneously (1, 2). Immune suppression may interfere with immune-mediated mechanisms implicated in the control and eradication of the parasite, leading to a reactivation of a latent parasitic disease (3, 4, 5). A polarized Th1 response with reduced Th2 cytokine activity has been associated with infection resistance and disease outcome (5).
Over the past decade, an increased number of biologic drugs have been introduced in the therapeutic armamentarium of different inflammatory and immune-mediated diseases. Treatments targeting tumour necrosis factor-α (TNFα) or interleukins have been identified as risk factors for the development and/or reactivation of several infectious diseases including leishmaniasis. Particularly, TNFα plays a critical role in host defence against various infectious diseases and its relevance in the immune response to Leishmania has already been investigated (6, 7, 8, 9, 10). TNFα has been implicated in the initial events of the infection, presenting a direct leishmanicidal activity, controlling parasite multiplication and promoting effective acquired immunity (4, 5, 6). Moreover, regulatory T cells and Th17 cells also appear to play significant roles in susceptibility and disease resistance (5, 7). In the literature, clinical manifestations of CL in patients receiving biologics have been described as atypical and more severe, frequently characterized by larger and more extensive ulcers, multifocal involvement and, in some cases, disseminated cutaneous disease (8, 9, 10, 11, 12, 13, 14, 15).
Additionally, the incidence of leishmaniasis is increasing in endemic regions such as the Mediterranean basin, probably in relation to factors like climate change, expanded travel and population migration (2, 3). A paradigmatic example is the large outbreak that occurred in the Madrid region between 2009 and 2012, which revealed the role of suburban transmission, wild hares as a key sylvatic reservoir of L. infantum and immunosuppressed hosts in the emergence of atypical and more complex clinical presentations (16).
This study aims to characterize the demographic and clinical profiles of CL in patients treated with biologic drugs, as well as the clinical approaches adopted by dermatologists in Spain, a country with endemic areas of the disease. Currently, there are no standardized clinical guidelines for the management of CL in patients undergoing biologic therapy, leading to variability in clinical decisions regarding whether to discontinue biologic treatment, perform additional extension studies or select appropriate therapies.
MATERIALS AND METHODS
Study design
We conducted a retrospective study of a prospectively collected multicentre database (CLINI-AEDV registry). Participating dermatology centres were required to include all prospective cases through the CLINI-AEDV platform, created by the Spanish Academy of Dermatology and Venerology (AEDV) (17). All centres followed a standardized data collection protocol implemented through this platform, including shared definitions. Data were collected and managed using the REDCap (Research Electronic Data Capture) system. CL was defined as the presence of one or more enlarging, slow-healing skin lesions caused by Leishmania, confirmed by one diagnostic method (histopathology, immunohistochemistry or specific qPCR amplification). MCL was defined as a form of leishmaniasis involving mucous membranes. Patient recruitment commenced on 20 March 2024 and concluded on 30 June 2024, spanning 14 weeks. Invitations to participate were sent to 87 CLINI-AEDV centres, encompassing both hospitals and private practices.
Study population
All patients with confirmed CL or MCL while receiving treatment with biologic agents were included in the study. Dermatologists enrolled patients during routine clinical practice. Both adult and paediatric patients were included in this series, with no upper or lower age limit for eligibility. All patients, or their legal guardians in the case of minors, provided their informed consent. Patients who consented to include clinical images signed a specific informed consent form.
Measures and variables
For each patient, data were collected on the type of biologic therapy, its indication and the duration of treatment. Clinical information recorded included the clinical form of leishmaniasis, number of lesions, size of the largest lesion, its anatomical location and whether there was mucosal or systemic involvement. Diagnostic methods, Leishmania species (if identified) and the extent of any diagnostic studies (if performed) were recorded. Treatment regimens and clinical outcomes at the last follow-up were also recorded, with particular attention to patients who discontinued biologic therapy. Treatment failure was defined as either nonresponse, indicated by persistent symptoms at the end of the treatment, or recurrence, defined as the reappearance of clinically compatible lesions within 6–12 months after apparent clinical cure, with microbiological confirmation when feasible.
Statistical analysis
Descriptive analyses were performed using absolute values and percentages for categorical variables, mean±standard deviation (SD) for normally distributed continuous variables, median (interquartile range, IQR) for non-normally distributed continuous variables. Comparisons of categorical variables were conducted using Fisher exact test, while continuous variables were compared using Student t-test (for means) or Wilcoxon test (for medians). p-Values <0.05 were considered statistically significant. Statistical analyses were conducted using STATA v.17.0 (Stata Corp. 2021. Stata Statistical Software: Release 17).
RESULTS
Seventy-one patients from 17 centres were included, predominantly from the Mediterranean area. Forty-nine patients (69%) were male, the mean age was 53 years (SD: 15.6). The most frequent underlying condition was inflammatory bowel disease (IBD) in 24 patients (33.8%), followed by psoriasis in 17 (23.9%). Most patients (94.4%) were receiving anti-TNFα therapy, with most therapies lasting for more than 12 months (87.3%). None of the patients included in the series presented immunosuppression related to HIV infection or solid organ transplantation. However, 58 out of 71 patients (81.7%) had received other immunosuppressive treatment in the context of their underlying inflammatory disease.
Sixty-three patients (88.7%) presented with CL, and 8 patients (11.3%) had MCL. One patient (1.4%) with CL subsequently developed visceral involvement. The most common lesion types were plaques in 29 (40.9%) and nodules in 28 cases (39.4%). The mean size of the largest lesion was 2.9 cm (range: 0.5–10 cm). The most affected areas were the arms and forearms, followed by the head and neck. Two or more lesions were observed in 27 out of 63 patients with CL (42.9%).
Leishmaniasis was diagnosed through direct observation of amastigotes in routine histopathological examination in 59 patients (81.9%) and by Leishmania polymerase chain reaction (qPCR) positivity in skin samples in 46 patients (64.8%). In 29 patients (40.9% patients), the species of Leishmania involved could be identified, reflecting limited access to species-level PCR in some centres and the use of alternative diagnostic methods. As expected, the most frequently found species was L. infantum in 26 patients (36.6%), followed by L. donovani in 3 patients (4.2%). Extension studies varied upon clinicians: complete blood tests in 54 (76.1%), abdominal ultrasound in 31 (43.7%) and bone marrow aspiration in 7 patients (9.9%).
The most common local therapy was intralesional antimonials in 49 patients (69%), followed by cryotherapy in 6 (8.5%). Cryotherapy was mainly used as an adjunctive treatment rather than as monotherapy (in 7 out of 8 patients). Among systemic treatments, amphotericin B (36.6%) and parenteral pentavalent antimonials (9.9%) were the most frequently used agents. Clinical resolution after the first treatment course was achieved in 57 cases (80.3%). The median disease-free follow-up period was 39 months (IQR: 10–60 months). Additional epidemiological, clinical and therapeutic details are summarized in Table I.
Table I. Demographic, clinical, therapeutic and outcome features after a median disease-free follow-up of 39.4 months (SD: 40.9 months)
| N (71) | %/SD | |
|---|---|---|
| Gender | ||
| Male | 49 | 69.0 |
| Female | 22 | 31.0 |
| Age (years), mean (SD) | 52.9 | 15.6 |
| Geographic area | ||
| Mediterranean areaa | 64 | 90.1 |
| Others (Madrid and Canary Island) | 7 | 9.9 |
| Disease for which receiving biologic | ||
| Inflammatory bowel disease | 24 | 33.8 |
| Rheumatologic disease other than psoriasis | 23 | 32.4 |
| Psoriasis | 17 | 23.9 |
| Others (hidradenitis, atopic dermatitis…) | 7 | 9.9 |
| Type of biologic | ||
| Anti-TNFα | 67 | 94.4 |
| Othersb | 4 | 5.6 |
| Duration of the treatment | ||
| Less than 6 months | 3 | 4.2 |
| >6 and <12 months | 5 | 7.1 |
| More than 12 months | 62 | 87.3 |
| Unknown | 1 | 1.4 |
| Type of lesion | ||
| Plaque | 29 | 40.9 |
| Nodule | 28 | 39.4 |
| Papule | 12 | 16.9 |
| Others (erysipela…) | 2 | 2.8 |
| Number of lesions | ||
| 1 | 42 | 60.0 |
| 2 | 12 | 17.1 |
| 3 | 5 | 7.2 |
| 4 or more | 10 | 15.7 |
| Size of the largest lesion in each patient (cm), mean (SD) | 2.9 | 2.3 |
| Location | n=88 | |
| Typical exposed areasc | 61 | 69.3 |
| Atypical hidden areas | 27 | 30.7 |
| Type of leishmaniasis | ||
| Cutaneous | 63 | 88.7 |
| Mucocutaneous | 8 | 11.3 |
| Visceral | 1 | 1.4 |
| Local treatment | ||
| Intralesional antimonials | 49 | 69.0 |
| Photodynamic therapy | 1 | 1.4 |
| Cryotherapy | 6 | 8.5 |
| Systemic treatment | ||
| Amphotericin B | 26 | 36.6 |
| Parenteral antimonials | 7 | 9.9 |
| Miltefosine | 6 | 8.5 |
| Fluconazole | 5 | 7.0 |
| Itraconazole | 3 | 4.2 |
| Discontinuation of biological treatment | ||
| No | 33 | 46.5 |
| Yes | 38 | 53.5 |
| Outcome at last follow-up | ||
| Complete cure | 57 | 80.3 |
| Persistence (no initial cure) | 3 | 4.2 |
| Recurrent course (initial cure and relapse) | 6 | 8.5 |
| Unknown | 5 | 7.0 |
| Number of recurrences | ||
| 1 | 5 | 1.4 |
| 2 | 1 | 2.8 |
| Worsening of underlying disease | ||
| No | 48 | 67.6 |
| Yes | 18 | 25.3 |
| Unknown | 3 | 4.2 |
| Disease-free follow-up time (months), mean (SD) | 39.4 | 40.9 |
|
aMediterranean endemic area includes Catalonia, Valencian community, Balearic Islands and Murcia. bOther biologic drugs included anti-IL 12/23 (1), anti-IL 23 (1) and anti-IL 4 (1). cTypical areas exposed to mosquito bites included arms and forearms, head and neck, legs and hands and wrists. Hidden and atypical areas of LC included thighs, back, abdomen, breast and axilla, buttocks and feet. It is important to note that patients presented with more than one lesion in different locations (total of lesions: 88). |
||
Nine patients (5 with CL and 4 with MCL) experienced treatment failure, defined either as recurrent (6 cases) or persistent disease (3 cases), as illustrated in Table II. Treatment failure was observed in 5 of 63 CL cases (7.9%) and in 4 of 8 cases of MCL (50%). All patients were receiving anti-TNFα therapy, often for prolonged periods (88.9%). All recurrences underwent an extension study, consisting of blood test and/or abdominal ultrasound. Systemic therapy, alone or combined with local treatments, was prescribed in all patients presenting treatment failure. In 4 of 9 cases, biologic therapy was discontinued, with half of these patients experiencing worsening of their underlying immune-mediated condition. Notably, in 2 patients, reintroduction of biologic therapy led to recurrence of CL.
Table II. Characteristics of failures: persistent and recurrent cases
| Pat. No. | Course of failure | Gender | Age at diagnosis | Underlying disease | Type of biologic therapy | Duration of biologic therapy (months) | Lesions, n | Type of leishmaniasis | Identified species | Local treatment | Systemic treatment | Treatment withdrawal |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Recurrent | Female | 42 | Other | Anti-TNF | >12 | 1 | MCL | L. infantum | IAM | Mitefosine, IvAM | No |
| 2 | Recurrent | Female | 57 | Nonpsoriatic rheumatologic disease | Anti-TNF | >12 | 1 | MCL | L. infantum | IAM | AnfB | No |
| 3 | Recurrent | Male | 50 | Inflammatory bowel disease | Anti-TNF | >12 | 1 | MCL | L. infantum | IAM | AnfB | Yes |
| 4 | Recurrent | Male | 53 | Inflammatory bowel disease | Anti-TNF | >12 | 3 | CL | L. infantum | None | Mitefosine, IvAM | No |
| 5 | Persistent | Male | 79 | Non-psoriatic rheumatologic disease | Anti-TNF | >12 | 5 | CL | L. donovani | IAM | Fluconazole | No |
| 6 | Persistent | Male | 73 | Psoriasis | Anti-TNF | >12 | 2 | CL | No | IAM | None | Yes |
| 7 | Recurrent | Male | 74 | Nonpsoriatic rheumatologic disease | Anti-TNF | >12 | 4 | CL | No | IAM | AnfB | No |
| 8 | Recurrent | Female | 64 | Psoriasis | Anti-TNF | >12 | 1 | MCL | No | None | Miltefosine | Yes |
| 9 | Persistent | Male | 56 | Inflammatory bowel disease | Anti-TNF | 6–12 | 1 | CL | No | IAM, cryotherapy | None | Yes |
|
AnfB:amphotericin B; CL:cutaneous leishmaniasis; IAM:intralesional antimonials; IvAM:intravenous antimonials; MCL:mucocutaneous leishmaniasis. |
||||||||||||
Following diagnosis, biological therapy was continued in 46.5% of patients and withdrawn in 53.5%. As presented in Table III, among those who achieved complete cure (26 who continued and 31 who discontinued biologic treatment), no statistically significant differences in outcomes were observed between the two groups (p=0.868). However, worsening of the underlying immune-mediated disease was reported in 17 of 38 patients who discontinued biologic therapy (44.7%).
Table III. Comparison between patients who continued and discontinued biologic therapy
| Variables | Continuation of biologic therapy | Discontinuation of biologic therapy | |||
|---|---|---|---|---|---|
| N=33 | % | N=38 | % | p-value | |
| Undergoing disease | |||||
| Inflammatory bowel disease | 13 | 39.4 | 11 | 29.0 | 0.071 |
| Non psoriatic rheumatic disease | 8 | 24.2 | 15 | 39.5 | |
| Psoriasis | 6 | 18.2 | 11 | 29.0 | |
| Others (atopic dermatitis, hidradenitis…) | 6 | 18.2 | 1 | 2.6 | |
| Type of biologic | |||||
| Anti-TNF | 30 | 90.9 | 37 | 97.4 | 0.239 |
| Othersa | 3 | 9.1 | 1 | 2.6 | |
| Duration of biologic treatment | |||||
| Less than 6 months | 0 | 0 | 3 | 7.9 | 0.248 |
| >6 and <12 months | 3 | 9.1 | 2 | 5.3 | |
| More than 12 months | 29 | 87.9 | 33 | 86.8 | |
| Unknown | 1 | 3.0 | 0 | 0.00 | |
| Number of lesions | |||||
| 1 | 18 | 54.5 | 24 | 64.9 | 0.121 |
| 2 | 4 | 12.1 | 8 | 21.6 | |
| 3 or more | 11 | 33.3 | 5 | 13.5 | |
| Type of leishmaniasis | |||||
| Cutaneous | 30 | 90.9 | 32 | 84.2 | 0.544 |
| Mucocutaneous | 3 | 9.1 | 5 | 13.2 | |
| Visceral | 0 | 0 | 1 | 2.6 | |
| Treatment | |||||
| Local | 23 | 69.7 | 26 | 68.4 | 0.908 |
| Amphotericin B | 12 | 36.4 | 14 | 36.8 | 0.967 |
| Parenteral antimonials | 2 | 6.1 | 5 | 13.2 | 0.317 |
| Miltefosine | 4 | 12.1 | 2 | 5.3 | 0.300 |
| Fluconazole | 2 | 6.1 | 3 | 7.9 | 0.763 |
| Itraconazole | 2 | 6.1 | 1 | 2.6 | 0.474 |
| Result at last review | |||||
| Cure | 26 | 78.8 | 31 | 81.6 | 0.868 |
| Recurrent course | 3 | 9.7 | 3 | 8.6 | |
| Persistence | 2 | 6.4 | 1 | 2.8 | |
| Number of recurrences | |||||
| 1 | 3 | 100 | 2 | 66.7 | 0.273 |
| 2 | 0 | 0 | 1 | 33.3 | |
| Worsening of underlying disease | |||||
| No | 29 | 87.9 | 19 | 50 | <0.001 |
| Yes | 1 | 3.0 | 17 | 44.7 | |
| Unknown | 3 | 9.1 | 2 | 5.3 | |
|
aOther biologic drugs included anti-IL 12/23, anti-IL 23 and anti-IL 4. |
|||||
DISCUSSION
In our cohort, and in concordance with other series (9, 10, 11, 12, 13, 14, 15), TNFα inhibitors were the biologic agents most frequently associated with CL (94.4%). Treatment with anti-TNFα is usually prescribed for a wide range of immune-mediated disorders including IBD, rheumatic diseases or psoriasis. Interestingly, psoriasis itself has been linked to an increased risk of CL (18).
This large multicentre study of CL and MCL in patients receiving biologic therapy within an endemic region provides a representative overview of this growing patient population. CL was by far the most reported form of leishmaniasis in patients receiving biologics (88.7%), with previous series ranging from 60 to 90%, followed by MCL (11.3%), varying from 10 to 32% in prior reports (9, 10, 11, 12). VL has been reported at variable frequencies, ranging from 1.4 to 33%.19, 20 In agreement with our results, a recent Spanish cohort of patients with IBD who developed leishmaniasis also identified CL as the predominant phenotype (85%), with a small proportion of VL (11%) (15).
Regarding risk factors for VL, age has been identified as the only independent predictor of visceral involvement, whereas biologic therapy, mainly anti-TNFα agents, has been more strongly associated with cutaneous or mucocutaneous forms (15). Together with our findings, this suggests that host factors, particularly advanced age, play a greater role than exposure to biologic therapy in determining visceral tropism, while patients receiving biologics in Mediterranean endemic settings predominantly develop CL or MCL.
A notable finding in our series was the high proportion of extensive and multifocal lesions. Almost half of patients with CL (42.9%) presented two or more lesions, and in most cases (94.4%), lesion size exceeded 1 cm in diameter, with a mean of 2.9 cm. Disseminated forms – defined as the presence of lesions distributed across separate body areas (21) – were observed in 15.7% of patients.
In previous series, immunosuppressed patients frequently exhibited multiple lesions, often exceeding four in number, whereas immunocompetent individuals typically presented with discrete solitary papules or plaques measuring 5–10 mm in diameter (22, 23, 24, 25, 26). Similarly, patients treated with anti-TNFα drugs have been reported to develop unusually large, multifocal lesions with little or no spontaneous regression (8, 15).
Accordingly, Palacios-Díaz et al. (8) observed that lesions in biologic-treated patients were predominantly ulcerated plaques (92%), of larger median size (2.5 cm) and requiring a higher number of intralesional meglumine antimoniate infiltrations than controls not exposed to biologics. Another retrospective review of 49 Mediterranean cases of leishmaniasis under anti-TNFα therapy found that 10 of 28 patients with CL presented with multifocal lesions (9). These consistent observations suggest that biologic-induced immunomodulation may alter lesion morphology and healing kinetics, without increasing the risk of visceral dissemination.
The diagnosis of CL or MCL can be particularly challenging, as its clinical presentation often mimics other ulcerative or granulomatous dermatoses, including syphilis, nontuberculous mycobacterial infections and deep mycoses, or even noninfectious conditions such as squamous cell carcinoma (27). The presence of atypical or multiple lesions in this patient population may further complicate the already difficult clinical recognition of leishmaniasis. Consequently, studies like the present one are crucial to better characterize the clinical spectrum of leishmaniasis in this specific setting, aiding clinicians in improving early diagnosis and management. Representative clinical and dermoscopic features of CL in patients with anti-TNFα therapy are illustrated in Fig. 1.
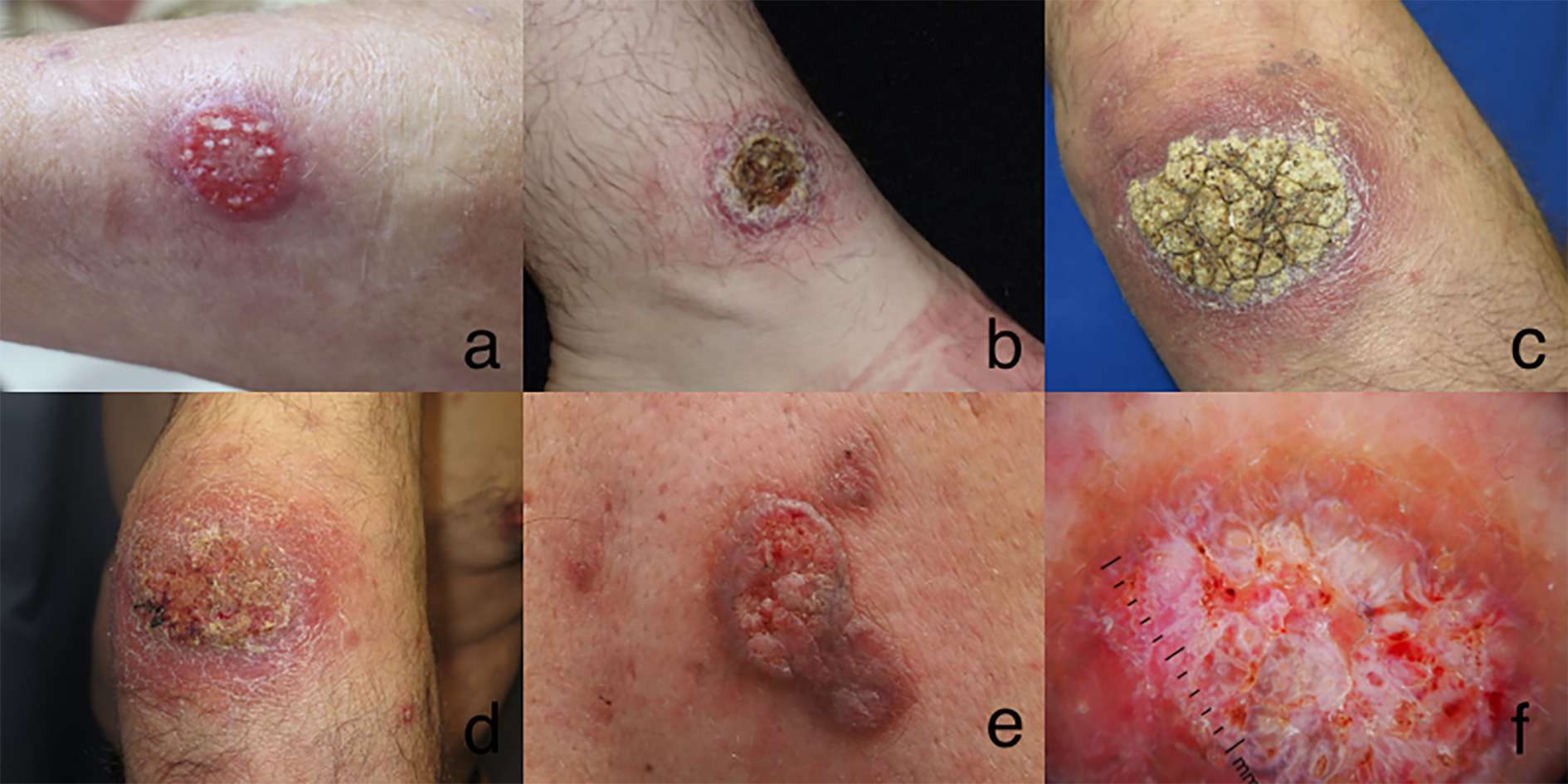
Fig. 1. Clinical features of CL infection. Clinical features of CL infection. (a) Single large plaque with erosive bleeding surface in left arm. (b) Crusted large plaque in right ankle. (c and d) Large hyperkeratotic crust plaque in the elbow of patient with Crohn’s disease in treatment with infliximab before and after treatment with intralesional antimonials. (e and f) Clinical and dermoscopic features of a large ulcerated plaque on the back of a patient with suppurative hidradenitis receiving treatment with adalimumab. Note the raised edge and satellite papules.
The natural history of CL caused by Old World species tends to heal spontaneously, being generally more prolonged for L. infantum, which predominated in our cohort, compared with other species such as L. major. Indeed, when lesions are smaller than 5 cm in diameter and are located in areas without risk of disfigurement or functional impairment, simple wound care alone is generally sufficient (1, 25). The reactivation of latent parasites within granulomatous lesions could provide a feasible explanation for the development of leishmaniasis under TNFα inhibitor treatment (3, 4, 5).
Although some authors have suggested that discontinuing TNFα therapy improves treatment outcomes (12, 13), our analysis revealed no statistically significant differences in cure rates between patients who continued (78.8%) and those who stopped biologic therapy (81.6%) (p=0.868). However, 44.7% of patients experienced a flare or worsening of their underlying immune-mediated disease after discontinuing biologics, indicating that, with careful supervision, continuation of biologics may be a safe and viable option for non-complicated CL. This observation aligns with other studies reporting no significant differences in clinical outcomes or relapse rates between patients who maintained or discontinued biologic therapy in cases of CL and MCL (8, 15).
There are notable discrepancies in treatment approaches for patients receiving TNFα blockers (8, 9, 10, 11, 12, 13, 14, 15). Some reports describe successful outcomes with intralesional antimonials and liposomal amphotericin B used as monotherapy, both leading to lesion resolution without relapses (9). Conversely, other studies have reported poor responses to intralesional meglumine antimoniate in patients on TNFα inhibitors, requiring systemic therapies with intravenous liposomal amphotericin B or intramuscular antimonials (28). In our series, treatment approaches varied: intralesional antimonials were the most used local therapy (69%), while systemic treatments like amphotericin B (36.6%) and pentavalent antimonials (9.9%) were also frequently administered.
Follow-up data, with a median disease-free period of 39 months, indicate that treatments can be effective in many cases, although responses are generally less favourable than in immunocompetent populations (10, 11). Indeed, persistence (4.2%) and recurrence (8.5%) of disease underscore the need for careful long-term monitoring of leishmaniasis in patients on biologic therapies.
In our series, treatment failure, defined as persistent or recurrent lesions after treatment, occurred in 50% of patients with MCL compared to 8% of cases with CL. All MCL cases were treated with systemic therapies including liposomal amphotericin B, miltefosine or intravenous antimonials. Recurrences were consistently associated with prolonged anti-TNFα exposure. After ruling out visceral involvement through blood tests, abdominal ultrasounds and peripheral blood qPCR, a different systemic regimen, often combined with local therapies were the most common approaches for treatment failures. Two-thirds of the patients experienced a relapsing course with various recurrences (associated with biologic reintroduction), suggesting that reinitiating anti-TNFα therapy after a first relapse should be avoided.
For patients with noncomplicated CL, when biologic therapy remains clinically necessary, a reasonable strategy may involve adding a systemic agent such as liposomal amphotericin B or pentavalent antimonials, using local treatments as adjuncts, while maintaining biologic therapy. In patients with very localized CL, particularly when the lesion is small and located in low-risk anatomical areas, it is plausible that systemic antileishmanial therapy may not be necessary even if biologic treatment is maintained. In such cases, local therapies alone such as intralesional antimonials, cryotherapy or topical agents may provide satisfactory outcomes while avoiding the toxicity associated with systemic agents (29). This tailored approach is especially relevant for patients in whom discontinuation of biologic therapy could lead to significant worsening of their underlying immune-mediated disease. Additionally, close monitoring should be the rule to identify early relapses.
Switching to alternative immunomodulators, such as IL-17 or IL-23 inhibitors – which carry a lower risk of reactivating granulomatous infections – may offer improved outcomes and should be considered. This approach aligns with current recommendations for patients with psoriasis and latent tuberculosis infection who have contraindications for undergoing chemoprophylaxis (25).
Our study has some limitations. The sample size (n=71) limits the statistical power of comparative analyses and should be considered when interpreting subgroup results. Leishmania species identification was achieved in fewer than half of cases, and imported species may display distinct behaviour compared with the endemic strains in our region, potentially influencing outcomes. Furthermore, most cases were retrospectively collected from multiple centres, introducing possible selection and measurement bias. Finally, the relatively short follow-up period, particularly for MCL, limits our assessment of long-term relapse risk.
In conclusion, this large retrospective study from an endemic region illustrates current practices in the management of leishmaniasis in patients receiving biologic agents. Continuation of biologic therapy, combined with close and regular follow-up, may be appropriate for most cases of noncomplicated CL. However, consideration of switching to alternative immunomodulatory strategies such as IL-17 or IL-23 inhibitors may be warranted for MCL and CL cases with diffuse, locally complicated or recurrent lesions. Optimal strategies to balance the management of leishmaniasis and the underlying immunomodulatory disease remain to be defined.
ACKNOWLEDGEMENTS
The authors gratefully acknowledge the contribution of all collaborating centres and clinicians involved in the diagnosis and management of the reported cases.
REFERENCES
- Handler MZ, Patel PA, Kapila R, Al-Qubati Y, Schwartz RA. Cutaneous and mucocutaneous leishmaniasis: Clinical perspectives. J Am Acad Dermatol 2015; 73: 897–908. https://doi.org/10.1016/j.jaad.2014.08.051
- Di Muccio T, Scalone A, Bruno A, Marangi M, Grande R, Armignacco O, et al. Epidemiology of imported leishmaniasis in Italy: implications for a European endemic country. PLoS One 2015; 10: e0129418. https://doi.org/10.1371/journal.pone.0129418
- Foroutan M, Dalvand S, Khademvatan S, Majidiani H, Khalkhali H, Masoumifard S, et al. A systematic review and meta-analysis of the prevalence of Leishmania infection in blood donors. Transfus Apher Sci 2017; 56: 544–551. https://doi.org/10.1016/j.transci.2017.07.001
- Maspi N, Abdoli A, Ghaffarifar F. Pro- and anti-inflammatory cytokines in cutaneous leishmaniasis: a review. Pathog Glob Health 2016; 110: 247–260. https://doi.org/10.1080/20477724.2016.1232042
- Kaye P, Scott P. Leishmaniasis: complexity at the host-pathogen interface. Nat Rev Microbiol 2011; 9: 604–615. https://doi.org/10.1038/nrmicro2608
- Liew FY, Parkinson C, Millott S, Severn A, Carrier M. Tumour necrosis factor (TNF alpha) in leishmaniasis. I. TNF alpha mediates host protection against cutaneous leishmaniasis. Immunology 1990; 69: 570–573.
- Nascimento MSL, Carregaro V, Lima-Júnior DS, Costa DL, Ryffel B, Duthie MS, et al. Interleukin 17A acts synergistically with interferon γ to promote protection against Leishmania infantum infection. J Infect Dis 2015; 211: 1015–1026. https://doi.org/10.1093/infdis/jiu531
- Palacios-Diaz RD, Sahuquillo-Torralba A, Rocamora-Durán V, Unamuno-Bustos B de, Salavert-Lleti M, Santos-Alarcón S, et al. Clinicopathological characteristics of cutaneous and mucocutaneous leishmaniasis in patients treated with TNF-α inhibitors. J Dtsch Dermatol Ges 2023; 21: 473–480. https://doi.org/10.1111/ddg.15007
- Guedes-Barbosa LS, Pereira da Costa I, Fernandes V, Henrique da Mota LM, de Menezes I, Aaron Scheinberg M. Leishmaniasis during anti-tumor necrosis factor therapy: report of 4 cases and review of the literature (additional 28 cases). Semin Arthritis Rheum 2013; 43: 152–157. https://doi.org/10.1016/j.semarthrit.2013.01.006
- Bosch-Nicolau P, Ubals M, Salvador F, Sánchez-Montalvá A, Aparicio G, Erra A, et al. Leishmaniasis and tumor necrosis factor alpha antagonists in the Mediterranean basin. A switch in clinical expression. PLoS Negl Trop Dis 2019; 13: e0007708. https://doi.org/10.1371/journal.pntd.0007708
- Schneider P, Bouaziz JD, Foulet F, Duong TA, Allanore LV, Bagot M. Multifocal cutaneous leishmaniasis due to Leishmania infantum under adalimumab therapy. Ann Dermatol Venereol 2009; 136: 815–816. https://doi.org/10.1016/j.annder.2009.05.008
- Neumayr ALC, Morizot G, Visser LG, Lockwood DNJ, Beck BR, Schneider S, et al. Clinical aspects and management of cutaneous leishmaniasis in rheumatoid patients treated with TNF-α antagonists. Travel Med Infect Dis 2013; 11: 412–420. https://doi.org/10.1016/j.tmaid.2013.05.003
- Pérez-Sola MJ, Torre-Cisneros J, Pérez-Zafrilla B, Carmona L, Descalzo MA, Gómez-Reino JJ. Infections in patients treated with tumor necrosis factor antagonists: incidence, etiology and mortality in the BIOBADASER registry. Medicina Clínica2011; 137: 533–540. https://doi.org/10.1016/j.medcli.2010.11.032
- Zanger P, Kötter I, Kremsner PG, Gabrysch S. Tumor necrosis factor alpha antagonist drugs and leishmaniasis in Europe. Clin Microbiol Infect 2012; 18: 670–676. https://doi.org/10.1111/j.1469-0691.2011.03674.x
- Madero-Velázquez L, Mínguez A, Mayorga L, Ramírez JJ, Moreno N, Amorós C, et al. Leishmaniasis in patients with inflammatory bowel disease: a national multicenter study of GETECCU. United European Gastroenterol J 2025; 13: 674–684. https://doi.org/10.1002/ueg2.12740
- Aguado M, Espinosa P, Romero-Maté A, Tardío JC, Córdoba S, Borbujo J. Outbreak of cutaneous leishmaniasis in Fuenlabrada, Madrid. Actas Dermosifiliogr 2013; 104: 334–342. https://doi.org/10.1016/j.adengl.2013.03.005
- Academia española de Dermatología y Venereología. CLINI-AED case series collection platform. 2024. Available from: https://redcap.fundacionpielsana.es/surveys/?s=PKLPEJRT4XY4RKKR
- Colomba C, Saporito L, Bonura S, Campisi G, Di Carlo P, Panzarella V, et al. Leishmania infection in psoriasis. Journal of Infection 2020; 80: 578–606. https://doi.org/10.1016/j.jinf.2020.01.019
- De Leonardis F, Govoni M, Lo Monaco A, Trotta F. Visceral leishmaniasis and anti-TNF-alpha therapy: case report and review of the literature. Clin Exp Rheumatol 2009; 27: 503–506.
- Xynos ID, Tektonidou MG, Pikazis D, Sipsas NV. Leishmaniasis, autoimmune rheumatic disease, and anti-tumor necrosis factor therapy, Europe. Emerg Infect Dis 2009; 15: 956–959. https://doi.org/10.3201/eid1506.090101
- Marcoval J, Penín RM. Evolution of cutaneous leishmaniasis in the last 30 years in a tertiary hospital of the European Mediterranean coast. Int J Dermatology 2017; 56: 750–753. https://doi.org/10.1111/ijd.13588
- Herrador Z, Gherasim A, Jimenez BC, Granados M del sol, San Martín JV, Aparicio P. Epidemiological changes in leishmaniasis in Spain according to hospitalization-based records, 1997-2011: raising awareness towards leishmaniasis in non-HIV patients. PLoS Negl Trop Dis 2015; 9: e0003594. https://doi.org/10.1371/journal.pntd.0003594
- Daudén E, García C, Zarco C, López S, Iglesias L. Cutaneous leishmaniasis: 20 years’ experience in a Spanish tertiary care hospital. Actas Dermosifiliogr 1990; 81: 395–404.
- Urrutia S, García C, Schoendorff C, Sáez A, Olivares M, García Almagro D. Cutaneous leishmaniasis in the Toledo province. A study of 43 patients. Actas Dermosifiliogr 2000; 91: 1–8.
- Torres T, Chiricozzi A, Puig L, Lé AM, Marzano AV, Dapavo P, et al. Treatment of psoriasis patients with latent tuberculosis using IL-17 and IL-23 inhibitors: a retrospective, multinational, multicentre study. Am J Clin Dermatol 2024; 25: 333–342. https://doi.org/10.1007/s40257-024-00845-4
- Garrido-Jareño M, Sahuquillo-Torralba A, Chouman-Arcas R, Castro-Hernández I, Molina-Moreno JM, Llavador-Ros M, et al. Cutaneous and mucocutaneous leishmaniasis: experience of a Mediterranean hospital. Parasit Vectors 2020; 13: 24. https://doi.org/10.1186/s13071-020-3901-1
- Chaudhary RG, Bilimoria FE, Katare SK. Diffuse cutaneous leishmaniasis: co-infection with human immunodeficiency virus (HIV). Indian J Dermatol Venereol Leprol 2008; 74: 641–643. https://doi.org/10.4103/0378-6323.45111
- Giavedoni P, Iranzo P, Fuertes I, Estrach T, Alsina Gibert M. Cutaneous leishmaniasis: 20 years’ experience in a Spanish tertiary care hospital. Actas Dermosifiliogr 2015; 106: 310–316. https://doi.org/10.1016/j.ad.2014.12.003
- Buffet PA, Rosenthal É, Gangneux JP, Lightburne E, Couppié P, Morizot G, et al. Therapy of leishmaniasis in France: consensus on proposed guidelines. Presse Med 2011; 40: 173–184. https://doi.org/10.1016/j.lpm.2010.09.023