ORIGINAL REPORT
Noninvasive Skin Surface Sampling Identifies Elevated IL-1β and IL-6 in Paediatric Atopic Dermatitis Patients
Amir HOREV1,2† , Sapir GABAY2,3†
, Sapir GABAY2,3† , Yuliya VALDMAN-GRINSHPOUN2,3
, Yuliya VALDMAN-GRINSHPOUN2,3 , Topaz ALFER4
, Topaz ALFER4 , Amir SZITENBERG5
, Amir SZITENBERG5 and Navit OGEN-SHTERN*
and Navit OGEN-SHTERN*
1Pediatric Dermatology Service, Soroka University Medical Center, Beer-Sheva, Israel, 2Faculty of Health Sciences, Ben-Gurion University of the Negev, Beer-Sheva, Israel, 3Department of Dermatology, Soroka University Medical Center, Beer-Sheva, Israel, 4The Skin Research Institute, the Dead Sea and Arava Science Center, Masada, Israel, and 5Ilana and Pascal Mantoux Institute for Bioinformatics, the Nancy and Stephen Grand Israel National Center for Personalized Medicine, Weizmann Institute of Science, Rehovot, Israel
†These authors contributed equally to this work.
Corr: Navit Ogen-Shtern, The Skin Research Institute, the Dead Sea and Arava Science Center, Masada, Israel, and Ben-Gurion University of the Negev, Eilat campus, Israel, Eilat. *Email: navit@adssc.org
Key words: Atopic dermatitis; Cytokines; Non-invasive sampling; paediatric.
Citation: Acta Derm Venereol 2026; 106: adv-2025-0010. DOI: https://doi.org/10.2340/actadv.v106.adv-2025-0010.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Sept 8, 2025. Accepted after revision: Mar 10, 2026.
Published: Apr 22, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
The data obtained in this study have not been deposited in a public database due to restrictions imposed by the IRB. Nevertheless, upon proper request from the corresponding author, we will be able to share the data.
The study was approved by the Ethics Review Committee of the Soroka University Medical Center (SUMC), Beer-Sheva, Israel (protocol #SCRC22018). The study was conducted in accordance with the Declaration of Helsinki and all appropriate amendments [Approval number: 0134–22-sor]. Written informed consent was obtained from one or both parents. Patients aged 16 and above (between 16 and 18) signed separate informed consent forms.
Despite extensive research, the molecular signature in atopic dermatitis (AD) remains underexplored. While previous studies have explored the expressions of pro-inflammatory markers in AD skin using biopsies and tape-stripping techniques, noninvasive sampling from the skin surface in paediatric AD patients has not been examined. This study aimed to identify and compare cytokine expression on the skin surface of children with AD and healthy controls. We enrolled 50 children, 40 with mild-to-moderate AD and 10 without AD as controls. Samples were collected using sticky bandages soaked in sterile phosphate-buffered saline, and concentrations of 15 cytokines were measured by a multiplex immunoassay. The effect of various factors on cytokine expression was assessed by redundancy analysis, while significance among groups was identified by factorial ANOVA. We found higher levels of IL-1β in nonlesional areas of AD patients compared to healthy subjects and increased expression of IL-1β and IL-6 in lesional areas compared to nonlesional skin. Factors such as skin folds and sex were found to affect cytokine secretion. This study demonstrates the feasibility of noninvasive cytokine sampling from the skin surface in paediatric AD patients. It underscores its potential for identifying additional biomarkers, complementing research on deeper skin layers.
SIGNIFICANCE
This study offers a painless, child-friendly method to understand atopic dermatitis (AD) in children. Instead of using invasive skin biopsies, we used special bandages to collect samples, making it much more comfortable for young patients. Our findings showed that certain inflammatory molecules, like IL-1β and IL-6, are consistently elevated in children with AD, which helps us better understand the disease’s underlying biology. The research also reveals that these molecular patterns differ based on individual characteristics like age and gender, and different areas of the body. Ultimately, this new method provides a valuable tool for physicians to track the disease and develop future personalized monitoring and treatment strategies, with reduced discomfort to children.
Although comprehensive research has been conducted on the pathogenesis of AD, the immune-molecular pathway remains under investigation, and the disease is understood to be a heterogeneous clinical condition that evolves over time. Several studies have demonstrated strong type 2 immune responses in AD patients (1). Moreover, additional activation of TH22, TH17/IL-23 and TH1 cytokine pathways has been identified depending on the subtype of AD. Understanding the broad molecular milieu of AD is a necessary step toward future management and targeted therapy for AD patients.
Up to now, cytokine and chemokine expression in the skin of adult AD patients has been evaluated mainly through invasive and prolonged procedures (punch biopsy, blister fluid and scraping) (2, 3). Minimal invasive methods are needed to evaluate levels of inflammatory mediators in paediatric patients. The tape-stripping technique allows a more accessible sampling of biomarkers from the outermost layer of the skin, the stratum corneum (2, 4, 5). Yet, applying the method on perturbed skin, to begin with, on children and infants may cause discomfort for the patient. Recently, a skin wash sampling technique was developed to quantify skin cytokines and other players in the skin secretome using a noninvasive method in psoriatic and AD adult patients (6, 7, 8). To date, no studies have investigated skin surface biomarkers of the paediatric population. Thus, the primary aim of the present study is to quantify cytokines on the skin surface of paediatric AD patients in lesional and nonlesional skin, using a modified noninvasive method inspired by skin surface washing sampling (9, 10, 11). A panel of 15 cytokines from the major inflammatory axes known to participate in AD was selected to demonstrate the method's feasibility. The keratinocyte-derived IL-1α/β and IL-6, which participate in innate/epithelial activation and play an upstream role in AD pathogenesis (9, 10). IL‑4, IL‑5, IL‑13 (type 2 immunity cytokines). IFN‑γ, IL‑12p70, IL‑17A, IL‑23 (type 1 and type 17 pathways), IL‑10 as part of the regulatory signaling and TNF‑mediated responses (TNF‑α, TNF‑β (10, 11). A secondary aim of the research is to compare the overall cytokine concentration between the skin of paediatric AD patients and healthy paediatric controls.
MATERIALS AND METHODS
Study design
A prospective study was conducted to identify key inflammatory cytokines present in the skin surface of paediatric AD patients. The study was approved by the Ethics Review Committee of the Soroka University Medical Center (SUMC), Beer-Sheva, Israel (protocol #SCRC22018). The study was conducted in accordance with the Declaration of Helsinki and all appropriate amendments [Approval number: 0134–22-sor]. Written informed consent was obtained from one or both parents. Patients aged 16 and older (between 16 and 18) signed separate informed consent forms. Patients were sampled at a single time point.
Study population
Patients were enrolled in the paediatric dermatology clinic at SUMC between August and November 2022. We included 50 children (≤18 years), 40 children diagnosed with mild-to-moderate AD [Investigator Global Assessment (IGA) scale score of 2–3] as a case group, and 10 paediatric patients without AD as a control group. Inclusion criteria included children between 0 and 18 years old, diagnosed clinically with AD, and having IGA scores between 2 and 3. Exclusion criteria included systemic or biological therapy or phototherapy within 28 days of the trial, the use of topical corticosteroids or calcineurin inhibitors within 7 days of the trial, and pregnancy. The mean age of the AD group was 5.86 ± 3.99 years, comparable to the healthy control group (6.81 ± 3.92 years; p=0.505). Gender distribution was balanced across groups, confirming that demographic characteristics did not differ significantly between AD and control participants. A summary of the demographic and Clinical Characteristics of the Study Population is exhibited in Table SI.
Sample collection
A dermatologist carried out a complete medical history and skin examination upon arrival. Additional essential data were collected, including age, gender, ethnicity, atopy history (respiratory asthma, food allergy), family history of atopy, breastfeeding and Fitzpatrick skin type (1–6). Samples were obtained from two locations: 1. Lesional skin and 2. unaffected healthy skin from the same anatomical region of the lesional skin and/or, if not available, at least 10 cm farther from the lesional sample. In addition, samples (lesional sample in the case group and healthy sample from the control group) were subclassified into fold vs nonfold and anatomical location (arm, leg, head and neck, and trunk).
Samples were taken noninvasively by placing a sticky bandage (shockproof bandages, 7.5×5, Life) soaked in sterile phosphate-buffered saline (PBS) from a 2×2 cm skin area for 15–20 min. After the time mentioned, the bandages were removed and stored at −40°C until processing.
Cytokine extraction and quantification
The PBS was extracted from all bandages by squeezing using sterile tweezers. The fluids were stored in microcentrifuge tubes at −80°C until analysed. Measurements of concentrations of 15 cytokine (IFNγ, IL-1α, IL-1β, IL-2, IL-4, IL-5, IL-6, IL-10, IL-12p70, IL-13, IL-15, IL-17A, IL-23, TNFα and TNFβ) were determined by multiplex analysis (Quansys Biosciences, London).
Statistical analysis
Three data sets were analyzed separately: 1) nonlesional patient+control samples, 2) lesional+nonlesional patient samples and 3) lesional+control samples. Each dataset was normalized with standard scaling, (Scikit Learn 1.3), and extreme outliers (samples 88, 28, 87, 86, 33, 65, and 40) were excluded, leaving 9 control, 40 non‑lesional, and 34 lesional samples.
To assess the effect of clinical and sampling factors on cytokine levels, we used redundancy analysis (RDA) approach (12) to relate cytokine matrices to metadata and estimate the variance explained by each factor (patient vs control, lesional vs non‑lesional, body site, fold vs non‑fold, with additional covariates). Principal component analysis (PCA; Scikit‑learn 1.3) Scikit Learn 1.3 (13) was used to visualize overall variance structure (14).
To identify cytokines whose expression changes significantly across the groups, we performed a factorial ANOVA for each cytokine separately, as implemented in statsmodels version 0.14. This test was carried out for each subset. For each subgroup, the model considered was the same as the one used for the RDA.
RESULTS
Fifty children were enrolled, including 40 with mild-to-moderate AD and 10 without AD as controls. We used RDA to quantify the contribution of factors to the variance between the control and lesional samples taken from patients (12). The factors included in the analysis were whether the sample was taken from a control group (healthy subjects), body site, skin type, age and gender. The analysis reveals that the model accounts for 61% of the variance (p=0.001). Around 12.4% of the differences are attributed to whether the sample is taken from a control individual or from lesional areas in AD patients (p=0.005). Additionally, significant factors explaining the variance are skin type (23.8%, p=0.004), age (11.9%, p=0.032) and body site (11.4%, p=0.09). A principal component analysis (PCA) reveals that IL-6, IL-1α and IL-1β were the primary variance factors along principal component 2 (PC2). The remaining cytokines only explained principal component 1 (PC1) (Fig. 1a).
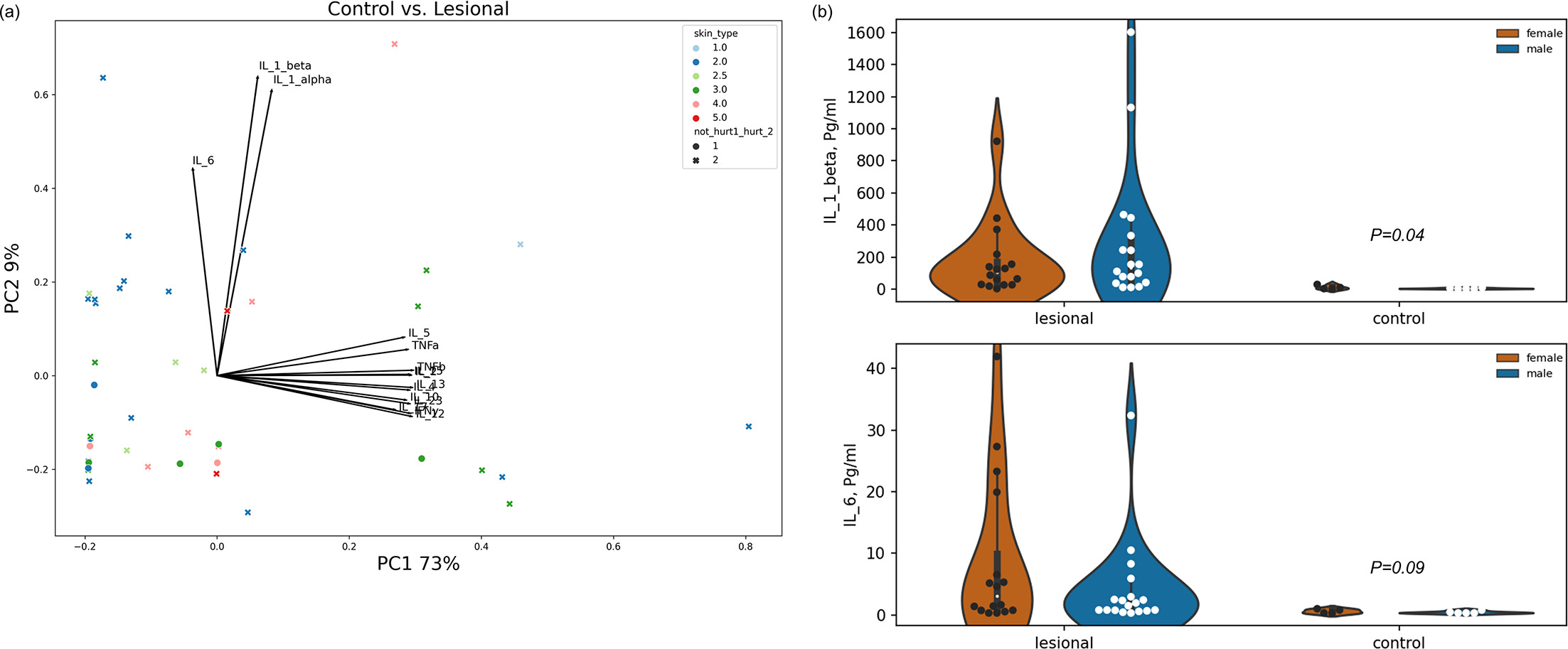
Fig. 1. Differences in cytokine expression at the surface of lesional skin areas compared to the control group. (A) PC1 and PC2 represent 73% and 9% of the total variance in cytokine expression, respectively. Skin types are colour-coded as indicated in the colour key (top right). Circular markers represent control samples, and x-markers represent lesional samples. Arrow lengths and directions represent the loading of each cytokine along each of the PCs. (B) The violin plots exhibit the distribution of expression of IL-1β and IL-6 in lesional samples compared to the control group. The calculated variances between the two groups are indicated above the control group. Each circular marker represents a patient or a healthy volunteer.
Accordingly, we examined the differences between the cytokines detected on the skin surface of healthy individuals and lesional areas in AD patients. The significance was calculated by using a factorial ANOVA to each cytokine. The analysis shows that IL-1β is significantly overexpressed on the skin surface of lesional areas in AD patients compared to healthy individuals (p=0.04). IL-6 also exhibited a similar pattern, although it was not significant (p=0.08, Fig. 1b).
When examining the differences in cytokine amounts between lesional and nonlesional areas of paediatric AD patients, the quantified contribution of factors to the variance by RDA explains 88% of the variance (p=0.001) with a breakdown of patient 17.6% (p=0.001); skin type 6.9% (p=0.002); breastfeeding 5.9% (p=0.001); gender 3.2% (p=0.002); age 2.6% (p=0.026); fold vs nonfold 1.8% (p=0.012); and family history 1.8% (p=0.075). Other factors did not contribute significantly to the total variance. A PCA to detect differences in cytokine measurements between nonlesional and lesional areas shows that IL-1α, IL-1β and IL-6 were the main factors of the variance along PC2 (8%). The remaining cytokines explained only PC1 (69%, Fig. 2a). We tested differences in the concentrations of individual cytokines between lesional vs nonlesional skin areas, as previously described, using factorial ANOVA. This analysis revealed significantly higher amounts of IL-1β and IL-6 in lesional areas compared to nonlesional skin surface (Fig. 2b). The remaining cytokines did not exhibit significant differences in expression.
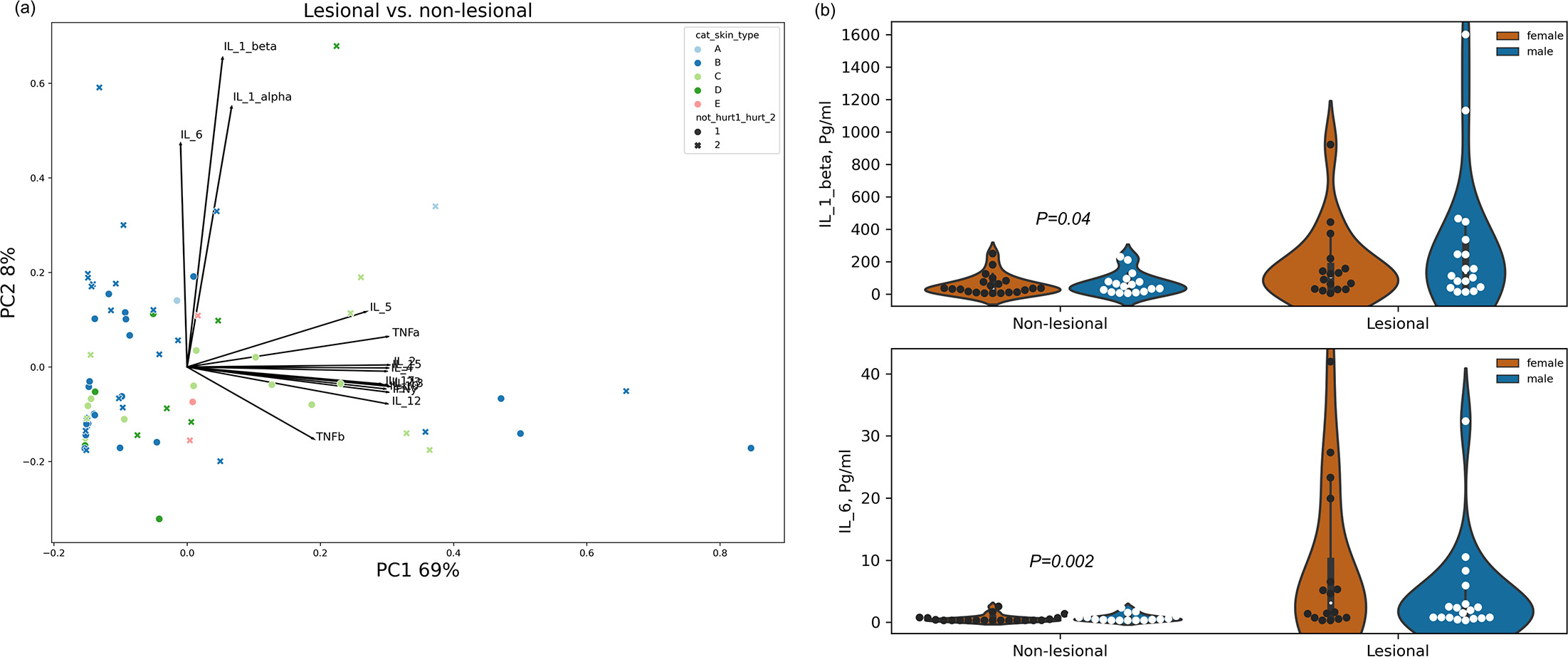
Fig. 2. Differences in cytokine expression at the surface of lesional skin areas compared to nonlesional skin areas. (A) A PCA analysis. PC1 and PC2 represent 69% and 8% of the total variance in cytokine expression, respectively. Skin types are colour-coded as indicated in the colour key. Circular markers represent nonlesional samples, and x-markers represent lesional samples. Arrow lengths and directions represent the loading of each cytokine along each of the PCs. (B) The violin plots exhibit the distribution of expression of IL-1β and IL-6 in lesional samples compared to nonlesional group. The calculated variances between the two groups are indicated above the nonlesional group. Each circular marker represents a patient or a healthy volunteer.
Differences were also observed in cytokine measurements on the skin surface of the control group compared to samples from nonlesional areas. An RDA analysis taking account of control vs patient group, body site, skin type, age and gender explains only 28% of the variance, out of which 10.2% of the differences are attributed to whether the sample is taken from a control individual or nonlesional areas in AD patients (p=0.021). Other factors did not contribute significantly to the total differences. A PCA reveals that IL-1α and IL-1β are the primary factors contributing to the variance along PC2, alongside IL-5 and TNFβ. The remaining cytokines only explained PC1 (Fig. 3a). The statistical significance of differences in cytokine levels between the study groups indicates that IL-1β is significantly overexpressed on the skin surface of nonlesional areas in AD patients compared to healthy individuals (p=0.032). IL-1α, IL-5 and TNF-β presented a similar pattern; however, the differences were insignificant (p>0.05, Fig. 3b).
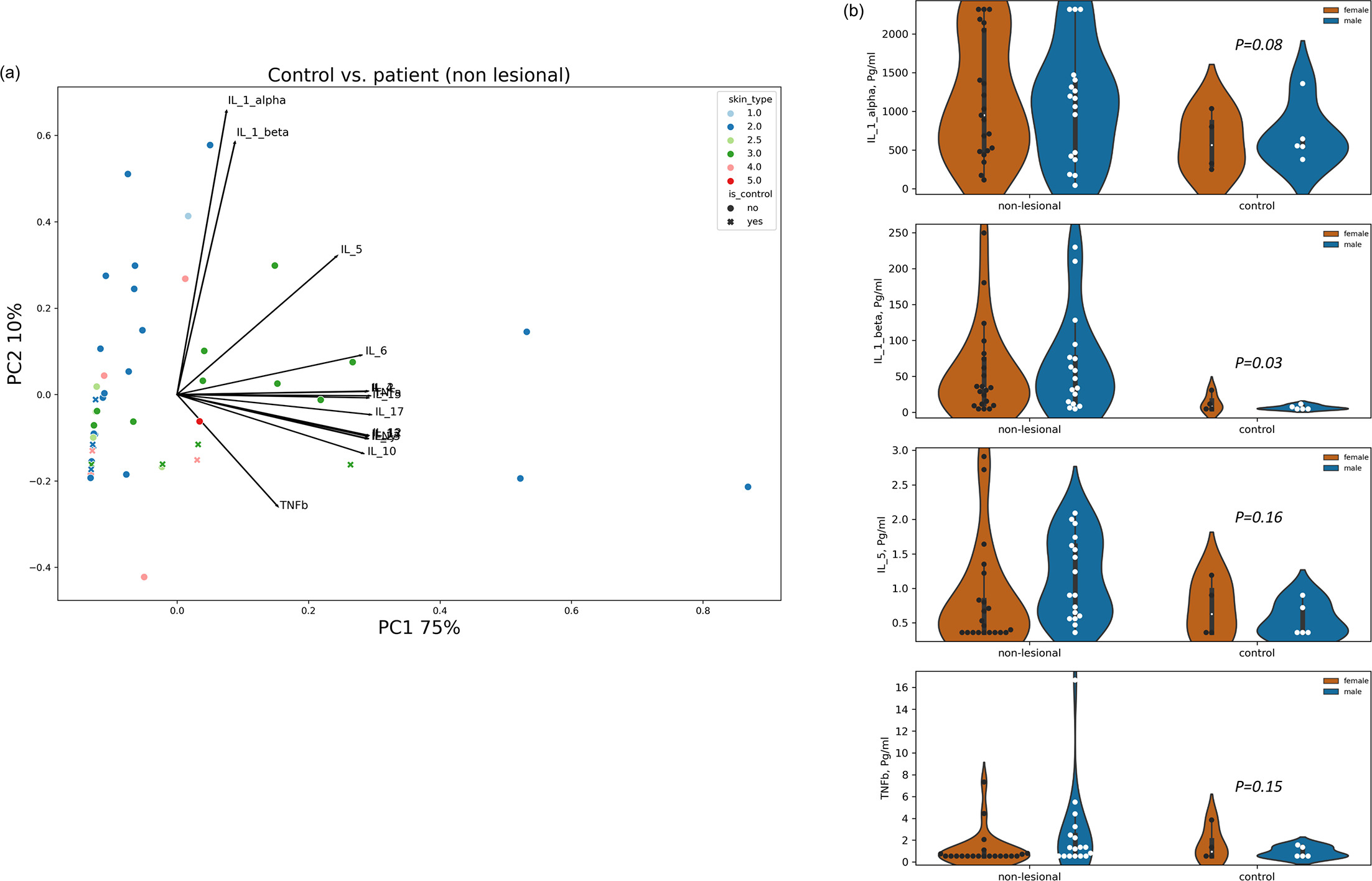
Fig. 3. IL-1β is overexpressed on the skin surface of nonlesional areas of AD patients compared to the control. (A) A PCA analysis. PC1 and PC2 represent 75% and 10% of the total variance in cytokine expression, respectively. Skin types are colour-coded as indicated in the colour key. Circular markers represent nonlesional samples and x-markers represent control samples. Arrow lengths and directions represent the loading of each cytokine along each of the PCs. (B) The violin plots exhibit the distribution of expression of IL-1β and IL-1α, IL-5 and TNFα in nonlesional samples compared to the control group. The calculated variances between the two groups are presented above the control group. Each circular marker represents a patient or a healthy volunteer.
Next, we attempted to determine whether various characteristics of each sample (sampling site, fold vs nonfold, gender, atopic history, family history, ethnicity, skin type, age and IgA) could account for the variation in cytokine concentration on the skin surface of nonlesional areas in AD patients. Considering these factors, an RDA analysis explains 70% of the variance (p=0.07). The skin type contributed most to the variance in total cytokine expression (24%, p=0.022), and the gender factor may also contribute to the differences (5.9%, p=0.06). Yet, when examining the effect of these characteristics on the detection of individual cytokines on the surface of nonlesional skin in AD patients, the most significant impact is attributed to the fold vs nonfold factor. We observed substantial differences in the measurements of eight cytokines (IL-2, IL-4, IL-5, IL-6, IL-15, IL-17, TNF-α and TNF-β) between folded and nonfolded areas of the nonlesional skin surface, where the average cytokine concentrations and variability among different individuals were both higher in the folded areas (Fig. 4). A more significant variation was observed between fold and nonfold areas in female patients. In contrast, less difference was evident among boys, which may be explained by higher cytokine levels expressed in nonfold areas in male patients compared to female patients (Fig. 4).
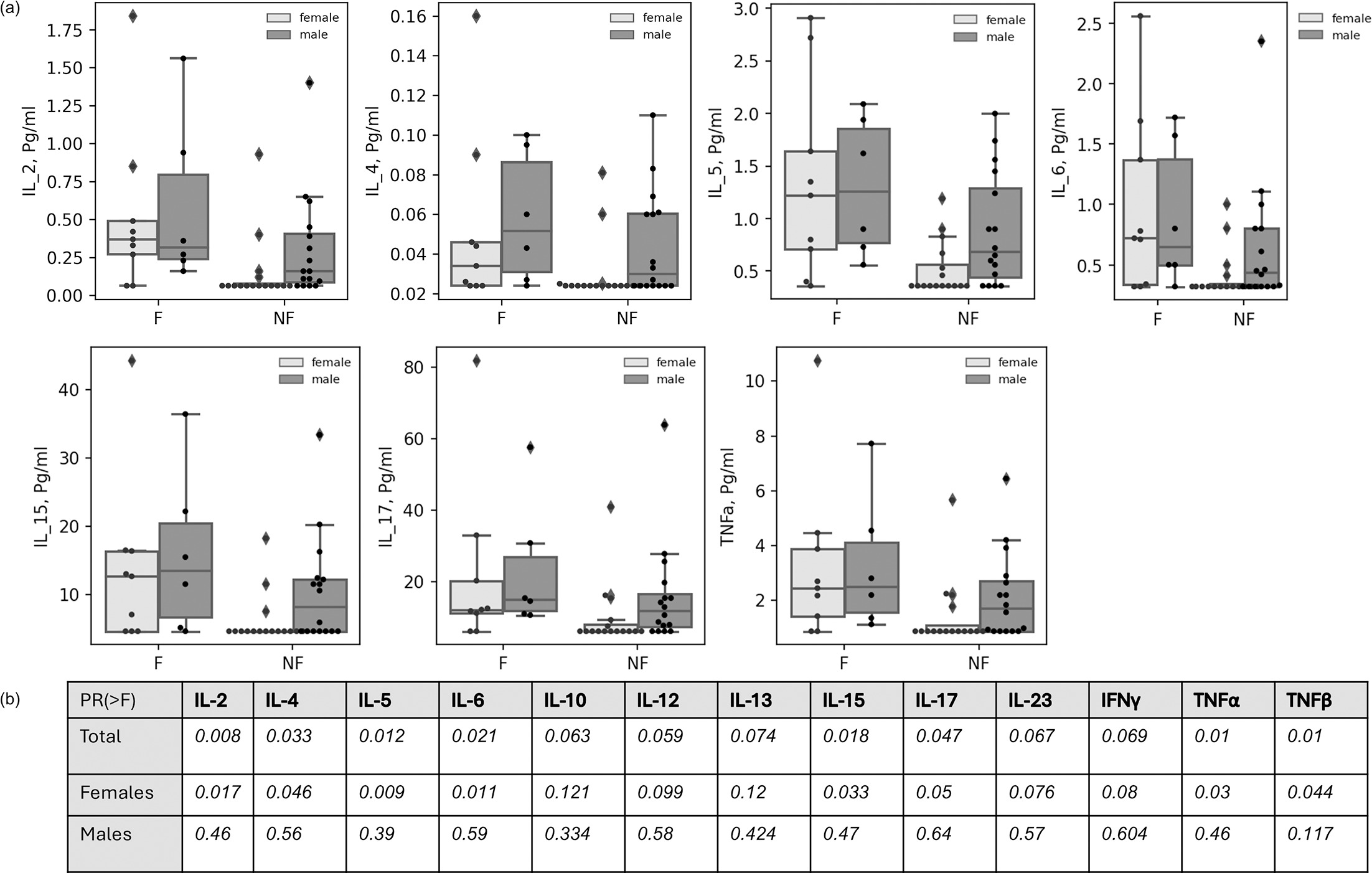
Fig. 4. Pro-inflammatory cytokines are overexpressed at folded nonlesional skin areas compared to nonfolded sites. (A) Boxplots of cytokine expression on the skin surface of nonlesional sites of AD patients. Dark boxplots are for samples taken from male patients. Light boxplots are for samples taken from female patients. Each circular marker represents a data point. Diamonds are for sections of distribution that extend the whisker. (B) Significance for differences in cytokine expression of folded compared with nonfolded skin areas. F, fold; NF, nonfold.
As for the variance for the lesional group, when analysing the differences of cytokine concentration for each cytokine separately, a decisive factor effect of fold vs nonfold factor is observed on the expression of IL-2 (p=0.06), IL-4 (p=0.023), IL-5 (p=0.029), IL-13 (0.058), IL-15 (p=0.012) and TNF (p=0.048), as seen in Fig. 5. The differences are less related to patients' gender than those observed in nonlesional areas.
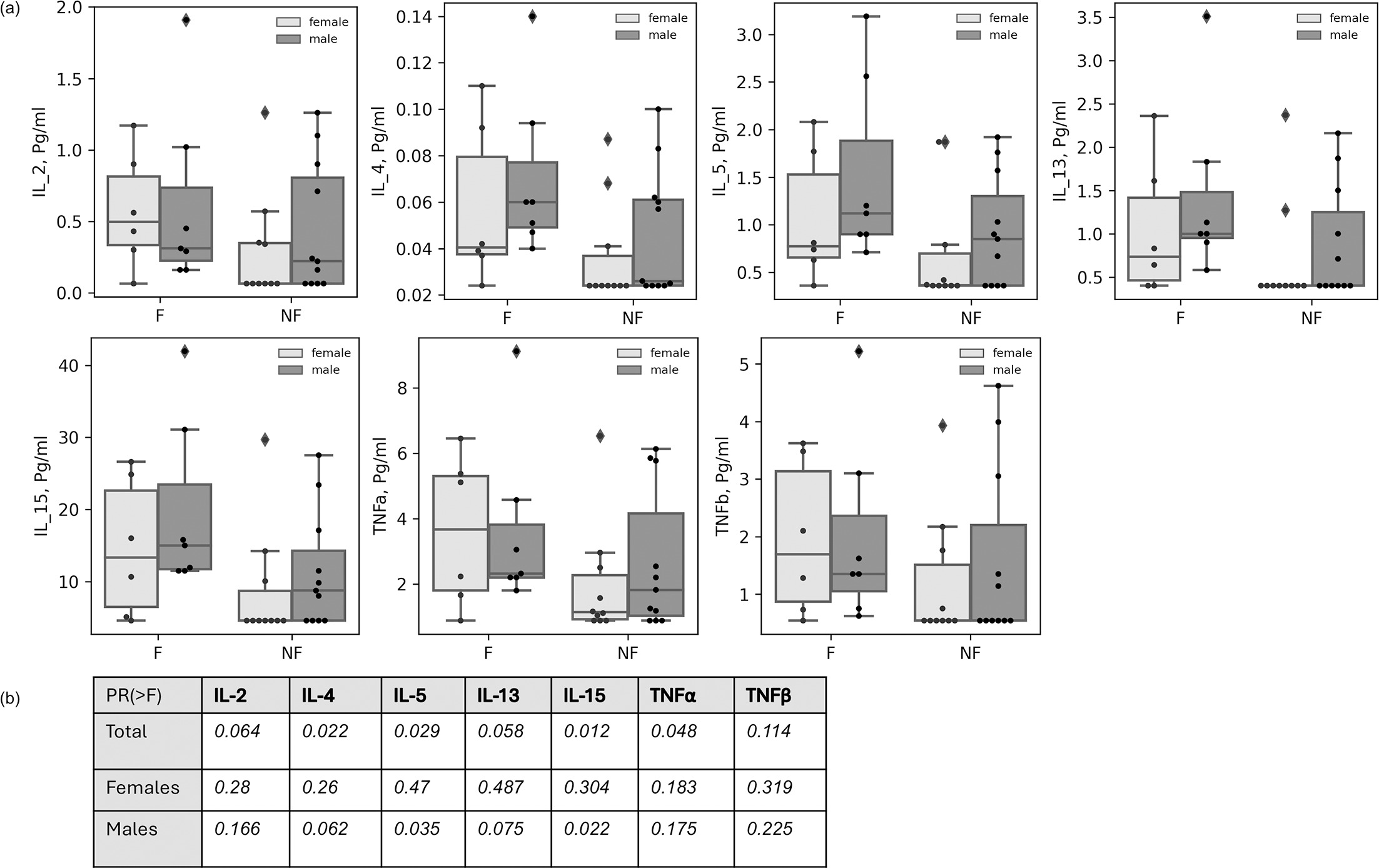
Fig. 5. Differences in cytokine expression at folded lesional skin compared to nonfolded areas. (A) Boxplots of cytokine expression. Dark boxplots are for samples taken from male patients. Light boxplots are for samples taken from female patients. Each circular marker represents a data point. Diamonds are for sections of distribution that extend the whisker. (B) Significance for differences in cytokine expression of folded compared with nonfolded skin areas. F, fold; NF, nonfold.
DISCUSSION
The current study aimed to identify specific cytokines at the skin surface of paediatric patients as unique biomarkers of AD. Measuring cytokines and other inflammatory markers at the protein level directly on the skin surface of pediatric AD patients is important to capture the actual local inflammatory activity at the site of the disease. Previous studies demonstrated the ability to differentiate healthy skin secretome from diseased skin, such as ectodermal dysplasia (15) or psoriasis (9). We decided to measure the abundance of 15 key-inflammatory cytokines using multiplex analysis and detect possible factors that may shape the composition of topical cutaneous cytokines in children with AD.
When we quantified the contribution of factors to the total variance between the control and nonlesional groups, 10% of the variance was explained by whether the sample was taken from an AD patient or a healthy individual. Other factors, such as body site, skin type, gender or age, did not contribute to the total variance, suggesting that most of the variance likely originates from the subjects rather than any common factor. Variance in of IL-1β and IL-1α contributed most to the total variance explained by AD. The nonlesional secretome contained significantly higher levels of IL-1β compared to the control group.
Moreover, IL-1β increased in lesional skin compared to nonlesional skin. IL-1α was also increased; however, it was not statistically significant. IL-1 family members are known to be involved in skin diseases in general (9, 16) and in inflammatory skin diseases. Overexpression of IL-1β was observed in samples taken from the stratum corneum of lesional skin from the upper arm of paediatric AD patients, along with IL-8 and IL-18 (4). IL-1β was also positively correlated with a higher disease severity score and a perturbed skin barrier as tested by transepidermal water loss (17).
Interestingly, in the same study, IL-1α levels were decreased compared to the control group, which is opposite to our observation. This may be due to high variations in IL-1α levels at the skin surface (presented in Fig. S1). Changes in the presence of IL-1 cytokines at the skin surface may partly be explained by changes in the skin microbiome or mycobiome in AD patients. Dysbiosis of the skin microbiome, mainly by overgrowth of Staphylococcus aureus (S. aureus), is one of the hallmarks of AD progression (18, 19). Possibly, the presence of the bacterium in AD skin is not only a result of the disease but may also contribute to the development of the disease by further inducing inflammatory reactions mediated, in part, by IL-1 cytokines, which are known to be involved in the pathogenesis or progression of inflammatory skin diseases, including AD (16). Brandwein et al. demonstrated that microbiota samples from AD patients’ nonlesional and lesional skin sites exhibit similar features (20). This supports our observation of overexpressed cytokines in both nonlesional and lesional skin sites, with particular emphasis on IL-1 cytokines.
Additionally, the expression of IL-6 is increased in the secretome of lesional sites, compared to that of nonlesional sites or healthy individuals. IL-6 is known to participate in the pathogenesis and progression of the disease along with other innate cytokines (21). Tocilizumab, a biologic drug that suppresses the IL-6 receptor and is used to treat rheumatoid arthritis, was also investigated as a possible treatment for AD (22). IL-6 was also found to be expressed at higher levels in the stratum corneum (obtained by tape stripping) and whole skin (obtained by punch biopsy) of adult AD patients (23). Moreover, IL-6 is overexpressed by different cells isolated from AD or allergic asthma patients (24, 25, 26). Apart from the clinical findings reported by others and us, the cytokine is used as a standard marker for the induction of inflammation in 2D or 3D laboratory skin models. Stimulation of keratinocytes or reconstructed skin equivalents by AD-like conditions (such as cytokine cocktails or exposure to skin pathogens) results in increased secretion of IL-6 (27, 28, 29).
Interestingly, but not surprisingly, we found that the tested panel of cytokines revealed the surface of paediatric atopic skin is characterized by overexpression of cytokines associated with the innate immune response. At the same time, we also observed significant differences in the expression of Th2-type cytokines (IL-4, IL-5 and IL-13) and IL-17 in fold areas (e.g. popliteal fossa) compared to unfolded areas of the skin. This phenomenon is observed for both lesional and nonlesional skin. Many variables may contribute to this finding. The expression of cytokines in folded versus nonfolded areas, when the data are separated by sex, reveals a trend in which, in girls, fewer cytokines are expressed in non-folded areas than in boys. This difference disappears in folded areas. We assumed that the general difference in cytokine expression mainly stems from differences between males and females. Changes in lipid composition, microbiome population, humidity and other factors are likely responsible for differences in the skin secretome between folded and unfolded areas. These questions remain to be elucidated in future studies.
Our study possesses several strengths that underscore its scientific relevance. A primary strength is its novel use of a noninvasive method to collect data in children, an area with limited existing data. Additionally, our prospective analysis, along with comparison to the control group, enhances the robustness of our findings by offering valuable insights into AD surface cytokines. However, there are some limitations to consider. We did not quantify all relevant cytokines or chemokines known to be involved in AD pathogenesis. Nevertheless, we demonstrated the ability to characterize unique biomarkers in the skin secretome of paediatric AD patients. In addition, the single-centre design may limit the generalizability of the findings to broader populations.
Our findings confirm that noninvasive skin surface sampling is a reliable approach to characterize the inflammatory profile of paediatric AD. Using this method, we identified significant overexpression of IL-1β and IL-6, not only in lesional but also in nonlesional skin, suggesting that inflammatory activity extends beyond visibly affected areas. Furthermore, we demonstrated that cytokine expression is shaped by individual and anatomical characteristics, including skin type, sex, age and the presence of skin folds, highlighting the heterogeneity of the disease. These insights underscore the potential of surface biomarkers to reflect both systemic and localized disease processes. Beyond providing a painless, child-friendly sampling strategy, this approach could facilitate longitudinal monitoring of disease activity, potentially improve early detection of flares and guide the development of more personalized treatment strategies in paediatric AD. Future studies involving larger, more diverse populations will be crucial for validating these biomarkers and exploring their integration into clinical practice. Furthermore, this method can be further developed and applied to test multiple panels of proteins and molecules for various skin conditions, providing complementary insights into protein expression in deeper skin layers.
ACKNOWLEDGEMENTS
Experimental design: AH, NOS, Sampling and experiments: SIG, TA, data analysis: AS, NOS, writing: AH, SIG, NOS, supervision: YVG. Manuscript review and approval: All authors.
REFERENCES
- Brunner PM, Guttman-Yassky E, Leung DYM. The immunology of atopic dermatitis and its reversibility with broad-spectrum and targeted therapies. J Allergy Clin Immunol 2017; 139: S65–S76. https://doi.org/10.1016/j.jaci.2017.01.011
- Gros E, Bussmann C, Bieber T, Förster I, Novak N. Expression of chemokines and chemokine receptors in lesional and nonlesional upper skin of patients with atopic dermatitis. J Allergy Clin Immunol 2009; 124: 753–60. https://doi.org/10.1016/j.jaci.2009.07.004
- Koppes SA, Brans R, Ljubojevic Hadzavdic S, Frings-Dresen MHW, Rustemeyer T, Kezic S. Stratum corneum tape stripping: monitoring of inflammatory mediators in atopic dermatitis patients using topical therapy. Int Arch Allergy Immunol 2016; 170: 187–193. https://doi.org/10.1159/000448400
- Andersson AM, Sølberg J, Koch A, Skov L, Jakasa I, Kezic S, et al. Assessment of biomarkers in pediatric atopic dermatitis by tape strips and skin biopsies. Allergy 2022; 77: 1499–1509. https://doi.org/10.1111/all.15153
- Del Duca E, Dahabreh D, Kim M, Bar J, Da Rosa JC, Rabinowitz G, et al. Transcriptomic evaluation of skin tape-strips in children with allergic asthma uncovers epidermal barrier dysfunction and asthma-associated biomarkers abnormalities. Allergy 2024; 79: 1516–1530. https://doi.org/10.1111/all.16060
- Røpke MA, Mekulova A, Pipper C, Eisen M, Pender K, Spee P, et al. Non‐invasive assessment of soluble skin surface biomarkers in atopic dermatitis patients—effect of treatment. Skin Res Technol 2021; 27: 715–722. https://doi.org/10.1111/srt.13006
- Portugal-Cohen M, Kohen R. Non-invasive evaluation of skin cytokines secretion: an innovative complementary method for monitoring skin disorders. Methods 2013; 61: 63–68. https://doi.org/10.1016/j.ymeth.2012.10.002
- Portugal-Cohen M, Horev L, Ruffer C, Schlippe G, Voss W, Ma’or Z, et al. Non-invasive skin biomarkers quantification of psoriasis and atopic dermatitis: cytokines, antioxidants and psoriatic skin auto-fluorescence. Biomed Pharmacother 2012; 66: 293–299. https://doi.org/10.1016/j.biopha.2011.12.009
- Iznardo H, Puig L. IL-1 family cytokines in inflammatory dermatoses: pathogenetic role and potential therapeutic implications. Int J Mol Sci 2022; 23: 9479. https://doi.org/10.3390/ijms23169479
- Fania L, Moretta G, Antonelli F, Scala E, Abeni D, Albanesi C, et al. Multiple roles for cytokines in atopic dermatitis: from pathogenic mediators to endotype-specific biomarkers to therapeutic targets. Int J Mol Sci 2022; 23: 2684. https://doi.org/10.3390/ijms23052684
- Yamamura Y, Nakashima C, Otsuka A. Interplay of cytokines in the pathophysiology of atopic dermatitis: insights from Murin models and human. Front Med 2024; 11: 1342176. https://doi.org/10.3389/fmed.2024.1342176
- Sankhyā: The Indian Journal of Statistics, Series A (1961-2002). JSTOR; [cited 2024 August 12]. Available from: https://www.jstor.org/journal/sankhyaseriesa
- Pedregosa F, Varoquaux G, Gramfort A, Michel V, Thirion B, Grisel O, et al. Scikit-learn: machine learning in Python. J Mach Learn Res 2011; 12: 2825–2830.
- Pearson K. LIII. On lines and planes of closest fit to systems of points in space. Philos Mag J Sci 1901; 2: 559–572. https://doi.org/10.1080/14786440109462720
- Burian M, Velic A, Matic K, Günther S, Kraft B, Gonser L, et al. Quantitative proteomics of the human skin secretome reveal a reduction in immune defense mediators in ectodermal dysplasia patients. J Invest Dermatol 2015; 135: 759–767. https://doi.org/10.1038/jid.2014.462
- Gómez-García F, Ruano J, Gay-Mimbrera J, Aguilar-Luque M, Sanz-Cabanillas JL, Hernández Romero JL, et al. A scoping review protocol to explore the use of interleukin-1-targeting drugs for the treatment of dermatological diseases: indications, mechanism of action, efficacy, and safety. Dermatol Ther 2018; 8: 195–202. https://doi.org/10.1007/s13555-018-0235-4
- Lyubchenko T, Collins HK, Goleva E, Leung DYM. Skin tape sampling technique identifies proinflammatory cytokines in atopic dermatitis skin. Ann Allergy Asthma Immunol 2021; 126: 46–53. https://doi.org/10.1016/j.anai.2020.08.397
- Nakatsuji T, Gallo RL. The role of the skin microbiome in atopic dermatitis. Ann Allergy Asthma Immunol 2019; 122: 263–269. https://doi.org/10.1016/j.anai.2018.12.003
- Bjerre RD, Bandier J, Skov L, Engstrand L, Johansen JD. The role of the skin microbiome in atopic dermatitis: a systematic review. Br J Dermatol 2017; 177: 1272–1278. https://doi.org/10.1111/bjd.15390
- Brandwein M, Fuks G, Israel A, Sabbah F, Hodak E, Szitenberg A, et al. Skin microbiome compositional changes in atopic dermatitis accompany Dead Sea climatotherapy. Photochem Photobiol 2019; 95: 1446–1453. https://doi.org/10.1111/php.13119
- Peng W, Novak N. Pathogenesis of atopic dermatitis. Clin Exp Allergy 2015; 45: 566–574. https://doi.org/10.1111/cea.12495
- Joshi P, Dhaneshwar SS. An update on disease modifying antirheumatic drugs. Inflamm Allergy Drug Targets 2014; 13: 249–261. https://doi.org/10.2174/187152811304140915152102
- Del Duca E, He H, Liu Y, Pagan AD, David E, Cheng J, et al. Intrapatient comparison of atopic dermatitis skin transcriptome shows differences between tape-strips and biopsies. Allergy 2024; 79: 80–92. https://doi.org/10.1111/all.15845
- Toshitani A, Ansel JC, Chan SC, Li SH, Hanifin JM. Increased interleukin 6 production by T cells derived from patients with atopic dermatitis. J Invest Dermatol 1993; 100: 299–304. https://doi.org/10.1111/1523-1747.ep12469875
- Wong CK, Ho CY, Ko FW, Chan CH, Ho AS, Hui DS, et al. Proinflammatory cytokines (IL-17, IL-6, IL-18 and IL-12) and Th cytokines (IFN-gamma, IL-4, IL-10 and IL-13) in patients with allergic asthma. Clin Exp Immunol 2001; 125: 177–183. https://doi.org/10.1046/j.1365-2249.2001.01602.x
- Wang Y, Wu Y, Gu C, Wang S, Yin H, Zhu R, et al. Peripheral blood mononuclear cell- transcriptome signatures of atopic dermatitis and prediction for the efficacy of dupilumab. J Dermatol Sci 2023; 111: 83–92. https://doi.org/10.1016/j.jdermsci.2023.06.002
- Gallegos-Alcalá P, Jiménez M, Cervantes-García D, Salinas E. The keratinocyte as a crucial cell in the predisposition, onset, progression, therapy and study of atopic dermatitis. Int J Mol Sci 2021; 22: 10661. https://doi.org/10.3390/ijms221910661
- Lee SH, Bae IH, Choi H, Choi HW, Oh S, Marinho PA, et al. Ameliorating effect of dipotassium glycyrrhizinate on an IL-4- and IL-13-induced atopic dermatitis-like skin-equivalent model. Arch Dermatol Res 2019; 311: 131–140. https://doi.org/10.1007/s00403-018-1883-z
- Morgner B, Tittelbach J, Wiegand C. Induction of psoriasis- and atopic dermatitis-like phenotypes in 3D skin equivalents with a fibroblast-derived matrix. Sci Rep 2023; 13: 1807. https://doi.org/10.1038/s41598-023-28822-7