SHORT COMMUNICATION
A Case of Transient Nodules with Venous Calcification during Acute Exacerbation of Chronic Kidney Disease
Satoko YOKOYAMA1, Teruasa MURATA1* and Nobuo KANAZAWA1
1Department of Dermatology, Hyogo Medical University, 1-1 Mukogawa-cho, Nishinomiya, Hyogo 663-8501, Japan. *Email: te-murata@hyo-med.ac.jp
Citation: Acta Derm Venereol 2026; 106: adv-2026-0478. DOI: https://doi.org/10.2340/actadv.v106.adv-2026-0478.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Mar 9, 2026. Accepted after revision: Mar 12, 2026.
Published: Apr 1, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
This work was supported by JSPS KAKENHI grant number 25K11593.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Vascular calcification is common in patients with chronic kidney disease (CKD). We report a rare case of venous calcification in a patient with CKD, in which painful nodules appeared during renal crisis, followed by spontaneous resolution.
A 40-year-old Japanese man with a history of chronic kidney disease (CKD), hypertension, diabetes mellitus and hyperuricemia was admitted with fever. Acute renal failure was diagnosed, and haemodialysis was initiated. Two weeks later, painful nodules appeared around both ankles. As treatment with loxoprofen and topical ketoprofen was ineffective, the patient was referred to our department.
On examination, subcutaneous indurations were found on the medial and lateral aspects of both ankles (Fig. 1a–c). Histopathology showed a round focus of calcium deposition in the subcutaneous fat (Fig. 1d–f). This deposition was adjacent to an artery (Fig. 1g) and surrounded by foam cells and capillaries (Fig. 1e). Based on the round morphology of the calcium deposition and its localization adjacent to an artery, we suspected that the lesion involved a companion vein, showing occlusion, calcification and secondary organization. Intact parathyroid hormone was elevated (846 pg/mL; reference range [RR], 10–65 pg/mL), whereas calcium was within the normal range (9.2 mg/dL; RR, 8.5–10.5 mg/dL) and phosphorus was elevated (8.6 mg/dL; RR, 2.5–4.5 mg/dL), indicating hyperphosphataemia associated with secondary hyperparathyroidism. Eight days after the skin biopsy, all subcutaneous nodules showed a tendency to regress. Six months later, the nodules completely disappeared.
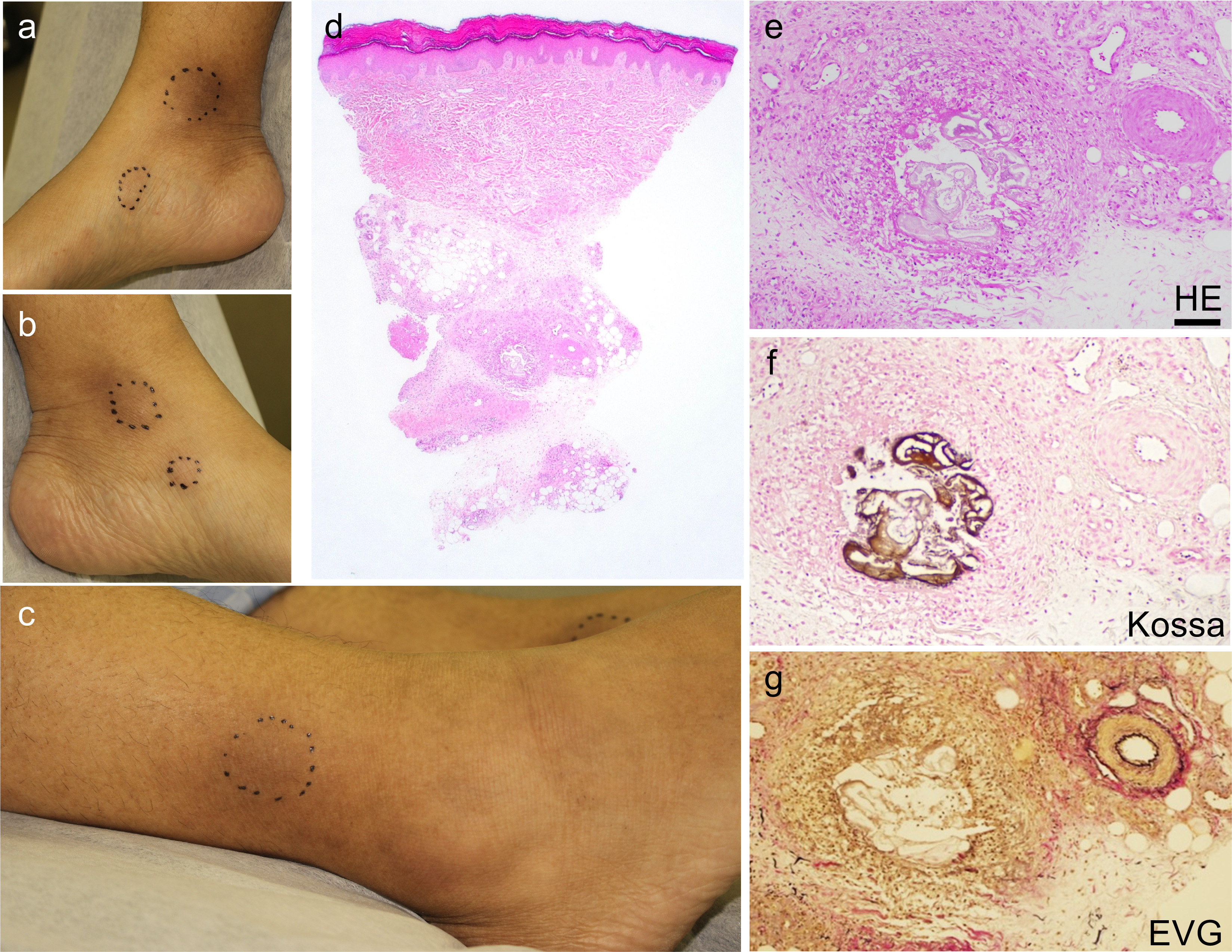
Fig. 1. Clinical and histopathological findings. (a–c) Subcutaneous indurations on the medial aspects of the right (a) and left (b) ankles and on the lateral aspect of the right ankle (c). (d) Histopathology showed inflammation and fibrosis with a round focus of calcium deposition in the subcutaneous fat. (e) The calcium deposition was surrounded by foam cells and capillaries. (f) Kossa staining. (g) Elastica-van Gieson staining. Scale bar: 100 µm.
Metastatic calcinosis cutis shows diffuse or perivascular calcification. Dystrophic calcinosis cutis is caused by trauma, tumours, collagen diseases, and other skin diseases. Idiopathic calcinosis cutis shows calcium deposition in the dermis surrounded by a fibrous capsule (1). Thus, none of these types of cutaneous calcinosis matched the characteristics of calcification in this case.
The clinical and histopathological features of this case also differed from the known entities of vascular calcification. Calciphylaxis, which primarily affects small arteries, is extremely resistant to treatment and often leads to death due to infection from skin ulcers (2). Atherosclerotic calcification occurs in arteries. Calcification of the cutaneous or subcutaneous vein walls was detected in multiple anatomical locations in 38% of patients with advanced CKD/end-stage kidney disease (3). However, in the present case, calcium deposition was much more extensive than that in previously reported venous wall calcification. It remained unclear whether the calcification involving the vascular wall occurred secondary to embolic occlusion, or whether primary, severe calcification of the vascular wall led to vascular occlusion. In addition, definitive identification of the structure as an occluded vein was not possible because of secondary changes.
Taken together, it is difficult to explain this case with known disease entities. We suspected that hyperparathyroidism secondary to CKD caused marked calcification in subcutaneous small veins resulting in tender nodules that regressed spontaneously. We tentatively refer to this type of lesion as transient nodules with venous calcinosis. Additional cases are needed to better understand this condition.
ACKNOWLEDGEMENTS
We thank Dr. Yoshihiro Ide for his histopathological assessment.
REFERENCES
- Elder DE. Lever’s Histopathology of the Skin. 12th ed. Philadelphia: Wolters Kluwer; 2022: 276–277 pp.
- Dobry AS, Ko LN, St John J, Sloan JM, Nigwekar S, Kroshinsky D. Association between hypercoagulable conditions and calciphylaxis in patients with renal disease: a case-control study. JAMA Dermatol 2018; 154: 182–187. https://doi.org/10.1001/jamadermatol.2017.4920
- Ruderman I, Hewitson TD, Smith ER, Holt SG, Wigg B, Toussaint ND. Vascular calcification in skin and subcutaneous tissue in patients with chronic and end-stage kidney disease. BMC Nephrol 2020; 21: 279. 10.1186/s12882-020-01928-0