RESEARCH LETTER
Severe Ulceronecrotic Evolution during Mogamulizumab Treatment in a Patient with Primary Cutaneous Peripheral T-cell Lymphoma
Quentin SAMARAN1,2,3* , Marilou DUBOËLLE4, Anaïs MARQUE1, Virginie BRES4
, Marilou DUBOËLLE4, Anaïs MARQUE1, Virginie BRES4 and Olivier DEREURE1,3
and Olivier DEREURE1,3
1Department of Dermatology, Montpellier University Hospital and Montpellier University, Montpellier, France, 2Epilogy (Epidemiology in Dermatology and Evaluation of therapeutics), IMRB, INSERM, Univ Paris-Est Créteil, Créteil, France, 3PCCEI, Univ Montpellier, INSERM, Univ Antilles, CHU Montpellier, Montpellier, France, and 4Pharmacovigilance Regional Centre, Department Of Medical Pharmacology And Toxicology, Montpellier University Hospital, Montpellier, France. *Email: dr.quentin.samaran@gmail.com
Citation: Acta Derm Venereol 2026; 106: adv-2025-0279. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0279.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Dec 18, 2025. Accepted after revision: Apr 8, 2026.
Published: Apr 20, 2026.
Competing interests and funding: The authors have no financial or material support to report linked to this manuscript.
Data that support the findings of this study are available from the corresponding author upon reasonable request.
The patient described in this manuscript has provided written informed consent to publish his case details. This manuscript does not report experiments involving human subjects. Therefore, IRB approval was not required for the described case.
Pr. DEREURE reports having served as a speaker and investigator for Kyowa Kirin Pharma and having received logistical support for attendance at scientific conferences from Kyowa Kirin Pharma. All remaining authors declare no conflicts of interest.
To the Editor,
Mogamulizumab, an anti-CCR4 monoclonal antibody, is currently registered in the treatment of primary cutaneous T-cell lymphomas (Mycosis Fungoides and Sézary Syndrome). Its approval was based on the MAVORIC phase III trial, showing significantly improved progression-free survival compared with vorinostat (1). Drug-induced eruptions are reported as “very common” in the Summary of Product Characteristics (SmPC) (2), but with no mention of ulceronecrotic evolution of skin lesions. We hereby report a case of severe ulceronecrotic evolution occurring during mogamulizumab in a patient with primary cutaneous peripheral T-cell lymphoma not otherwise specified (pcPTCL-NOS).
A 55-year-old man was followed for nodular skin lesions of clinical vascular appearance, predominantly affecting the lower limbs and evolving over several years, with no extracutaneous involvement. Pathological examination repeatedly showed an atypical CD3+CD4+CD7- CD30- lymphocytic infiltrate with a dominant cutaneous T-cell clone and a strong vascular component, consistent with a pcPTCL-NOS (3). HTLV-1 serology was negative, and repeated whole-body CT scans remained unremarkable. These lesions mostly responded to iterative electron-beam radiotherapy.
Owing to a final increasing number, size, and frequency of lesions, still without extracutaneous involvement, and an unchanged histological pattern with no large cell transformation, several systemic treatments were attempted, without success and/or with significant toxicity (methotrexate, liposomal pegylated doxorubicin, pegylated interferon and bendamustine). Because of the continuous and relentless clinical progression of the cutaneous disease (Fig. 1), mogamulizumab was introduced as fifth-line therapy on the basis of CCR4 expression by tumoral cells. Mogamulizumab was administered according to the approved regimen for Mycosis Fungoides and Sézary Syndrome: 1 mg/kg weekly for the first 4 weeks, followed by administration every 2 weeks.
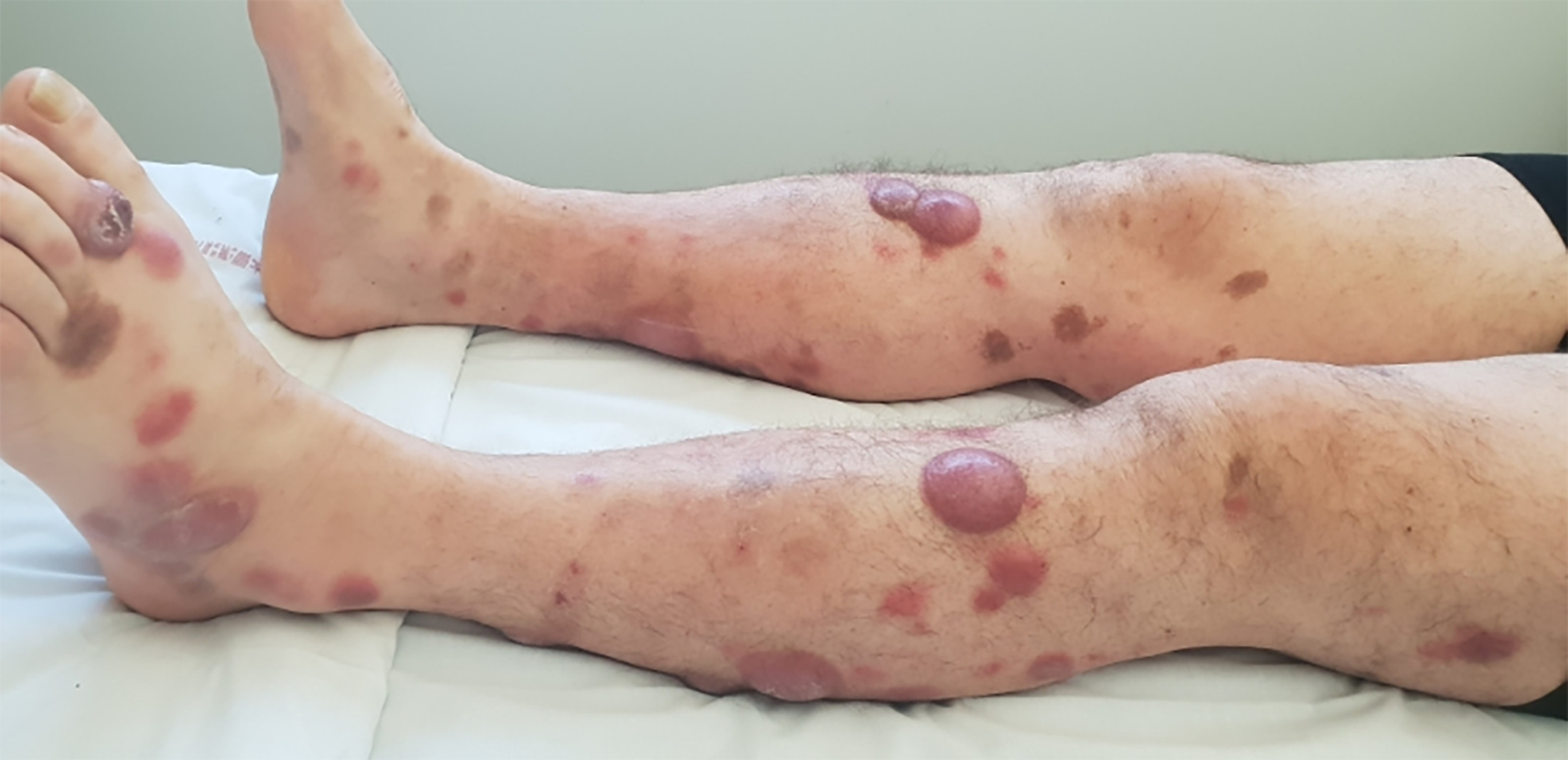
Fig. 1. Tumoural lesions of primary cutaneous peripheral T-cell lymphoma not otherwise specified at initiation of mogamulizumab.
From day 3 after the first mogamulizumab infusion onward, some nodules evolved toward progressive and painful ulceronecrotic lesions, while the remaining lesional areas either remained unchanged or kept enlarging. No systemic symptoms or deterioration in general condition were observed. No concomitant systemic treatments were administered during mogamulizumab therapy, apart from analgesics. This pattern progressively worsened over the following weeks (Fig. 2), leading to permanent discontinuation of mogamulizumab after 8 infusions (2 months). Two months later, the ulcerated lesions persisted, some of them requiring skin grafting. BV-CHP (brentuximab vedotin, cyclophosphamide, doxorubicin and prednisone) was then initiated, resulting in progressive re-epithelialization and flattening of most tumours.
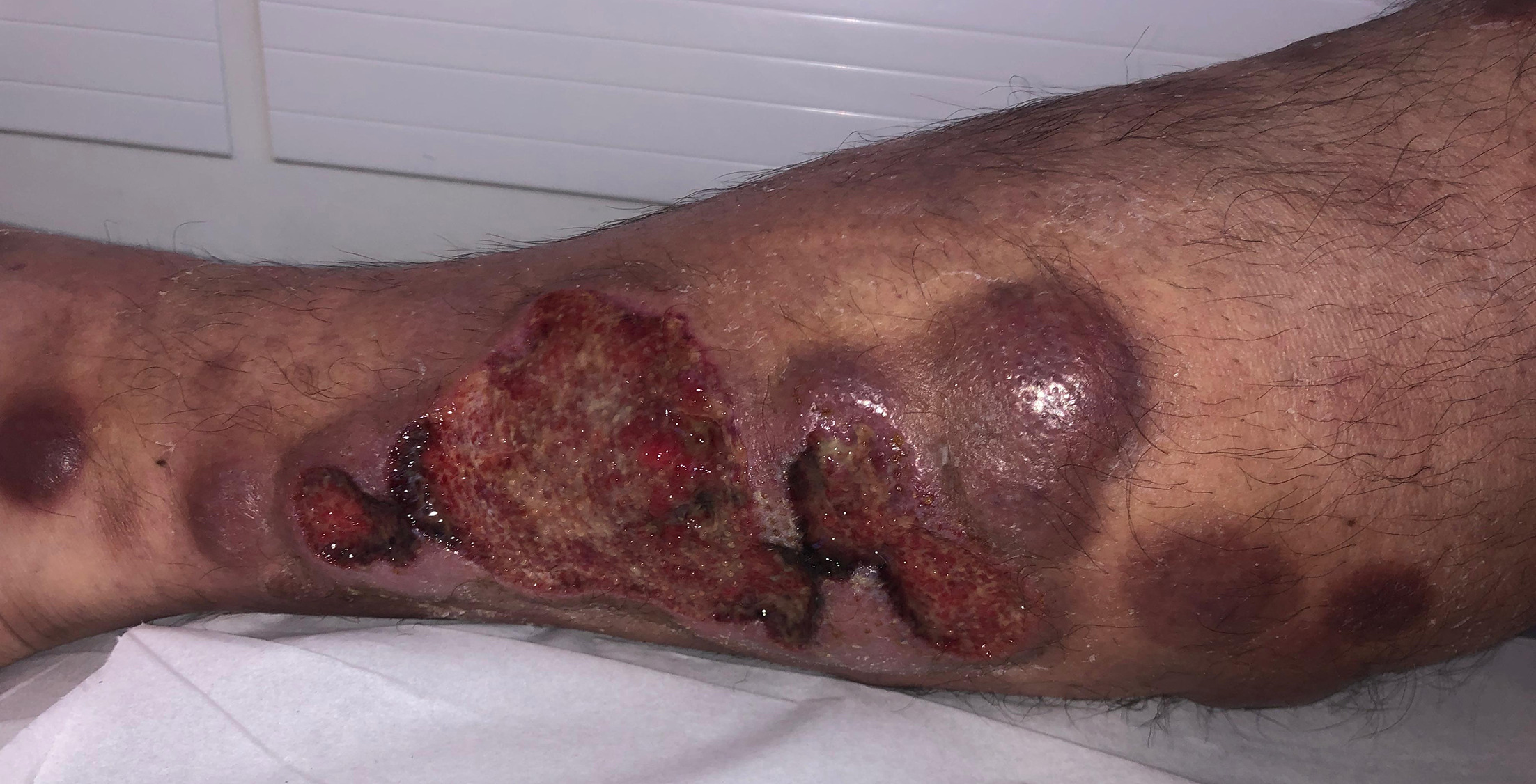
Fig. 2. Ulceronecrotic evolution of part of the tumoral lesions shown here 6 weeks after the last mogamulizumab infusion.
Mogamulizumab-associated rashes (MAR) are frequent and feature variable clinical and histological presentations, but are typically benign and non-necrotic (4, 5). Rare cases of Stevens-Johnson syndrome and toxic epidermal necrolysis have been reported both in the mogamulizumab SmPC (2) and in the published literature (4). However, to our knowledge, ulceronecrotic evolution of lymphomatous skin lesions has not previously been reported with this drug. Only one case of ecthyma gangrenosum has been described in the literature, with a favourable outcome under antibiotic therapy and without treatment discontinuation (6). A query in VigiBase, the WHO global database of reported drug adverse events, using the MedDRA High-Level Term (HLT) “skin and subcutaneous tissue ulcerations” and the Preferred Term (PT) “cutaneous necrosis”, identified nine additional cases, including 2 cases of ecthymas, 2 skin erosions, 2 cutaneous necrosis and one perineal ulceration.
Regarding the present case, the intrinsic causality score was assessed as 2/6 according to the French pharmacovigilance method (7). Although spontaneous evolution cannot be excluded, mogamulizumab was considered a possible contributor to the ulceronecrotic lesions, supported by temporal association. Mogamulizumab may elicit autoantibodies against keratinocytes and melanocytes, leading to complement-dependent cytotoxicity in the skin through regulatory T-cell depletion and loss of peripheral immune checkpoints (8). Such immune dysregulation might similarly have contributed to the ulceronecrotic evolution of skin tumours. Additional hypotheses include a particularly aggressive MAR or a direct cytotoxic effect due to high local CCR4 expression in selected lesions.
This case illustrates the possibility of a rare but severe cutaneous adverse event of mogamulizumab in CCR4-expressing, non-epidermotropic cutaneous lymphomas, and highlights the importance of early and protracted close clinical monitoring in this setting.
ACKNOWLEDGEMENTS
As with any pharmacovigilance database, establishing a causal relationship between a reported adverse event and a suspected drug may be challenging due to the inherent limitations of the available data. The information, results, and conclusions derived from VigiBase do not represent the opinions of the Uppsala Monitoring Centre, the WHO Collaborating Centre for International Drug Monitoring, or the World Health Organization.
REFERENCES
- Kim YH, Bagot M, Pinter-Brown L, Rook AH, Porcu P, Horwitz SM, et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): an international, open-label, randomised, controlled phase 3 trial. Lancet Oncol 2018; 19: 1192–1204. https://doi.org/10.1016/S1470-2045(18)30379-6
- Poteligeo. European Medicines Agency (EMA). 2018. [cited 2025 November 21]. Available from: https://www.ema.europa.eu/en/medicines/human/EPAR/poteligeo
- Kempf W, Mitteldorf C, Cerroni L, Willemze R, Berti E, Guenova E, et al. Classifications of cutaneous lymphomas and lymphoproliferative disorders: An update from the EORTC cutaneous lymphoma histopathology group. J Eur Acad Dermatol Venereol 2024; 38: 1491–1503. https://doi.org/10.1111/jdv.19987
- Avallone G, Roccuzzo G, Pileri A, Agostinelli C, Maronese CA, Aquino C, et al. Clinicopathological definition, management and prognostic value of mogamulizumab-associated rash and other cutaneous events: a systematic review. J Eur Acad Dermatol Venereol 2024; 38: 1738–1748. https://doi.org/10.1111/jdv.19801
- Hansen I, Abeck F, Menz A, Schneider SW, Booken N. Mogamulizumab-associated rash - case series and review of the literature. J Dtsch Dermatol Ges 2024; 22: 1079–1086. https://doi.org/10.1111/ddg.15432
- Barré M, Valois A, Okhremchuk I, Sair M, Masbou J, Abed S, et al. Ecthyma gangrenosum complicating mogamulizumab treatment of Sézary syndrome. Ann Dermatol Venereol 2021; 148: 63–65. https://doi.org/10.1016/j.annder.2020.09.576
- Bégaud B, Evreux JC, Jouglard J, Lagier G. Imputation of the unexpected or toxic effects of drugs. Actualization of the method used in France. Therapie 1985; 40: 111–118.
- Suzuki Y, Saito M, Ishii T, Urakawa I, Matsumoto A, Masaki A, et al. Mogamulizumab treatment elicits autoantibodies attacking the skin in patients with adult T-cell leukemia-lymphoma. Clin Cancer Res 2019; 25: 4388–4399. https://doi.org/10.1158/1078-0432.CCR-18-2575