SHORT COMMUNICATION
Successful Treatment of Pityriasis Rubra Pilaris with Upadacitinib: Report of Two Cases
Yanyu DU1,2†, Xurong LIU1†, Lian ZHANG1, Jing ZHONG1* and Renliang HE1*
1Department of Dermatologic Surgery and Dermatologic Oncology, Guangzhou, China, Dermatology Hospital of Southern Medical University, Guangdong Provincal Dermatology Hospital, and 2The First School of Clinical Medicine, Southern Medical University, Guangzhou, China. *Emails: hahada2008@outlook.com; zshrl2006@126.com
†These authors contributed equally to this work and should be considered as co-first authors.
Citation: Acta Derm Venereol 2026; 106: adv-2025-0149. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0149.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Nov 2, 2025. Accepted after revision: Mar 4, 2026.
Published: Mar 26, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
This work was supported by the research project of the Basic and Applied Basic Research Foundation of Guangdong Province. (2025A1515010822).
The data that support the findings of this study are available from the corresponding author upon reasonable request.
This study was approved by the Institutional Review Board of Dermatology Hospital of Southern Medical University, strictly adhering to the Declaration of Helsinki and the International Council for Harmonisation - Good Clinical Practice (ICH-GCP) guidelines, with informed consent obtained from all patients. The patients in this manuscript have given written informed consent to publication of their case details.
Pityriasis rubra pilaris (PRP) is a rare, chronic inflammatory dermatosis characterized by erythematous plaques with islands of sparing, follicular hyperkeratotic papules and palmoplantar keratoderma. The pathogenesis is thought to involve dysregulation of the interleukin (IL)-23/Th17 axis (1). Therapeutic options are limited, with conventional systemic agents like retinoids often yielding suboptimal results. Biological agents targeting IL-17 or IL-23 have shown promise, yet a subset of patients remains refractory (2, 3, 4). Recently, small molecule Janus kinase (JAK) inhibitors, such as the selective JAK1 inhibitor upadacitinib, have emerged as potential alternatives, with a few case reports suggesting efficacy in recalcitrant PRP (5, 6, 7, 8). Here, we report two cases of PRP that responded favourably to upadacitinib after insufficient response to prior therapies.
CASE REPORTS
Case 1: A 24-year-old Asian male patient presented with a sudden onset of diffuse erythematous papules and scaly plaques on the face, trunk and extremities for 2 weeks. The lesions initially appeared as follicular papules on the dorsal hands, rapidly spreading and coalescing into orange-red, well-defined plaques. The patient reported skin tightness and mild pruritus, with no personal or family history of psoriasis or pityriasis rubra pilaris . Physical examination revealed widespread erythematous and scaly plaques involving approximately 30% of the body surface area, facial lesions at baseline are shown in Fig. 1aa,with typical “islands of sparing” on the chest(Fig. 2a) . Skin biopsy from the facial lesion showed histopathological features consistent with PRP. The patient was initially treated with acitretin (30 mg/day) and topical corticosteroids, but there was no improvement after 4 weeks, and the disease continued to progress. The treatment regimen was switched to upadacitinib 15 mg once daily. After 4 weeks of therapy, the patient exhibited significant improvement, with thinning of plaques and reduction in erythema on the face and chest (Figs. 1b and 2b) . At the 5-month follow-up, the skin had completely cleared, leaving only post-inflammatory hyperpigmentation (Figs. 1c and 2c) . Upadacitinib was well tolerated, with no laboratory abnormalities, and the patient has remained in remission on continued 15 mg daily therapy.
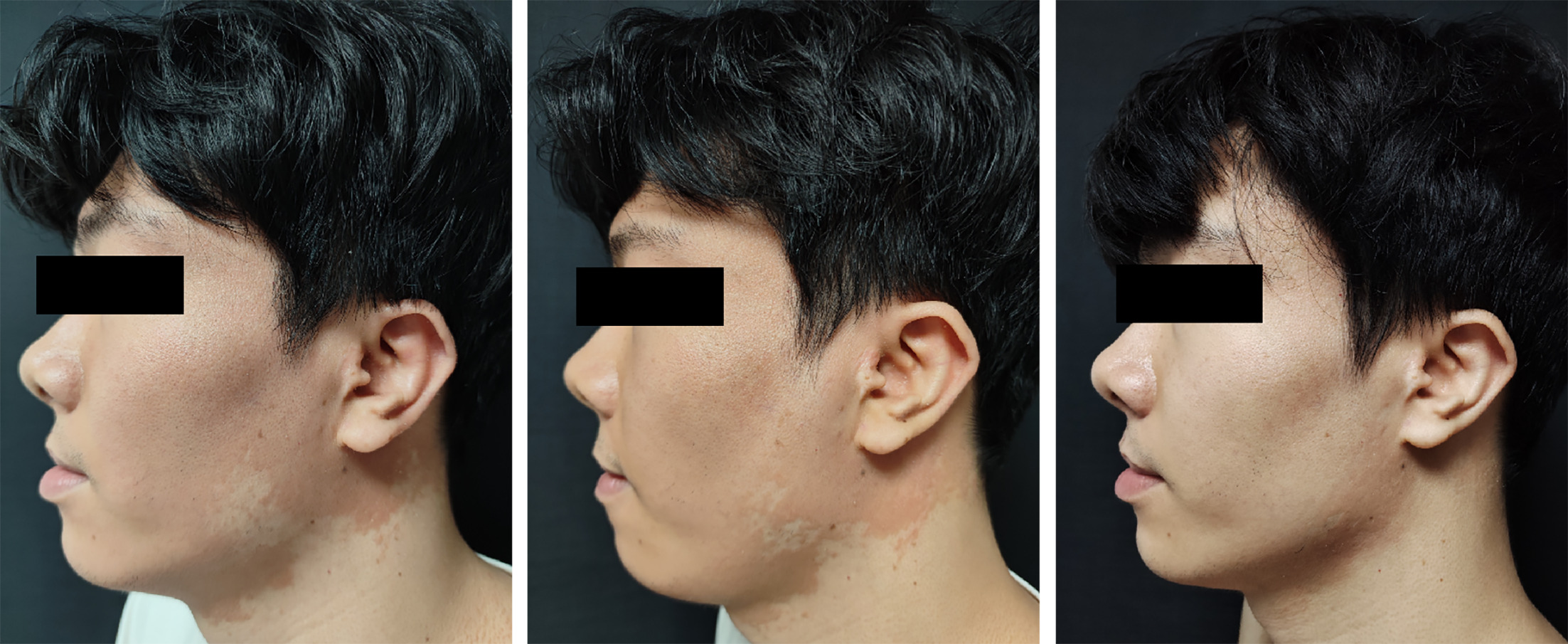
Fig. 1. Case 1. Clinical course of facial lesions. At baseline (Fig. 1a), diffuse orange-red scaly plaques involved the face with islands of spared skin. After 4 weeks of upadacitinib 15 mg daily (Fig. 1b), marked improvement was observed, with only faint residual erythema. At 5 months (Fig. 1c), the lesions had completely cleared.
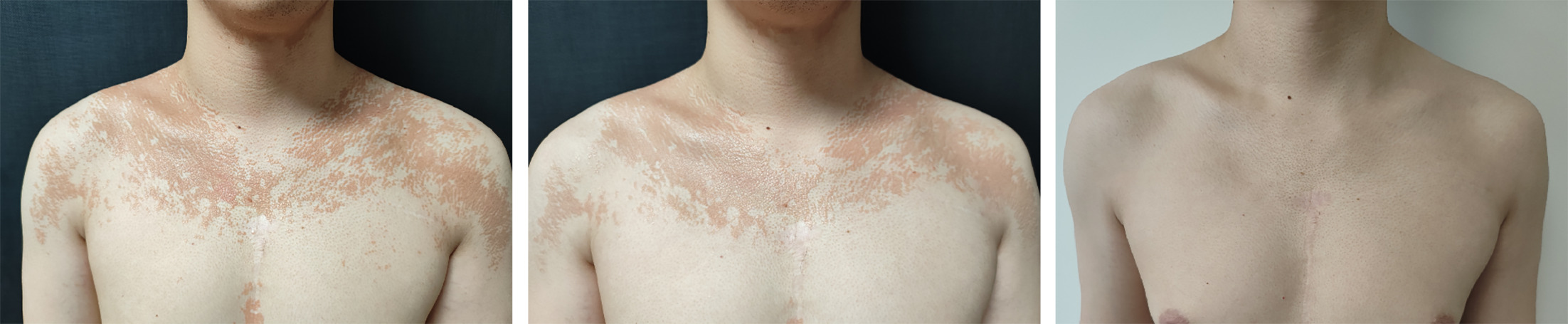
Fig. 2. Case 1. Clinical course of chest lesions. At baseline (Fig. 2a), diffuse orange-red scaly plaques involved the chest with islands of spared skin. After 4 weeks of upadacitinib 15 mg daily (Fig. 2b), marked improvement was observed, with only faint residual erythema. At 5 months (Fig. 2c), the lesions had completely cleared.
Case 2: A 25-year-old female patient had a 5-year history of recurrent, persistent hyperkeratotic erythematous plaques. The lesions initially appeared on both lower legs as scaly red patches and gradually increased in extent. She exhibited marked palmoplantar keratoderma with waxy thickening and painful fissures, as well as diffuse follicular plug-like papules on the dorsal fingers, producing a rough “nutmeg grater” texture. Multiple skin biopsies confirmed a diagnosis of PRP. Previous treatments included methotrexate (15 mg weekly for 6 months), acitretin (25–35 mg daily for 8 months) and adalimumab (>4 months), all of which produced limited or transient improvement. On examination, hyperkeratotic follicular papules and plaques were noted on the elbows and knees, while the palms and soles showed prominent yellowish keratoderma with fissures (Fig. 3a) . Given the refractory nature of her disease, upadacitinib 15 mg once daily was initiated. After 4 weeks, the lesions improved markedly: erythema and scaling decreased by approximately 50%, plaques became thinner, palmoplantar keratoses softened and pain was relieved(Fig. 3b) . At 4 months, the skin lesions had nearly completely resolved , and palmoplantar hyperkeratosis had almost disappeared, greatly improving mobility(Fig. 3c) . The patient continued maintenance therapy with upadacitinib, experiencing only mild, occasional acne without other adverse effects, and laboratory results remained within normal limits.
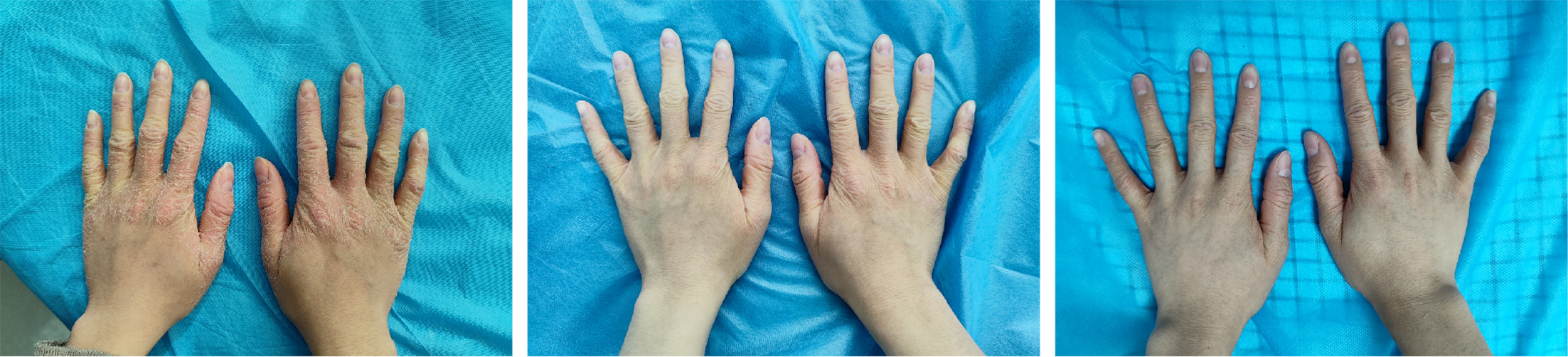
Fig. 3. Case 2. Clinical course of palmoplantar lesions. At baseline (Fig. 3a), a 25-year-old woman with longstanding PRP presented with palmoplantar keratoderma. After 1 month of upadacitinib (Fig. 3b), scaling and erythema on the hands were significantly reduced. After 4 months (Fig. 3c), the plaques had completely resolved, with restoration of normal skin texture.
DISCUSSION
The management of PRP remains challenging. Traditional systemic therapies, including retinoids and methotrexate, often have variable efficacy and side-effect profiles. Biologics, particularly IL-17A inhibitors like secukinumab and ixekizumab, have become important options, supported by clinical trials and numerous case reports (2, 3). However, resistance can occur, potentially linked to persistent elevation of other cytokines such as IL-17C (9). JAK inhibitors offer a broader upstream mechanism by interfering with the signalling of multiple cytokines involved in inflammatory pathways (10).
The efficacy of upadacitinib in our patients aligns with emerging literature. A recent systematic review identified upadacitinib as an effective small-molecule drug for PRP, with reported cases showing complete or significant clearance, often in patients refractory to biologics (8). Case reports have documented successful use of upadacitinib in both erythrodermic PRP and PRP coexisting with generalized pustular psoriasis, with rapid and sustained responses (5, 6). Another report highlighted its utility in an elderly patient with refractory disease (7). The mechanism is hypothesized to involve the inhibition of JAK/STAT pathways, thereby suppressing a wider array of pro-inflammatory cytokines (e.g. IL-6, IL-12, IL-23) implicated in PRP pathogenesis compared to monoclonal antibodies targeting single cytokines (7, 10). This broader inhibition may explain its effectiveness in cases where more targeted biologic therapies fail.
Our cases add to the growing body of evidence supporting upadacitinib as a viable therapeutic alternative for PRP. Both patients, who had an inadequate response to first-line retinoid therapy, achieved near-complete clearance with upadacitinib monotherapy within a few months, demonstrating a favourable safety profile. While larger, controlled studies are needed to definitively establish its efficacy and safety, these observations suggest that upadacitinib warrants consideration for the treatment of PRP, especially in cases refractory to conventional systemic agents or specific biologics.
REFERENCES
- Feldmeyer L, Mylonas A, Demaria O, Mennella A, Yawalkar N, Laffitte E, et al. Interleukin 23-helper T cell 17 axis as a treatment target for pityriasis rubra pilaris. JAMA Dermatol 2017; 153: 304–308. https://doi.org/10.1001/jamadermatol.2016.5384
- Boudreaux BW, Pincelli TP, Bhullar PK, Patel MH, Brumfiel CM, Li X, et al. Secukinumab for the treatment of adult-onset pityriasis rubra pilaris: a single-arm clinical trial with transcriptomic analysis. Br J Dermatol 2022; 187: 650–658. https://doi.org/10.1111/bjd.21708
- Haynes D, Strunck JL, Topham CA, Ortega-Loayza AG, Kent G, Cassidy PB, et al. Evaluation of ixekizumab treatment for patients with pityriasis rubra pilaris: a single-arm trial. JAMA Dermatol 2020; 156: 668–675. https://doi.org/10.1001/jamadermatol.2020.0932
- Napolitano M, Abeni D, Didona B. Biologics for pityriasis rubra pilaris treatment: a review of the literature. J Am Acad Dermatol 2018; 79: 353–359. https://doi.org/10.1016/j.jaad.2018.03.036
- Li Y, Chen Z, He M, Tan X, Nie S, Zeng Y, et al. Successful treatment of erythrodermic pityriasis rubra pilaris with upadacitinib. J Eur Acad Dermatol Venereol 2024; 38: e593–e595. https://doi.org/10.1111/jdv.19762
- Xiaoyuan P, Lin S, Xinyu Z, Qianya S, Mei C, Fei W, et al. Successful treatment of generalized pustular psoriasis coexisting with pityriasis rubra pilaris with upadacitinib. Indian J Pediatr 2024; 91: 1296. https://doi.org/10.1007/s12098-024-05214-x
- Song EJ, Al-Saedy MA, Bouché N. Refractory pityriasis rubra pilaris treated with upadacitinib. JAAD Case Rep 2023; 35: 112–114. https://doi.org/10.1016/j.jdcr.2023.03.004
- Zhang X, Wei K, Song H, Chen X, Yang J, Zhao J, et al. Efficacy and safety of small molecule drugs in the treatment of pityriasis rubra pilaris—a systematic review. Front Med 2025; 12: 1544197. https://doi.org/10.3389/fmed.2025.1544197
- Strunck JL, Cutler B, Rajpal B, Kent G, Haynes D, Topham CA, et al. Pityriasis rubra pilaris response to IL-17A inhibition is associated with IL-17C and CCL20 protein levels. J Invest Dermatol 2022; 142: 235–239. https://doi.org/10.1016/j.jid.2021.06.009
- Virtanen A, Spinelli FR, Telliez JB, O’Shea JJ, Silvennoinen O, Gadina M. JAK inhibitor selectivity: new opportunities, better drugs? Nat Rev Rheumatol 2024; 20: 649–665. https://doi.org/10.1038/s41584-024-01153-1