RESEARCH LETTER
Solitary Oral Progression in a Well-controlled Sézary Syndrome
Gaspard FERRER1, Olivier DEREURE1,2 and Quentin SAMARAN1,2,3*
and Quentin SAMARAN1,2,3*
1Department of Dermatology, Montpellier University Hospital and Montpellier University, Montpellier, France, 2Pathogenesis and Control of Chronic and Emerging Infections (PCCEI), Inserm, University of Montpellier, Montpellier, France, and 3Epidemiology in Dermatology and Evaluation of Therapeutics (EpiDermE) EA7379, University Paris-Est Créteil Val de Marne, Inserm, Créteil, France. *Email: dr.quentin.samaran@gmail.com
Citation: Acta Derm Venereol 2026; 106: adv-2026-0449. DOI: https://doi.org/10.2340/actadv.v106.adv-2026-0449.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Feb 25, 2026. Accepted after revision: Mar 4, 2026.
Published: Mar 26, 2026.
Competing interests and funding: Data that support the findings of this study are available from the corresponding author upon reasonable request.
The patient described in this manuscript has provided written informed consent to publish his case details.
OD reports having served as a speaker and investigator for Kyowa Kirin and having received logistical support for attending scientific meetings from Kyowa Kirin, the company manufacturing and marketing mogamulizumab. The other authors have no conflicts of interest to declare.
To the Editor,
Sézary syndrome (SS) is a leukaemic and aggressive rare variant of primary cutaneous T-cell lymphomas (CTCLs) characterized by generalized erythroderma, lymphadenopathy, circulating malignant T-cells (1) and a poor prognosis, particularly in advanced stages (2). Mucosal involvement has been exceptionally reported in CTCL and usually occurs in advanced, leukaemic or transformed diseases (3). The oral cavity is the most frequently reported site, with gingival, lingual, buccal or perioral lesions (3, 4). Few cases of large cell transformation have also been described in the oral cavity, generally associated with poor prognosis (5). We hereby report an original case of SS well-controlled by mogamulizumab, with secondary solitary oral mucosal progression successfully treated with pegylated liposomal doxorubicin.
A 62-year-old male patient with no significant medical history was diagnosed with SS with predominant haematological involvement. Initial treatment with methotrexate was quickly discontinued due to cutaneous hyperesthesia and worsening skin involvement. Mogamulizumab combined with extracorporeal photochemotherapy (ECP) was subsequently implemented, leading to a complete haematological response and a near-complete cutaneous response after a few months, despite a transient and mild mogamulizumab-associated rash with no other significant adverse events. Twenty months after mogamulizumab initiation, painful proliferative and erosive lesions developed in the oral cavity, initially involving the right upper gingiva. Direct immunofluorescence, viral PCR testing and serological screening for antibodies of autoimmune bullous dermatoses were negative. The lesions rapidly evolved to a large intrabuccal tumoural mass invading the whole right side of the mouth, including upper gingiva, hard palate and inner cheek mucosa, associated with ipsilateral cheek oedema (Fig. 1A). While a sustained complete haematological response was confirmed, (18)F-FDG PET/CT revealed intense hypermetabolism of the oral lesion and newly appeared right cervical satellite lymphadenopathies (Fig. 1B). Histological examination of multiple oral biopsies identified a dense dermal infiltrate of medium-sized, highly atypical CD3+CD4+CD8- PD-1+lymphoid cells with epitheliotropism, with approximately 20% of CD30+cells; the already known tumoural T-cell clone was present, supporting the diagnosis of specific oral mucosal involvement of SS. Mogamulizumab was discontinued, and the patient received 8 cycles of pegylated liposomal doxorubicin (20 mg/m² every 3 weeks), achieving a complete clinical response (Fig. 2A) confirmed by PET/CT (Fig. 2B). Mogamulizumab and ECP were subsequently resumed as maintenance therapy.
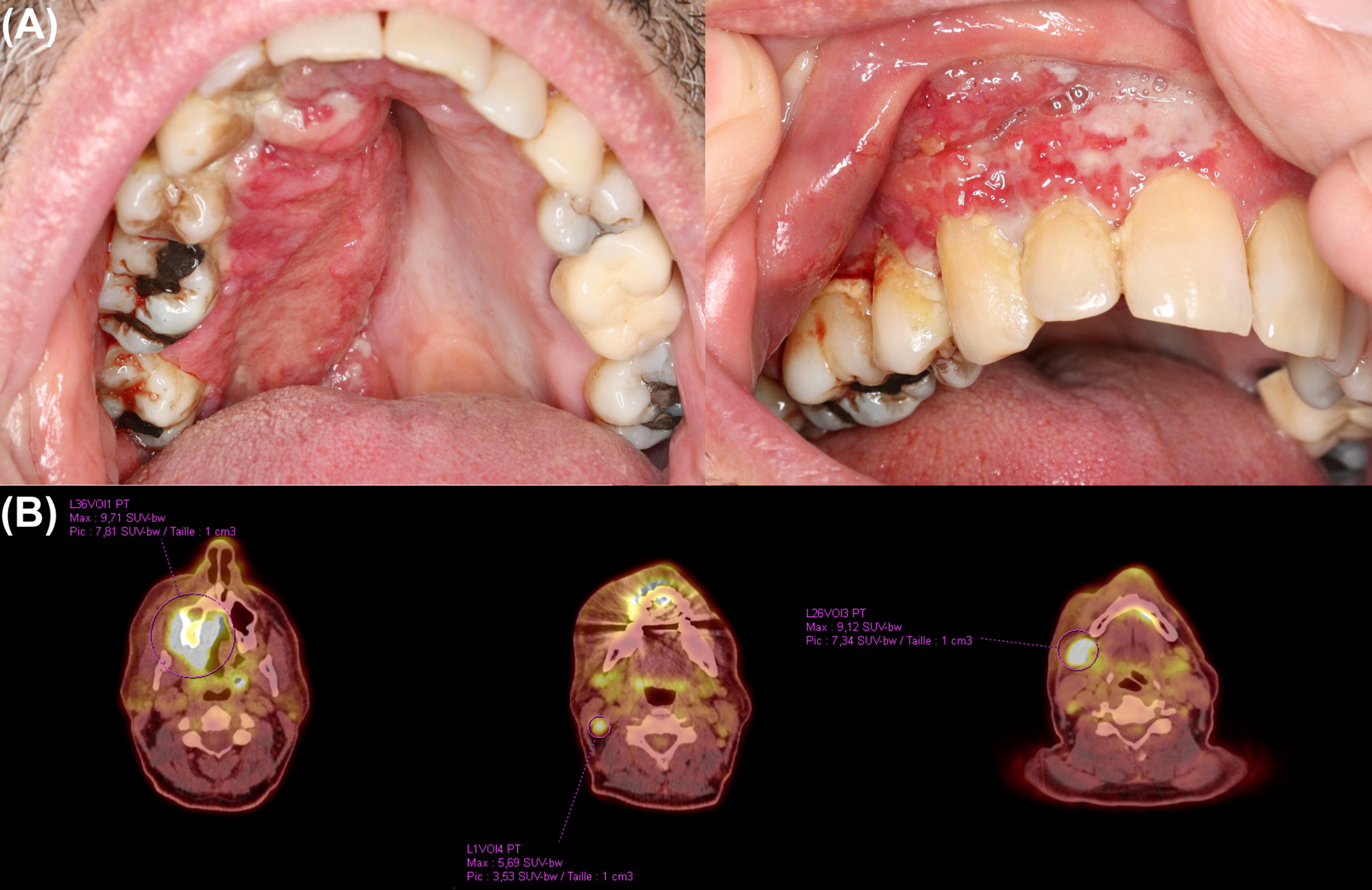
Fig. 1. (A) Unilateral tumoural oral mucosal involvement affecting the right upper gingiva, hard palate and inner cheek mucosa; (B) (18)F-FDG PET/CT showing intense hypermetabolic activity of the oral mucosal lesion with right cervical satellite lymphadenopathies.
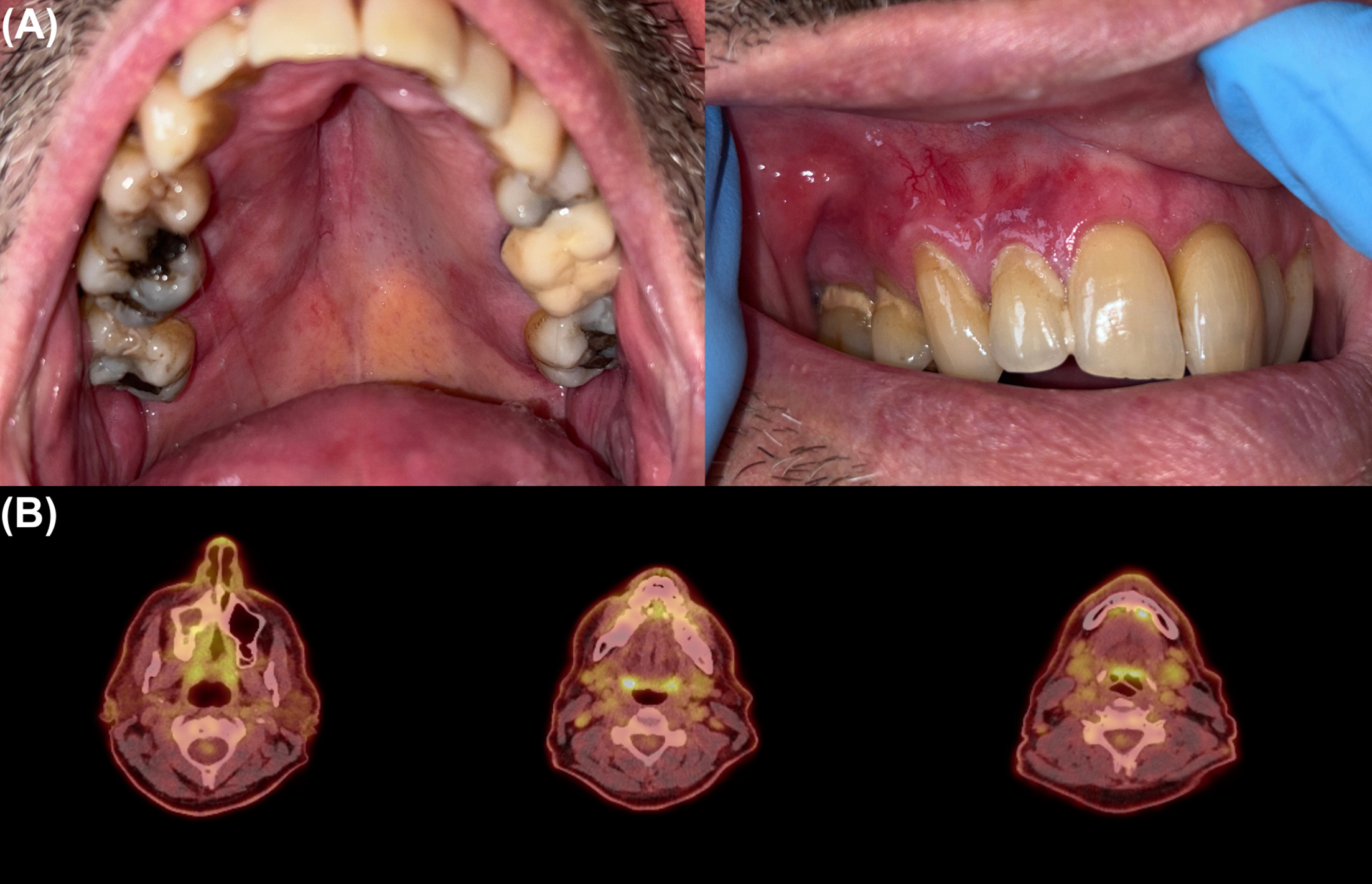
Fig. 2. (A) Complete clinical resolution of oral mucosal lesions after 8 cycles of pegylated liposomal doxorubicin; (B) (18)F-FDG PET/CT confirming complete metabolic response of the oral lesion and cervical lymph nodes
This case highlights the importance of systematic mucosal surveillance in addition to cutaneous monitoring and of histological assessment of any atypical lesion in this setting, even in the absence of haematological progression. Reported cases of CTCL with mucosal involvement remain rare and predominantly occur in mycosis fungoides patients, with clinical presentations ranging from erythematous plaques to ulcerative or tumoural lesions of the gingiva, tongue, palate or inner cheek mucosa (3, 4, 5). In most reports, mucosal involvement affects patients with advanced disease and is frequently associated with nodal dissemination or large cell transformation with poor overall outcome (3, 4, 5). Conversely, our case illustrates an unusual clinical pattern, characterized by a localized, strictly unilateral oral mucosal progression with no skin relapse, associated with regional lymphadenopathies of dubious nature despite sustained complete haematological response under mogamulizumab. This observation highlights a potential dissociation between blood and tissue compartments in SS, emerging data already observed with mogamulizumab in this setting.
Mogamulizumab, an anti-CCR4 monoclonal antibody, can induce prolonged and high-quality responses in SS even after treatment discontinuation, but secondary and sometimes heterogeneous resistance may occur after initial response, potentially related to loss of CCR4 expression (6, 7, 8). Recent data suggest that tumour heterogeneity reshaping under mogamulizumab may result in the selection of CCR4-negative resistant subclones within tissue compartments, potentially explaining localized disease progression despite ongoing systemic response (8).
REFERENCES
- Willemze R, Cerroni L, Kempf W, Berti E, Facchetti F, Swerdlow SH, et al. The 2018 update of the WHO-EORTC classification for primary cutaneous lymphomas. Blood 2019; 133: 1703–1714. https://doi.org/10.1182/blood-2018-11-881268
- Scarisbrick JJ, Prince HM, Vermeer MH, Quaglino P, Horwitz S, Porcu P, et al. Cutaneous Lymphoma International Consortium study of outcome in advanced stages of mycosis fungoides and Sézary syndrome: effect of specific prognostic markers on survival and development of a prognostic model. J Clin Oncol 2015; 33: 3766–3773. https://doi.org/10.1200/JCO.2015.61.7142
- Rosebush MS, Allen CM, Accurso BT, Baiocchi RA, Cordell KG. Oral mycosis fungoides: a report of three cases and review of the literature. Head Neck Pathol 2019; 13: 492–499. https://doi.org/10.1007/s12105-018-0923-5
- Rodrigues-Fernandes CI, Vargas PA, de Aquino IG, Lopes MA, Santos-Silva AR. Sézary syndrome: report of a rare case with perioral manifestation and review of the literature. Oral Oncol 2022; 134: 106134. https://doi.org/10.1016/j.oraloncology.2022.106134
- Sultan AS, Mostoufi B, Papadimitriou JC, Koka R, Basile J, Younis RH. Large cell transformation of oral mycosis fungoides. Head Neck Pathol 2018; 12: 247–251. https://doi.org/10.1007/s12105-017-0840-z
- Kim YH, Bagot M, Pinter-Brown L, Rook AH, Porcu P, Horwitz SM, et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): an international, open-label, randomised, controlled phase 3 trial. Lancet Oncol 2018; 19: 1192–1204. https://doi.org/10.1016/S1470-2045(18)30379-6
- Tzoumpa S, Ingen-Housz-Oro S, de Masson A, Pham-Ledard A, El Aarbaoui T, Dereure O, et al. Evolution of patients with Sézary syndrome after mogamulizumab discontinuation for any cause except progression: a multicentre retrospective study (Moga-stop Study). Br J Dermatol 2024; 191: 143–144. https://doi.org/10.1093/bjd/ljae153
- Roelens M, de Masson A, Andrillon A, Ram-Wolff C, Biard L, Boisson M, et al. Mogamulizumab induces long-term immune restoration and reshapes tumour heterogeneity in Sézary syndrome. Br J Dermatol 2022; 186: 1010–1025. https://doi.org/10.1111/bjd.21018