SHORT COMMUNICATION
Successful Treatment of Crohn Disease and Concomitant Alopecia Totalis with Upadacitinib: Case Report and Systematic Review of the Literature
Yuki M. F. ANDERSEN1* , Farzad SEYED-ALINAGHI1, Malene F. KRISTIANSEN2 and Simon F. THOMSEN1
, Farzad SEYED-ALINAGHI1, Malene F. KRISTIANSEN2 and Simon F. THOMSEN1
1Department of Dermatology, Bispebjerg Hospital, University of Copenhagen, Copenhagen, Denmark, and 2Department of Gastroenterology, Bispebjerg Hospital, University of Copenhagen, Copenhagen, Denmark. *Email: yuki.egeberg@gmail.com
Citation: Acta Derm Venereol 2026; 106: adv-2025-0156. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0156.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Nov 4, 2025. Accepted after revision: Mar 30, 2026.
Published: Apr 20, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
Crohn disease (CD) is a chronic inflammatory bowel disease (IBD) characterized by immune-mediated inflammation of the gastrointestinal tract. Disease pathophysiology is complex; however, abnormal activation of immune pathways, particularly involving tumour necrosis factor-alpha (TNF-α), interleukin (IL)-12, IL-23/IL-17 and interferon-gamma (IFN-γ) plays a crucial role in sustaining intestinal inflammation.(1, 2).
Alopecia areata (AA) is an autoimmune condition characterized by nonscarring hair loss, ranging from small patches on the scalp to complete loss of scalp and body hair. The disease is mediated by autoreactive cytotoxic CD8+T cells targeting hair follicles, driven by inflammatory cytokines such as IFN-γ and IL-15, which disrupt the immune privilege at the follicular level, leading to hair loss.(3).
Janus kinase (JAK) inhibitors have emerged as effective treatments by blocking intracellular signalling of multiple inflammatory cytokines implicated in inflammatory conditions. The JAK-STAT pathway mediates critical cytokine signalling in both CD and AA, making JAK inhibitors a promising therapeutic option (4, 5). Here, we describe the successful use of upadacitinib in a patient with concomitant CD and AA and perform a systematic review of the literature.
CASE
An 18-year-old male was treated at the Department of Gastroenterology and Department of Dermatology for CD and AA, respectively. The patient had a previous history of atopic dermatitis (AD), allergic rhinoconjunctivitis and seborrheic dermatitis. Family history included alopecia totalis (AT) and allergic rhinoconjunctivitis (father) and myxoedema (mother).
At age 13, he developed his first symptoms of AA with small patches on the scalp and lower legs that followed a spontaneous regrowth and relapse pattern without any treatment. From age 15, he experienced abdominal discomfort, diarrhoea, bloating and perianal fissures. Initial evaluation suggested lactose intolerance, but symptoms persisted. At age 17, further evaluation revealed elevated faecal calprotectin (358, then 466 μg/g), terminal ileitis on colonoscopy and gastroduodenitis on gastroscopy. Biopsies confirmed the diagnosis of CD. Initial treatment with oral budesonide failed to control inflammation, with calprotectin rising to 1990 μg/g and the patient developed mucus in stool. During this period, his hair loss worsened, progressing to AT, significantly impacting his quality of life. Although adalimumab, an anti-TNF treatment, is first choice for CD in Denmark, given the dual diagnosis and patient’s hair loss concerns, a multidisciplinary team initiated upadacitinib 45 mg/day to target CD, AA and AD.
Baseline Severity of Alopecia Tool (SALT) score was 100, corresponding to 100% scalp hair loss (Fig. 1A). The patient tolerated upadacitinib well, with mild to moderate facial acne as the only side effect. At 3 months, gastrointestinal symptoms improved markedly, with calprotectin decreasing to 17 μg/g and showed signs of hair regrowth with a SALT score improving to 75 (Fig. 1B). Topical minoxidil was added twice daily, and the upadacitinib dose was reduced to a maintenance dose of 15 mg/day. At 6 months the dose was increased to 30 mg/day due to insufficient hair growth. At 10-month follow-up, the patient achieved complete hair regrowth (SALT 0), with sustained remission of gastrointestinal symptoms (Fig. 1C). The patient’s symptoms of AD similarly improved during the course of treatment with upadacitinib.
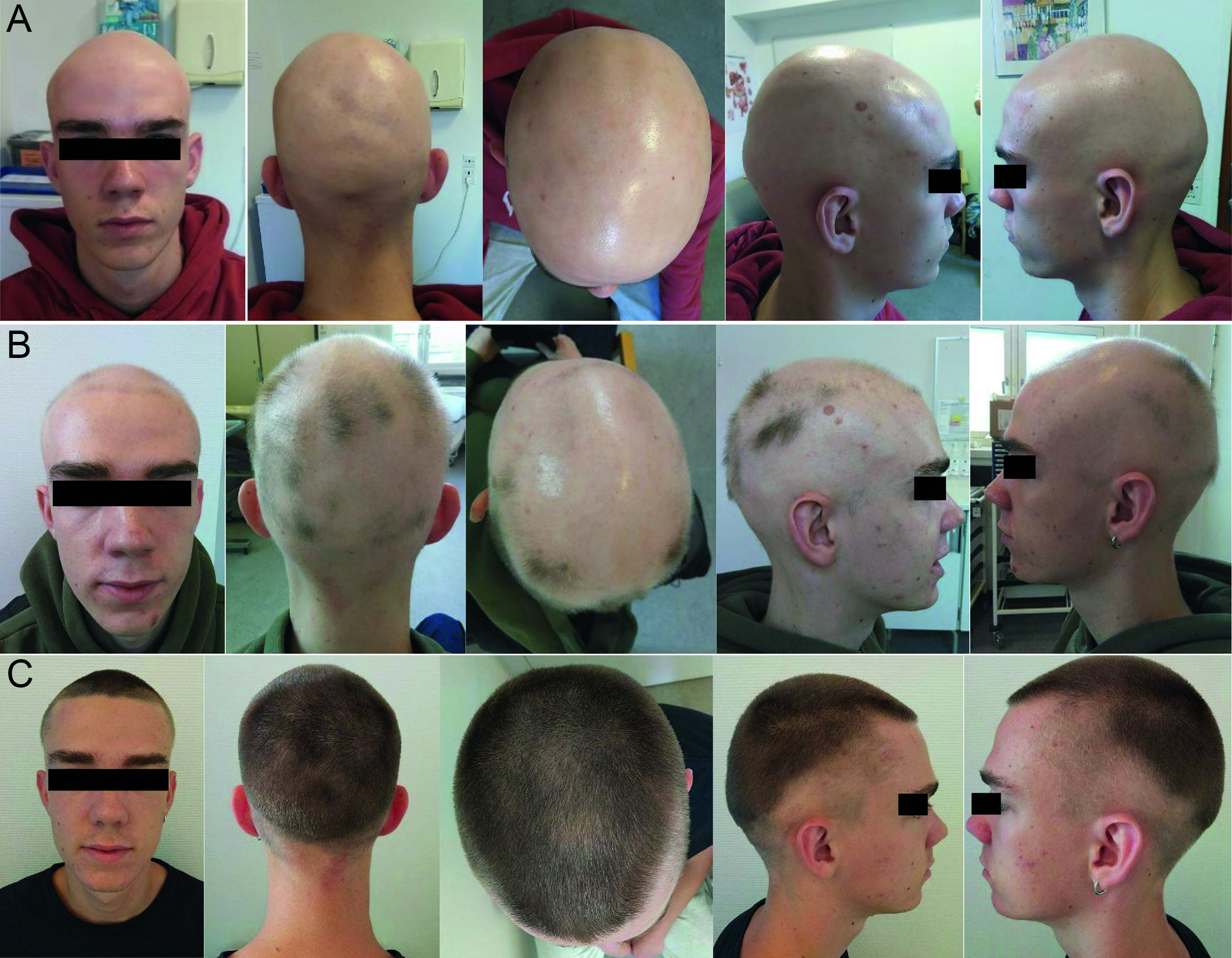
Fig. 1. Clinical photos (A) at baseline (SALT100), (B) after 3-month treatment with upadacitinib (SALT 75), and (C) after 10-month treatment with upadacitinib (SALT 0).
LITERATURE REVIEW
We systematically searched (search string: alopecia areata, Crohn, upadacitinib) for previously reported cases of upadacitinib treatment for concomitant CD and AA in the literature and found the following published cases.
A 20-year-old male with CD developed alopecia universalis (AU) after one year of treatment with adalimumab which only provided partial CD symptom control. At clinical examination, he was further diagnosed with AD. Treatment with upadacitinib (45 mg/day induction for 8 weeks followed by 30 mg/day maintenance) and oral minoxidil (10 mg/ day) led to complete hair regrowth within 11 weeks and histologic remission of CD was confirmed via colonoscopy (6).
A 23-year-old male with CD developed AA while on adalimumab. He was switched to ustekinumab for CD and was treated with prednisolone (3 week tapering course starting with 40 mg/day) and methotrexate (20 /week) for 6 weeks with limited signs of hair growth. He was then treated with upadacitinib (30 mg/day) while ustekinumab was maintained and achieved near-complete hair regrowth after 7 months as well as clinical remission of CD (7).
A 23-year-old male with a history of CD and AU was treated with upadacitinib given the concurrent diseases. The treatment was initiated at 45 mg/day for 12 weeks, followed by a maintenance dose of 30 mg/day. Hair regrowth was observed after a month and kept improving over the following months. His CD symptoms and follow-up colonoscopy findings also improved (8).
A 28-year-old male with severe AD and AA (SALT 100) refractory to treatment with systemic cyclosporine, azathioprine and dupilumab, as well as severe CD refractory to infliximab, adalimumab and ustekinumab. The patient received treatment with upadacitinib 30 mg/day and achieved a SALT 0 and almost clearance of AD after one month. His CD also improved with a normalized ileal mucosa by colonoscopy after 9 months of treatment (9).
DISCUSSION
Clustering of autoimmune conditions is a well-described phenomenon, and in a systematic review and meta-analysis, AA was found to be associated with IBD, although the literature is still limited (10). In patients with inflammatory comorbidities, a holistic and multidisciplinary approach can lead to effective treatments that can target more than one condition, which is highly beneficial for the patient. Furthermore, certain pitfalls may be avoided when selecting therapy to avoid side effects in patients with a specific risk profile. For example, while anti-TNF agents are commonly used for CD, they may paradoxically induce or exacerbate AA (11). A thorough medical history is therefore warranted in this patient group.
Upadacitinib, a second-generation JAK inhibitor targeting JAK1, is currently FDA-approved for treatment of AD, IBD, psoriatic arthritis, rheumatoid arthritis and ankylosing spondylitis (12). Other indications such as AA, vitiligo and hidradenitis suppurativa have also been investigated (13, 14, 15). Our systematic literature review identified four previously published cases of concomitant CD and AA successfully treated with upadacitinib, supporting the efficacy observed in our case. Importantly, due to publication bias, negative results and cases with lack of response are less likely to be published. Additional investigation and clinical trials are therefore warranted to further elucidate the indication.
In the presented case, upadacitinib’s dual efficacy allowed for resolution of intestinal inflammation and hair regrowth, improving the patient’s quality of life significantly. This adds to the growing evidence that JAK inhibitors are promising options for patients with concomitant CD and AA. The case also illustrates the importance of a multidisciplinary approach in clinical decision-making when treating comorbid patients with inflammatory diseases.
REFERENCES
- Neurath MF. Cytokines in inflammatory bowel disease. Nat Rev Immunol 2014; 14: 329–342. https://doi.org/10.1038/nri3661
- Dolinger M, Torres J, Vermeire S. Crohn’s disease. Lancet 2024; 403: 1177–1191. https://doi.org/10.1016/S0140-6736(23)02586-2
- Strazzulla LC, Wang EHC, Avila L, Lo Sicco K, Brinster N, Christiano AM, et al. Alopecia areata: Disease characteristics, clinical evaluation, and new perspectives on pathogenesis. J Am Acad Dermatol 2018; 78: 1–12. https://doi.org/10.1016/j.jaad.2017.04.1141
- O’Shea JJ, Kontzias A, Yamaoka K, Tanaka Y, Laurence A. Janus kinase Inhibitors in autoimmune diseases. Ann Rheum Dis 2013; 72. https://doi.org/10.1136/annrheumdis-2012-202576
- Sandborn WJ, Feagan BG, Loftus EV Jr, Peyrin-Biroulet L, Van Assche G, D’Haens G, et al. Efficacy and safety of upadacitinib in a randomized trial of patients with crohn’s disease. Gastroenterology 2020; 158: 2123–2138. https://doi.org/10.1053/j.gastro.2020.01.047
- Levine J, Sharma D, Morsia S, Ungar B. Management of comorbid Crohn’s disease and alopecia universalis with upadacitinib and oral minoxidil. J Dermatolog Treat 2025; 36: 2528704. https://doi.org/10.1080/09546634.2025.2528704
- Johnston LA, Lu C, Poelman SM. Successful treatment of concomitant alopecia universalis and Crohn’s disease with upadacitinib: A case report. SAGE Open Med Case Rep 2023; 11: 2050313X231160914. https://doi.org/10.1177/2050313X231160914
- Robinson BL, Yoo BS, Lodhia NI. S3886 concurrent management of Crohn’s Ileitis and Alopecia Universalis With Upadacitinib. Am J Gastroenterol 2024; 119: S2536–S2536. https://doi.org/10.14309/01.ajg.0001044912.80416.c8
- Novielli D, Foti C, Principi M, Mortato E, Romita P, Dell’Aquila P, et al. Upadacitinib in concurrent Crohn’s disease, atopic dermatitis and alopecia areata: A case report. Acad Dermatol Venereol 2024; 38. https://doi.org/10.1111/jdv.19377
- Maghfour J, Olson J, Conic RRZ, Mesinkovska NA. The Association between Alopecia and Inflammatory bowel disease: a systematic review and meta-analysis. Dermatology (Basel) 2021; 237: 658–672. https://doi.org/10.1159/000512747
- Perez SM, Kabashima K, Reich K, Gilliet M, Gilhar A, Paus R. Lessons from TNF-α Inhibitor-Induced alopecia areata: Does TNF-α operate as a hair follicle immune privilege guardian under inflammatory conditions? J Invest Dermatol 2025; 145: 2410–2416. https://doi.org/10.1016/j.jid.2025.04.024
- Padda IS, Bhatt R, Patel P. Upadacitinib. Pharma-Kritik ; 41: 45–48.
- Stirrat T, Thakker S, Bejugam D, Goh C, Lipner SR. Upadacitinib for alopecia: current evidence and clinical insights. Skin Appendage Disord 2025; 11: 320–334. https://doi.org/10.1159/000544868
- Ferreira C, King B, Torres T. JAK inhibitors for the treatment of vitiligo: current evidence and emerging therapeutic potential. Drugs (Abingdon Engl) 2025; 85: 1521–1534. https://doi.org/10.1007/s40265-025-02246-1
- Ackerman LS, Schlosser BJ, Zhan T, Prajapati VH, Fretzin S, Takahashi H, et al. Improvements in moderate-to-severe hidradenitis suppurativa with upadacitinib: Results from a phase 2, randomized, placebo-controlled study. J Am Acad Dermatol 2025; 92: 1252–1260. https://doi.org/10.1016/j.jaad.2024.12.046