ORIGINAL REPORT
Sex Differences in Loss in Life Expectancy in Primary Cutaneous Melanoma, a Swedish Cohort Study
Anna OKSANEN1, Elisavet SYRIOPOULOU1, Kari NIELSEN2,3, Hanna ERIKSSON4,5*† and Therese M-L ANDERSSON1†
and Therese M-L ANDERSSON1†
1Department of Medical Epidemiology and Biostatistics, Karolinska Institutet, Stockholm, Sweden, 2Department of Dermatology, Skåne University Hospital, Lund, Sweden, 3Department of Clinical Sciences, Dermatology, Lund University Skin Cancer Research Group, and Lund Melanoma Study Group, Lund University, Lund, Sweden, 4Department of Oncology and Pathology, Karolinska Institutet, Stockholm, Sweden, and 5Theme Cancer, Unit of Head-Neck-, Lung- and Skin Cancer, Skin Cancer Center, Karolinska University Hospital, Stockholm, Sweden
†Shared last authors.
Corr: Hanna Eriksson, Department of Oncology and Pathology, Karolinska Institutet, Stockholm, Sweden. *Email: hanna.eriksson.4@ki.se
Keywords Cutaneous malignant melanoma; Life Expectancy; Mortality.
Citation: Acta Derm Venereol 2026; 106: adv-2025-0227. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0227.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
Submitted: Nov 25, 2025. Accepted after revision: Mar 17, 2026.
Published: Apr 20, 2026.
Competing interests and funding: This study was supported by the Swedish Research Council (HE, TA), the Swedish Cancer Society (HE, TA), Region Stockholm (HE), the Radiumhemmet Research funds (HE), the Swedish Medical Society (HE), Karolinska Institutet fund (HE)s, Castenbäck funds (HE), and the Cancer-Allergy Fund (HE) Swedish Research Council for Health Working Life and Welfare (FORTE) (ES)
The data used for this study is protected by GDPR to preserve individual patient integrity. Access can only be provided within the context of the obtained ethical approval and approval from the data owner.
The study was register-based and no patient consent was required. An ethical permit was given by Regional Ethical Review Board Lund (Regionala Etikprövningsnämnden Lund) (decision number 2015-630) with ammendment approved by The Swedish Ethical Review Authority (Etikprövningsmyndigheten) (decision number 2021-03275).
The authors have no conflicts to declare except for HE; lecturer for BMS, Novartis and Janssen, honouraria from Janssen, and research funding from SkyLine Dx.
Sex differences in cutaneous melanoma (CM) survival are pronounced. It is unclear how these differences impact the loss in life expectancy (LLE). A population-based cohort from the Swedish Melanoma registry was used to analyze sex differences in life expectancy (LE) and LLE following a CM diagnosis. The cohort included 12,893 men and 12,572 women aged 40 years or above diagnosed with a first invasive CM in Sweden between 2000 and 2014. The average remaining LE at CM diagnosis was 15.45 years (95% CI 15.21, 15.7) for men and 20.14 years (95% CI 19.89, 20.43) for women, and the LLE was 2.95 years (95% CI 2.70, 3.19) and 2.4 years (95% CI 2.11, 2.67), respectively. If men had the same relative survival as women, the average LE for men would instead be 17.33 years (95% CI 17.03, 17.65) and the LLE 1.07 years (95% CI 1.07, 1.38). Men lose more years due to CM than women. Baseline age, stage or location are not sufficient to explain the sex differences.
SIGNIFICANCE
We calculated how many years women and men loose after being diagnosed with cutaneous melanoma, when compared to life expectancies in the general population. It was observed that men lose on average more years than women following a melanoma diagnosis, despite having a shorter life expectancy in the general population. This means that the survival advantage women have in melanoma is greater than the difference in general population life expectancies. The differences were not explained by disease severity, age at diagnosis or location of the melanoma.
The sex differences in cutaneous melanoma (CM) survival are well-described with women having prolonged CM survival across different stages (1, 2, 3). These survival discrepancies are still not fully understood (3, 4). Different behavioural and biological rationales have been suggested (3), and in some previous reports, behavioural patterns have been considered insufficient to explain the observed differences, suggesting that biological factors are at least part of the explanation (1, 3). Known prognostic factors for primary CM besides sex include age, tumour thickness, ulceration status and stage at diagnosis but also for example socioeconomic status (5). The site of the primary CM is not only a prognostic factor but also strongly sex specific (6, 7).
Life expectancy (LE) varies between men and women also in the general population. A large majority of the primary CMs are diagnosed at stages I and II, in which a high proportion of individuals are cured (8). Therefore, it is of interest to complement studies concentrating on relative survival or melanoma-specific survival (MSS) with a measure that takes background life expectancy differences into account. Loss in life expectancy (LLE) quantifies the impact of CM diagnosis with respect to background LE. It can be interpreted as mean years lost following a CM diagnosis.
In this study, we estimated the differences in the remaining LE and LLE for men and women following a diagnosis of a first CM overall, as well as within different stages at diagnosis and location of primary tumour. The outcome measures expand current understanding of CM by capturing the impact of CM diagnosis in relation to the remaining life expectancy.
MATERIALS AND METHODS
The initial cohort consisted of all individuals diagnosed with first invasive CM between the years 2000 and 2014 in Sweden (35,847 individuals) and was extracted from the Swedish Melanoma Registry (SweMR)(9), which has a coverage of close to 100% compared to the National Cancer Register. The SweMR was linked to the Swedish Cause of Death Register for information on time of death. The time period (2000–2014) was chosen to provide sufficient follow-up for the estimation of LE, including enough information on the relationship of general population mortality and cancer mortality to reliably extrapolate life-course in the cancer cohort (10). Patients diagnosed before the age of 40 were excluded (3,828 individuals) to increase estimation robustness; the estimation of LE is difficult for younger individuals (10). This cut-off was drawn low to keep as large a population as possible. Other exclusion criteria were missing information on stage (1,980 individuals) or tumour site (123 individuals). The final cohort included 29,916 patients diagnosed with first primary CM (Fig. 1). The outcome was defined as time from diagnosis until death by any cause. If death was not observed, the individual was administratively censored on 31st of December 2020.
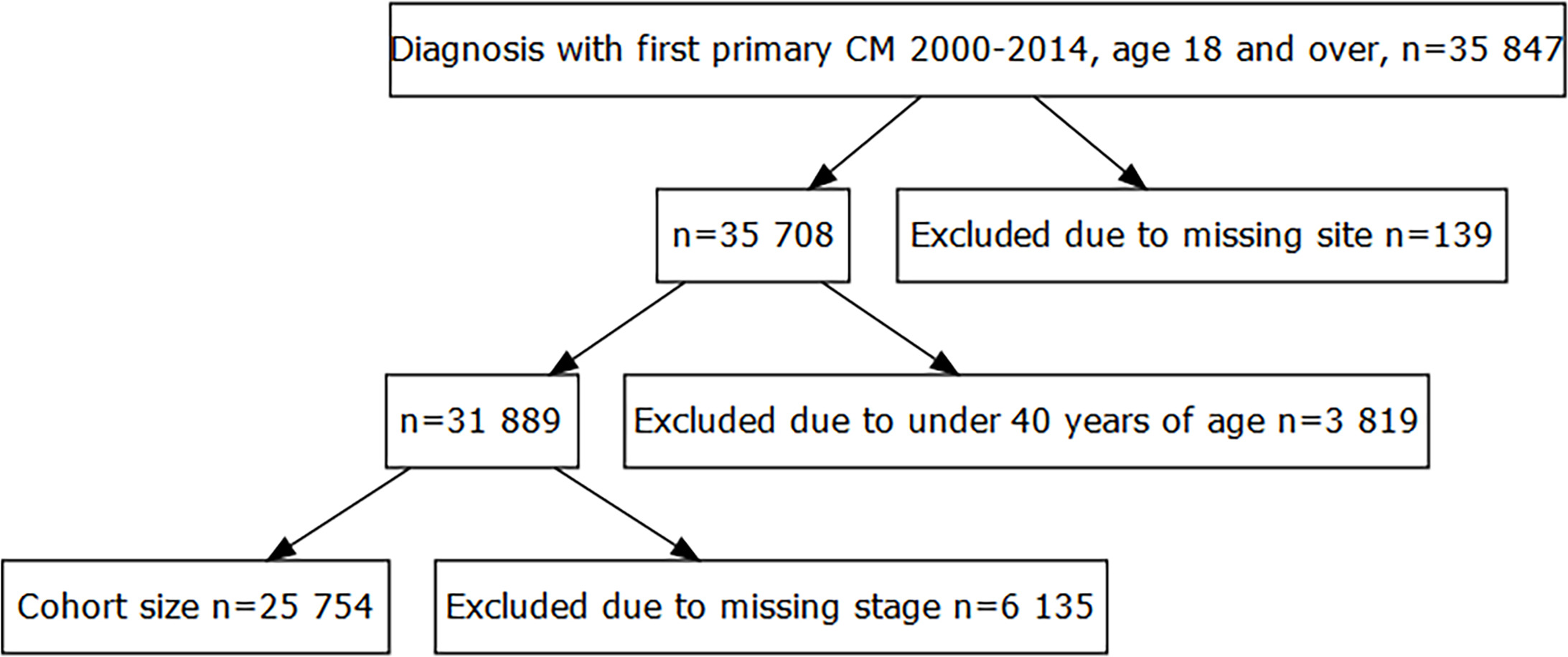
Fig. 1. Flowchart of the cohort extraction. CM: cutaneous melanoma.
The stage at primary diagnosis was categorized according to the 8th American Joint Committee on Cancer staging for CM (AJCC) where stages I and II include nonmetastatic cancer, stage III indicates lymph node or satellite metastasis and stage IV distant metastasis (11). Location of the primary tumour was categorized into three groups: extremities (upper and lower extremities and acral CMs), head/neck and trunk. Acral CMs were few (413 individuals) and were expected to have little impact on results.
Our estimands of interest were LE and LLE. The LLE was defined as the difference in LE that would have been observed in the absence of CM and the LE following CM. LLE is calculated by comparing the observed mortality in the cancer cohort to the expected mortality of individuals with the same birth year, sex and age. Population mortality rates were used as a proxy for the mortality rates in the absence of melanoma. Differences in sex, age or year of diagnosis directly impacted the LE and LLE and are therefore used in matching. The stratified population mortality rates for the Swedish population were extracted from the Human Mortality Database.
Statistical analysis
Baseline statistics were presented as counts (percentages), mean (standard deviation) and median (25th and 75th percentiles). Differences of sex versus stage and location at diagnosis were tested with a χ2 test. A flexible parametric relative survival model (FPRM) was used to estimate the survival after CM diagnosis (10). The model included sex, stage at diagnosis, location of CM and a natural spline for age at diagnosis. Interactions of sex with age at diagnosis, stage and location were included in the model. Modelling was done on the log cumulative excess hazard scale, using a natural spline. Time-varying effects were allowed for sex, stage, location and age at diagnosis. The final model was chosen based on both theoretical and clinical considerations.
The outcomes are the mean predicted LE and LLE, calculated by averaging over all individual predictions within the relevant subcohort. These results are referred to as the marginal estimates. The age, stage and location differences were expected to drive some of the marginal results. To enable fair comparisons, we also standardized the age and stage and/or location distributions. Standardization was done by replacing the relative survival estimates of one group, with those of another group. First, standardization was done using women’s relative survival, but baseline age, stage and location distributions, and expected survival of men. These results are reported as the “men under women’s relative survival”. They can be compared to the marginal estimates of men to see how much they would change if men had the relative survival of women’s. Similarly, the “men under women’s covariate structure” refers to estimates using the women’s baseline covariate distribution but relative and expected survival of men. Effectively, it is LE and LLE of men in a population weighted to resemble that of the women.
Estimation was done in Stata 18 (StataCorp. 2023. Stata Statistical Software: Release 18. College Station, TX: StataCorp LLC.), using the stpm3 -command to fit the FPRM and standsurv for the standardized survival probabilities. As the study was register-based, no informed consent was required. The study was approved by the Lund regional ethical committee. The study was descriptive, and no pre-specified hypothesis testing was done.
RESULTS
Women were diagnosed younger than men, with median age of 64 (p25 : 53, p75 : 76) and 67 (p25 : 58, p75 : 76) years for women and men respectively (Table I). The most common CM location for women was extremities (56%), and for men trunk (56%). Head and neck cancers were approximately equally common in men and women (14% and 13%, respectively). Sex differences in the stage distribution were statistically significant (Table I). Location and stage were not statistically independent in neither men nor women, indicating that they are related (Table SI).
Table I. Clinico-pathological characteristics for patients 40 years and older, diagnosed with a first primary invasive cutaneous melanoma in Sweden in 2000–2014
| Men N=15,418 |
Women N=14,498 |
Total N=29,916 |
p-value | |
|---|---|---|---|---|
| Clinico-pathological characteristics | ||||
| Stage | ||||
| I | 10,076 (65.4%) | 10,302 (71.1%) | 20,378 (68.1%) | <0.001 |
| II | 4,972 (32.2%) | 3,938 (27.2%) | 8,910 (29.8%) | |
| III | 215 (1.4%) | 153 (1.1%) | 368 (1.2%) | |
| IV | 155 (1.0%) | 105 (0.7%) | 260 (0.9%) | |
| Location | ||||
| Head/Neck | 2,192 (14.2%) | 1,818 (12.5%) | 4,010 (13.4%) | <0.001 |
| Extremities | 4,661 (30.2%) | 8,335 (57.5%) | 12,996 (43.4%) | |
| Trunk | 8,565 (55.6%) | 4,345 (30.0%) | 12,910 (43.2%) | |
| Year of diagnosis | ||||
| 2000–2004 | 3,599 (23.3%) | 3,402 (23.5%) | 7,001 (23.4%) | 0.13 |
| 2005–2009 | 4,837 (31.4%) | 4,687 (32.3%) | 9,524 (31.8%) | |
| 2010–2014 | 6,982 (45.3%) | 6,409 (44.2%) | 13,391 (44.8%) | |
| Age (mean) | 66.6 (12.67) | 64.77 (14.19) | 65.71 (13.47) | <0.001 |
| Age (median) | 67 (58–76) | 64 (53–76) | 66 (55–76) | <0.001 |
| Data characteristics | ||||
| Deaths, number of events | 6,643 (43.1%) | 4,654 (32.1%) | 11,297 (37.8%) | - |
| Follow-up time | ||||
| Cumulated person years | 118543.4 | 122830.2 | 241373.6 | - |
| Mean years | 7.69 (3.81) | 8.47 (3.55) | 8.07 (3.71) | - |
Sex differences in LE and LLE
Women had almost 5 years longer marginal LE at the time of diagnosis, compared to men (women: 21.45 years; 95% CI 21.13, 21.77, men: 16.57 years; 95% CI 16.28, 16.88, Table II). Women’s average LLE (1.94 years; 95% CI 1.61, 2.26, Table III) was approximately half a year shorter compared to men, despite their longer expected LE (men LLE: 2.38 years; 95% CI 2.07, 2.67, Table III). The LLE did not change for men when standardized using the baseline covariate distributions of women, even though the LE increased by 1.93 years. The standardized estimates showed that LE would have been about 0.6 years longer for men if they had the same relative survival as women (Table II, Fig. 2).
Table II. Life expectancy (LE) following a cutaneous melanoma diagnosis in Sweden among individuals diagnosed at age 40 and above between 2000 and 2014, by sex, stage at diagnosis and location
| LE | Women | Men | Men under women’s relative survival | Men under women’s covariate structure |
|---|---|---|---|---|
| All | 20.14 (19.86, 20.42) | 15.45 (15.21, 15.7) | 17.33 (17.02, 17.65) | 16.05 (15.85, 16.26) |
| Location head/neck | ||||
| I | 16.85 (16.58,17.13) | 15.18 (14.93,15.44) | 15.37 (15.16,15.58) | 14.67 (14.42,14.94) |
| II | 7.95 (7.44,8.51) | 7.82 (7.32,8.34) | 8.74 (8.18,9.33) | 6.32 (5.94,6.72) |
| III | 7.19 (5.63,9.19) | 7.05 (5.87,8.47) | 6.63 (5.28,8.33) | 7.19 (6,8.6) |
| IV | 4.59 (2.91,7.25) | 3.27 (2.42,4.41) | 2.42 (1.58,3.71) | 4.88 (3.44,6.92) |
| Location extremity | ||||
| I | 23.78 (23.49,24.08) | 19.34 (18.99,19.69) | 19.82 (19.62,20.03) | 20.8 (20.4,21.22) |
| II | 12.7 (12.25,13.16) | 10.14 (9.64,10.66) | 12.38 (11.97,12.79) | 9.29 (8.83,9.78) |
| III | 11.73 (10,13.76) | 8.25 (7.07,9.63) | 9.23 (7.97,10.68) | 9.29 (7.85,10.99) |
| IV | 5.58 (4.19,7.43) | 4.74 (3.65,6.16) | 5.18 (3.94,6.8) | 4.58 (3.51,5.98) |
| Location trunk | ||||
| I | 25.39 (24.91,25.89) | 19.24 (18.89,19.59) | 19.44 (19.16,19.73) | 22.59 (22.11,23.08) |
| II | 13.3 (12.53,14.1) | 9.54 (9.16,9.94) | 10.4 (9.9,10.93) | 10.87 (10.34,11.44) |
| III | 9.16 (7.25,11.58) | 9.39 (7.94,11.11) | 8.37 (6.72,10.44) | 9.34 (7.86,11.1) |
| IV | 4.03 (2.66,6.11) | 4.84 (3.66,6.39) | 3.49 (2.35,5.18) | 5 (3.75,6.66) |
|
Men under women’s relative survival used women’s relative survival and men’s expected survival in the male cohort. Men under women’s covariate structure used the relative and expected survival of men, but weighting the cohort age and location and/or stage distribution to be like that of the women. All results present the average of the group specific predictions. |
||||
Table III. Loss in life expectancy (LLE) after a cutaneous melanoma diagnosis in Sweden among individuals diagnosed at age 40 and above in 2000–2014, by sex, stage at diagnosis and location
| LLE | Women | Men | Men under women’s relative survival | Men under women’s covariate structure |
|---|---|---|---|---|
| All | 2.4. (2.11, 2.67) | 2.95 (2.70, 3.19) | 1.07 (1.07, 1.38) | 2.35 (2.14, 2.55) |
| Location head/neck | ||||
| I | 0.63 (.41,.84) | 0.81 (.55,1.07) | 0.77 (.5,1.04) | 0.81 (.55,1.07) |
| II | 4.27 (3.68,4.83) | 4.13 (3.72,4.5) | 4.14 (3.59,4.66) | 5.19 (4.66,5.68) |
| III | 8.99 (7.29,10.35) | 8.46 (7.04,9.64) | 10.54 (8.54,12.11) | 8.57 (7.15,9.75) |
| IV | 10.12 (8.83,10.96) | 13.3 (11.27,14.74) | 15.81 (13.15,17.49) | 9.27 (8.13,10.12) |
| Location extremity | ||||
| I | 0.61 (0.41,.81) | 1.3 (0.88,1.7) | 0.92 (0.62,1.22) | 1.1 (.75,1.44) |
| II | 4.03 (3.61,4.44) | 5.74 (5.25,6.2) | 4.42 (3.95,4.87) | 6.27 (5.75,6.77) |
| III | 7.65 (6.2,8.91) | 10.09 (8.38,11.52) | 10.05 (8.01,11.78) | 8.63 (7.26,9.81) |
| IV | 10.01 (8.39,11.25) | 10.18 (8.78,11.25) | 11.24 (9.39,12.63) | 10.45 (9.02,11.54) |
| Location trunk | ||||
| I | 0.95 (0.65,1.23) | 1.56 (1.06,2.04) | 1.47 (.97,1.95) | 1.15 (0.8,1.49) |
| II | 5.45 (4.93,5.95) | 7.68 (7.11,8.21) | 7.6 (6.79,8.36) | 6.31 (5.91,6.7) |
| III | 11.96 (9.9,13.62) | 11.06 (9.3,12.55) | 13.7 (11.28,15.61) | 10.94 (9.22,12.4) |
| IV | 13.57 (11.89,14.71) | 12.73 (11.07,13.98) | 16.02 (13.94,17.39) | 12.23 (10.68,13.4) |
|
Men under women’s relative survival used women’s relative survival and men’s expected survival in the male cohort. Men under women’s covariate structure used the relative and expected survival of men, but weighting the cohort age and location and/or stage distribution to be like that of the women. All results present the average of the group specific predictions. |
||||
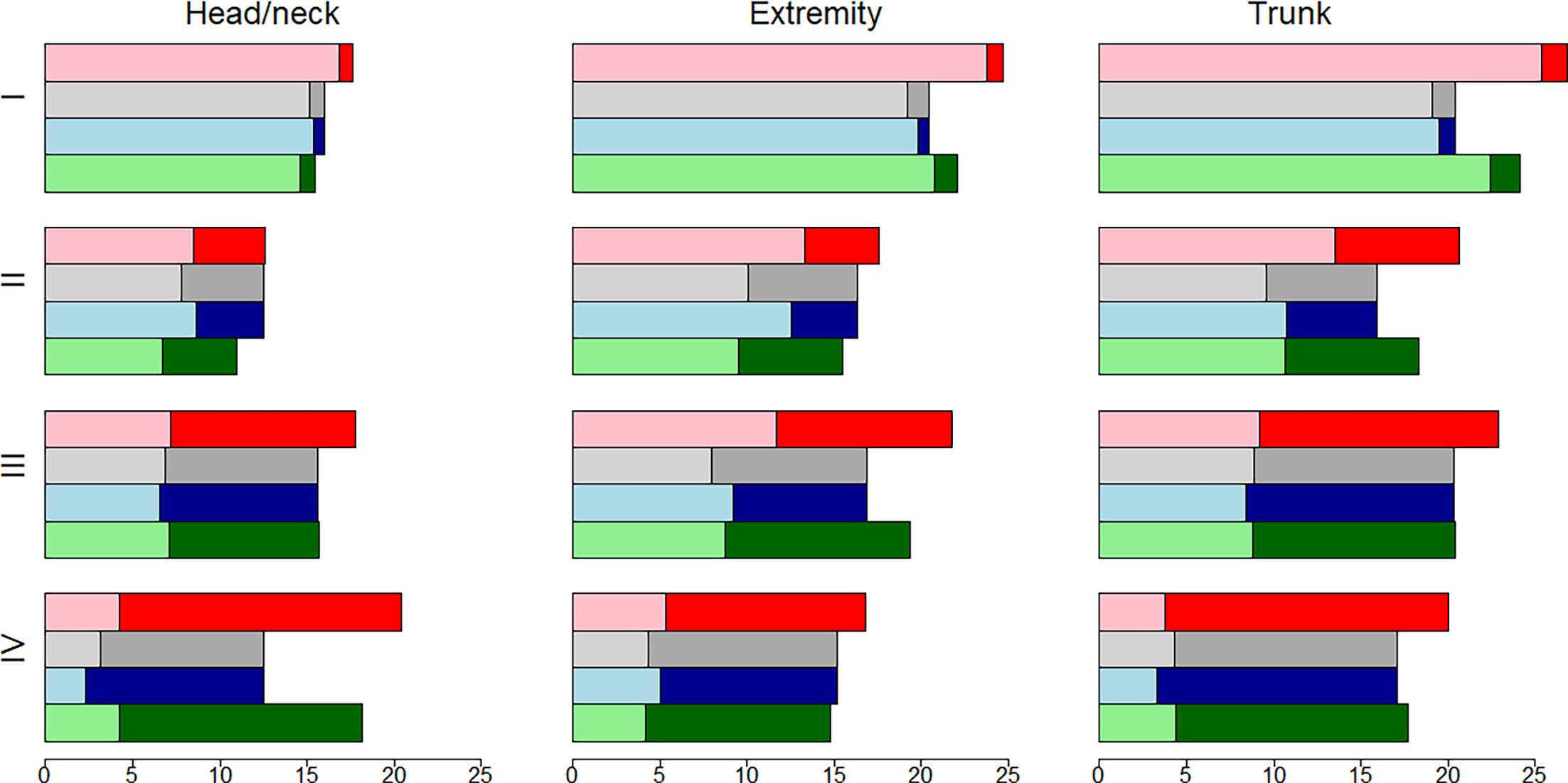
Fig. 2. Life expectancy (LE) and loss in life expectancy (LLE) after a cutaneous melanoma diagnosis in Sweden among individuals diagnosed at age 40 and above in 2000–2014, by sex, stage at diagnosis and location. Red is women’s marginal estimates, gray is men’s marginal estimates, blue is men under women’s relative survival, green is men with women’s covariate structure. The lighter (transparent) colors show the LE and the LLE is shown as darker colors.
Women had longer LE compared to men within all cancer stages and locations, except for stage III and IV CM of the trunk (Table II, Fig. 2). However, women’s LLE was greater than men’s in a third of the subgroups (Table III, Fig. 2). Patterns in LE of CM patients did not translate into patterns in LLE, as LLE also depends on the expected survival in absence of CM. LE decreased and LLE increased with increasing stage within all locations. For both men and women, head and neck CM had the shortest LE and greatest LLE within all stages.
LE and LLE by sex and tumour site
Within head/neck CM, women had a longer marginal LE than men within all stages. The LLE of men was slightly greater for stage I and IV, but for stages II and III, men lost less years, even if the differences were minor. Standardization for age shortened men’s LE in stages I and II but increased it for stages III and IV. These estimates are very close to the women’s estimates, indicating that within this group, men’s and women’s LE are approximately the same if age differences are taken into account. The differences between the men’s marginal and age standardized LLE estimates were under or around 1 year, except for stage IV, where the LLE difference is 4 years (Table III). When applying women’s relative survival to men, the LE increased at stages I and II (difference: 0.19, 0.92 years) but shortened for stages III and IV (difference 0.42 and 0.85 years).
As with head/neck, marginal LE for CM of the extremity was longer for women in all stages. The marginal LLE in CM of extremity was lower for women at stages I and IV, but higher in stages II and III (Table III), compared to men. In stages I and III, baseline age seems to explain some of the marginal difference between men and women (LE differences: 1.46 and 1.04 years, respectively, Table II) but not so for stage II and IV CM (difference: −0.85 and −0.17 years, respectively, Table II). The corresponding changes in LLE were small, except stage III where men would lose 1.46 years more if they had the baseline covariate structure of women (Table III).
The largest marginal LE differences between the sexes for stages I and II are observed at the CM of the trunk: 6.15 and 3.76 years respectively (Table II). Weighting men to align with the age structure of women increased LE by 3.35 and 1.33 years in stages I and II but had very small impact for stages III and IV (−0.05 and 0.16 years, Table II). Men would have reduced LE for stages III and IV if they had women’s relative survival but not so for stages I and II (differences: 0.20, 0.86,–1.02 and −1.35 years, stages I to IV respectively, Table II), leading to a larger LLE for stage III and IV if men would have the relative survival of women.
DISCUSSION
In this large, population-based study with high-coverage CM data from the SweMR, we found that despite the generally longer LE of women, LLE difference between men and women varied between subgroups. Standardization for age differences partly explained the differences in LE and LLE in some subgroups, for example stage I and II LE head/neck CM, yet was found to accentuate them in others, like LE of stage III and IV head/neck. In stages I and II, worse relative survival of men contributed to shorter LE compared to women.
This study demonstrated that women had longer average LE at diagnosis, but, despite the longer general population LE giving them more years to lose, they on average lost fewer years to CM than men. These findings are in line with previous studies on primary CM LLE by Vikström et al. (12) and Syriopoulou et al. (13), which both found that women lost fewer years (12, 13). According to Syriopoulou et al. (13), CM was the only cancer among those studied where women lose less years than men. Moreover, Thiam et al. (14) provided a comparison of the LLEs between not only the sexes, but also countries, using always the relative survival estimates from the same (US) population. Their overall results were in line with ours, but they reported larger differences, potentially driven by the use of US relative survival and secondary data.
Although direct comparisons of different outcome measures are difficult, women have been observed to have a survival advantage at different cohorts and different geographical locations (1, 2, 4, 15, 16). Joosse et al. found women to have a better survival with a consistent hazard ratio of 0.7 (1, 2). Similar results have been found by others over the years (4, 17). Our overall results were driven by stage I and II CM, which constitute the majority of the primary CMs at diagnoses in Sweden (97.5%, Table I). Like in our data, Vikström et al. (12) observed a switch from lower LLE for women in stage II CM to higher LLE for women in stage III CM. This finding was also supported by Thiam et al. (14), who found women to lose more years when combining stage IIIB/C and IV CM diagnosis (14) and Syriopoulou, who observed women to lose less years when all CM diagnoses are investigated (13). On the other hand, Vikström et al. also observed that women consistently lost a smaller proportion of their remaining life (12). This underlines the complexity of measuring survival differences; women can simultaneously have longer LE yet lose more years as their expected LE is longer but also lose less of their remaining expected life percentagewise.
The marginal LE and LLE estimates did not take the age, location and stage differences into consideration. Several studies have found that men are initially diagnosed older and with more severe disease (15, 18, 19, 20, 21, 22), which we also observed (Table I). In addition to the marginal estimates, we presented standardized estimates where covariate structures and then relative survival estimates were made to be comparable between the sexes. Equalizing relative survival increased men’s LE in some stage and location groups. Similarly, aligning the age distributions brought the LE of men closer to that of women for most groups, but not all.
The inability of age, location and stage differences to thoroughly explain the observed sex differences in CM LE and LLE indicated that other factors, like biological differences, are probably involved (1, 2, 3). It is also conceivable that the shorter LLE for women within stage I was due to a larger portion of the women having lost no life years to CM. CM incidence has increased over the decades without corresponding changes in mortality (23, 24), and several authors have raised the possibility of over-diagnosis (25, 26), potentially more common in women (27).
The major strengths of this study were the population-based nationwide comprehensive CM data which, together with advanced methodology, enable modelling of the sex differences in a flexible and realistic way. On the other hand, potential limitations were the uncertainty inherent to extrapolation, particularly for the younger patients whose survival has to be extrapolated for a longer period. Also, the cohort was limited to Swedish patients and generalizability to other populations is uncertain. Another limitation was that, to guarantee sufficient follow-up, we were unable to use information on more recently diagnosed patients who have received new treatments not yet in use on a national level in Sweden.
This study showed that on average, men have a shorter life expectancy after a CM diagnosis and lose more years due to a CM diagnosis than women. Marginally, women had longer LE in all subgroups except stage IV CM of the trunk, and, after considering age, also in stage IV CM of the head/neck. Despite the clear pattern in LE differences, LLE difference between men and women varied in length and direction between the subgroups.
REFERENCES
- Joosse A, Collette S, Suciu S, Nijsten T, Lejeune F, Kleeberg UR, et al. Superior outcome of women with stage I/II cutaneous melanoma: pooled analysis of four European Organisation for Research and Treatment of Cancer phase III trials. J Clin Oncol 2012; 30: 2240–2247. https://doi.org/10.1200/JCO.2011.38.0584
- Joosse A, Collette S, Suciu S, Nijsten T, Patel PM, Keilholz U, et al. Sex is an independent prognostic indicator for survival and relapse/progression-free survival in metastasized stage III to IV melanoma: a pooled analysis of five European organisation for research and treatment of cancer randomized controlled trials. J Clin Oncol 2013; 31: 2337–2346. https://doi.org/10.1200/JCO.2012.44.5031
- Schwartz MR, Luo L, Berwick M. Sex differences in melanoma. Curr Epidemiol Rep 2019; 6: 112–118. https://doi.org/10.1007/s40471-019-00192-7
- White LP. Studies on melanoma. II. Sex and survival in human melanoma. N Engl J Med 1959; 260: 789–797. https://doi.org/10.1056/NEJM195904162601601
- Eriksson H, Lyth J, Månsson-Brahme E, Frohm-Nilsson M, Ingvar C, Lindholm C, et al. Low level of education is associated with later stage at diagnosis and reduced survival in cutaneous malignant melanoma: a nationwide population-based study in Sweden. Eur J Cancer 2013; 49: 2705–2716. https://doi.org/10.1016/j.ejca.2013.03.013
- Crombie IK. Distribution of malignant melanoma on the body surface. Br J Cancer 1981; 43: 842–849. https://doi.org/10.1038/bjc.1981.123
- El Sharouni MA, Witkamp AJ, Sigurdsson V, van Diest PJ, Louwman MWJ, Kukutsch NA. Sex matters: men with melanoma have a worse prognosis than women. J Eur Acad Dermatol Venereol 2019; 33: 2062–2067. https://doi.org/10.1111/jdv.15760
- Andersson TML, Eriksson H, Hansson J, Månsson-Brahme E, Dickman PW, Eloranta S, et al. Estimating the cure proportion of malignant melanoma, an alternative approach to assess long term survival: a population-based study. Cancer Epidemiol 2014; 38: 93–99. https://doi.org/10.1016/j.canep.2013.12.006
- Swedish melanoma registry (SweMR–Svenska Melanomregistret). 2025 Feb 13. Available from: Https://Cancercentrum.Se/Samverkan/Cancerdiagnoser/Hud-Och-Melanom/Malignt-Melanom/Kvalitetsregister/
- Andersson TML, Dickman PW, Eloranta S, Lambe M, Lambert PC. Estimating the loss in expectation of life due to cancer using flexible parametric survival models. Stat Med 2013; 32: 5286–5300. https://doi.org/10.1002/sim.5943
- Gershenwald JE, Scolyer RA, Hess KR, Sondak VK, Long GV, Ross MI, et al. Melanoma staging: evidence-based changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J Clin 2017; 67: 472–492. https://doi.org/10.3322/caac.21409
- Vikström S, Syriopoulou E, Andersson TML, Eriksson H. Loss in life expectancy in patients with stage II-III cutaneous melanoma in Sweden: a population-based cohort study. J Am Acad Dermatol 2024; 90: 963–969. https://doi.org/10.1016/j.jaad.2023.12.053
- Syriopoulou E, Bower H, Andersson TML, Lambert PC, Rutherford MJ. Estimating the impact of a cancer diagnosis on life expectancy by socio-economic group for a range of cancer types in England. Br J Cancer 2017; 117: 1419–1426. https://doi.org/10.1038/bjc.2017.300
- Thiam A, Zhao Z, Quinn C, Barber B. Years of life lost due to metastatic melanoma in 12 countries. J Med Econ 2016; 19: 259–264. https://doi.org/10.3111/13696998.2015.1115764
- Jung KW, Park S, Shin A, Oh CM, Kong HJ, Jun JK, et al. Do female cancer patients display better survival rates compared with males? Analysis of the Korean National Registry data, 2005-2009. PLoS One 2012; 7: e52457. https://doi.org/10.1371/journal.pone.0052457
- Magnus K. Prognosis in malignant melanoma of the skin. Significance of stage of disease, anatomical site, sex, age and period of diagnosis. Cancer 1977; 40: 389–397. https://doi.org/10.1002/1097-0142(197707)40:1<389::aid-cncr2820400155>3.0.co;2-i
- Rampen FHJ. Sex differences in survival from cutaneous melanoma. Int J Dermatology 1984; 23: 444–452. https://doi.org/10.1111/ijd.1984.23.7.444
- Malec E, Eklund G. The changing incidence of malignant melanoma of the skin in Sweden, 1959-1968. Scand J Plast Reconstr Surg 1978; 12: 19–27. https://doi.org/10.3109/02844317809010476
- Olsen CM, Thompson JF, Pandeya N, Whiteman DC. Evaluation of sex-specific incidence of melanoma. JAMA Dermatol 2020; 156: 553–560. https://doi.org/10.1001/jamadermatol.2020.0470
- Shaw HM, McGovern VJ, Milton GW, Farago GA, McCarthy WH. Histologic features of tumors and the female superiority in survival from malignant melanoma. Cancer 1980; 45: 1604–1608. https://doi.org/10.1002/1097-0142(19800401)45:7<1604::aid-cncr2820450715>3.0.co;2-o
- Smith AJ, Lambert PC, Rutherford MJ. Understanding the impact of sex and stage differences on melanoma cancer patient survival: a SEER-based study. Br J Cancer 2021; 124: 671–677. https://doi.org/10.1038/s41416-020-01144-5
- Yuan TA, Lu Y, Edwards K, Jakowatz J, Meyskens FL, Liu-Smith F. Race-, age-, and anatomic site-specific gender differences in cutaneous melanoma suggest differential mechanisms of early- and late-onset melanoma. Int J Environ Res Public Health 2019; 16: 908. https://doi.org/10.3390/ijerph16060908
- Cosman B, Heddle SB, Crikelair GV. The increasing incidence of melanoma. Plast Reconstr Surg 1976; 57: 50–56. https://doi.org/10.1097/00006534-197601000-00010
- Erdmann F, Lortet-Tieulent J, Schüz J, Zeeb H, Greinert R, Breitbart EW, et al. International trends in the incidence of malignant melanoma 1953-2008--are recent generations at higher or lower risk? Int J Cancer 2013; 132: 385–400. https://doi.org/10.1002/ijc.27616
- Welch HG, Mazer BL, Adamson AS. The rapid rise in cutaneous melanoma diagnoses. N Engl J Med 2021; 384: 72–79. https://doi.org/10.1056/NEJMsb2019760
- Rubin R. Melanoma diagnoses rise while mortality stays fairly flat, raising concerns about overdiagnosis. JAMA 2020; 323: 1429. https://doi.org/10.1001/jama.2020.2669
- Kurtansky NR, Dusza SW, Halpern AC, Hartman RI, Geller AC, Marghoob AA, et al. An epidemiologic analysis of melanoma overdiagnosis in the United States, 1975-2017. J Invest Dermatol 2022; 142: 1804–1811. https://doi.org/10.1016/j.jid.2021.12.003